Abstract
Keywords
Introduction
Of the acute stroke patients who require rehabilitation, 55% to 75% will have impairment of function of the hemiplegic arm 3 to 6 months poststroke and only 5% to 20% will gain complete, or near complete, functional recovery.1,2
Motor cortical plasticity and relearning of function is encouraged by functional practice of meaningful tasks aimed at acquiring a practical skill rather than constant repetition of movement that lacks purpose and context.3,4 Functional electrical stimulation (FES) combined with task-specific practice could be effective in improving outcomes in patients with moderate to severe dysfunction. 5
FES may improve outcomes for patients, including reduction in impairment and function at all stages of recovery.6-9 However, these studies have not examined the effects on activities of daily living (ADL) or quality of life. Few studies assess for sustained relearning effects. Many studies into the effect of FES on recovery of the upper limb following stroke have focused mainly on the application of cyclical stimulation for exercise. It has been proposed that patients may gain greater improvement in functional task performance if they were able to control stimulation volitionally by triggering it “on demand,” using the device as an orthosis to assist ADL.6,10 This technique allows patients to experience appropriate sensory and proprioceptive feedback timed to motor commands if the stimulation is controlled by the patient.4,11
Studies have demonstrated modest improvements in functional outcome measures using electromyography (EMG) as a trigger to initiate electrical stimulation to produce wrist extension.12-15 A literature review of 19 clinical trials evaluating the effect of electrical stimulation on motor control concluded that triggered stimulation may be more effective than cyclical stimulation but that its effect was not influenced by specific parameters of stimulation. 16 However, a more recent study by the same authors comparing cyclic with EMG-triggered stimulation found no significant difference between the 2 methods with respect to improvement in motor function. 17 EMG-triggered stimulation, however, may increase spasticity in the antagonist muscle group to that being stimulated, due to the effort required to produce a signal. 18 This may limit the number of patients for whom this means of control is suitable. An alternative method of triggering electrical stimulation to augment intensive task-oriented arm movements is by means of a push button switch operated by the therapist to initiate hand opening and grasp and release of an object.19,20
An accelerometer is a means of controlling electrical stimulation in which the trigger for stimulation is directly associated with initiation of forward reach and grasp. 21 Since active wrist and finger extension is not necessary to trigger the stimulation it may be that less increase in flexor tone is generated than in EMG-based systems.
Our hypothesis was that accelerometer-triggered electrical stimulation under volitional control would be feasible to assist upper limb recovery of function following stroke.
Methods
Participants
Participants were recruited from patients referred for electrical stimulation treatment for the upper limb to the National Clinical FES Centre at Salisbury District Hospital, UK. Ethical approval was obtained from the Swindon Research Ethics Committee.
Inclusion criteria included first stroke leading to hemiplegia of more than 6 months duration; age >18 years; a minimum of 45° forward shoulder flexion; able to initiate elbow extension; able to pick up and release a 2.5-cm cube (the easiest task in the Grasp section of the Action Research Arm Test); elbow, wrist, and finger extension in response to electrical stimulation; able to give informed consent; and ability to understand and comply with assessment procedures. Exclusion criteria included an orthopedic condition affecting the hemiplegic upper limb, cardiac pacemaker, shoulder pain, and grade of more than 3 on the Modified Ashworth Scale for the elbow, wrist, and finger flexor muscles.
Device
We used the Odstock 2-channel Programmable Stimulator (O2PS) from the Department of Clinical Science and Engineering, Salisbury District Hospital, UK. Figure 1 shows the device. A biaxial accelerometer detects movement of the arm as it attempts to reach forward. This movement is interpreted as a change of angle between the axis of the device and the Earth’s gravitational field. The device is powered by a 9-V battery, and delivers stimulation with an asymmetric biphasic waveform, current at 20 to 100 mA, and a pulse duration with a positive phase between 10 and 350 µs. A stimulation frequency of 20 Hz or 40 Hz can be selected and stimulation envelope ramps chosen for each channel between 0.1 and 6.5 seconds. The stimulator was worn on the upper arm held in place by a Velcro strap. The O2PS can be used for cyclic exercise or in triggered mode to assist function.
