Abstract
Background
Electrical stimulation has been employed as a safe and effective therapy for improving arm function after stroke. Contralaterally controlled functional electrical stimulation (CCFES) is a unique method that has progressed from application in small feasibility studies to implementation in several randomized controlled trials. However, no meta-analysis has been conducted to summarize its efficacy.
Objective
To summarize the effect size of CCFES through measures of upper extremity motor recovery compared with that of neuromuscular electrical stimulation (NMES).
Methods
The PubMed, Cochrane Library, EMBASE, Scopus, and Google Scholar databases were searched. Randomized controlled trials (RCTs) were selected and subjected to meta-analysis and risk of bias assessment.
Results
6 RCTs were selected and 267 participants were included. The Upper Extremity Fugl-Meyer assessment (UEFMA) was included in all studies, the Box and Blocks test (BBT) and active range of motion (AROM) were included in 3 and 4 studies, respectively. The modified Barthel Index (mBI) and Arm Motor Abilities Test (AMAT) were included in 2 and 3 studies, respectively. The CCFES group demonstrated greater improvement than the NMES did in UEFMA (SMD = .42, 95% CI = .07–.76), BBT (SMD = .48, 95% CI = .10–.86), AROM (SMD = .54, 95% CI = .23–.86), and mBI (SMD = .54, 95% CI = .12–.97). However, the results for AMAT did not differ significantly (SMD = .34, 95% CI = −.03–.72).
Conclusion
Contralaterally controlled functional electrical stimulation produced greater improvements in upper extremity hemiplegia in people with stroke than NMES did. PROSPERO registration number: CRD42021245831
Keywords
Introduction
Stroke causes neurological deficit attributed to an acute focal injury of the central nervous system due to a vascular cause, which may lead to disability or death. An estimated 9.6 million ischemic strokes and 4.1 million hemorrhagic strokes occur globally each year. 1 Stroke is ranked as the second leading cause of death worldwide, with an annual mortality rate of approximately 5.5 million. 2 A substantial proportion of stroke survivors have very poor health-related quality of life, and stroke is strongly associated with disability.3,4 Specifically, approximately 65% of stroke survivors have hemiparesis. 5 Post-stroke upper extremity impairments cause functional limitations, which then cause difficulty in the performance of activities of daily living.
Neuromuscular electrical stimulation (NMES) is widely used in clinical rehabilitation for stroke and spinal cord injury and is based on artificially controlling human muscles or muscle nerves. 6 Neuromuscular electrical stimulation stimulates lower motor neurons to activate paralyzed or paretic muscles. 7 Cyclic NMES and electromyography (EMG)-triggered NMES are frequently used to aid motor recovery in people with stroke. 8 In cyclic NMES, a stimulator automatically turns on and off to stimulate the participant’s paretic hand for preset durations and intensities; EMG-triggered NMES of the wrist or finger extensor muscles is initiated by a volitional EMG signal from the same or nearby muscles and remains turned on for a preset duration. 9 These 2 forms of NMES have demonstrated several favorable effects, such as effective facilitation of motor recovery, reduction of shoulder subluxation, and pain-free range of motion, especially among acute stroke survivors.6,7,10
Contralaterally Controlled Functional Electrical Stimulation (CCFES), first introduced by Knutson et al. in 2007, is an innovative therapy for improving recovery of hand function in stroke survivors. 11 CCFES therapy electrically stimulates the extensor muscles of the paretic fingers and thumb to achieve hand opening. CCFES and NMES differ in that in CCFES, the intensity of the stimulation is controlled by the user’s contralateral unimpaired hand and is directly proportional to the degree to which the user opens the unimpaired hand. 12 Knutson et al. used CCFES to facilitate motor relearning in chronic hemiplegia, and their results suggest a positive effect on hand impairment and function. 13 Studies have reported that CCFES may produce more favorable effects in people with stroke than the more widely used NMES does; this is because CCFES might have some specific features such as promoting neuroplasticity and disinhibition by bilateral symmetrical movement.14–16
However, to the best of our knowledge, no meta-analysis has yet compared the effects of both CCFES and conventional NMES in treating upper extremity hemiplegia. A meta-analysis may provide a more precise estimate of the effect size of CCFES. Therefore, we compared the clinical effectiveness of CCFES and NMES in facilitating recovery in hemiplegic upper extremities in people with stroke.
Methods
Study Protocol
We conducted this review in accordance with the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) guidelines. 17 The PRISMA checklist is presented in Supplementary Appendix A. The protocol was prospectively registered with the International Prospective Register of Systematic Reviews (PROSPERO; registration number: CRD42021245831).
Eligibility Criteria
We included randomized controlled trials (RCTs), including pilot RCTs; the PICO model used to identify eligible RCTs is as follows:
(P) involved participants with any phase of stroke of any sex, age, or ability level
(I) employed CCFES intervention
(C) compared the results of CCFES intervention with other intervention therapy or placebo
(O) reported outcomes for the upper extremities
Randomized controlled trials that assessed only outcomes of lower extremities and diseases other than stroke were excluded.
Data Sources and Retrieval
The following electronic databases were searched until December 30, 2020: PubMed, the Cochrane Library, EMBASE, Scopus, and Google Scholar. Several keywords for the disease and intervention in combination were used: ((Contralateral* AND (electrical and stimulation or electric and stimulation or electrostimulation or ES)) AND (stroke or hemipare* or infarct*) AND ((upper and extremit*) or hand or arm or UE). The corresponding MeSH terms were searched in the PubMed database. Two of the authors of this study (M.S. Loh and Y.C. Kuan) independently screened the RCTs according to their titles, abstracts, and full texts. Decisions for inclusion or exclusion of RCTs were compared and discussed to reach a consensus. Any persisting disagreements were resolved through discussion with the third author (H.C. Chen).
