Abstract
Introduction
Transcutaneous neuromuscular electrical stimulation (TNMES) has succeeded in facilitating therapy for individuals with neuromuscular disorders.1-5 For example, for individuals with neurologically incomplete spinal cord injury (SCI), combined use of TNMES and locomotor training has been shown to be more effective in improving their ambulation skills than other clinical approaches.6,7 In individuals with complete SCI, TNMES has been demonstrated to enhance muscle strength and mass.8-11 A critical limitation with these rehabilitative approaches is the rapid onset of muscle fatigue during repeated contractions.1-3,12 The functional consequence of this rapid muscle fatigue during TNMES is early force decay,13,14 which is a critical issue during therapies, such as muscle strengthening and cardiovascular fitness exercises, aimed to promote physiological and functional improvement in paralyzed limbs.1-7 One of the main reasons of the increased fatigability with TNMES is believed to be a localized nerve excitation, repeatedly and synchronously activating only certain subset(s) of motor units using fixed parameters.12,15,16 Another reason for the fatigability may be a reversal of the size principle of recruitment when larger axons that innervate the more easily fatigable fibers are recruited at low stimulus magnitudes and the smaller axons follow with increased stimulation levels, 12 but this reason is controversial. 4 In addition, in individuals with neuromuscular disorders, muscle fatigue can be exacerbated with depletion of substances, accumulation of catabolites, and problems in excitation–contraction coupling.17-20 Consequently, the paralyzed muscles show greater fatigability than healthy muscle.19,21-25
Because synchronous activation of an entire muscle is believed to be one of the principle causes of rapid muscle fatigue during TNMES,2,12 an approach utilizing activation of several muscle subcomponents independently seems feasible in reducing fatigue. This approach was implemented invasively in animal experimental models using spinal stimulation,26-28 intrafascicular stimulation,29,30 interfascicular stimulation, 31 epineural stimulation, 27 and intramuscular stimulation.32-34 However, observations on this approach in humans are limited. Pournezam et al 35 applied sequential stimulation to 3 knee extensor muscles in 2 individuals using 3 active surface electrodes distributed over these muscles. Malešević and colleagues 36 investigated fatigue reduction using sequential stimulation of the knee extensor muscles through 4 active surface electrodes distributed over quadriceps as compared with 1 active electrode. Decker et al 37 sequentially stimulated knee extensor muscles in functional electrical stimulation (FES) cycling in individuals with SCI. In all 3 studies, the method requires activation of several synergistic muscles independently, which makes this method feasible only for a large group of synergistic muscles, such as the knee extensors.
We have developed a method called spatially distributed sequential stimulation (SDSS) to reduce muscle fatigue by distributing the center of electrical field over a wider area within a single stimulation site, using an array of surface electrodes. 38 Our method is unique in a sense that, while the stimulation is interleaved in a similar manner to other studies,35-37 it is not applied to different muscles but instead is distributed between multiple active surface electrodes that are collocated at the same site and over the same area as during stimulation with a single active electrode. Thus, this method can be applied when it is difficult or not possible to distribute stimulation between synergistic muscles in contrast to previous studies.35-37 Indeed, the same method was tested successfully for a small finger flexor by Maneski et al. 39
The feasibility of SDSS was tested in a pilot study where the paralyzed plantarflexors of an individual with complete SCI were stimulated. 38 SDSS showed a drastic fatigue reduction effect in 2 minutes of isometric plantarflexion. More recently, we demonstrated that SDSS can reduce muscle fatigue in plantarflexors in the able-bodied population and investigated the mechanism of SDSS. 40 We demonstrated that different sets of muscle fibers are activated alternately by different electrodes, which is closer to physiological activation and effective in fatigue reduction. The purpose of the present study was to extend the previous findings and to prove the feasibility of the method by exploring the fatigue-reducing ability of SDSS for other lower limb muscle groups including thigh muscles in the able-bodied population, as well as in individuals with SCI. Testing with thigh muscles and testing the effectiveness of SDSS in individuals with SCI are important steps for translating this method into a clinical application, since thigh muscles are the target muscles of muscle strengthening as well as cardiovascular exercises where fatigue-reduction is most valuable.
Materials and Methods
Study Participants
Experiments were conducted in 11 able-bodied and 17 SCI participants (Table 1). The number of participants analyzed for each muscle group varied according to data availability (see Results). Each participant gave written informed consent to the experimental procedure. Able-bodied participants were free from any lower-limb injury in the previous 6 months and had no lower extremity surgery in the 2 years prior to study participation. None of the participants had any history of neurological or circulatory disorders. Individuals who were admitted to the SCI rehabilitation program at Toronto Rehabilitation Institute-UHN and met all the inclusion and exclusion criteria were invited to participate. The inclusion criteria were the following: SCI ranging from cervical 4 to thoracic 12 spinal segments; at least 12 months postinjury; ability to sit up on a chair with a backrest. We excluded individuals who suffer from serious cognitive or psychological impairments; with bone fractures following the injury, and/or associated with decreased bone mineral density; required medication for the prevention or treatment of autonomic dysreflexia or orthostatic hypotension; had injuries, open wounds, or rashes at the sites where the electrodes would be placed; had severe contractures in lower extremities. All participants were requested to refrain from strenuous exercise for 24 hours prior to and between the testing sessions, and all were naïve for FES on leg muscles. This study was approved by the local ethics committee in accordance with the Declaration of Helsinki on the use of human subjects in experiments.
Demographic and Anthropometric Characteristics of Able-Bodied and SCI Participants.