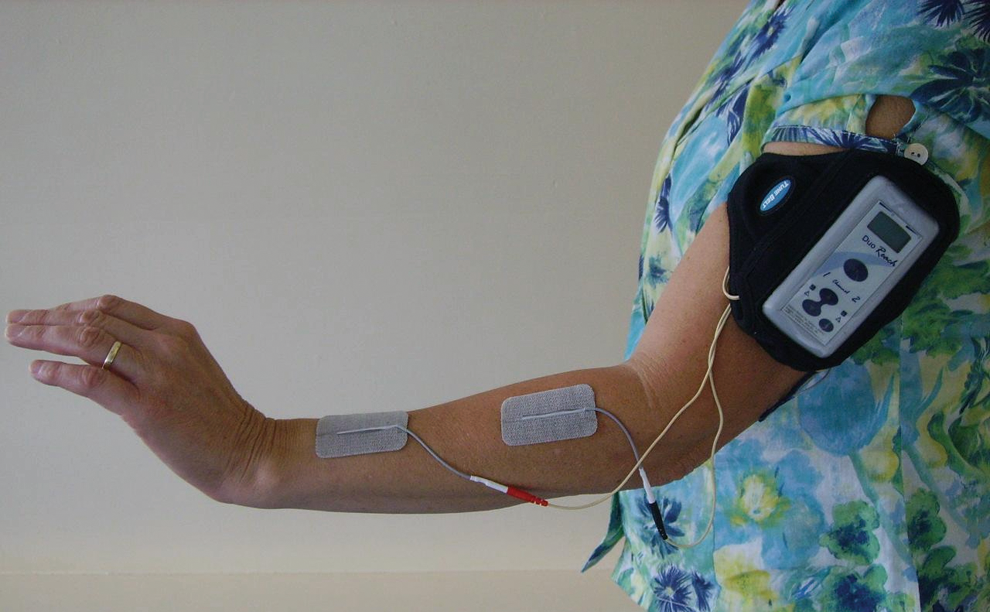
Stimulation of elbow, wrist, and finger extensors with the Odstock 2-channel Programmable Stimulator (O2PS).
Study Design
The study was a longitudinal case series design. An initial baseline period of 4 weeks was followed by 2 weeks of exercise stimulation carried out at home without using the trigger function. This allowed participants to practice placement of the electrodes, adjust to the sensation of stimulation, and to familiarize themselves with the device. After 2 weeks, participants were taught how to use triggered stimulation at home for the following 10 weeks. The device was returned to the clinic. Assessments were conducted at the beginning and end of baseline (weeks −4 and 0); weeks 2, 6, and 12 of treatment; and week 24, which was 12 weeks after treatment was withdrawn.
Procedure
Following the baseline period the O2PS was set up for 2 weeks of exercise stimulation. Pulse duration typically ranged between 150 and 350 microseconds, frequency was 40 Hz, and the output ramped at 2 seconds at the beginning and end of each cycle of stimulation. Participants were taught to apply stimulation to the extensor muscles of the elbow, wrist, and fingers. The stimulation was delivered via PALS skin surface self-adhesive 5-cm2 electrodes for the elbow extensors and 3.3 cm × 5.3 cm electrodes for the wrist and finger extensors. The first channel stimulated the triceps brachii muscle to elicit elbow extension. Electrodes were placed on the back of the upper arm (Figure 1). The second channel stimulated the extensor carpi radialis, extensor digitorum communis, extensor pollicis longus, and abductor pollicis longus muscles to elicit wrist and finger extension. Electrodes were placed on the back of the forearm (Figure 1). The electrodes were positioned to produce the most effective forward reach and hand opening although each participant was able to use any active glenohumeral flexion and abduction to acquire objects not directly in front of them.
In the 2-week conditioning period, stimulation of all muscle groups was synchronous with a duty cycle of 8 seconds on and 6 seconds off. Stimulation output was adjusted for individual participants according to their sensitivity to stimulation and the degree of antagonist muscle spasticity, to give an effective forward reach with hand opening without discomfort. A digital photograph, retained with each participant’s records and copied for the subject’s reference at home, was used to document the electrode positions. Participants were asked to apply stimulation twice daily for 10 minutes a session for the first 5 days, 15 minutes twice daily for days 5 to 10, and 20 minutes twice daily for the remaining 4 days. During this period of familiarization, participants were asked to work in synchrony with the stimulation while practicing the reaching movements produced.
