Abstract
Background:
Muscle weakness and fatigability, the prominent symptoms of autoimmune myasthenia gravis (MG), negatively impact daily function and quality of life (QoL). It is currently unclear as to what extent symptoms limit activity and whether physical activity (PA) behaviours are associated with reduced QoL.
Objectives:
This study aimed to describe habitual PA patterns and explore relationships between PA metrics, clinical MG characteristics, and health-related QoL (HRQoL).
Methods:
PA data from a tri-axial trunk accelerometer worn for seven days, was collected from females with generalized, stable MG and compared to control subjects. MG-specific evaluations, the six-minute walk test and knee extension strength were assessed in individuals with MG (IwMG). Mann-Whitney tests were used to study between-group differences. Spearman rank correlation coefficient was performed to explore relationships between variables.
Results:
Thirty-three IwMG (mean (SD) age 45 (11) years) and 66 control subjects were included. IwMG perform less vigorous-intensity PA than control subjects (p = 0.001), spend more time sedentary (p = 0.02) and engage in less and shorter durations of moderate-vigorous-intensity PA (MVPA). For IwMG, habitual PA correlated positively with 6 min walking distance (rho = 0.387, p = 0.029) and negatively with body mass index (rho = –0.407, p = 0.019). We did not find any association between PA or sedentary behaviour and; HRQoL, symptom severity nor lower limb strength.
Conclusions:
Individuals with stable MG perform less PA, at lower intensities, and are more inactive than control individuals. Further research is warranted to understand factors influencing PA patterns in MG and whether interventions could be successful in increasing PA quantity and intensity in IwMG.
Keywords
INTRODUCTION
Myasthenia gravis (MG) is an autoimmune disease in which antibodies alter the normal functioning of the neuromuscular junction of skeletal muscles causing reduced neuromuscular transmission efficiency. Consequently, individuals experience fatigability and weakness, which leads to functional impairments and presumably contributes to physical inactivity [1, 2]. However, due to the fluctuating nature of MG symptoms, typical point-in-time measures fail to capture the overall impact of symptoms on daily life. Self-report measures such as the MG-Activities of Daily Living score (MG-ADL) suffer from a floor effect in highly functioning individuals and is heavily weighted towards oculobulbar symptoms [3]. Wearable activity trackers or accelerometers have been used in multiple patient populations as they provide quantitative information of physical activity (PA) and sedentary behaviour (SB) and therefore could indirectly inform of the impact of MG on daily life [4–6].
With recent research investigating the benefits of exercise as an adjunct therapy in MG, it is essential to thoroughly understand habitual PA patterns, their relationships with symptoms and consequences of MG such as health-related quality of life (HRQoL) [7–9]. Not only could this information provide insight into feasibility and aid in designing appropriate exercise interventions but it could also assist in identifying and differentiating between causes of inactivity, reduced HRQoL and limitations in ADLs in MG.
Two recent studies evaluated daily PA in individuals with MG (IwMG) [7, 10]. Westerberg et al. reported high volumes of sedentary behaviour (SB) from data of 11 participants however, no relationship with MG symptoms was investigated and PA patterns were self-reported [7]. Using accelerometer-derived data in a larger cohort of 27 IwMG, O’Connor et al. reported relatively high levels of PA (average 181±158 metabolic equivalent of task (MET) min/day), with 78% of the cohort fulfilling the recommended minimum average of 64 MET min/day [10]. Thus, suggesting that IwMG are more active than previously thought. Comparisons with control populations in the latter study remain uncertain as data was not recorded with the same device [10]. Surprisingly, the authors did not find any correlation between PA or SB, and MG clinical status [10]. Relationships between PA patterns and HRQoL were not explored, nor was walking capacity or, muscle weakness.
Therefore, the aim of this current study was to specifically investigate free-living PA patterns in IwMG by quantifying and comparing daily PA of differing intensities and SB, to a control population. Any potential relationships between PA metrics and HRQoL, muscle strength, functional capacities, and other clinical variables in IwMG were also investigated.
