Abstract
BACKGROUND:
Biosurfactants are surfactants derived from several types of microorganisms such as bacteria, yeasts and fungi as membrane components or secondary metabolites.
OBJECTIVE:
To increase BS productivity as a biocompatible, low-toxic substitute for chemical surfactants employed in modern industry and the huge variety of applications they are used in.
METHODS:
Different media were used in isolation of the biosurfactant BS producers. The most potent bacterial isolate was analysed by 16S-rRNA. Plackett Burman and Box-Behnken Designs were used for optimization conditions. BS was purified by and characterized.
RESULTS:
Only one strain demonstrated great BS productivity, excellent emulsifying capability (54.5±0.1%) and oil spreading activity which analysed as Bacillus cereus AHMNAZ1 with accession number OP714421.1. The BS was lipopeptide in nature, identified as a surfactin which was stable and has Critical Micelle Concentration of 60 mg/L. Isolated surfactin showed excellent activity against Colon Carcinoma and Breast Carcinoma cell lines and can be used as antimicrobial agent.
CONCLUSION:
The study found a cheaper way of creating biosurfactants from agro-industrial wastes, delivering a twofold benefit of lowering environmental pollution and manufacturing useful biotechnological products (biosurfactants) with high activity and thermostability. Moreover, it can be used for the bioremediation of oil-polluted soils and in medical fields.
Introduction
Biosurfactants are microbial substances that have a distinct surface activity [1]. Surfactants are amphipathic molecules which lower the surface tension between water and hydrocarbon interfaces. The majority of surfactants on the market available are petroleum compounds which are extremely toxic and non-degradable [2]. A variety of microorganisms, including bacteria, yeasts, and fungi, produce surfactants as membrane components or secondary metabolites [3, 4]. Microbial surfactants can be divided into two groups: (i) low molecular weight BS, which involve glycolipids, lipopeptides, peptides, fatty acids and neutral lipids, and (ii) high molecular weight BS consist of polysaccharides, proteins, lipopolysac-charides, lipoproteins or complex combinations of various biopolymers [5]. As a result of the global preference for renewable products, microbial surfactants have garnered more attention due to their diversity, selectivity, low toxicity, biodegradability, ecological acceptance, efficiency at high pH and temperatures, the possibility to be produced through fermentation, and broad applicability [6, 7]. Recently, BS have been found to inhibit biofilm formation. BS are used in modern industries such as medical fields, food industries, and agriculture. They have a number of interesting properties of medical importance, bioremediation of petroleum pollutants, management, and enhancing crude oil recovery [6, 8]. BS can be created with inexpensive, widely accessible raw materials. An issue that affects the environment more and more is oil pollution. One of the primary causes of soil pollution is the steadily increasing demand for oil and oil-related products [9]. Due to the low solubility of liquids in soil, the biodegradation of oil is restricted [10, 11]. In the present study, a biosurfactant-producing bacterium, Bacillus cereus AHMNAZ1 strain, was isolated from Cook-top of kitchen stove. Moreover, optimization of bacterial growth conditions using Plackett Burman factorial design followed by Box-Behnken Design to enhance the BS production and characterization of the product for its chemical nature were carried out, moreover several applications of extracted surfactin were detected in the field of pharmacy and bioremediation like removal of crude oil from clothes.
Materials and methods
Isolation sources and culture medium
A total of 16 solid samples were collected from different oil contaminated regions in Egypt for isolation of the BS producers. Nutrient agar medium [12] and Minimal salts medium (MSM) (g/l) [13] are utilized for isolation, purification and maintenance of bacterial isolates. The purified isolates have been screened for BS production and the most potent isolate has been selected for further investigations.
Modified production medium, Bushnell and Hass (BHM-B) medium [14] which contain (BHM); glucose,1%; molasse,2; Bagasse,4% has been used for BS production.
Screening methods for BS producers
Microbial isolates have been screened for BS production using: Oil displacement method according to Satpute et al. [15], Emulsification activity according to Patel and Desai [16] and Emulsification index (E24) as reported by Sarubbo et al. [17], Drop collapse method according to Morais et al. [18], Cetyl trimethyl ammonium bromide (CTAB) agar plate assay as stated by Siegmund and Wagner [19], Hemolytic activity according to Igbonekwu et al. [20], Surface tension (SFT) measurement using du Nouy ring method as reported by Pornsunthorntawee et al. [21], Lipolytic activity according to Kumar et al. [22], Phenol sulfuric acid reaction according to Dubois et al. [23], Penetration method as recorded by Maczek et al. [24], Solubilization of Crystalline Anthracene according to Willumsen and Karlson [25].