After 2 weeks, the trigger was set up using the same stimulation parameters used in the previous 2 weeks. The stimulator was strapped to the outer side of the hemiplegic upper arm. Participants were asked to position the arm by their side and then to initiate forward reach. When they reached their limit of active shoulder flexion, the angle of the shoulder with the vertical was detected by the device and used as the trigger threshold at which stimulation would be initiated. On exceeding the threshold angle, the sequence began with stimulation of elbow extension followed 1 second later by stimulation of wrist and finger extension to open the hand. Stimulation was maintained for sufficient time for the participant to be able to position his or her hand around an object, typically about 3 seconds. Stimulation of both channels ended simultaneously, allowing the object to be grasped. When the participant was ready to release the object, hand opening was triggered by exceeding the threshold angle once again.
At this stage participants were shown by the physiotherapist (GM) how to practice 4 functional tasks at home using the hemiplegic arm with triggered stimulation for 2 sessions of 30 minutes a day with no more than a 5-minute rest period during a single session. These tasks included picking up a bath sponge and attempting to wash the opposite arm with it, picking up a small juice can and pouring, picking up a flat-sided beaker and attempting to drink, and holding a 4-ounce jar of coffee while unscrewing the lid with the unaffected hand. The tasks were initially done seated, but participants were encouraged to also try them in standing and when appropriate during the day to use the system to assist them in their ADL. The amount of time spent practicing ADL tasks was left to the individual. At the 6-week assessment, task performance was monitored by the therapist and was the only other face-to-face interaction with participants during the 10 weeks of triggered stimulation. Participants or their carers were encouraged to contact the therapist for advice. After the end of the treatment phase, participants were encouraged to continue to use the hemiplegic arm as much as possible during functional activities for the next 12 weeks.
Outcome Measures
The Action Research Arm Test (ARAT) 22 comprises 4 subtests. A total score is calculated from individual scores for each task. Scores range from 0, unable to attempt the task, to 3, performs task perfectly. Maximum scores for each category are grasp, 18; grip, 12; pinch, 18; gross movement, 9, for a total possible score of 57.
The Modified Ashworth Scale (MAS) 23 was used to measure passive resistance of elbow, wrist, and finger flexors while subjects sat on an upright chair without arms. The Canadian Occupational Performance Measure (COPM) 24 measures participants’ perception of their performance of a task and their level of satisfaction with that performance for 10 ADLs that they wish to improve and to rate them in order of importance on a scale of 1 to 10. The 5 activities scored highest are selected and rated 1 to 10, then averaged. A change in score of 2 or more is considered clinically significant. The Psychosocial Impact of Assistive Devices Scale (PIADS) 25 measures the impact of an assistive device. The 27 questions address 3 indicators of quality of life: competence (ability to perform), adaptability (adapt to different tasks), and self-esteem (confidence in performance). The questionnaire is scored from −3 to +3, with a 0 indicating no change and a positive score an improvement in quality of life. Use of Device Questionnaire (UDQ) was devised by the researchers and assesses the ease of donning and doffing the stimulation device, frequency and length of time used, and its degree of usefulness to the individual. Each participant completed a daily diary about use of the stimulator.
Data Analysis
Comparisons were made for the ARAT and MAS between the median total scores and those for the individual categories of grasp, grip, pinch, and gross movement at the beginning and end of baseline, from end of baseline and week 2 (to detect any change due to a possible treatment effect during the conditioning period), between end of baseline and the end of treatment, between the end of baseline and the end of the study, and from end of treatment and the end of the study. COPM median performance and satisfaction scores were compared between end of baseline and the end of treatment and end of treatment and follow-up. PIADS scores were collected at the end of the treatment.
Statistical analysis was performed using the Wilcoxon signed ranks test for nonparametric data. An α of .05 was selected to indicate statistical significance and left unadjusted for multiple testing in accordance with Rothman. 26
Results
Twenty-five volunteers with chronic stroke were assessed for suitability. Of these, 10 were not recruited because of too little active forward flexion movement at the shoulder or inability to pick up the 2.5-cm block. The remaining 15 agreed to take part, including 5 female and 10 male volunteers with a mean age of 57.9 years (SD = 14.4; range = 26-79 years) and mean time since stroke of 4.1 years (SD = 3.6; range = 1-13 years). Ten had left-sided and 5 right-sided hemiplegia. One was left handed. The hemiplegic upper limb was the dominant side in 5.