MATERIALS AND METHODS
Study design, ethics and study participants
Prospective, cross-sectional study design using the STROBE guidelines for reporting of observational studies [11] (Supplementary information). Data for IwMG are from a prospective clinical trial (MGEX), prior to exercise intervention. Full details of participant recruitment, eligibility criteria have been previously published [9]. Briefly, to be eligible participants required a confirmed diagnosis of stable, generalised, mild-moderately severe autoimmune MG without contra-indication to exercise. The MGEX study was approved by an ethics committee in accordance with French regulations (Comité de Protection des Personnes Île-de-France XI #13064 on 13/12/2013) and authorised by the Agence National de Sécurité du Médicament et des Produits de Santé on 11/10/2013. The study was conducted in accordance with the Helsinki Declaration [12]. Written, informed consent was obtained from all participants. Only women were considered for this cross-sectional study to favour homogeneity. Control data was obtained from the accelerometer manufacturer database where participants self-reported no significant medical condition, were willing to wear the accelerometer for 7 days and provided consent for data use. Controls were matched 2:1 to MG patients in order to minimize the Mahalanobis distance computed on height, weight and age, using the optmatch package of R software [13].
Accelerometer-derived data
Free-living PA and SB were measured using a triaxial accelerometer (Version 3.0 DynaPort Movemonitor, McRoberts, The Netherlands) attached to the lumber spine with an elastic belt. The accelerometer weighs 55 g, measures 106.6×58×11.5 mm and measures acceleration at a sample rate of 100 Hz with a range of –6 g to + 6 g [14]. This accelerometer has been previously shown to be valid in several populations with chronic disease [15–17] and has been used in two cohorts of individuals with MG [7, 10]. Raw data are stored on a microSD card, which is retrieved, for analysis, at the end of the planned wear period. Proprietary algorithms were used to quantify PA and SB by measuring body posture and movement intensity from acceleration signals [18, 19]. To classify different activity intensities, proprietary energy expenditure algorithms previously validated using indirect calorimetry were used [18]. Participants were asked to wear the device during waking hours for 7 consecutive days, except when bathing or swimming, as the device was not waterproof. To account for day-to-day variation multiple days were monitored. Participants were supplied with a pre-paid envelope to mail the device back to the coordinating centre after the 7-day monitoring period. Data for IwMG was collected throughout the year thus covering all four seasons.
Accelerometer-derived data analysis
A valid day was defined as ≥12 waking hours (between 7am–11pm) wear-time. Non-wear time was detected using a proprietary algorithm meeting six different criteria based on vertical, medio-lateral and anterior-posterior accelerations (see supplementary data for details). Recordings with ≥4 valid days were considered for analysis and only valid days were included in the analysis irrespective of week and weekend days [20, 21]. For all individuals who met minimum wear-time criteria, valid data were averaged to obtain a mean daily value for all pre-selected accelerometry variables. PA metrics included total volume PA (all durations and intensities) when the device was worn, expressed in counts [22]. Time (minutes) spent in different PA intensities and time spent sedentary were normalised to wear time. PA intensity was expressed as multiples of the METs where 1 MET was defined as the resting metabolic rate [23]. Light-intensity PA (LPA) was defined as within 1.5 and 3 METs, moderate-intensity PA (MPA) as within 3 and 6 METs, and vigorous-intensity PA (VPA) as within 6 and 9 METs. Moderate-to vigorous-intensity PA (MVPA) was PA > 3 METs. Based on previous health recommendations, MVPA is commonly measured in bouts of 10 minutes [24]. PA bouts are periods of sustained activity of a specified minimum duration and intensity. In this study, we evaluated bouted MVPA (MVPA ≥10 mins), defined as sustained MVPA for at least 10 minutes with PA < 3 METs not lasting more than 1 minute over the 10-minute period. We also analysed cumulated sporadic MVPA, i.e. MVPA of any duration. SB was defined as time (minutes) spent in activities or postures where energy expenditure was < 1.5 METs.