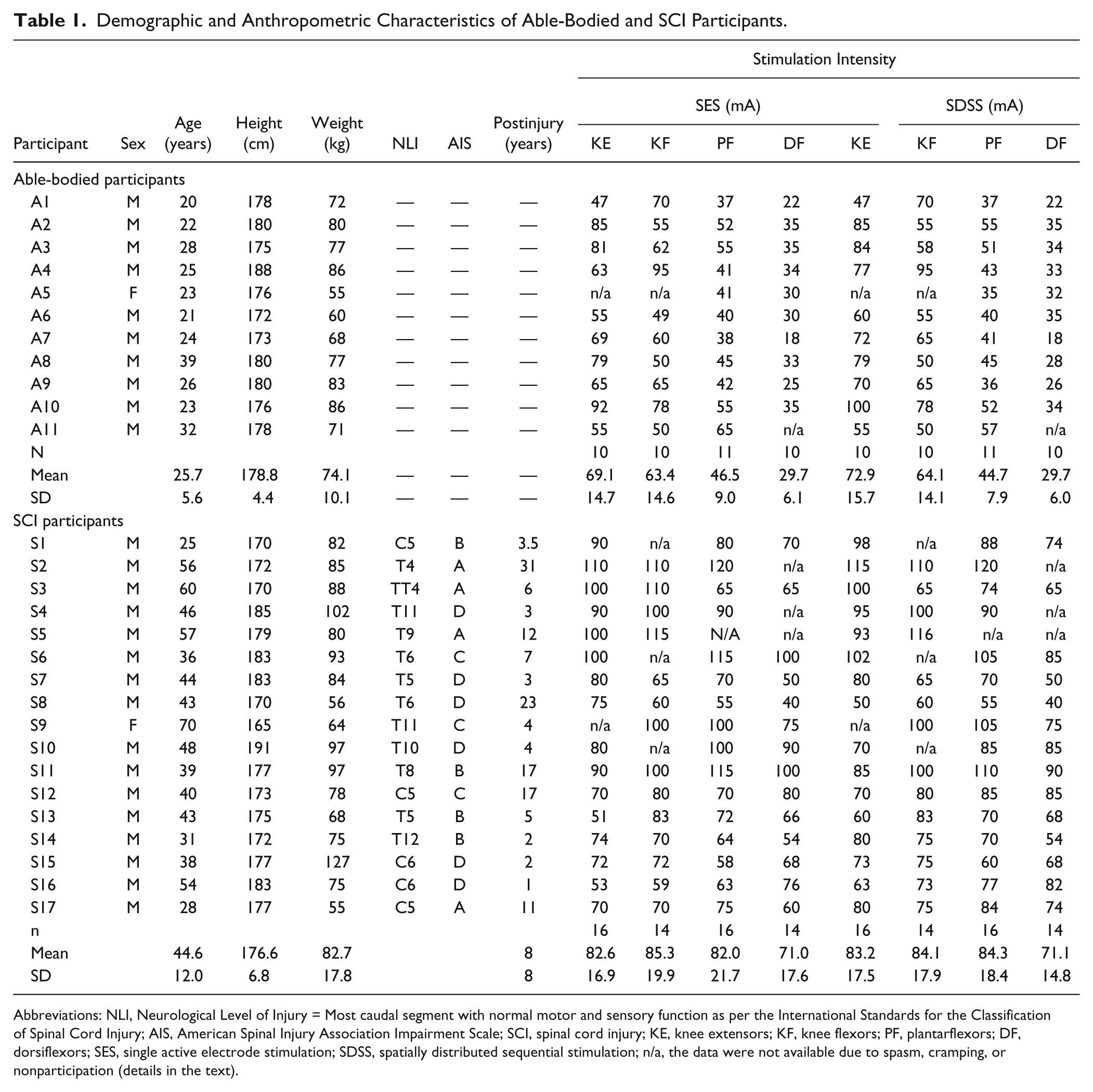
Abbreviations: NLI, Neurological Level of Injury = Most caudal segment with normal motor and sensory function as per the International Standards for the Classification of Spinal Cord Injury; AIS, American Spinal Injury Association Impairment Scale; SCI, spinal cord injury; KE, knee extensors; KF, knee flexors; PF, plantarflexors; DF, dorsiflexors; SES, single active electrode stimulation; SDSS, spatially distributed sequential stimulation; n/a, the data were not available due to spasm, cramping, or nonparticipation (details in the text).
Transcutaneous Neuromuscular Electrical Stimulation
A programmable 4-channel neuromuscular electrical stimulator (Compex Motion, Compex SA, Switzerland) was used to deliver transcutaneous electrical stimulation to 4 muscle groups: knee extensors, knee flexors, plantarflexors, and dorsiflexors. Self-adhesive gel electrodes (ValuTrode, Denmark) were placed over the proximal (active electrode) and distal (reference electrode) parts of the right muscle groups (Figure 1). Standard procedure in clinical setups for lower limb muscles is to stimulate a group of muscles simultaneously using relatively large electrodes that cover the group of muscles to be targeted by TNMES instead of stimulating individual muscles at each motor point.
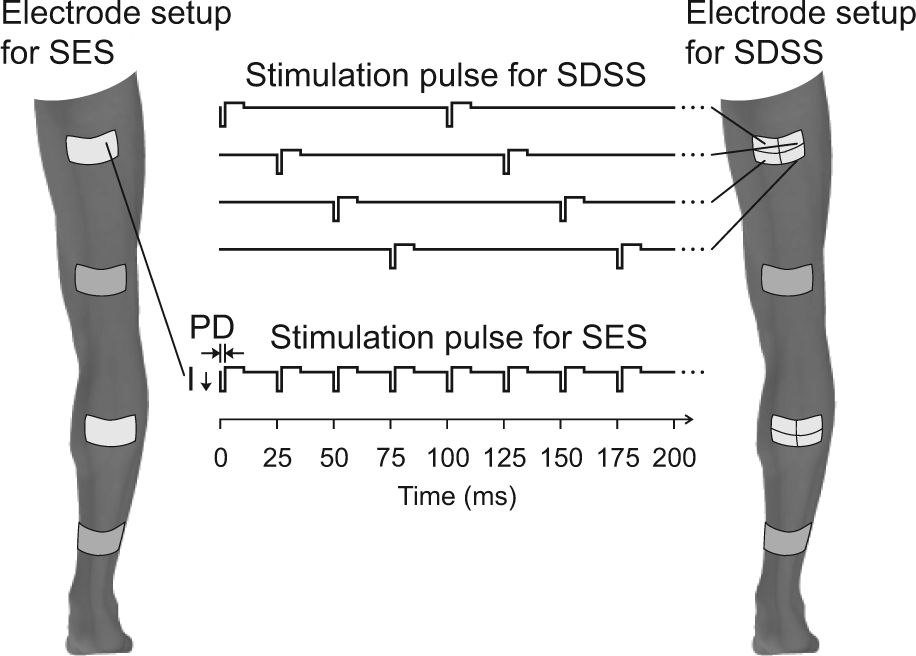
Schematic representation of SES and SDSS electrodes placements. Stimulation pulse timing is shown as well.