Molecular identification of the most potent bacterial isolate
The most promising BS producer has been identified using 16 S rRNA analysis by Sigma company (https://sigmaeg-co.com) using universal primers, (5-AGAGTTTGATCCTGGCTCAG-3) (5-GGTTACCTTGTTACGACTT-3) as forward and reverse primer respectively for DNA amplification.
Optimization using PB factorial design
Plackett Burman (PB) factorial design was used for selection of the most significant factors that affect BS production by the most potent bacterial isolate, also Box-Behnken (Response-surface method) was used for modeling of the production system and prediction of the optimum conditions for maximum BS production. Analysis of variance (ANOVA) was used for data analysis and p < 0.05 was used as the criterion for statistical significance. Design Expert® (Version 7.0) software, State-Ease Inc., Minneapolis, MN, USA, was used for construction and analysis of the data obtained from both PB and Box-Behnken designs. Data represented in Table 1 shows the factors used in the (PB) design with their ranges and data represented in Table 2 shows Box-Behnken design used for modeling BS production by the most potent isolate.
Factors used for Plackett Burman design and their range of study
Factors used for Plackett Burman design and their range of study
Box-Behnken design used for modelling BS production by the most potent isolate
A summary of all biosurfactant screening assay techniques for BP isolate
Extraction, PURIFICATION AND CHARACTERIZATION of BS
The produced BS was extracted as stated by Varadavenkatesan and Murty [26]. Briefly, the culture broth was centrifuged in a refrigerated centrifuge (OHAUS USA FRONTIER 5706) at 5000 rpm for 20 min at 4°C to obtain a cell-free supernatant. BS was acid precipitated at pH 2 using 6 N HCl allowed overnight at 4°C. The precipitate collected by centrifugation at 10000 rpm for 30 min at 4°C. The precipitate was extracted thrice with a (2 : 1, v/v) chloroform-methanol mixture. The organic phase was removed and evaporated at 40°C. The solvents were evaporated leaving behind relatively BS extract as a viscous light brown matter which was weighed BS extract was characterization for its type by TLC using elution solvent chloroform: methanol: water (65 : 25 : 4), spots were revealed by ninhydrin specific for amino acid and stored at –20°C until use. BS was purified using silica gel column (25*2 cm) using stepwise elution with methanol and water ranging from 0 : 100 to 100 : 0 (v/v) at room temperature. Fractions (5 ml) were collected and evaluated for oil displacement zone. The activity of each peak was further evaluated using oil displacement zone, emulsification index and surface tension of fractions and the highest peak was analyzed by FTIR, GC/MS and tandem mass spectrometry LC/MS.
Fourier Transform Infra-Red Spectroscopy (FTIR)
The Infra-Red absorption spectrum of purified BS was obtained and recorded using Thermo Scientific company (USA) FTIR spectrophotometer (4000–400 cm-1) model Nicolettrademark iS50/iS50 R FTIR Spectrometer and performed at Regional Centre For Mycology and Biotechnology (RCMB), Nasr City, Cairo, Egypt.
Gas chromatography–mass spectrometry (GC-MS) analysis
The chemical composition of BS sample was performed using Trace GC-TSQ mass spectrometer (Thermo Scientific, Austin, TX, USA) with a direct capillary column TG–5MS (30 m x 0.25 mm x 0.25μm film thickness) according to [27].
Mass spectrometry
The molecular weight of the components of the surfactant was determined by negative- and positive-ion mode electrospray ionization (ESI) analyses. BS in the methanolic extract was examined by LC–ESI–MS by using UPLC MS/MS “Agilent” 642 [28].
Study the physical properties of extracted BS
Determination of critical micelles concentration (CMC) of BS
The CMC of the produced BS was determined through change in surface tension reduction measurements at various concentrations of 0–200 mg/L prepared in distilled water [29].
Biosurfactant stability studies
The stability (activity) of the BS was examined at a various range of temperatures (4–100°C), pH values (2–12) and NaCl concentrations (0–12%, w/v) for 30 min [30]. Stability studies were carried out using 0.1% (w/v) BS solution in 0.1M-phosphate buffer, pH7.0 [31]. The emulsification indexes of each treatment were determined at the conclusion of each period.
