Abstract
Background:
Physical activity (PA) in patients with myasthenia gravis (MG) is considered safe and beneficial, and an active lifestyle is required to obtain the health benefits of exercise. However, as the disease leads to physical impairments an insight into the overall PA habits in this patient population is relevant but lacking.
Objective:
To measure habitual physical activity in a Danish cohort of patients with MG measured by accelerometer and questionnaire, and to determine relevant predictors for PA intensities.
Methods:
Habitual physical activity was assessed by; 1) the accelerometer ActiGraph in a cohort of patients recruited from our neuromuscular clinic, 2) the International Physical Activity Questionnaire (IPAQ) in a web-based survey. PA levels were compared to international recommendations. Predictors for PA (age, sex, body mass index, disease severity and duration) were included in the regression analyses.
Results:
Habitual physical activity was measured by accelerometer for 7 days in 69 patients and by questionnaire in 691 patients. Measured by the accelerometer, 46%of the patients did not meet the international recommendations for PA at moderate/vigorous intensity and 57%were below the recommendations for steps per day. Measured by the IPAQ, 48%did not meet the recommendations. Disease severity and age were predictors for PA intensities.
Conclusions:
This study found that around half of the included patients did not meet the recommendations for PA. This is a concern, as it increases the risk of life-style related diseases. Disease severity and age may be taking into consideration when counseling the patients about PA.
Keywords
INTRODUCTION
Habitual physical activity refers to the physical activity (PA) that is integrated in peoples’ everyday life in their natural environment, and thus covers the activity level of a whole day instead of dedicated exercise sessions. Habitual physical activity is a relevant and important outcome measure in research, as lifelong PA at recommended intensities is important to obtain the health benefits of being active.
Myasthenia gravis (MG) is a chronic, autoimmune disease with a well-known pathogenesis. Blocking of signal transduction and destruction of the synaptic components, result in fluctuating strength of voluntary muscles, and distinct muscular fatigue with repeated muscle contractions. The Danish prevalence of MG is estimated to 180 per million people [1] with an annual incidence of 9.2 per million [2]. Recent trials [3, 4] as well as older case-studies of patients with MG participating in endurance exercise [5–7], or resistance training [8, 9], suggest that exercise training is beneficial, especially by increasing muscle strength without apparent side effects.
Over the last decades, new technology, such as accelerometers, has been developed that facilitates objective assessment of habitual physical activity. Accelerometers are considered objective but with some technical user problems or limitations. An alternative to accelerometry is self-reported outcomes such as questionnaires, which are simple to use and cost-efficient, but can be flawed by information bias. Habitual physical activity has previously been assessed in neuromuscular diseases by a wide range of accelerometers and questionnaires [10].
In patients with MG, habitual physical activity has been measured in three recent cross-sectional studies by either accelerometry [11, 12] or questionnaire [13], finding that patients with MG were less active than non-MG persons. Two interventional exercise studies examined baseline PA patterns in 10 and 11 patients with both accelerometry and questionnaire, even though this was a minor part of the studies’ analyses [4, 14].
The purpose of the present study was to measure habitual physical activity in patients with MG by both accelerometer (ActiGraph) and a standardized questionnaire (the International Physical Activity Questionnaires, IPAQ), and to identify important predictors for different PA intensities.
MATERIALS AND METHODS
Patients
Habitual physical activity was measured by questionnaire (IPAQ) in a web-based survey and by accelerometer in patients (≥18 years) recruited from the neuromuscular clinic at Copenhagen University Hospital –Rigshospitalet.
The survey was conducted from June to November 2019 and has been described in a recent study [13]. Patients were recruited from the Danish National Registry of Patients, which is a registry of all Danish hospital in- and outpatient discharges since 1977. The inclusion criteria were; resident of Denmark and a diagnosis code for MG according to International Classification of Diseases in either the eight edition (ICD-8: 733.09, 1971–1993) or tenth edition (ICD-10; G.70.0, from 1994) in the period from 1977 to the end of 2018. Also, patients should subscribe to e-Boks, which is a personal, digital mailbox, connected to the patient’s 10-digit civil registration number. Patients who met these criteria were invited to participate in the web-based survey, using the software REDCap (© 2018 Vanderbilt University). All responses to the survey were automatically stored in a secure database. As a part of the survey, patients completed the IPAQ, and the MG-ADL (MG Activities of Daily Living profile) as a measure for disease severity. Also, background information was registered; age, sex, height and weight (to calculate the body mass index, BMI), MG treatment and MG duration.