Two modes of stimulation were compared: SES and SDSS (Figure 1). During SES, pulses were delivered conventionally through one active electrode at 40 Hz. For plantarflexors and knee extensors and flexors, both active and reference electrodes were 9 cm by 5 cm; for dorsiflexors, the electrodes were 5 cm by 5 cm. During SDSS, pulses of the same amplitude were sequentially distributed among 4 active electrodes (4.5 cm by 2.5 cm for plantarflexors and knee extensors and flexors; 2.5 cm by 2.5 cm for dorsiflexors) placed with a minimum gap between each other so that they together covered the same area as the active electrode during SES, while the reference electrode was of the same size and at the same location as during SES.38,40 This stimulation was delivered by sending a stimulation pulse to each of the 4 electrodes, one after another. Individual electrodes were being stimulated at 10 Hz with a phase shift of 90° between successive electrodes, giving a resultant stimulation frequency of 40 Hz to the muscle group as a whole (Figure 1).38,40 These stimulation frequencies were selected because 40 Hz is often used for FES applications and ensured activation of most muscle fibers while 10 Hz still ensured activation of most slow muscle fibers. Other stimulation frequencies have not been tested comprehensively and is a task for the future. The stimulation current had a rectangular asymmetric biphasic pulse waveform with a pulse duration of 300 µs. A bout of fatiguing stimulation was delivered consisting of 120 trains, each composed of 12 pulses and spaced 1 second apart, resulting in 120 muscle contractions. This protocol somewhat mimicked cyclic activation in FES applications such as walking and cycling. During each test, the stimulation was delivered for approximately 2 minutes.
The tests were performed with SES followed by SDSS. Before the SES test, the stimulation intensity was increased to reach the maximal initial torque or the maximum tolerable intensity, whichever was less. Participants were warned that the constant stimulation intensity would need to be sustained for approximately 2 minutes, so should not cause severe discomfort. For all able-bodied participants, as well as some SCI participants, the stimulation intensity was determined by the maximum tolerable intensity. Thus, the exerted torque was not necessarily equivalent to the physiological maximal torque, since our intention was only to induce ensured fatigable torque instead of physiologically maximal torque. Then, before the SDSS test, the amplitude of the stimulation was adjusted to produce the same initial torque as during SES. This protocol was decided because we noticed that SDSS was less uncomfortable for the majority of subjects in our preliminary study and we had a concern that, if SDSS was performed first, we might not be able to induce the same torques. During SDSS, stimulation amplitudes were increased simultaneously for all electrodes and set at the same level during the test. In able-bodied participants, the tests were performed with an interval between them of at least 30 minutes. In participants with SCI, the tests were performed on different days, with at least 1 day of rest in between to reduce the possible cumulating effects of fatigue. Electrode positions were marked with a permanent marker to ensure that electrode placement was identical across tests.
Experimental Setup and Analysis
During the experiments, all participants were seated in an adjustable chair with arms crossed, and straps were used to stabilize the pelvis and trunk. In the able-bodied group, a Biodex Isokinetic Dynamometer (Biodex Medical Inc, Shirley, NY) was used to measure torque in all muscles. During the assessment of knee extensors and flexors, each participant was seated on the dynamometric chair with the hip and knee joints at 90° of flexion. The calf was secured by a strap above the malleoli to the dynamometer arm. The dynamometer axis rotation was aligned with the flexion/extension knee joint axis, and the resistance pad was fixed at the distal end of the thigh. During the assessment of plantarflexors and dorsiflexors, the participant was seated on the dynamometric chair with the seatback reclined 10° from vertical (slightly back from upright), right hip and knee positioned at 140°and 60° of flexion based on the anatomical frame, respectively, so that the thigh was elevated and the shank was positioned parallel to the floor. The participant’s foot was tightly fixed in a holder attached to the dynamometer, and the ankle joint center was aligned with the axis of the dynamometer.
In the SCI group, the assessment of knee extensors and flexors was performed as described above. Because conventional positioning for the ankle torque measurements at the Biodex dynamometer was not comfortable for some participants with SCI, their plantarflexors and dorsiflexors were assessed using a custom-built device with a reaction torque transducer (TS11, Interface, Inc, Scottsdale, AZ). The positions of the hip and knee joints were set to 90° of flexion and that of the ankle joints to neutral position (0° dorsi-/plantar-flexion).38,40 Both measurement devices were calibrated to ensure equivalently accurate torque measurements.
We identified 2 variables of interest. To indicate muscle force decay14,41,42 during the fatiguing stimulation, we calculated fatigue index (FI) and torque peak mean (TPM). FI was defined as the ratio between the mean peak torque values of the last 5 stimulus trains and those of the initial 5 stimulus trains. FI characterized the difference between the torque values at the beginning and end of the stimulation and indicates the ability to maintain the given torque for a certain period where higher values indicate greater fatigue resistance. TPM was calculated as the mean of peak torques throughout the whole bout of fatiguing stimulation, normalized to the mean peak torque values of the initial 5 stimulus trains. TPM was meant to assess the entire torque profile, characterizing the amount of contractile work during repetitive contractions, and presents the overall performance throughout the session.
The clinically meaningful difference for each outcome measure was assessed by the smallest real difference, SRD = 1.96√2×.
Statistics
To identify significant differences in the analyzed parameters during SDSS and SES, Wilcoxon signed-rank tests were performed (α = .05) for each participant group, since the normality was not confirmed for 5 distributions out of 32 distributions. As a secondary analysis, we performed a mixed model 2-way analysis of variance (ANOVA) comparing the groups (SCI and able-bodied individuals) and the stimulation methods (SDSS and SES). We performed this parametric analysis in this case because this is a secondary analysis and groups with nonnormal distribution were few among all sample groups (5 of 32).
Results
In the able-bodied group, A5 was not able to participate in knee joint measurements due to intolerance to higher-intensity (above 60 mA) electrical stimulation that yielded only small contraction in knee extensors, and A11 had muscle cramping during dorsiflexion after about 1 minute at 55 mA; therefore, their data were excluded from the analysis. In the SCI group, some subjects had spasm and/or withdrawal reflex during SES and/or SDSS and therefore were excluded from the analysis. Those were S1 during knee flexors, S2 during dorsiflexors, S4 during dorsiflexors, S5 during plantarflexors and dorsiflexors, S6 during knee flexors, S9 during knee extensors, and S10 during knee flexors. Thus, the number of subjects was different for each comparison and is summarized in Table 1.
In both experimental groups, the stimulation intensity utilized during the test of each muscle did not differ significantly between SDSS and SES protocols, except for knee extensors in the able-bodied group (69.1 ± 14.7 vs 72.9 ± 15.7 mA during SES and SDSS,
Mean Peak Torque Values of Initial 5 Stimulus Trains During SES and SDSS Protocols in Able-Bodied and Spinal Cord Injury Participants.