Clinical data
Clinical MG history, Myasthenia Gravis Foundation of America (MGFA) classification and current medications were recorded by an expert neurologist who also administered the Myasthenia Muscle Score (MMS) and the MG-ADL score. The MMS is a clinical score routinely performed to assess muscle force and fatigability, i.e. symptom severity at the time of evaluation [25]. It evaluates limb, axial, bulbar and ocular symptoms with a maximum score (corresponding to no detectable deficit) of 100. The MG-ADL score consists of 8 items and measures the impact of MG on activities of daily living (ADLs) where zero indicates no functional impairment [26]. IwMG also completed a self-reported MG-specific HRQoL questionnaire (MGQOL-15-F). The MGQOL-15-F has been shown to be valid and reliable [27]. Higher scores (maximum score of 60) represent a lower perceived HRQoL. To explore functional walking capacity and lower limb strength, IwMG also performed the 6-minute walk test (6MWT) and maximal isometric knee extension strength (right-side) using an isokinetic dynamometer (Biodex System 3 Pro, Biodex Medical Systems, Shirley, USA). Full details of the protocol have been described previously [9]. Six-minute walking distance (6MWD) and knee extension strength were also expressed as percentage of predicted values using previously published equations [28, 29].
Statistical analysis
Demographic and clinical characteristics of participants were described as mean (standard deviation) or median (interquartile range) for continuous variables and, number (percentage), for categorical variables. Between-group differences were assessed using a Mann-Whitney test. Bivariate analyses were performed to evaluate relationships between habitual PA (total volume), SB and personal factors (age, body mass index), disease characteristics (duration and symptom severity: MMS), MG-specific QoL (MGQOL-15-F), functional walking capacity (6MWD) and knee extension strength. Spearman-rank-order correlation coefficient was used to investigate relationships between continuous variables. Graphpad Prism 8.4.3 and R version 3.6.3 (The R Foundation for Statistical Computing, Vienna, Austria) were used for analyses, p values < 0.05 were considered significant.
RESULTS
Of 41 accelerometer recordings returned, eight did not meet eligibility criteria (five had insufficient wear-time and three were male participants). Thus, data from 33 women with MG were included and compared to data from 66 female control subjects, matched for height, weight and age. Demographic characteristics are presented in Table 1. For IwMG, median (Q1; Q3) disease duration was 11 (5; 21) years with average (SD) disease onset at 32 (15) years old. Clinical characteristics are presented in Table 2. Accelerometer data are displayed in Figs. 1-2 and Table 3. For IwMG; the median times of LPA, MPA, VPA, MVPA, MVPA ≥10 mins and SB were 71, 80, 11, 95, 12 and 689 min/day, respectively. IwMG spent on average 80% of wear time sedentary and 8.7%, 9.4% and 1.9%, engaged in LPA, MPA and VPA, respectively (see supplementary data Table 1).
Demographic information of individuals with myasthenia gravis and control subjects
Data are mean (SD). IwMG: individuals with myasthenia gravis.
Clinical characteristics of individuals with myasthenia gravis
Data are number (n), median, interquartile range (Q1; Q3). EOMG: Early onset myasthenia gravis, LOMG: Late onset myasthenia gravis, MGFA: Myasthenia Gravis Foundation of America classification, AchR: muscle nicotinic acetylcholine receptor, MuSK: muscle-specific tyrosine kinase, MMS: Myasthenia Muscle Score, ADLs: Activities of Daily Living, Quality of Life (QoL), 6MWD: Six-minute walking distance, DM 2: Diabetes Mellitus Type 2, BMI: Body Mass Index.