A group of patients were recruited from the neuromuscular clinic in the period November 2018 until February 2020. Patients arriving for their regular follow-up at the neurologist, were informed about the project, and, if meeting the inclusion criteria, invited to participate. The inclusion criteria were a MG diagnosis verified by a typical clinical history and symptom improvement with acetylcholinesterase inhibitors coupled with either positive acetylcholine receptor antibodies and/or significant decrement/increased jitter on electromyography. The included patients had a history of symptoms corresponding to categories II-IV on the Myasthenia Gravis Foundation of America clinical classification [15]. If patients met the inclusion criteria and agreed to participate, they were instructed to wear the accelerometer at home, 24 hours a day (unless when showering and swimming) for 7 full days. While in the clinic, patient completed the MG-ADL and the IPAQ, and background information was registered; age, sex, height and weight (to calculate the body mass index, BMI), MG treatment and duration.
Measurements
The ActiGraph wGT3X-BT accelerometer (ActiGraph, LLC, Pensacola, FL) was used to measure the patients’ PA level. Acceleration intensity and duration of hip motion (e.g., steps and other movements such as rising from a chair) were monitored in three axes (up–down, left–right, forward–backward). The accelerometer (weight 19 g, dimension 4.6 cm ×3.3 cm ×1.5 cm) was worn in an elastic belt on the right side of the patient’s waist. The ActiGraph monitors were set to activity counts in triaxial mode, using a 10-second epoch. The number of counts increased with the frequency and intensity of movement. Patients with 7 days of at least 10 hours of recording during daytime were included in the analyses. Wear time was defined by subtracting non-wear time from 18 h because all data between 12:00 a.m. and 6:00 a.m. were registered and excluded as sleeping time as done in other studies on habitual PA using accelerometry [16, 17]. Non-wear time was defined as intervals of at least 60 consecutive minutes of zero counts [18]. Light, moderate, and vigorous intensities were extracted, and were defined as: light = 100–1951 counts per minute (CPM); moderate = 1952–5724 CPM; vigorous ≥5725 CPM in bouts of minimum 10 minutes [19]. CPM ≤99 were defined as sedentary time. CPM were converted to minutes per week (m/w) of light, moderate and vigorous physical activities. Sedentary time was reported in minutes per day (m/d). Steps per day were also reported.
IPAQ short form [20] was used to measure self-reported time spent on PA undertaken across a comprehensive set of domains; leisure time PA, domestic and gardening activities, work-related PA, and transport-related PA. IPAQ assessed three specific types of activity; walking, moderate-intensity activities and vigorous-intensity activities. Patients were asked to report number of days, hours and minutes spent on these activity intensities during the last 7 days. Also, the number of hours and minutes of sitting on one of these days were reported. From these data, the minutes per week (m/w) in each intensity were calculated. For sitting time, minutes per day (m/d) were calculated. The PA level of the patients was determined according to the following international recommendations: 1) ≥150 min at a moderate intensity or ≥75 min at vigorous intensity per week [21], 2) a minimum of 10,000 steps per day [22].
The MG-ADL is an 8-item patient-reported questionnaire where higher score indicates higher MG severity (total score range 0–24) [23]. The scale assesses common MG-symptoms and dysfunctions, including questions of ocular, bulbar, respiratory, and extremity functions. The MG-ADL is a validated outcome measure in research and clinical practice [24].
The study was approved by the ethics committee of the Capital Region of Denmark (approval H-18031231). Written informed consent was obtained from all patients, but according to the ethics committee of the Capital Region of Denmark, written informed consent was not required for the survey. Permission was obtained from the Danish Health Data Authority to extract patient contact information from the Danish National Registry of Patients.
Analyses
Age, BMI and disease duration were presented by means and standard deviations (SD). MG-ADL, IPAQ and ActiGraph results were presented by medians and inter-quartile ranges (IQR) due to non-normal distributions. Normality was assessed visually by histograms and boxplots. Sex and treatment were presented by numbers and percentages. Differences in variables with continuous data where investigated by either unpaired t test or Mann-Whitney test, depending on normality. Differences in categorical data were investigated by Fisher’s exact test.