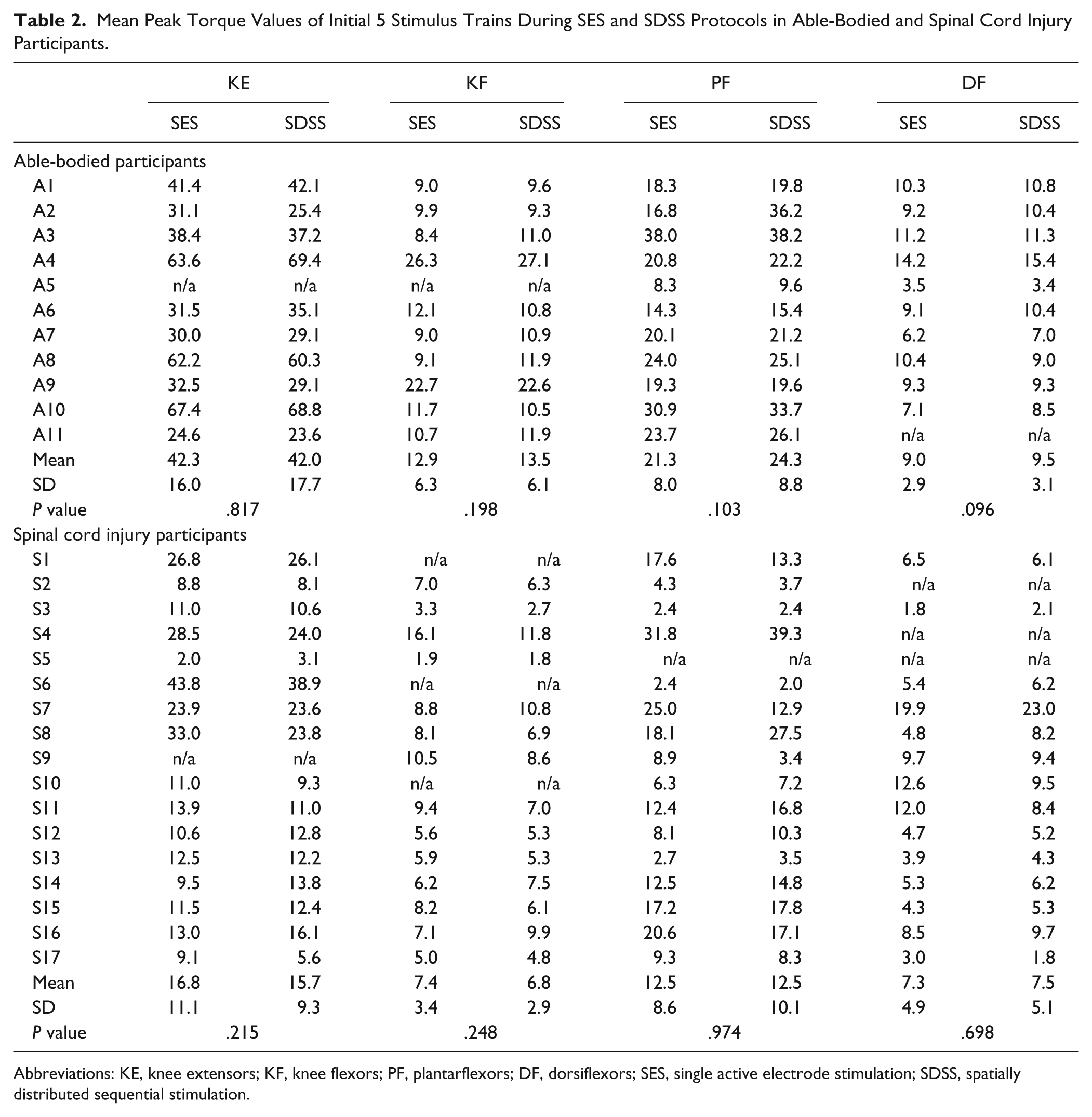
Abbreviations: KE, knee extensors; KF, knee flexors; PF, plantarflexors; DF, dorsiflexors; SES, single active electrode stimulation; SDSS, spatially distributed sequential stimulation.
An example of the torque time series of the fatiguing stimulation in one individual with SCI appears in Figure 2 with corresponding FI and TPM. The figure demonstrates inevitable muscle decay during TNMES utilizing both protocols. During SES, the torque values started decreasing monotonically shortly after the onset of stimulation, and deteriorated dramatically in some cases (see knee flexors). During SDSS, the torque decline was slowed down, and sometimes was even preceded by a muscle potentiation (see knee extensors and knee flexors during first 20 seconds). In knee flexors, plantarflexors, and dorsiflexors, it can be seen that the torque reduction during SDSS was less pronounced at the end of the fatiguing stimulation, as compared with SES. In knee extensors, although the FI value in knee extensors appears to be lower during SDSS, the peak torques values throughout the test, and ultimately the TPM, were higher indicating better performance as compared with SES.