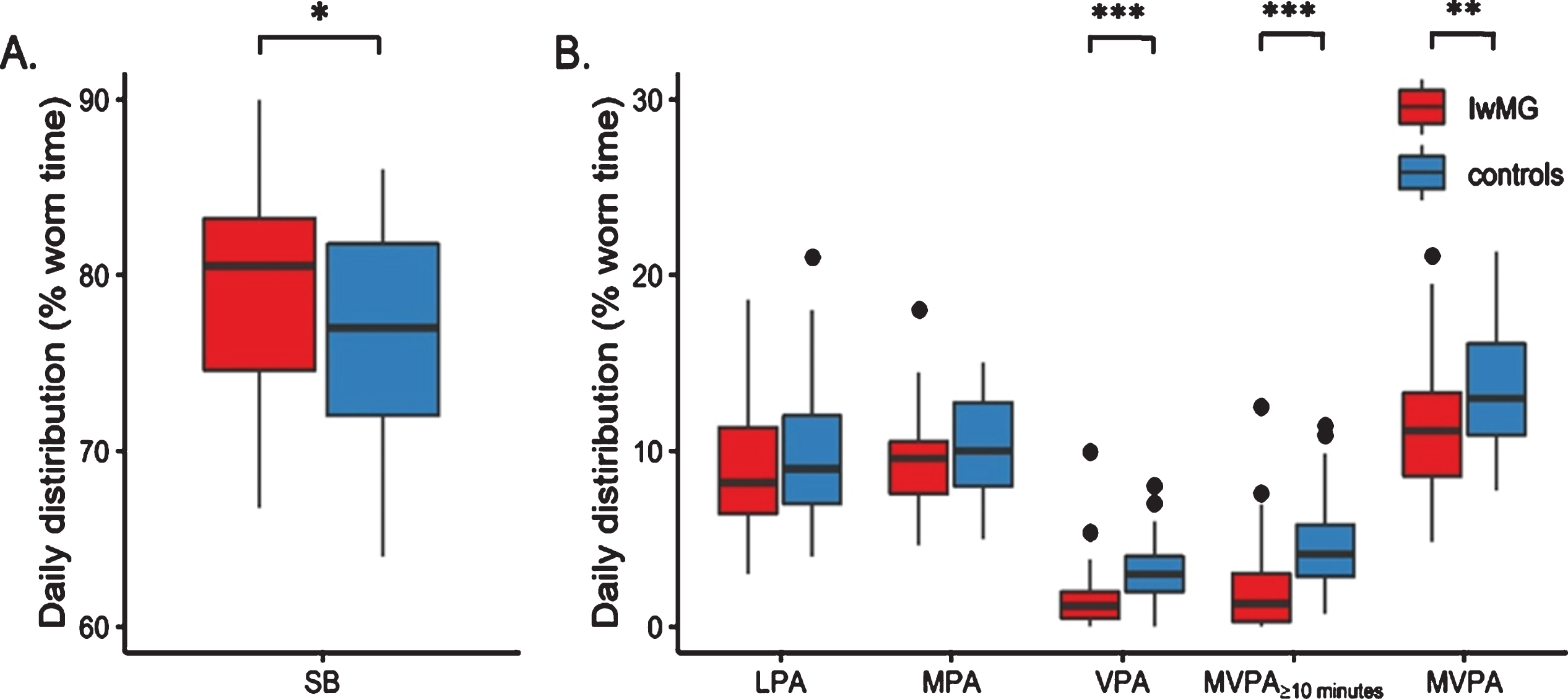
Distribution of the percentage of mean daily time spent in sedentary behavior and in physical activity according to intensity. Percentage of accelerometer worn time spent sedentary (panel A). Percentage of accelerometer worn time spent in different intensities of physical activity (panel B). The box-and-whiskers plots present the median (thick black lines), the first and third quartiles (box). The whiskers represent data points that do not extend from the boxes by more than 1.5 times the interquartile (IQR) range, and filled circles represent data points that lie beyond 1.5 IQR from the box. SB: sedentary behaviour, LPA: light-intensity physical activity, MPA: moderate-intensity physical activity, VPA: vigorous-intensity physical activity, MVPA ≥10 mins: bouted moderate-to-vigorous-intensity physical activity, MVPA: sporadic (i.e. any duration) moderate-to-vigorous-intensity physical activity. *p = 0.024, **p < 0.001, ***p = 0.003.