Data Items
We included population diagnosed with stroke, and the following parameters were obtained from each RCT: number of participants, phase of stroke, follow-up duration, baseline therapy for both intervention and control groups, number of electrodes, placement and duration of treatment, and appraised outcome measure.
Outcomes
For classification of outcome measurements, we used the International Classification of Functioning, Disability, and Health (ICF). The ICF is a World Health Organization framework for measuring health and disability at both the individual and population levels. The ICF classifies human functioning as containing 3 levels, namely, body function, activity, and participation, with activity including capacity and performance. 18 Outcome measures that were included in at least 2 RCTs were assessed in our study, and the results with the longest follow-up period were analyzed.
Risk-Of-Bias Assessment
The methodological quality of each study was independently assessed by 2 authors (M.S. Loh and Y.C. Kuan) by using the Cochrane risk of bias tool, version 2.0, which is widely used to assess the risk of bias in RCTs.19,20 6 domains of this tool were evaluated: randomization process, deviations from intended interventions, missing outcome data, measurement of the outcome, selection of the reported result, and overall bias. The results were classified as low risk, some concerns, or high risk, in accordance with the guidelines provided by the Cochrane Collaboration. 21 Any ambiguities were resolved through discussion with the third author (H.C. Chen).
Statistical Analysis
Data analysis was performed using Review Manager version 5.4.1 (The Cochrane Collaboration, London, UK). 22 All relevant data with different scales were converted to the same scale using the standard mean difference (SMD) for meta-analysis. The precision of effect sizes was reported as 95% confidence intervals (CI). I 2 statistics were used to evaluate statistical heterogeneity; I 2 > 50% was regarded as indicating substantial heterogeneity, thus requiring a sensitivity analysis to confirm the effect. Statistical significance was set at P < .05.
Results
After the search terms were applied, 2229 articles were initially retrieved. Next, 519 duplications were removed using EndNote 20. A further1689 were excluded because of irrelevance. Among the remaining 21 articles, 2 were excluded because their full texts were unavailable, and 13 were excluded because they were non-RCTs. Finally, 6 studies were included. The selection flowchart is detailed in Figure 1.
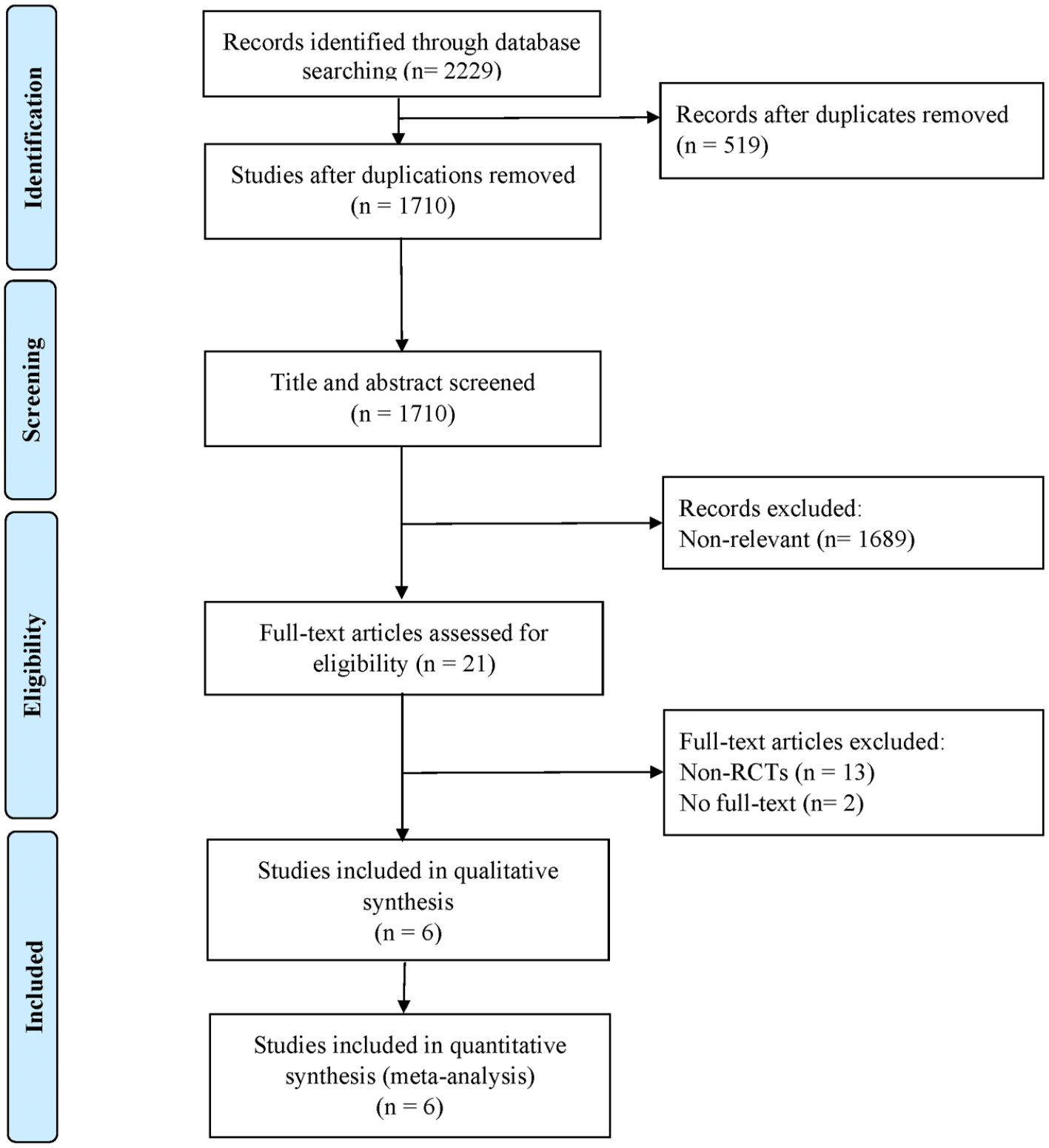
Flowchart of the study selection 180 × 198 mm (300 × 300 DPI).