Applications of extracted BS
Removal of crude oil from clothes
The potential use of the BS for Microbial Enhanced Oil Recovery (MEOR) was evaluated
using artificially contaminated clothes with crude oil. Evaluation of the oil recovered
from artificially contaminated clothes was carried out using the approch of Zhang et al.
[32]. For a custom-designed oil removal
test, small pieces of cloths were weighed before (m) and after being
covered with drop of crude oil (m1). The oil-covered cloths
were transferred to 100 ml conical flasks with 20 ml of BS solutions. The flasks were
shaken at 35°C, 120 rpm for different incubation period (1, 2, 3, 4 days). Control was
prepared in the same way with addition of water instead of BS solution at the end of
incubation period, the cloths were taken out and dried in air to constant weight, then
weighed (m2). The crude oil removal efficiency (RE%) was
described as follows:
Where m is the mass of empty cloths, m1 is the mass of cloths covered with crude oil, and m2 is the mass of oil-covered cloths after BS treatment.
Antimicrobial activity of the produced BS
The derived BS has been assayed for its antimicrobial activity against bacterial strains: Staphylococcus aureus (ATCC 25923), Bacillus subtilis (NRRL B-543) and Methicillin-Resistant Staphylococcus aureus (MRSA 2658RCMB), Bacillus cereus RCMB 027, Staphylococcus epidermidis RCMB 009, Enterococcus faecalis (ATCC 29212), Escherichia coli (ATCC 25922), Proteus vulgaris (ATCC 13315) and the fungal strains: Aspergillus fumigatus (RCMB 002008) and Candida albicans (RCMB 05031) using agar well diffusion assay [33]. Well diameter was 6.0 mm, Gentamycin (4μg/ml) and ketoconazole (100μg/ml) were used as positive standard antibacterial and antifungal [34].
Minimum inhibitory concentration
MIC of the BS was determined as stated by Meir et al. [35].
Antitumor activity of the BS
The derived BS was assayed for its antitumor activity against MCF-7 cells (human breast cancer cell line) and HCT-116 (colon carcinoma), for its cytotoxic activity against VERO (Mammalian normal cells from African Green Monkey Kidney) according to [36, 37] at the Regional Center for Mycology and Biotechnology (RCMB), Al-Azhar University, Cairo, Egypt.
Cytotoxicity evaluation using viability assay
Cytotoxicity of the BS was assayed according to Mosmann [38] where the 50% inhibitory concentration (IC50) and the cytotoxic concentration (CC50), the concentration required to cause toxic effects in 50% of tumor cells and intact cells, respectively, were estimated from graphic plots of the dose response curve for each conc. using GraphPad Prism software (San Diego, CA. USA).
Results
Isolation and purification of biosurfactant (BS) producers
The isolation, purification and screening of BS results showed that from Sixty-three (63) isolates (49 bacteria and 14 fungi), only one purified bacterium coded BP was the most potent BS producer. This bacterium was isolated from Cook-top of kitchen stove, Nasr City, Cairo, Egypt on Modified Bushnell and Hass medium (BHM) at 35°C for 48 h.
Screening methods for BS producers
A summary of all biosurfactant screening assay techniques that had represented in Table 2a which showed that isolated BP was the most potent biosurfactant isolate with oil spreading zone 11.26, emulsification index 54.5±0.1, emulsification activity 1.18±0.035, positive drop collapse and blood hemolysis as well as 37.53 reduction of surface tension and has ability for solubilization of crystalline anthracene and penetration activity compared with control. These results are comparable with those reported in literature.
Identification of the most potent BS producer
The isolate BP had been identified using 16 S rRNA and analyzed using the advanced BLAST search program at the NCBI (http://www.ncbi.nlm.nih.gov). The BLAST analysis of the 16 S rRNA gene sequence of the selected isolate BP revealed alignments of this sequence with reported 16 S rRNA genes in the GenBank. The phylogenetic tree was constructed by neighbor-joining method [39] Fig. 1. On the basis of phylogenetic data obtained, the isolate BP showed maximum similarity 97.73% with Bacillus cereus. The nucleotide sequences of 16 S rRNA of Bacillus cereus strain AHMNAZ1 was submitted in the NCBI GenBank and given the accession number OP714421.1.

Phylogenetic tree showing genetic relationship between Bacillus cereus strain AHMNAZ1 and other reference Bacillus species in gene bank.