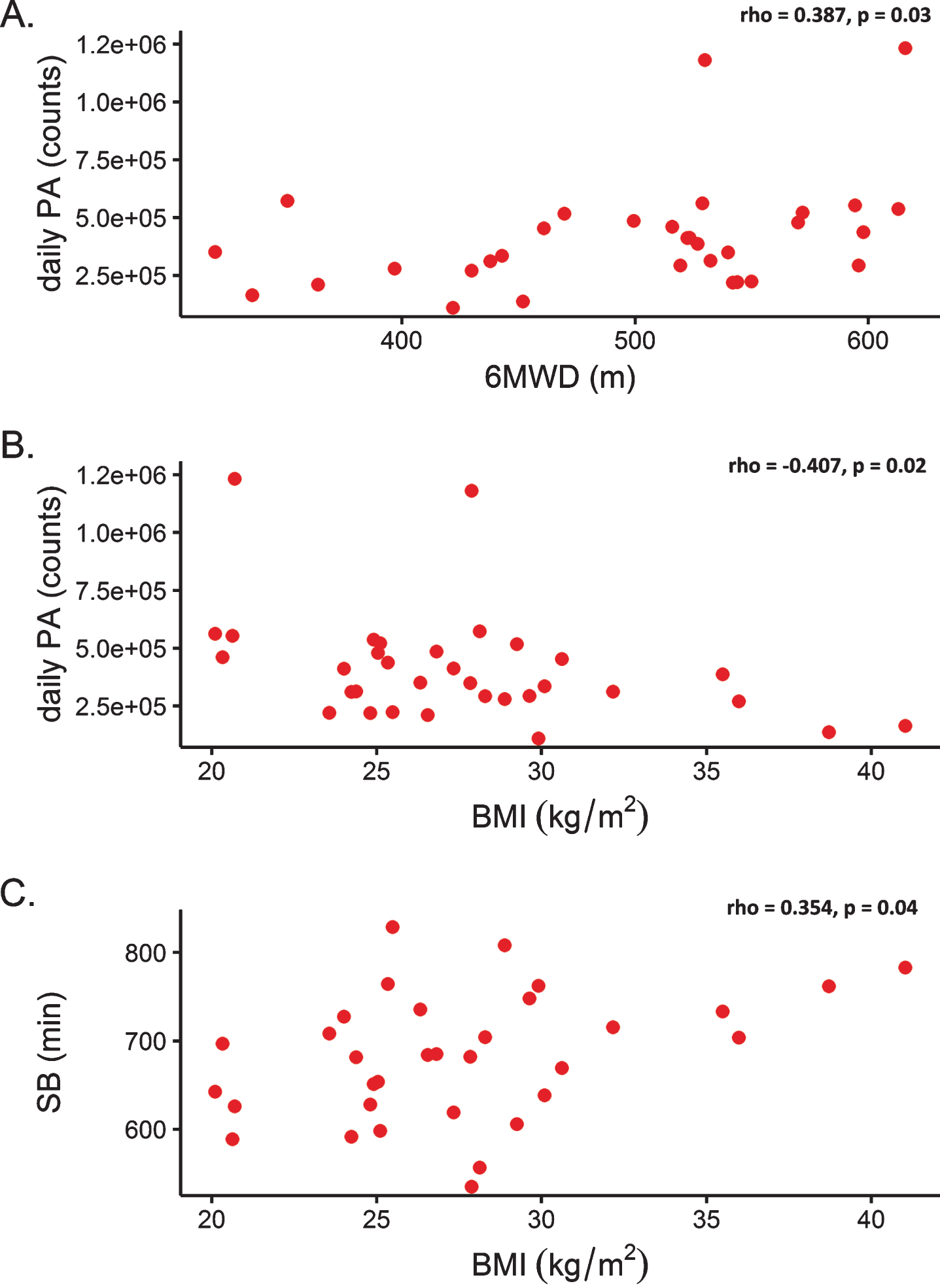
Relationship between variables. Relationship between daily physical activity and walking capacity (six-minute walking distance) for individuals with myasthenia gravis (panel A), rho = 0.387, p = 0.03; Relationship between daily physical activity and body mass index for individuals with myasthenia gravis (panel B), rho = –0.407, p = 0.02; Relationship between sedentary behaviour and body mass index for individuals with myasthenia gravis (panel C), rho = 0.354, p = 0.04. PA: physical activity, 6MWD: six-minute walking distance, BMI: body mass index.
Accelerometer wear-time data
Data are median, interquartile range (Q1; Q3). Total daily recording time = 16 h, between 7am and 11pm. IwMG: individuals with myasthenia gravis.
Compared to control subjects, IwMG spent more time sedentary and engaged in less vigorous PA, less overall MVPA and less bouted MVPA (i.e. less MVPA ≥10 mins) (Fig. 1). Whilst all IwMG undertook more than 21.4 min of MVPA/day (i.e. > 150 min/week), only 30% engaged in 21.4 min/day of 10-minute bouts of MVPA (i.e. MVPA ≥10 mins) compared to 58% for control subjects.
For IwMG, neither symptom severity, HRQoL, disease duration, lower limb strength nor age were associated with PA metrics, including SB (see supplementary data Table 2). There was a positive correlation between PA and 6MWD (rho = 0.387, p = 0.03, Fig. 2A) but not with SB (rho = –0.183, p = 0.32). Body mass index (BMI) was negatively associated with daily PA (rho = –0.407, p = 0.02) and positively associated with SB (rho = 0.354, p = 0.04), Fig. 2B-C.