Study Characteristics
The selected 6 studies were published between 2012 and 2020. In all studies, the control group received cyclic NMES. A total of 267 participants (137 participants in the CCFES group and 130 in the cyclic NMES group) were included. A study by Knutson et al. that was included in our meta-analysis originally classified participants into 3 groups: 23 arm and hand CCFES, hand CCFES, and arm and hand NMES. However, we included only the arm and hand CCFES and arm and hand NMES groups in our meta-analysis. Of the 6 studies included in our meta-analysis, 1 investigated acute phase stroke, 24 1 explored chronic phase stroke, 25 3 examined subacute phase stroke,14,26,27 and 1 studied subacute and chronic phase stroke. 23 The first 2 weeks post-stroke, 3–24 weeks post-stroke, and more than 24 weeks post-stroke were defined as the acute, subacute, and chronic phases, respectively. 28 The participants in both intervention groups in each study received treatment and background intervention for the same duration. The main characteristics of the 6 RCTs are summarized in Table 1.
Characteristics of Included Studies.
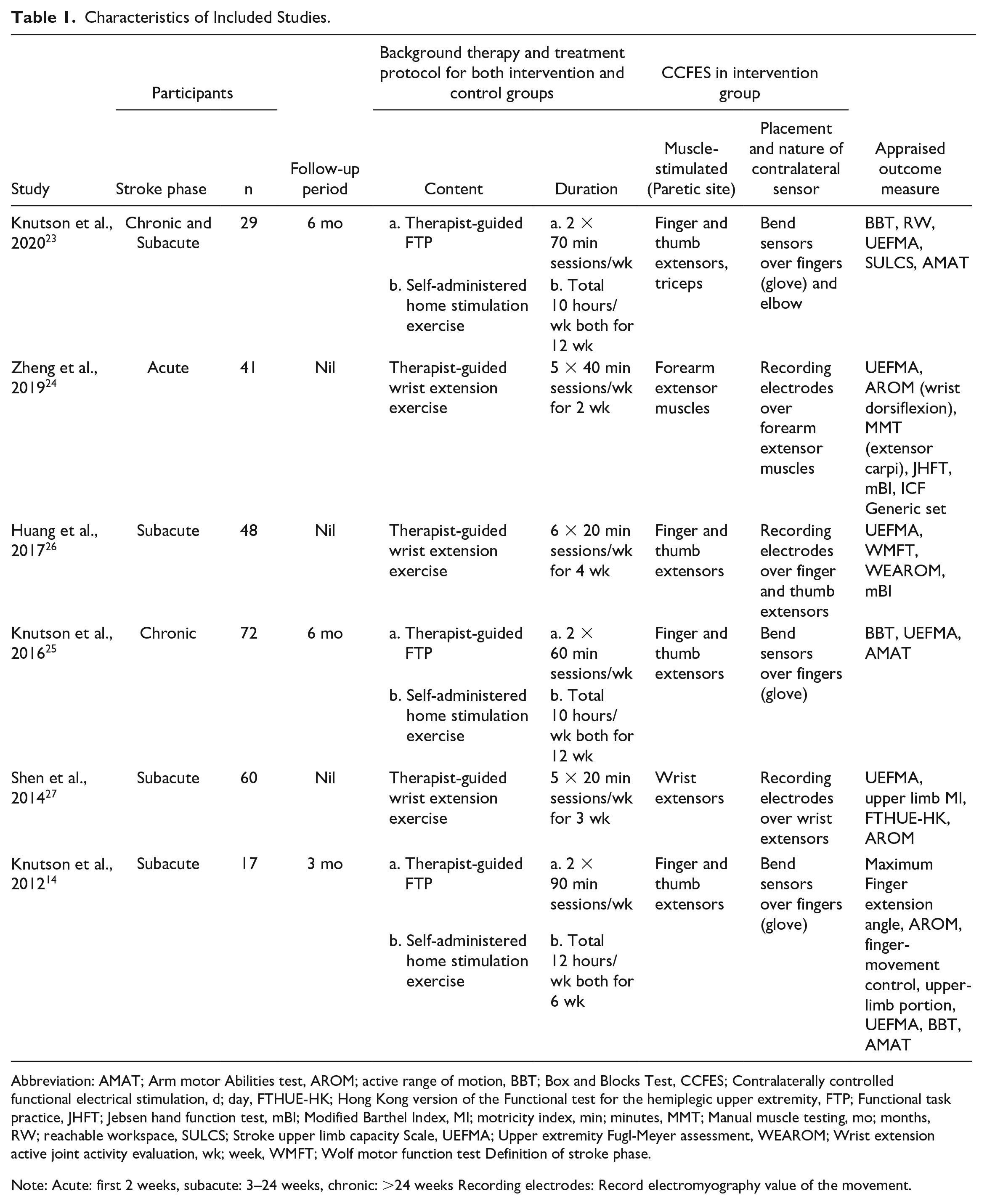
Abbreviation: AMAT; Arm motor Abilities test, AROM; active range of motion, BBT; Box and Blocks Test, CCFES; Contralaterally controlled functional electrical stimulation, d; day, FTHUE-HK; Hong Kong version of the Functional test for the hemiplegic upper extremity, FTP; Functional task practice, JHFT; Jebsen hand function test, mBI; Modified Barthel Index, MI; motricity index, min; minutes, MMT; Manual muscle testing, mo; months, RW; reachable workspace, SULCS; Stroke upper limb capacity Scale, UEFMA; Upper extremity Fugl-Meyer assessment, WEAROM; Wrist extension active joint activity evaluation, wk; week, WMFT; Wolf motor function test Definition of stroke phase.