A commercially available software, ActiLife v6.11.6, was used for the analyses of raw accelerometer data. The 10-second epochs were for analyses collapsed into 60-second epochs, which was required by the ActiGraph software to set sleep time periods.
For analyses, minutes spent on moderate and vigorous activities were merged into one outcome; MVPA (moderate-vigorous physical activity), as many patients reported zero minutes spent on vigorous activities. The outcomes of the analyses were the activity levels measured by accelerometer (sedentary, light, MVPA, steps) and by IPAQ (sitting, walking, MVPA). Covariates were selected a priori and included in the general linear regression models as continuous variables (age, BMI, MG-ADL score, disease duration) or as categorical variable (sex). The number of covariates was limited to sample size divided by ten to avoid mass significance. For the same reason MG treatment was not included in the models. The following model assumptions were checked; independence of observations, linearity of covariates, homogeneous and normally distributed residuals. In case of violation, the analyses were conducted on log-10 transformed data.
For analyses, a p≤0.05 (2-tailed testing) was considered significant. All statistical analyses were carried out using SAS enterprise guide 7.1.
RESULTS
A total of 1745 persons were registered at the Danish National Registry of Patients with a diagnosis code of MG. N = 282 persons (164 women [58.2%]; mean [SD] age, 78.0 [12.6] years) did not subscribe to e-Boks and were excluded. N = 1463 persons received an information for the survey. N = 294 persons (173 women [58.8%]; mean [SD] age, 62.2 [15.1] years) contacted the study coordinator (LKA) due to uncertainty of the MG diagnosis and were excluded, and 390 persons (226 women [57.9%]; mean [SD] age, 55.0 [16.5] years) never responded to the survey. Of the 779 responders (413 women [53.0%]; mean [SD] age, 60.8 [15.5] years), there were missing data on IPAQ (n = 59), MG-ADL (n = 24), BMI (n = 1) and MG duration (n = 4), leaving 691 patients for analyses. Flowchart of in- and exclusion can be seen in Fig. 1.
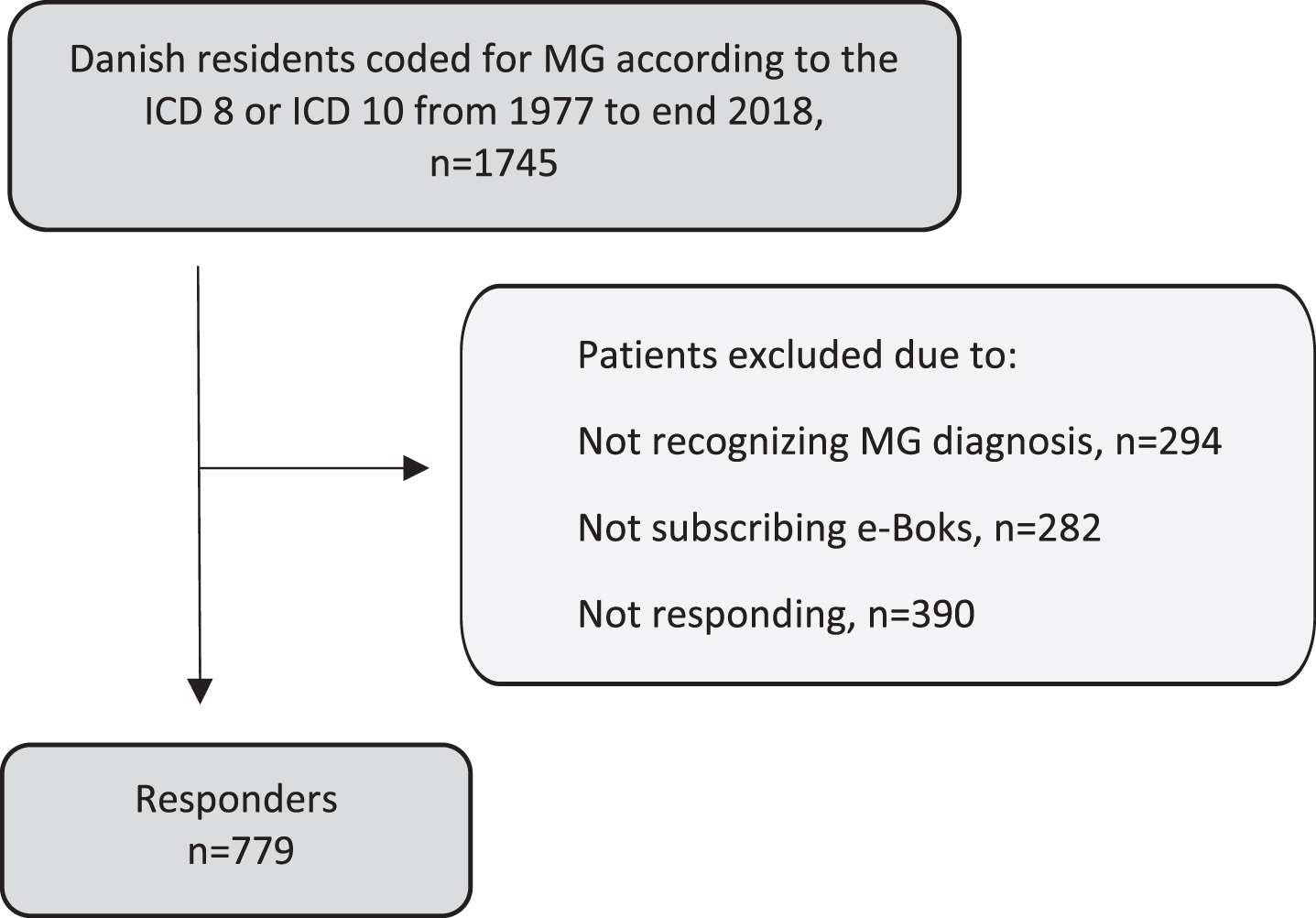
Flowchart of in- and exclusion of the survey.