All completed the full protocol and none required extra assistance by telephone.
Total ARAT scores (Figure 2) showed no statistically significant change in the baseline period (
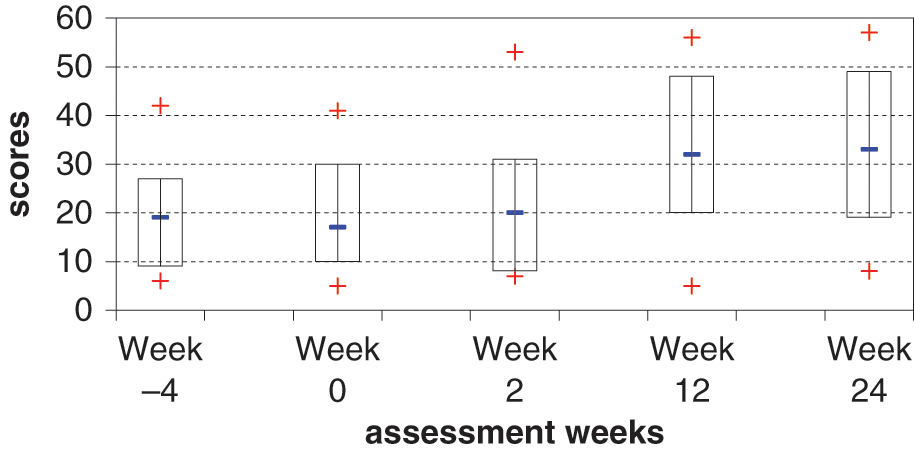
Action Research Arm Test. Median scores (−), box plot range 25th to 75th percentile range of scores (+).
Median performance scores for the COPM increased from 1.6 at baseline to 3.0 (
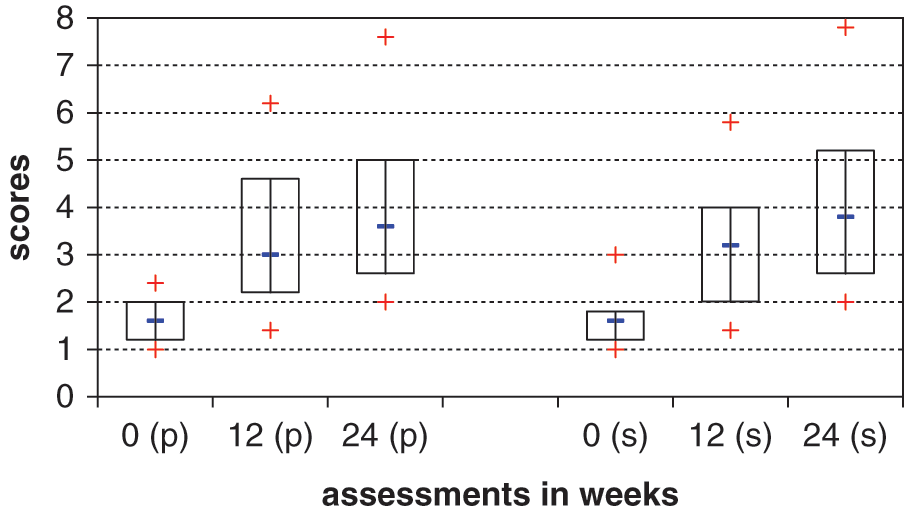
Canadian Occupational Performance Measure Median. scores (−), box plot range 25th to 75th percentile range of scores (+), p = performance, s = satisfaction.