Note: Acute: first 2 weeks, subacute: 3–24 weeks, chronic: >24 weeks Recording electrodes: Record electromyography value of the movement.
Risk of Bias Assessment
The Cochrane risk of bias tool was used to assess the quality of the RCTs. The assessment was then independently reviewed by 2 reviewers according to the guidelines of the Revised Cochrane Risk of Bias tool for randomized trials (RoB 2.0). The results are illustrated in Table 2.
Methodological Quality Assessment of Selected Trials.
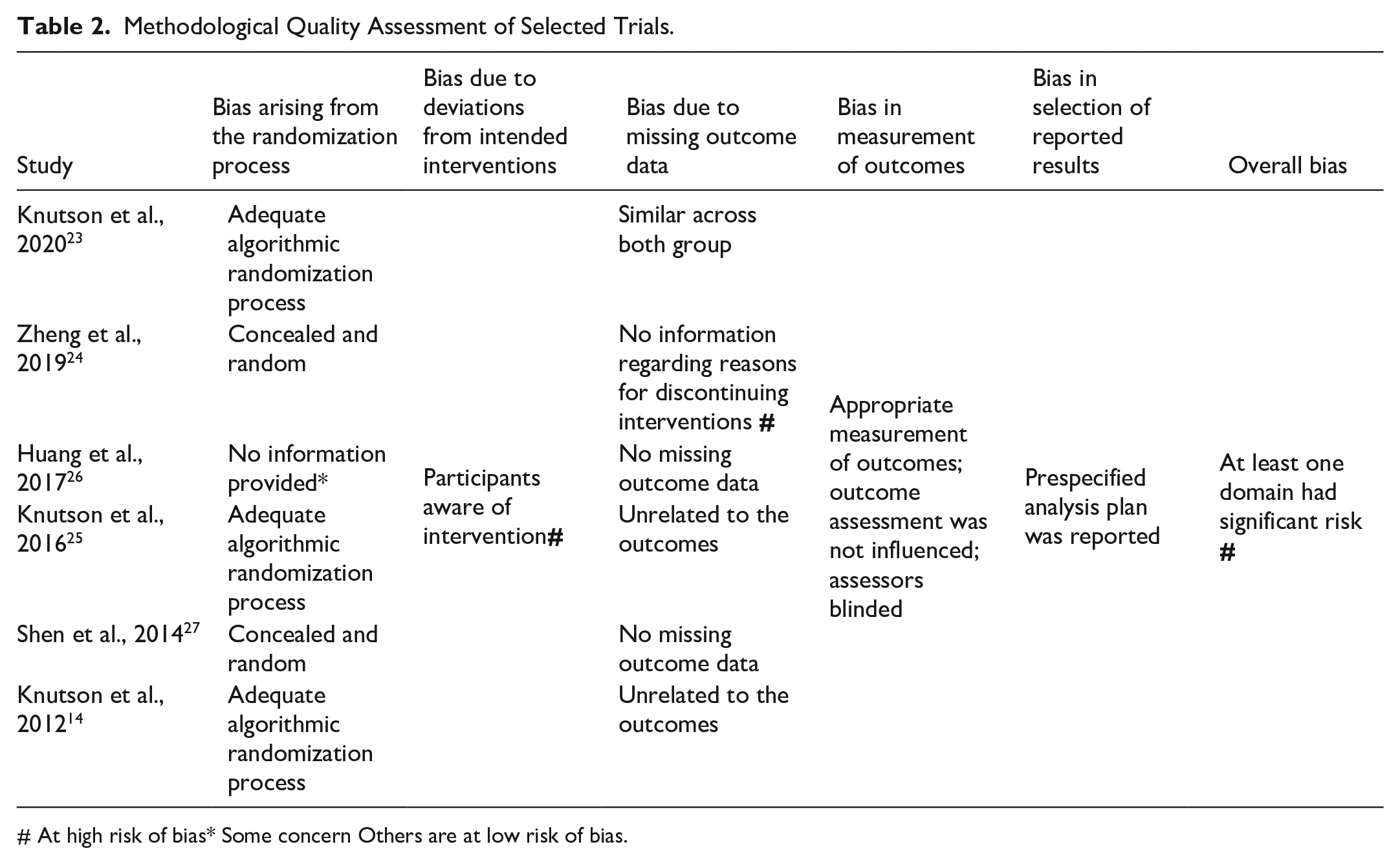
At high risk of bias* Some concern Others are at low risk of bias.