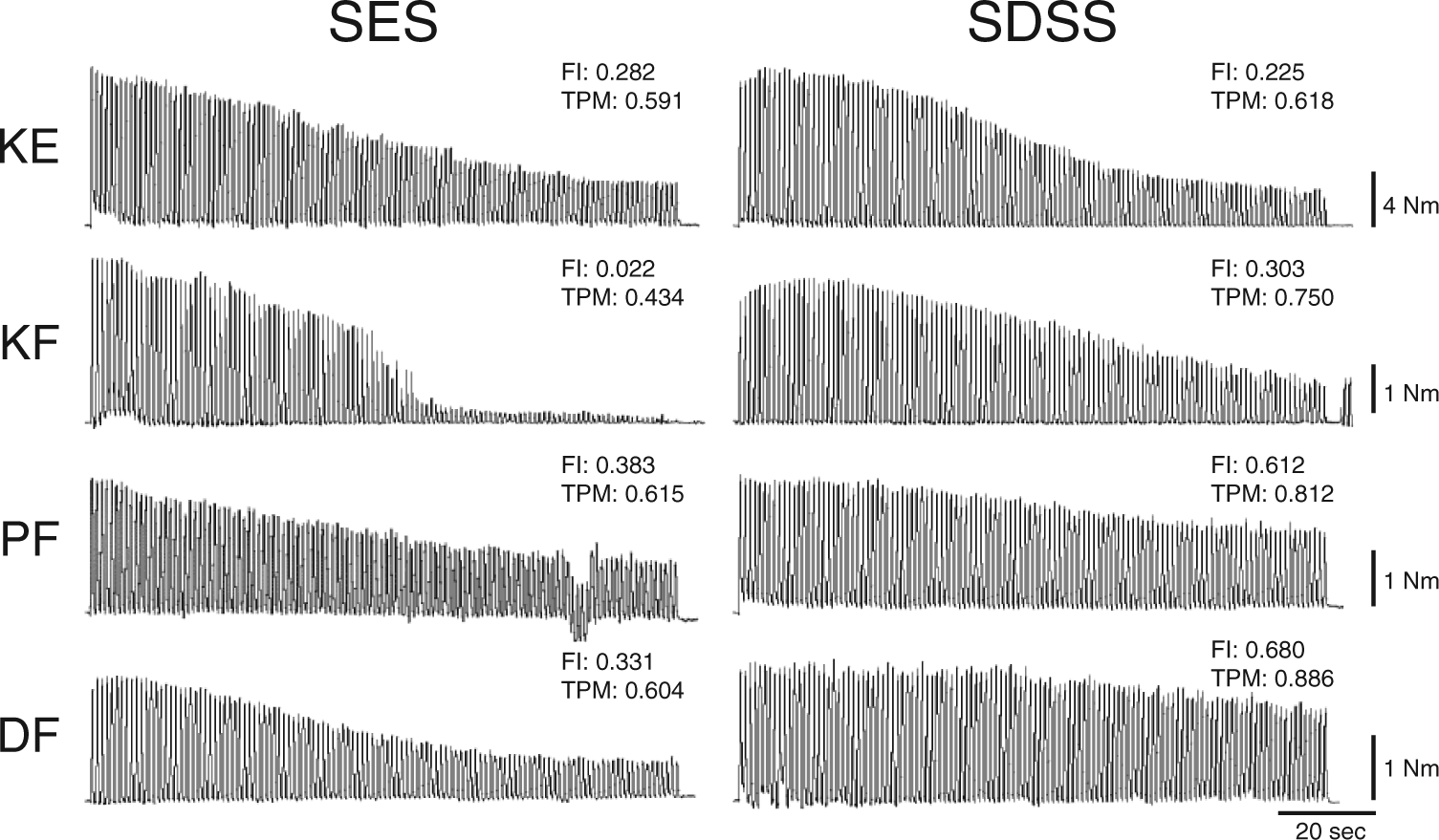
An example of the torque time series of the fatiguing stimulation during SES and SDSS protocols in one individual with spinal cord injury and the corresponding fatigue index (FI) and torque peak mean (TPM) for each time series.
Figures 3 and 4 show the differences between the 2 types of stimulation in terms of the force decay measures. Figure 3 compares the effects of the fatiguing stimulation on FI in the groups with and without SCI. In participants with SCI, FI showed significantly higher values for SDSS than for SES in all muscle groups (
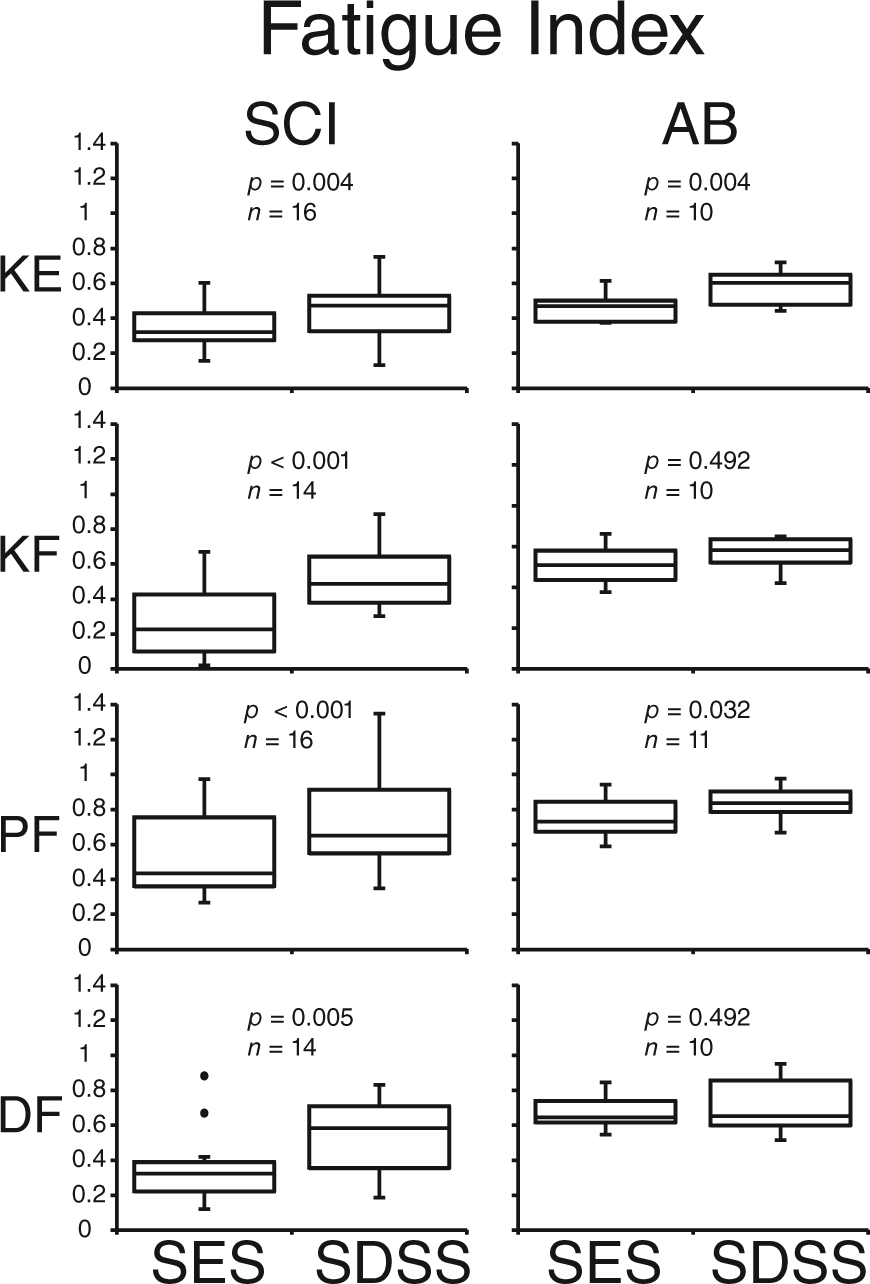
Fatigue index (FI) during SES and SDSS.