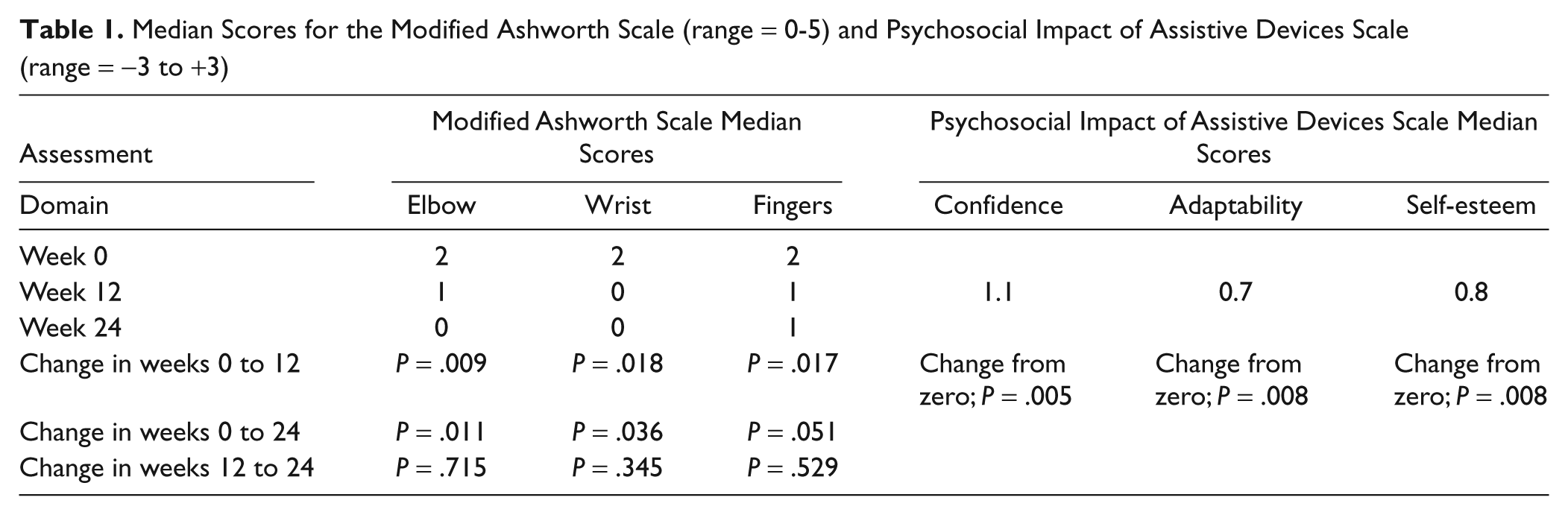
The Modified Ashworth Scale (MAS) demonstrated a reduction in resistance to passive movement at the elbow wrist and fingers at the end of treatment, which was maintained at follow-up (Table 1). Improvements were demonstrated in the 3 quality-of-life indicators of the PIADS assessment (Table 1).
No correlations were found between baseline scores and age, time since stroke, or side of stroke for any of the outcome measures.
Median Scores for the Modified Ashworth Scale (range = 0-5) and Psychosocial Impact of Assistive Devices Scale (range = −3 to +3)
The UDQ showed that 10 participants used the O2PS stimulator daily during the treatment period, 3 participants used it 5 days a week, and the remaining 2 participants used it for 4 and 3 days a week, respectively. The time spent using the device each day was between 1 and 12 hours (mean = 3 hours a day) shared between triggered practice of given functional tasks, practice of their selected tasks for the COPM assessment, and any other ADL they wished to attempt. The main reported changes in the hand as a result of using stimulation were increased awareness of the hemiplegic arm (all participants), reduced muscle stiffness (5 participants), increased movement (12 participants), increased sensation (1 participant), and reduced discomfort at the elbow (1 participant). All participants found the system useful to varying degrees: 3 fairly useful, 7 useful, 3 very useful, and 2 extremely useful. All participants reported that the device was more useful to them for its training effect on function than for its orthotic benefit while wearing it. Most participants or their carers found the system easy to don and doff, and none had problems with positioning the electrodes effectively. The stimulator itself worked reliably. However, all participants reported that the accelerometer triggering system lacked sensitivity and was therefore not always effective in assisting with function. Some participants therefore used the system in exercise mode at times to perform functional tasks. All but 2 participants reported that their hand function had improved when they were not wearing the device. Use of the O2PS stimulation device assisted improvement in a wide range of functional tasks that included picking up and carrying objects of different sizes, washing and dressing, 2-handed tasks such as opening and closing curtains, cooking, opening doors, turning on taps, hanging out washing, and computer keyboard skills. No adverse effects of the electrical stimulation treatment, such as skin irritation or hypersensitivity, were reported.
Discussion
This study investigated the feasibility and possible effects of accelerometer-triggered electrical stimulation on recovery of upper limb function in 15 subjects following stroke. The results demonstrated significant improvements in scores of impairment, function, and quality of life during the 12-week treatment period. These were maintained at follow-up 12 weeks later. The 100% compliance of the volunteers in this study may indicate acceptability of the intervention and the importance of upper limb recovery and rehabilitation to this patient group.