Regarding the overall risk of bias, 4 studies were identified as low risk, one as having some concerns, and 1 as high risk. As previously discussed, the study by Knutson et al. classified participants into 3 groups; however, randomization of the NMES group was discontinued to achieve statistical power and enable comparison with the 2 CCFES groups. While Knutson et al. are to be commended for their transparency in reporting, discontinuing the randomization of the NMES group resulted in the sample size of the NMES group being less than half of that of the CCFES group, which may have negatively influenced the statistical power for that comparison. Furthermore, all included studies exhibited high risk for domain 2 (i.e., bias due to deviations from intended interventions).
Body Function Level Assessment
Three outcomes were classified under level of body function: Upper Extremity Fugl-Meyer assessment (UEFMA), Box and Blocks test (BBT), and active range of motion (AROM).
We assessed UEFMA in all the RCTs. The results revealed significantly higher levels of improvement in the CCFES group than in the NMES group (SMD = .42, 95% CI = .07–.76, n = 267, I2 = 43%). The forest plot for UEFMA is presented in Figure 2A. Because half of the included RCTs were conducted by the same authors (Knutson et al), we performed a sensitivity analysis by excluding the data of these 3 studies. The results still revealed greater improvement in the CCFES groups (SMD = .60, 95% CI = .12–1.08, n = 149).
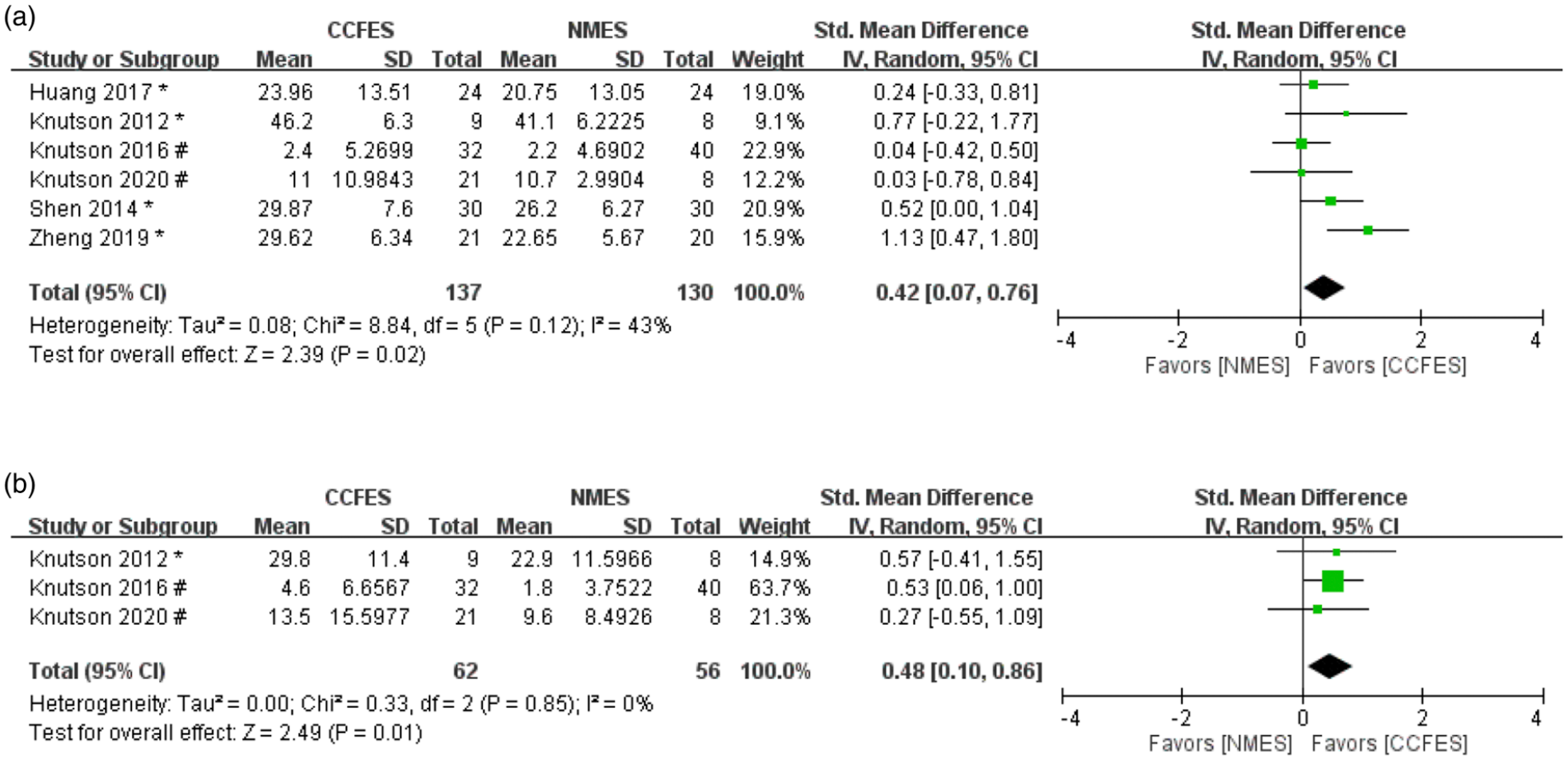
(A) Forest plot of upper extremity Fugl-Meyer assessment (UEFMA); (B) forest plot of box and blocks test (BBT) (#: Values of mean are mean change from baseline, *: Values of mean are values at study endpoint) 174 × 79 mm (300 × 300 DPI).