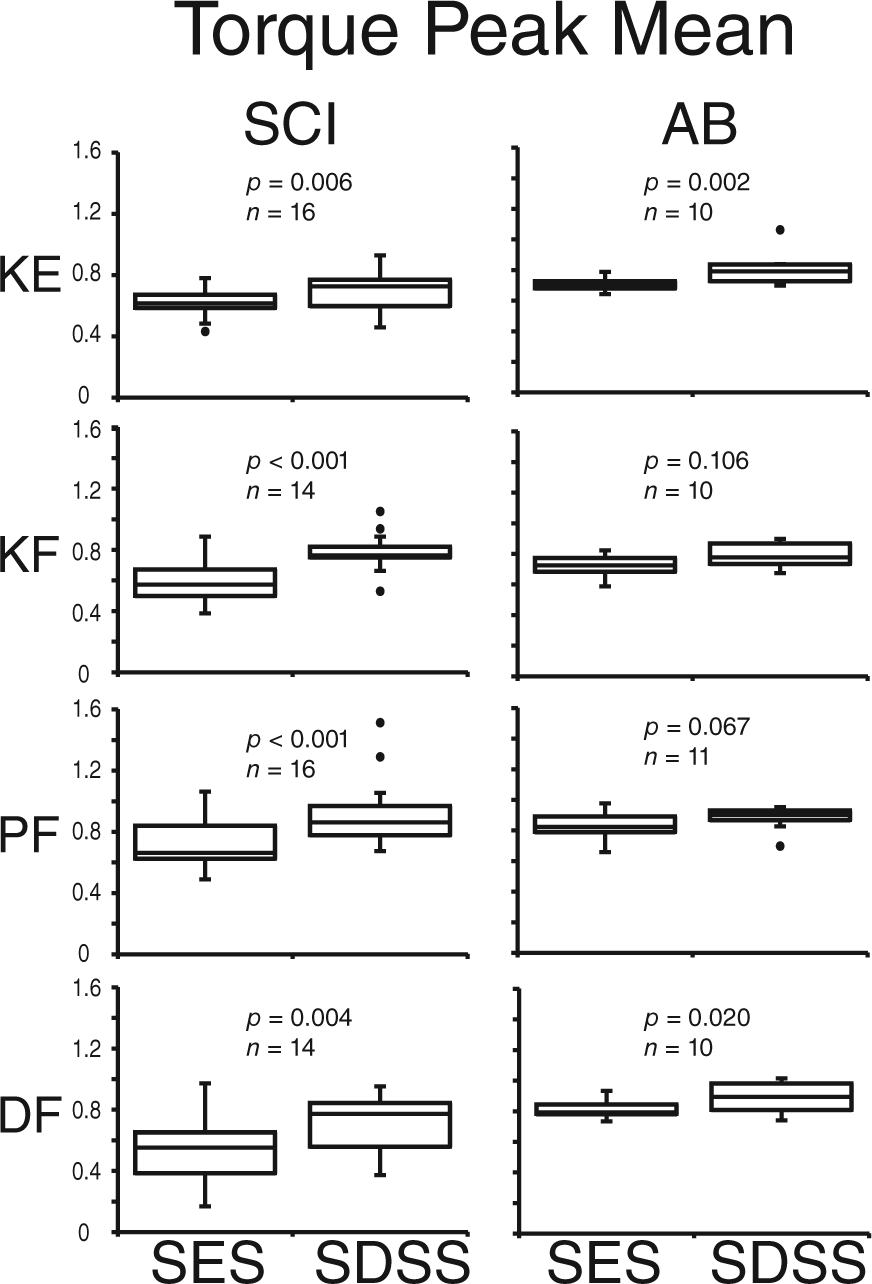
Torque peak mean (TPM) during SES and SDSS.
Figure 4 demonstrates the effects of the fatiguing stimulation on TPM in the 2 groups. In participants with SCI, TPM showed significantly higher values for SDSS than for SES in knee extensors (
We performed a mixed model 2-way ANOVA comparing the groups and the stimulation methods for each muscle and for both FI and TPM. In all ANOVAs, the main effects of stimulation methods were significant (
Table 3 summarizes the result of SRD for each muscle and for each group. In the able-bodied group, more than half of subjects showed larger values than SRD for either FI or TPM. In the SCI group, more than half of the subjects showed larger values than SRD for both measures.
SRD of Fatigue Index (FI) and Torque Peak Mean (TPM) for Each Muscle and for Each Group.
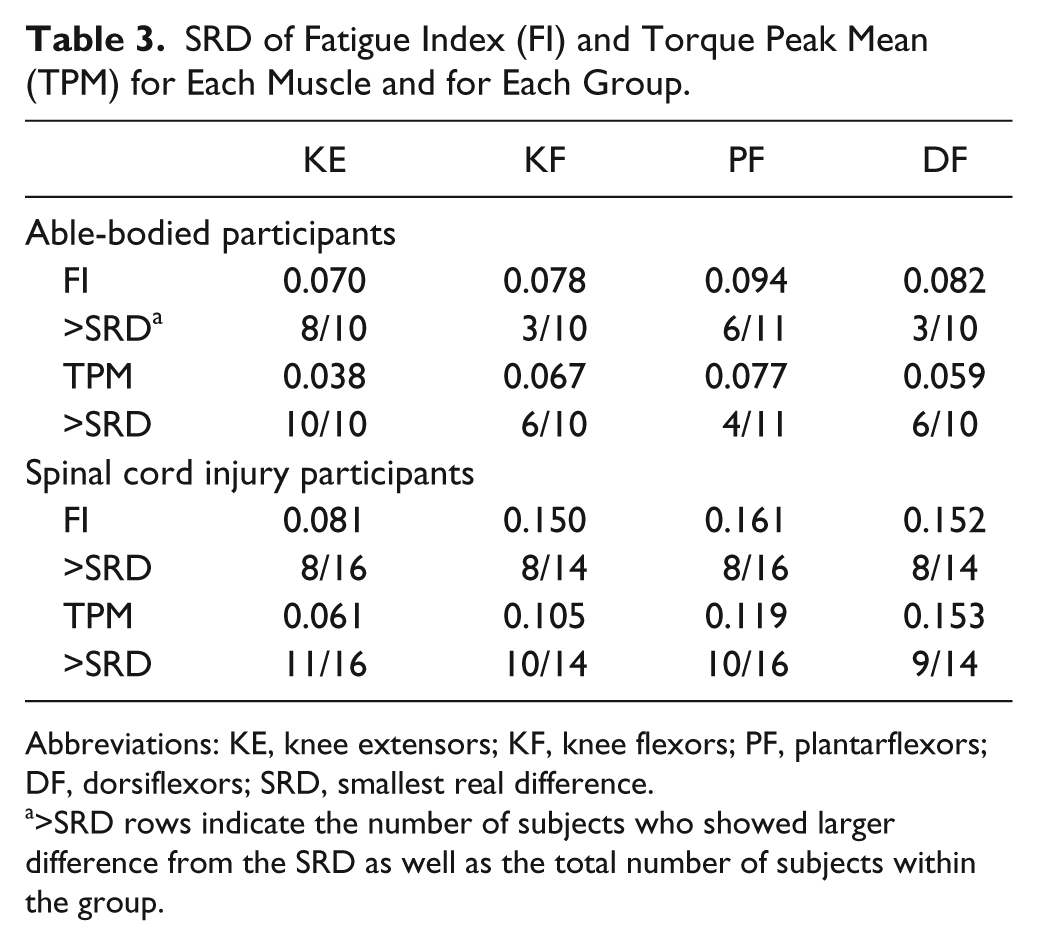
Abbreviations: KE, knee extensors; KF, knee flexors; PF, plantarflexors; DF, dorsiflexors; SRD, smallest real difference.