A total of 69 patients were included for accelerometer measurements from the clinic. Wear time was on average (mean±SD) 16±1.4 hours/day during the 7 days. There was missing data on MG-ADL (n = 5) and IPAQ (n = 6) due to lack of time for completing the questionnaires.
Patient demographics are shown in Table 1. The only difference between the two samples was MG duration (p < 0.001). The mean BMI indicated overweight (BMI≥25) in both samples.
Patient demographics
Age, BMI and MG duration are presented as means and standard deviations (SD). Sex and treatment are presented as number and percentages. MG-ADL scores are presented as medians and inter-quartile ranges (IQR). Abbreviations: MG-ADL = MG Activities of Daily Living profile. *Significant (p < .001) difference between the two samples. #Some patients were in a combination treatment with both pyridostigmine and immunosuppressive drugs. Immunosuppressive drugs: prednisolone, Azathioprine, Methotrexate, Mycophenolic acid, Tacrolimus, Rituximab.
Time spent in different activity intensities are shown in Table 2. The time spent on activities of moderate intensity was 157 minutes (m) per week (w) measured by accelerometer and 150 m/w measured by IPAQ. Time spent on vigorous activities was 0 m/w and 60 m/w measured by accelerometer and IPAQ, respectively. The proportion of patients not meeting the international recommendations of PA was high. Measured by accelerometer, patients that did not meet the recommendations of moderate intensity (≥150 m/w) (n = 32, 46%) were older (67 vs 52 years, p < 0.01) compared to patients, that met the recommendations. In the survey sample, patients that did not meet the recommendations of moderate intensity (n = 343, 48%) had a higher median score on MG-ADL (3 vs 2, p < 0.01) and were more often women (58 vs. 42%, p = 0.01) compared to patients, who met the recommendations.
Physical activity levels measured by accelerometer and questionnaire
Data are presented as medians and inter-quartile ranges (IQR). Abbreviations: m = minute, d = day, w = week, IPAQ = the International Physical Activity Questionnaires. *patients both wearing an accelerometer and completing the IPAQ (missing data: n = 6).
More than half of the patients assessed by accelerometry (n = 38, 57%) walked less than the recommended 10,000 steps per day. There were no significant differences between the patients that walked more or less than 10,000 steps per day regarding age, sex, BMI, disease severity and duration.