Box and Blocks test was assessed in 3 studies, including 62 participants in the CCFES group and 56 in the NMES group. The results revealed significantly higher improvements in the CCFES group than in the NMES group (SMD = .48, 95% CI = .10–.86, n = 118, I2 = 0%). The forest plot for BBT is shown in Figure 2B.
Active range of motion was assessed in 4 studies, including 84 participants in the CCFES group and 82 in the NMES group. Two ranges of motion were analyzed in these studies, namely finger extension and wrist dorsiflexion. The results reveal significant improvement in the CCFES group compared with the NMES group (SMD = .54, 95% CI = .23–.86, n = 166, I2 = 0%). The forest plot for AROM is shown in Figure 3.
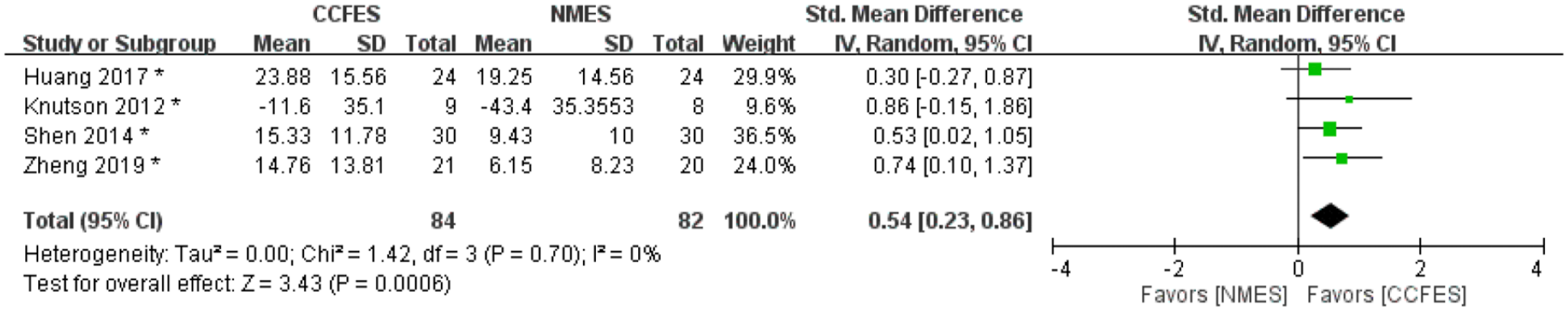
Forest plot of active range of motion (AROM) (*: Values of mean are values at study endpoint) 174 × 38 mm (300 × 300 DPI).
Activity Level Assessment
Ability to perform activities of daily living (ADL) was classified under activity level and was measured using 2 methods: the modified Barthel Index and the Arm Motor Abilities Test.
The modified Barthel Index was included in 2 RCTs comprising 45 participants in the CCFES group and 44 participants in the NMES group.24,26 The results revealed that the CCFES group had significantly greater improvement than the NMES group did (SMD = .54, 95% CI = .12–.97, n = 89, I2 = 0%). The forest plot for modified Barthel Index is displayed in Figure 4A.
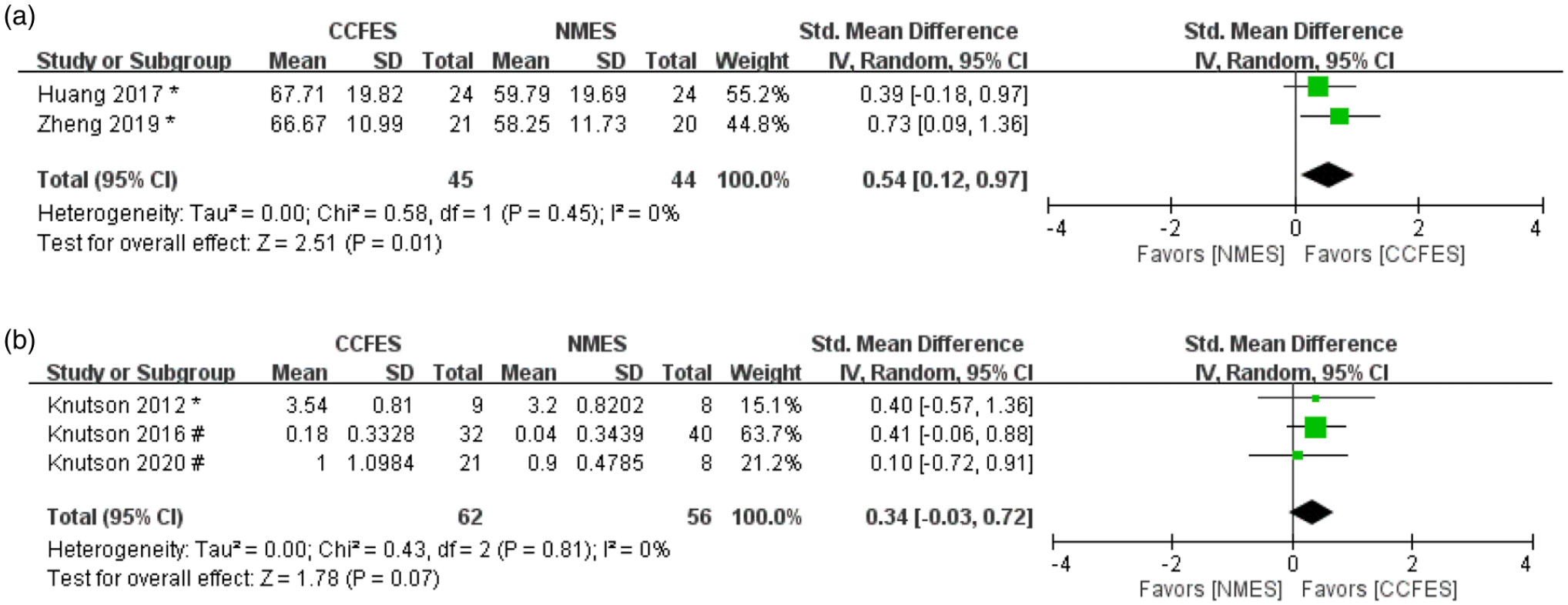
(A) Forest plot of modified Barthel index; (B) forest plot of arm motor abilities test (AMAT) (#: Values of mean are mean change from baseline, *: Values of mean are values at study endpoint) 174 × 63 mm (300 × 300 DPI).