The greatest mean improvements in ARAT scores occurred in the Grasp (6 points out of 18) and Grip (4 points out of 12) categories of the test. This is not unexpected as the muscles selected for stimulation would not have affected the dexterity required for the tasks in the Pinch category or the shoulder movements needed to perform the Gross Movement tasks. However, stimulation of the selected muscles to assist elbow extension and gross hand opening would have facilitated reaching and acquisition and release of the larger items in the grasp and grip categories.
Other studies that have combined task-specific training with triggered FES have also shown significant improvements in functional outcome measures in chronic stroke patients.27-31 Although other studies stated that ADL tasks were practiced in the home while wearing the FES system, they did not report outcomes of the effect on specific activities related to patient objectives.
In this study, the COPM was used to identify individual patient objectives. The significant changes in scores for both performance and satisfaction after the treatment period and at follow-up may indicate that improvements in ADL can be maintained by continued practice of these tasks. However, the reliance of this assessment on the subjective reports of study participants does give potential for bias in the results. All participants reported that they were able to achieve additional functional tasks to those they had selected. It has been suggested that when performance of one set of tasks improves, this improvement may be transferred to other tasks requiring different motor skills. 32
An inclusion criterion of this study was that participants could grasp and release a 2.5-cm cube. However, some were able to achieve this by positioning their hand over the cube to grasp it without using any active wrist and finger extension. This lack of active extension may have caused them to benefit less from treatment than those with greater movement at baseline. However, there was only a weak correlation between ARAT scores at baseline and change in scores after treatment.
The reduction in spasticity throughout the hemiplegic limb demonstrated in this study is supported by a number of other studies, but none conducted a follow-up assessment to establish how long that improvement was maintained.31,33,34 At our 3-month follow-up, reduction in spasticity was maintained in the wrist and fingers and had reduced further at the elbow. It may be that this was due to the fact that participants used their hemiplegic arms more for functional tasks or at least to assist the nonhemiplegic arm during this period, thus reducing the likelihood of muscle and soft tissue shortening.
Although there were no statistically significant changes in the ARAT or MAS scores between weeks 0 and 2, this may have been due to the short length of the stimulation period. It cannot be inferred that the improvements in scores observed between weeks 2 and 12 of the treatment period were due to the change to triggered stimulation rather than just the increased period of treatment. However, it is possible that subjects were more highly motivated to use the triggered device for ADL rather than exercise alone, leading to greater compliance and the higher reported use of the device.
All participants reported that stimulation increased awareness of their hemiplegic arm and that they used it more spontaneously to assist the nonhemiplegic arm in bimanual tasks. This observation is supported by Lafosse et al, 35 whose study in stroke patients with left hemispatial neglect showed that stimulation that produced muscle contraction was more effective in reducing neglect compared with stimulation at the submotor threshold. The additional somatosensory input derived from peripheral nerve stimulation prior to motor training of the hemiplegic limb in chronic stroke patients has also been shown to improve sensorimotor hand function.36,37
The accelerometer-controlled device used in this study triggered movement of the hemiplegic arm “on demand” either to perform a task or to assist the nonhemiplegic limb in performing that task. However, the triggering method did prove somewhat crude for some participants, particularly those who had weak shoulder flexion and may have needed several attempts to initiate the movement. Some refinement of this mechanism is needed. Nevertheless, the improvements in functional tests and ADL may have been derived not only from facilitation of movement provided “on demand” but also from reduced spasticity in the hand, increased limb awareness gained by enhanced sensory feedback and the cognitive input, and increased attention required to complete a desired task.
The 3-month follow-up assessment in this study detected a short-term training effect on ARAT scores and the ADL tasks chosen for the COPM assessment. However, a 6- or 12-month follow-up period would have been of interest. Further limitations to this uncontrolled study were that the outcome measures were not blinded and the inclusion criteria restricted us to only a very small subset of the chronic stroke population. The ARAT test was not carried out with the stimulation device and so no measure of a possible orthotic effect was taken.
Conclusions
We show that accelerometer-triggered electrical stimulation for the hemiplegic upper limb is feasible and may improve functional ability and quality of life during treatment and for at least 12 weeks after treatment. The method of triggering requires refinement and a comparison study with nontriggered stimulation is required to demonstrate efficacy and cost benefit.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: The authors gratefully acknowledge a grant from the UK Stroke Association, which funded this study.