Significant predictors for PA were MG-ADL score and age. High scores of MG disease severity increased time spent on sedentary activities (regression coefficient (β): 6.1, confidence interval (CI): 0.0; 12.1) and sitting time (β: 2.5, CI: 0.9; 4.0). Severe self-reported MG symptoms also decreased walking time (β: –3.6, CI: –6.5; –0.4). Higher age decreased time spent on MVPA (β: –4.0, CI: –6.3; –1.6) and steps per day (β: –90.8, CI: –168.0; –13.5) measured by accelerometer. Measured by IPAQ, increasing age decreased time spent in sitting (β: –0.5, CI: –0.8; 0.2) and increased time spent on MVPA (β: 0.7, CI: 0.1; 1.3) (Table 3).
Associations of physical activity, disease severity and demographics
β= regression coefficient, SE β = standard error of the regression coefficient, CI β = confidence intervals of the regression coefficient. Significance in bold. Abbreviations: MVPA = moderate-vigorous physical activity, MG-ADL = MG Activities of Daily Living profile, m = minute, d = day, w = week, BMI = Body mass index ¶ There was missing data on MG-ADL (n = 5) due to lack of time for completing the questionnaire, #measured in years
DISCUSSION
This is the first study to assess habitual physical activity measured by both accelerometer and questionnaire in a large cohort of patients with MG.
The study demonstrates that around half of the included patients with MG met the international recommendations for PA, which was found for both accelerometry and questionnaire assessments. These findings are in line with previous findings from our research group using another questionnaire (Saltin-Grimby Physical Activity Level Scale) in a large cohort of patients with MG [13].
Our study findings indicate a more active patient group, than previously reported [11, 12]. In those previous studies, only 22%and 30%of the patients met the recommendations for MVPA in bouts of ≥10 min, and only 22%met the 10,000 steps per day measured by accelerometer [11]. Time spent on sedentary behavior was almost similar across studies, but our study demonstrated more time spent on MVPA and more steps per day than previous findings [11, 14]. The disparity might be explained by the use of different accelerometers (O’Connor, Birnbaum, Westerberg used DynaPort Movemonitor) and thereby different algorithms, manufacturer software and cut-off points to determine time spent on PA intensities. Also, the wording of the recommendations is important. Patients meeting the recommendations of ≥150 min per week, might not meet the recommendations of ≥30 min/day for at least 5 days, as some patients accumulate their exercise in few days. However, our findings are strengthened by the combination of accelerometry and patient-reported outcomes, and by the large sample size. As we solely analyzed data (time spent on PA intensities) that were comparable between the IPAQ and accelerometry, we did not include other PA metrics. However, it might be relevant for future research to compare findings of other PA parameters, e.g. energy expenditure.
In a study of patients with myotonic dystrophy type 1 (n = 67) [25], around 50%of the patients met the recommendations for moderate intensity as measured by ActiGraph, which is in line with our results. Compared to a study of patients with mitochondrial diseases (n = 100) and healthy controls (n = 100) [26], MG patients walked more steps daily than the mitochondrial patients (mean±SD, 9,602±5,016 steps/day vs, 6,883±3,944 steps/day) and were closer to the results of healthy controls (9,924±3,944 steps/day). In the Copenhagen City Heart Study [27], the PA levels of 1670 adults (≥18 years) living in Copenhagen were measured by ActiGraph accelerometers for 24 hours/day, 7 consecutive days. This study found a median (IQR) of 579 (509–646) min/day spent on sedentary activities and a median (IQR) of 9288 (6932 –12003) steps/day, indicating only minor differences between patients with MG and the background population. However, in another Danish population study using IPAQ, only 29%didn’t meet the WHO recommendations [28], indicating that patients in the present study were less active. This emphasizes that comparisons with other patient- or healthy cohorts must be performed cautiously, due to differences in diseases, sampling (age, sex) and methods used.
Even though it is positive that patients with MG seem more active than reported earlier, it is still concerning that around half of the patients did not meet the recommendations of PA. Inactivity in MG is both associated with physical –and general fatigue [13], and as inactivity increases the risk of life-style related diseases [21] and disuse atrophy in all individuals, it is a concern if disuse atrophy exacerbates the disease-related fatigue/weakness in patients with MG. However, further research is needed to determine this.