In addition, the Arm Motor Abilities Test was performed in 3 studies comprising 62 participants in the CCFES group and 56 participants in the NMES group.14,23,25 The CCFES group achieved greater improvement than the NMES group did; however, the results were not significant (SMD = .34, 95% CI = −.03–.72, n = 118, I2 = 0%). The forest plot for the Arm Motor Abilities Test is presented in Figure 4B.
Discussion
Previous studies have indicated that NMES yields satisfactory results for post-stroke rehabilitation.8,29 Our findings reveal that CCFES is more effective than NMES in improving upper extremity motor function and the ability to perform ADL for hemiparesis after stroke. To the best of our knowledge, this is the first meta-analysis to compare the effects of both CCFES and NMES in treating people with stroke.
An NMES device consists of electrodes, which are connected to a stimulator, and a controller. The electrodes are placed over forearm muscles that open the hand or extend the wrist to deliver targeted pulses to induce muscle contractions. 30 By comparison, a CCFES device consists of a stimulator; stimulating electrodes, which are placed over forearm muscles that open the hand or extend the wrist; and a command glove or electrodes that are worn or placed over the nonparetic hand to record movements. 11 Volitional opening of the nonparetic hand acts as an input signal, producing a proportional intensity of stimulation to the paretic hand. 12 In the studies assessed, the participants were instructed to open or relax their hands with a timing identical to that of NMES.
Two functional outcomes were used in these studies, namely, UEFMA and BBT.14,23-27 FMA and BBT are reliable and widely used tools for assessing functional performance or repetitive movements in multiple clinical populations, including stroke participants.31,32 Overall, our results suggest that CCFES contributed to a greater extent to the positive functional outcomes in people with stroke than NMES did. Another notable finding was the AROM. Our results indicate that the CCFES group exhibited better improvements in finger or wrist extension. The results for level of body function indicate that CCFES may be more beneficial in regaining motor function. Furthermore, participants in the CCFES group had higher scores in the modified Barthel Index and Arm Motor Abilities Test, suggesting they may better perform their ADL. This may enable participants to have more active social lives. 33 However, although the results of the AMAT exhibited a potentially positive trend, the between-group difference did not reach significance (P = .07). This may be due to limited RCTs included in our study.
Previous studies have reported that therapies are more effective for participants in early phases of stroke because neuroplasticity is higher in these phases. 10 Among the selected RCTs, 3 trials included participants in the subacute phase of stroke.14,26,27 The data gathered in these trials indicate significant improvements in UEFMA and AROM in people with subacute phase stroke who underwent CCFES. The heterogeneity of the UEFMA was 42%, which differed from that in other outcomes. When the study by Zheng et al. was removed, 24 the I2 value became 0. This is because the effects of CCFES in Zheng et al., which was the only article to include participants with acute phase stroke, were more favorable than those in the other articles included in our study. This higher favorability may indicate that the effects of CCFES during the acute phase may be superior to those in the other phases, which is consistent with the findings of a previous study, in which electrical stimulation was found to be more effective during the acute phase of stroke. 10 However, because of the limited number of relevant articles and small sample sizes, we were unable to verify the effects of CCFES in different stroke recovery phases. Further study using larger RCTs involving various phases of stroke could provide more robust evidence.
Information on adverse events was provided in only 2 of the RCTs included in our study.23,25 In these RCTs, some nonserious, study-related, or potentially related adverse events, including numbness or tingling, skin irritation from the electrodes, or some transient discomfort from the stimulation, were reported in a few cases. These results are consistent with those of other studies, suggesting that electrical stimulation is a safe and feasible modality for stroke participants.34–36
Regarding the treatment protocols of the included studies, that of Knutson et al. comprised therapist-guided functional task practice and self-administered home stimulation exercise, whereas that in the other studies consisted of only therapist-guided extension or flexion exercises. Both protocols produced satisfying results, with no adverse events occurring, even when treatment was self-administered. This may indicate that these interventions are safe and easy to administer, even by nonhealthcare professionals. Furthermore, for contralateral control methods, both bend sensors in gloves and EMG recording electrodes were discussed in the included studies. The 2 methods were highly accurate and sensitive. 37 However, 1 study demonstrated that EMG signals may be altered by the position of electrodes, with 1 cm of difference potentially resulting in amplitude variations of 200%. 38 Therefore, gloves containing bend sensors may be more suitable for those considering self-administered home exercises.