>SRD rows indicate the number of subjects who showed larger difference from the SRD as well as the total number of subjects within the group.
Discussion
We investigated the effectiveness of the spatially distributed sequential stimulation (SDSS) technique in reducing muscle fatigue during electrical stimulation in major lower limb muscle groups in individuals with and without SCI. We demonstrated greater fatigue-reducing ability of SDSS compared with conventional single active electrode stimulation (SES), as revealed by larger values of fatigue index (FI) and/or torque peak mean (TPM). Thus, the effectiveness of SDSS was revealed in all muscles tested in individuals with SCI, and in all muscles except the knee flexors in able-bodied individuals.
Effectiveness of Spatially Distributed Sequential Stimulation
The analysis demonstrated that FI and/or TPM values were higher during SDSS in all muscle groups in participants with SCI (Figures 3 and 4), indicating higher capability to maintain torque in the course of fatiguing stimulation compared with SES. Thus, using similar measures describing muscle force decay during fatiguing stimulation as in our previous studies,38,40 we extended previous results obtained for plantarflexors and demonstrated that the SDSS strategy that alternates activation of subcompartments of muscles produces greater stimulation times and resultant torques than the conventional SES protocol in all tested muscles in SCI and able-bodied (except knee flexors) populations. Longer maintenance of a given torque in key lower limb joints during rehabilitation may enhance the efficacy of different exercises’ modality and promote physiological and functional improvements in persons with SCI. In the able-bodied population, the SDSS approach can be utilized during strength training. However, not all subjects showed a larger difference of SDSS from SES than the SRD, indicating that the obtained improvement was modest in those cases and might not be clinically meaningful at this stage in some cases.
In able-bodied individuals, the fatigue-reducing effects were in general less pronounced than in the individuals with SCI (significant in all but knee flexors compared with all muscle groups). The difference between populations may be due to the SCI population having less fatigue resistance compared with the able-bodied population10,12,19,42,43 due to morphological and physiological changes following SCI.19,20,44-46 When we specifically consider knee extensors, we did not see any large difference between SCI and able-bodied participants as well as between the current study and a closely related study by Malešević and colleagues. 36 As reported above, the increments of FI for SDSS compared with SES for knee extensors were 28% and 26% (SCI and able-bodied, respectively), which were very close. Furthermore, the increments of TPM were 13% and 14% (SCI and able-bodied, respectively), which were also very close. In the secondary analysis of ANOVA, we did not confirm the statistical difference between the groups for FI nor TPM. Thus, regarding knee extensors, the effects of SDSS were about the same between the 2 tested populations. Malešević and colleagues 36 reported that improvement of torque exertion time using their proposed method compared with a conventional method was 26% for knee extensors in 5 complete SCI participants. We cannot directly compare this value with ours since time was their outcome measure and their protocol was different from ours. However, this suggests that the improvement by their method must be close to ours. This might indicate that the mechanisms between their method and ours are not very different, that is, SDSS may activate different synergistic muscles in the case of knee extensors with the current electrode setup. Further investigation is definitely required to elucidate the mechanism. However, it should be emphasized that an advantage of SDSS compared with the method of Malešević and colleagues 36 is that it is easier to incorporate into clinical applications because set up is easier as individual electrodes for several synergists are not required.
In the able-bodied population, the initial torques of plantarflexors were larger in the present study than in our previous one, 40 that is, 22.8 ± 2.1 versus 10.2 ± 0.3, respectively. The larger target torques in the present study were obtained because the stimulation intensity was chosen to reach the maximal initial torque value or the maximum tolerable intensity for each muscle group, as opposed to the target torque of 8 to 12 N m in the previous study. Additionally, the duration of the stimulation in the present study was shortened to 2 minutes as opposed to 3 minutes in the previous study, since 2-minute stimulation was still sufficient to produce muscle fatigue with the larger target torque. Due to these differences, the increments of FI were considerably different between the 2 studies, that is, 11% and 30% for the current and the previous studies, respectively. This suggests that SDSS may be more advantageous in cases of exercises with relatively low torque exertion and longer period, such as cardiovascular exercise compared with muscle strengthening exercise requiring high-intensity torque exertion with short period. However, this should be investigated in future studies with direct comparisons between these 2 conditions within subjects.
Mechanisms
In our previous study, 40 we demonstrated that the mechanism for the effectiveness of SDSS was that the sequentially distributed electrodes activated different parts (branches or axonal terminals) of the motor nerve and, as a consequence, different subcomponents of the target muscle. We suggest that because the frequency for each subcomponent during SDSS was slower (10 Hz) than that for the corresponding subcomponent during SES (40 Hz), the accumulating muscle fatigue was less at each subcomponent as the increased time between subsequent activation of motor units allowed greater recovery. The exact mechanism is not completely revealed, and therefore further experimental and theoretical investigations are required. From a theoretical perspective, electric fields created by SES and SDSS need to be compared—which we currently work on.