We found that severe MG (MG-ADL) was associated with increasing time spent in sitting and sedentary activities. This finding has not been detected in the smaller, previous studies [11, 12], and might be explained by the large number of patients included in our study. However, no associations were found between disease severity and time spent on physical activities in any of the studies. This could be explained by the cross-sectional design of these studies, but also in the interventional exercise studies, MG severity was constant [3, 4] or only slightly decreased [14] after the intervention period. These findings indicate, that even though exercise improves muscle parameters and functional capacity in patients with MG, the commonly used MG-measurements might not be sensitive enough to detect these changes. Whether isolated symptoms of MG, e.g. muscle weakness in the leg, play a specific role on PA levels, can’t be ruled out in our study, as we used the total score of the MG-ADL. However, O’Connor[11] did not find any correlation between specified leg fatigue and PA levels.
Increasing age was associated with less time spent on MVPA and less steps per day measured by accelerometry. Contrary to these findings, increasing age was associated with less sitting time and increasing time on MVPA as measured by the IPAQ. These conflicting results could be due to bias in self-reported PA. The IPAQ has previously been found to underestimate sitting time and overestimate time on MVPA compared to accelerometry [29, 30], which might also be the case in the study (Table 2). This is also in line with the feedback from some patients, telling us that it was difficult to self-estimate time spent on sitting during a day. However, comparison of IPAQ and accelerometry should be performed with caution, as the cut-off points for intensity intervals assessed by accelerometry did not exactly correspond to the definitions of PA in the IPAQ. Also, as the IPAQ measured PA in certain PA domains (e.g. leisure time and transportation), accelerometry measured activities during the whole day. Social desirability bias might also explain the overestimation of vigorous activity measured by the IPAQ, compared to the accelerometer, but social desirability bias is found to be less pronounced in web-based surveys, where the online setting increases respondents’ perceived privacy [31, 32]. Also, it was not relevant to compare walking time in IPAQ with light activity from the ActiGraph, as walking was specified as one certain activity, whereas light activity was every activity in-between sleep, sedentary, moderate and vigorous activities throughout the 24 hours.
There are several study limitations. Based on the available data from the survey, it was not possible to determine the proportion of patients with ocular/generalized MG. This is a limitation of the study, as it could be relevant to determine if patients with a history of solely ocular impairments and no experiences of MG-related muscular fatigue in the extremities have a different pattern of habitual physical activity.
The accelerometer was worn at the waist, which we thought was the most convenient for the patients. However, as measurements in this position required hip movements, some activities e.g. bicycling where the hip was held still, may have been underestimated. Furthermore, the accelerometer did not measure the added strain of carrying a load, or walking uphill or upstairs, and arm movements were not recorded. A solution for a future study might be to add heart rate measurements to the accelerometer measures to register PA without or with minimal hip movements.
It might be that only the best functioning and most active patients agreed to wear an accelerometer, leading to selection bias. However, as baseline demographics such as age, BMI, and MG-ADL score did not differ between the clinic and the survey sample, selection bias was not likely.
The study was strengthened by the large number of patients included and the combination of patient-reported and objective data of PA.
The survey sample was considered representative of the Danish MG cohort in terms of demographic characteristics, except for the most disabled MG patients [13, 33]. Persons not subscribing to e-Boks (n = 282) were older than the survey sample, which could have introduced some selection bias based on age. However, as the non-responders (n = 390) were younger, the age- and sex distribution were almost similar between in- and excluded persons. As the 69 patients, who were assessed by accelerometry, resembled the survey sample regarding demographics and MG severity, and as the study findings were almost similar between the smaller clinical and the larger survey sample, we think these 69 patients are representative of the larger survey cohort.
We found that accelerometry was useful to measure habitual physical activity in patients with MG. Accelerometry could be relevant in future longitudinal studies to identify associations between changes in the patient’s disease and/or lifestyle and changes in the quantity or intensity of PA. Also, further research is needed to identify barriers to exercise for patients with MG, as this is not explained by MG severity. A previous, explorative study [34] identified, that barriers to exercise for patients with neuromuscular diseases were e.g. “lack of energy”. As fatigue is a core symptom of MG, and as fatigue and PA are previously found to be strongly associated [13], it is relevant to examine if changes in fatigue result in changes in PA habits. Also, it is relevant to identify motivational factors for PA. Music during exercise might be motivational as music during walking is found to improve walking distance in the 6MWT in patients with MG [35]. Further research is needed to elaborate this.
Footnotes
ACKNOWLEDGMENTS
LKA is grateful for a grant from the Research Foundation of Copenhagen University Hospital –Rigshospitalet.
CONFLICTS OF INTEREST
The authors have no conflict of interest to report.