The risk of bias in the studies included in this meta-analysis was assessed by the Cochrane risk of bias tool. One point of concern was that all of the RCTs included in our study lacked participant blinding; this might create bias in deviations from intended interventions. This may be due to the characteristics of CCFES treatment; the electrodes or instrumented gloves are placed or worn on participants’ bilateral hands. Furthermore, we had no method for ensuring that participants who underwent NMES did not open their contralateral unaffected hand in synchrony with the stimulation. This may influence the outcomes, such as influencing or reducing the difference between the NMES and CCFES groups. However, even under this circumstance that has the potential to blur the distinction between the 2 interventions, our study demonstrated satisfying results for CCFES. This indicates that the findings on the efficacy of CCFES may be even more robust if this bias was avoided. This observation is crucial for future research.
Although the underlying mechanism of CCFES improving motor recovery is not clearly understood, several features of the therapy have caused it to be more effective than NMES. First, functional task practice in the CCFES group was stimulation-assisted; participants could control the degree of opening and timing of the stimulation, and it was, therefore, more conducive to enabling completion of task-oriented hand function activities. 24 Moreover, because the participant controls the intensity of stimulation by using the unimpaired limb, synchronized bilateral symmetric movement must occur. A study by Wu et al. demonstrated that participants who underwent bilateral training more notably reduced compensatory trunk movement. This might be partly related to neurological reorganization in the motor cortex occurring while the participants were experiencing bilateral symmetric movement. 39 Exercise involving bilateral symmetric movement mediated by remodeling in the premotor cortex may be operative in CCFES. 16
In addition, the assisted practice of functional tasks that occurs in CCFES may contribute to its promotion of neuroplasticity; moving the bilateral hands symmetrically for a common purpose causes both hemispheres to act together, which then causes the undamaged, contralesional hemisphere to act as a template for the damaged, ipsilesional hemisphere, thus enabling the impaired upper limb to learn from the contralateral unimpaired upper limb.40,41 Mudie and Matyas reported that, if the bilateral upper limbs simultaneously perform asynchronous tasks, different templates are used in the hemispheres; thus, these tasks do not assist in restoring the neural network ring organization that controls action in the hemiplegic limb. 40 This suggests that CCFES may be a more effective form of intervention than NMES is.
Moreover, a previous study reported that central fatigue, which is caused by an increase in the excitability of inhibitory networks, may occur with NMES and may reduce corticospinal excitability. 42 Cunningham et al. 15 observed that ipsilesional output decreased after NMES was administered but remained relatively stable after CCFES was; they proposed that this was because the disinhibition induced by the bilateral movements of CCFES may have partially prevented fatigue-related diminution of ipsilesional output. 15
This study has several strengths. This is the first meta-analysis to verify and demonstrate the effect size of the positive effects of CCFES for people with stroke when compared with the widely used NMES. This research could also serve as a basis for future studies (e.g., the effect of CCFES on lower extremities). Multiple major databases without language restrictions were used for collecting RCTs on the basis of our wide inclusion criteria. Nevertheless, several limitations to this meta-analysis must be acknowledged. First, only 6 RCTs were included in this study, and one of our outcomes was reported in only 2 RCTs. We included participants regardless of the phase of recovery or the severity of impairment at baseline; however, the limited RCTs did not provide enough data to elucidate how the effects of CCFES are influenced by participant characteristics. Second, although our results were significant, more RCTs are required before definite conclusions can be drawn. Third, half of the included RCTs originated from the same authors;14,23,25 although the participant populations were dissimilar and the results passed the sensitivity analysis, this may still limit the applicability of our findings. Fourth, the comparison group in our study included only NMES. The effect of CCFES compared with that of usual care or other therapy not involving electrical stimulation could not be verified because no study has yet compared CCFES with other interventions. Finally, all of the included studies lacked participant blinding, which may have caused bias in deviations from intended interventions and have influenced the results of the analysis.
Clinical Implications
CCFES might be a safe and effective method for improving body function and activity in people with stroke. CCFES, in which participants are able to control stimulation using their unimpaired limbs, might be an alternative to conventional electrical stimulation. Because it reduces occurrence of central fatigue, CCFES may enable participants to train their hemiplegic limbs for longer periods and thereby improve their condition more rapidly. More RCTs with larger sample sizes and including different phases of stroke or other interventions are required for the effects of CCFES to be understood at every phase of stroke.
Conclusion
Contralaterally Controlled Functional Electrical Stimulation might be an alternative form of intervention that could facilitate recovery of upper extremity motor function and the ability to perform ADL. However, studies on this topic are limited. Our study has raised several questions that necessitate investigation. Therefore, further RCTs may be required to verify the effects and efficacy of CCFES. Further studies comparing CCFES with other interventions or modalities may be necessary to verify its role in stroke rehabilitation.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683221092647 – Supplemental material for Upper Extremity Contralaterally Controlled Functional Electrical Stimulation Versus Neuromuscular Electrical Stimulation in Post-Stroke Individuals: A Meta-Analysis of Randomized Controlled Trials
Supplemental material, sj-docx-1-nnr-10.1177_15459683221092647 for Upper Extremity Contralaterally Controlled Functional Electrical Stimulation Versus Neuromuscular Electrical Stimulation in Post-Stroke Individuals: A Meta-Analysis of Randomized Controlled Trials by Mei-Sean Loh, Yi-Chun Kuan, Chin-Wen Wu, Chun-De Liao, Jia-Pei Hong and Hung-Chou Chen in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
This manuscript was edited by Wallace Academic Editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