DISCUSSION
In this study using a triaxial trunk accelerometer to assess daily PA and SB of IwMG, compared to 66 control subjects, three main findings emerged. First, we observed that females with MG perform less VPA, less overall MVPA and less sustained MVPA (10-minute bouts) than control subjects. Secondly, IwMG are more sedentary than control subjects. Thirdly, PA was related to walking capacity but we did not observe any association between HRQoL, symptom severity, disease duration nor lower limb strength, with daily PA or SB, for IwMG.
The quantity of PA undertaken by IwMG in this study is well below that described by O’Connor et al. but well above that cited by Westerberg et al. who reported only a median of 30 minutes of strenuous exercise per week (evaluated by self-report) in IwMG [7, 10]. These discrepancies with our results could be due to bias in self-reported PA, a lack of sensitivity in the questionnaire or selection bias in the former study where participants were likely to be motivated to exercise [10, 31].
Furthermore, whilst the amount of moderate-intensity PA did not differ between the two populations in our study, IwMG undertook less vigorous-intensity PA suggesting that exercise intensity may be a very important factor to consider, as well as duration. Our data do not provide a direct explanation for less vigorous-intensity PA levels in IwMG but one potential explanation is that fatigability may have contributed to the shorter durations of MVPA. In this study, no fatigue-specific measure was performed. Surprisingly, our results do not show any association with symptom severity and PA volume. These data are important to consider when designing interventions for this patient population. Although more intense and longer durations of PA correlate directly with improved health outcomes, small amounts of PA provide protective health benefits and increased longevity [32, 33].
There are currently no specific recommendations with respect to PA for IwMG. However when considering the most recent public health guidelines for weekly aerobic MVPA (target range of 150–300 minutes), all participants met current recommendations [34, 35]. When considering previous guidelines which required MVPA to be sustained for 10 minutes, only 30% of IwMG compared to 58% of control subjects met recommendations [24]. This demonstrates that beyond a certain intensity, fragmented or sporadic activity (of shorter duration) may be more feasible for this population. Especially considering the fact that the vast majority of time is spent in SB, as per current recommendations even small amounts of activity in exchange for sedentary time should be encouraged to offset the negative impact of SB [36]. This may be a more realistic goal particularly for individuals whom are less active.
Our results demonstrated wide variability in PA patterns in IwMG yet the data does not provide evidence of one specific explanatory factor for these differences. Less overall PA was related to greater BMI, which is consistent with previously reported findings in the general population [37]. However, the cross-sectional nature of this study does not enable us to determine the direction of this relationship, i.e. whether reduced PA contributed to greater BMI or greater BMI contributed to decreased PA, if indeed a causal relationship exists. Further research is warranted to explore the role of BMI in this population. Past studies in MG have also reported elevated BMI [7, 8]. In this cohort, 49% of IwMG were overweight and 21% were obese. One potential explanation could be that greater BMI is related to corticotherapy treatment, which could in turn be related to symptom severity. However, we did not find any relationship between BMI and symptom severity and current corticosteroid treatment doses were low [mean 7 mg; min 0, max 15, n = 6≥10 mg]. Previous corticosteroid-induced weight gain could still be present despite reduced current dosage.
Furthermore, we found that lower PA volume was also related to shorter distance walked in the 6MWT, a test of functional walking capacity. This relationship was weak, thus whilst habitual PA appears to be linked to capacity in IwMG, there may also be a behavioural component. Behaviour can be influenced by social, environmental, psychological and genetic factors, possibly entwined with, but also possibly independent from MG-related factors. Given that there was a negative correlation of BMI with SB and a positive correlation of BMI with daily PA this could suggest that body weight may play a role in lifestyle behaviours for IwMG and warrants further study.
Surprisingly, we found that habitual PA and SB were not related to HRQoL. In the general population beyond the physical health benefits previously presented, regular PA also reduces feelings of anxiety and depression, improves sleep and QoL and, increased volume of PA is associated with higher levels of happiness [38]. In this cohort, walking capacity (6MWD) was not related to HRQoL either (data not presented). Self-perceived QoL is complex and multi-dimensional and it is difficult to predict influencing factors.