As the purpose of the current study was not to investigate the mechanism of SDSS, we do not have experimental evidence to provide a profound discussion on the topic. However, we can speculate somewhat with comparisons among muscles and between SCI and AB. First, if the above-mentioned potential mechanism is true, SDSS has the advantage that it can be used even for muscles without multiple-synergistic compartments, such as dorsiflexors (dominantly only tibialis anterior muscle) and small muscles of the upper limbs. Thus, the positive result in the current study on dorsiflexors supports this mechanism. Second, the knee extensors tended to show lower increments of FI and TPM in the SCI population compared with the other 3 muscles. Furthermore, as mentioned above, the tendency of the result for knee extensors was similar to Malešević and colleagues. 36 These might indicate that the mechanism of SDSS for knee extensors is different from other muscles and is similar to stimulating synergistic muscles alternately like Malešević and colleagues. 36 However, the effectiveness of SDSS for knee flexors, which has multiple-synergistic compartments similar to knee extensors, was not clearly shown in the able-bodies population. This difference between knee extensors and flexors may depend on the relation between the locations of electrodes and the innervating nerves, but in any case, further investigation of the mechanism focusing on these factors is required. Third, as we compared above, the fatigue-reducing effects were overall less pronounced in the able-bodied population than in the SCI population, which may be explained by the fact that individuals with SCI have less fatigue resistance compared with the able-bodied population. This may be especially due to morphological changes following SCI such as muscle fibers’ transformation to a fast-fatigable phenotype.19,44,46 Therefore, the results that the fatigue-reducing effects were pronounced in the SCI population and that the initial torque was much less in the SCI population than in the able-bodied population may support the speculation that SES uses 40 Hz stimulation that predominately activates fast muscle fibers, whereas SDSS uses 10 Hz for each electrode that effectively activates slow muscle fibers.
Comparison With Other Approaches in Human Studies
Several studies have suggested that manipulation of the characteristics of the stimulation train during TNMES, including pulse frequency, width, and amplitude,47-51 may potentially improve fatigue resistance. The results from these studies are not consistent: some studies showed such approaches decrease fatigue,48,51 and other studies showed no significant difference.47,49,50 Therefore, the effectiveness of these techniques for fatigue reduction is yet to be ascertained. Another approach is to stimulate afferent nerves as proposed by Bergquist et al. 4 It has been demonstrated that electrical stimulation delivered to the Ia-afferent nerve trunk of the tibial, common peroneal, or femoral nerve may orthodromically activate motor units projecting to different subcompartments of muscles through monosynaptic pathways. In this way, the evoked contractions were distributed over more muscle fibers and hence less fatigue occurred as compared with a conventional neuromuscular stimulation.4,52 The main limitations of such an approach across different muscle groups are accessibility of the stimulation site, electrode susceptibility to movement, contraction reliability, as well as stimulation comfort.4,52
Contrarily, the approaches incorporating delivery of TNMES through spatially distributed electrodes, each of which alternately targets specific compartments of a synergistic muscle group,35-37 have made evident their effectiveness for fatigue reduction. The results from these studies demonstrated significantly higher fatigue-resistant ability when synergistic muscles within quadriceps femoris were alternately activated. However, it was unclear whether such an approach would be feasible in a smaller muscle group or without multiple synergistic muscles. Maneski et al 39 have shown the effectiveness of applying a similar approach to the finger flexors. In the present study we demonstrated that such a method is effective in another muscle without multiple-synergistic compartments, the one-headed dorsiflexors (ie, tibialis anterior muscle), and compared the fatigue-reducing effects of SDSS in populations with and without SCI.
Limitations and Future Directions
In the able-bodied group, the 2 tests were performed with an interval greater than 30 minutes. There might be concern that this interval was not sufficient to allow full recovery from muscle fatigue. However, as neither stimulation intensities nor initial torques differed statistically between SES and SDSS protocols (as shown in Tables 1 and 2), it is unlikely that there were accumulating fatigue effects from previous trials. For the same reason, although the order of 2 trials was fixed, which might cause an order effect affecting the results, an order effect is unlikely. As mentioned in the Methods section, the reason why we decided to use the fixed order was due to concern that the same amount of torque in SES as in SDSS would not have been induced if SDSS had been performed first since SDSS was less uncomfortable. However, as shown in Tables 1 and 2, equivalent initial torque could be induced using the same stimulation intensity. Therefore, we now know that it is possible to perform SDSS first. Thus, we propose that the 2 trials be performed in random order in future studies. Also, we included both motor complete and incomplete SCI individuals within the SCI group, which may be a confounding factor when we interpret the effect of SDSS. Unfortunately, we were not able to analyze the patient population separately in this study as it would have required an increased number of subjects.
Further research is required to investigate and compare the SDSS and SES strategies in clinical settings, such as strength training or FES cycling during rehabilitation of individuals with SCI or other neurological disorders and motor impairments. The ultimate goal of these studies would be to determine the clinical impact and functional consequences of SDSS as compared with SES. In addition, although SDSS showed its effectiveness in the relatively small dorsiflexors, as well as in one study with finger flexors, 39 the feasibility of utilizing the SDSS strategy for different upper limb muscles is yet to be shown. More in-depth investigation of the mechanisms of SDSS is needed, as it may extend the current knowledge and provide the basis for even further improvements of the method. Finally, we plan to develop a simple device that incorporates the SDSS protocol for electrical stimulators out on the market that would make easier translation to the clinic as the new standard of care instead of using a highly sophisticated controller system. 39
Conclusion
Our study has revealed improvements in TNMES performance using a stimulation strategy, named SDSS, which alternates activation of subcompartments of muscles in participants with and without SCI and in most of the major lower limb muscle groups, except for knee flexors in able-bodied participants. The SDSS protocol has provided greater stimulation times with less decrement in mechanical output compared with the conventional SES protocol. Future applications of SDSS for TNMES-based training may be a means to promote longer training sessions and greater rehabilitative outcomes. As the effect of SDSS was confirmed in quasi-maximum conditions in this study, the findings indicate that SDSS would be especially useful for high-intensity exercise such as muscle strength training for leg muscles and cardiovascular exercise for individuals with SCI. Future research is required to investigate further the mechanism of SDSS in order to improve its stimulation pattern. Furthermore, since the effectiveness of SDSS was shown in a relatively small muscle (dosriflexor) without involving other muscle synergists, the results suggest that SDSS can be used in the smaller muscles of the upper limbs. As FES therapy has predominantly been shown to be effective in the upper limbs, the effectiveness of SDSS should be tested there in the near future. Additionally, it was shown that the obtained improvement was modest, and further modification might be required to make the SDSS method clinically meaningful.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Canadian Institutes of Health Research (MOP 111225) to KM and Russian Scientific Fund (14-45-00024) to DGS. The authors acknowledge the support of Toronto Rehabilitation Institute—University Health Network, which receives funding under the Provincial Rehabilitation Research Program from the Ministry of Health and Long-Term Care in Ontario.