With regards to comorbidities, twenty-four percent of IwMG had osteoporosis, which may be related to corticosteroid treatment and/or menopause [39]. This may be another reason to encourage PA in this population due to its multiple positive health benefits, including in osteoporosis [40]. The promotion of PA in MG is in its infancy. An important step in prescribing or promoting PA is to understand PA patterns and relationships (or lack of), with symptoms. These results provide important insights into PA patterns which could assist in designing future interventions as well as facilitating recommendations for clinical practice. Further research is needed to understand both barriers and facilitators to undertaking PA for IwMG and whether these results are applicable to males with MG and those with severe MG.
Regarding accelerometry compliance, wear-time during waking hours was superior to previous work and included a minimum of four valid days to account for possible day-to-day variation due to MG symptoms [41]. Both IwMG and control subjects spent the largest amount of waking time in SB. For both populations this was longer than that reported in the literature despite similar device wear-time [42]. This may be due to different accelerometer processing techniques and algorithms, different devices, shorter wear-time as well as different corrections for wear-time in other studies [43, 44].
Study limitations
There are several limitations in the current study. Due to the cross-sectional design only associations can be observed but no causal relationships can be confirmed or refuted. This cohort of IwMG were included as part of a clinical trial evaluating an exercise intervention, thus it is possible that there was selection bias. Included participants may have had a specific interest in exercise and therefore either perform more physical activity or on the contrary, perform less but with the desire to perform more, possibly within the safety of a clinical trial. Thus, this may be an over or underestimation of the PA and SB of the general population with MG. The majority of women in this cohort were overweight or obese however our figures are within the range of the French population [45]. More studies are required to explore whether this cohort is representative of all IwMG, especially considering there were only women included in this study. Concerning PA measurement; accelerometer type, wearing location and cut-points used to define PA and SB can influence results thus, comparison with other studies should be performed with caution [46]. A limit of accelerometry is the difference in wear-time which can be present. Wear-time variation can present a random bias if the participants takes off the device regardless of activity and a non-random bias if participants remove the device before undertaking an activity [47]. Non-wear time has been shown to mainly affect sedentary time estimates and due to the large wear-time inclusion criteria in this study we believe to have minimized any possible non-wear-time bias. Waterproof accelerometers are now available which can be worn non-stop and may help eliminate variation and facilitate analyses in future studies. Whilst the device itself has been validated in other chronic disease populations and has previously been used in two cohorts with MG, it has not specifically been validated in this population. Seasonal variation was not accounted for in this study as MG data was collected throughout the year. According to our analyses season did not affect PA metrics (Table S3), however this should be taken into consideration in future studies as temperature and environmental factors may have an influence on some physical activities and sedentary behaviours [48–50]. Finally, the control population in this study were from the Netherlands which has been shown to be the most active country in Europe, although activity levels in this group were well below what has been described elsewhere [51]. Nonetheless, this and cultural and environmental differences should be considered when comparing the two populations.
CONCLUSIONS
IwMG spend more time sedentary, undertake less VPA, less MVPA and shorter durations of MVPA than the general population as measured using trunk accelerometry. Habitual PA and SB are not related to self-perceived HRQoL nor symptom severity. PA monitoring in IwMG may be a useful tool to evaluate treatment effectiveness and appropriately target interventions, keeping in mind that shorter duration MVPA also provides health benefits and may be more feasible and appropriate particularly for less active individuals.
Footnotes
ACKNOWLEDGMENTS
We thank all participants for their participation and all administration and support staff who contributed to the conduct of the study. Special thanks to Alper Carras Ayaz and Aurélie Canal for assistance in data preparation.
A poster was presented at the World Conference of Physical Therapy, Geneva, May, 2019 and the World Muscle Society congress 2020.
FUNDING
The MGEX study was sponsored by the Assistance Publique - Hôpitaux de Paris “Direction de la Recherche Clinique et de l’Innovation” and funded through a grant from the Programme Hospitalier de Recherche Clinique - PHRC régional 2012 (Ministère de la Santé AOR12149), in collaboration with the Institute of Myology.
CONFLICT OF INTEREST
The authors SB, DB, RP, TS, JYH, PP, declare no conflict of interest.