The most efficient fermentation factors regulating the formation of BS could be predicted by the PB model. It estimated the significant variables on BS formation for reducing the cost and incubation time for production process [40]. Implementation of PB Design for BS production by B. cereus resulted in the data represented in Table 3 and Fig. 2. The predicted and actual results showed an excellent correlation coefficient (R2) of 0.9757 and reasonable agreement between adjusted-R2 of 0.9271 and Prediction-R2 of 0.874.
Experimental layout designed by Design Expert and the corresponding experimental and
predicted values of the responses
Experimental layout designed by Design Expert and the corresponding experimental and predicted values of the responses
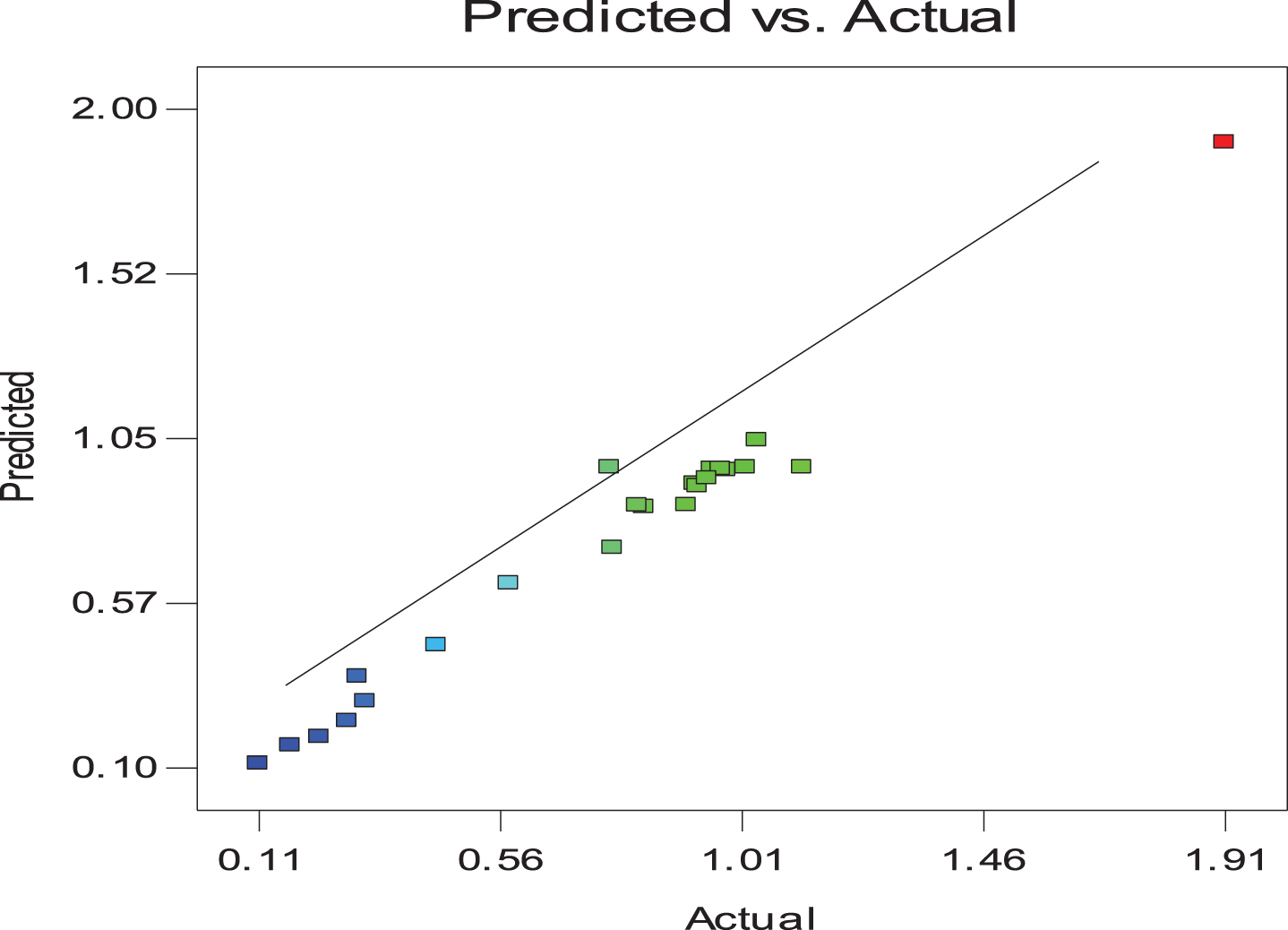
Relation between the actual and predicted results obtained from Plackett Burman Design.
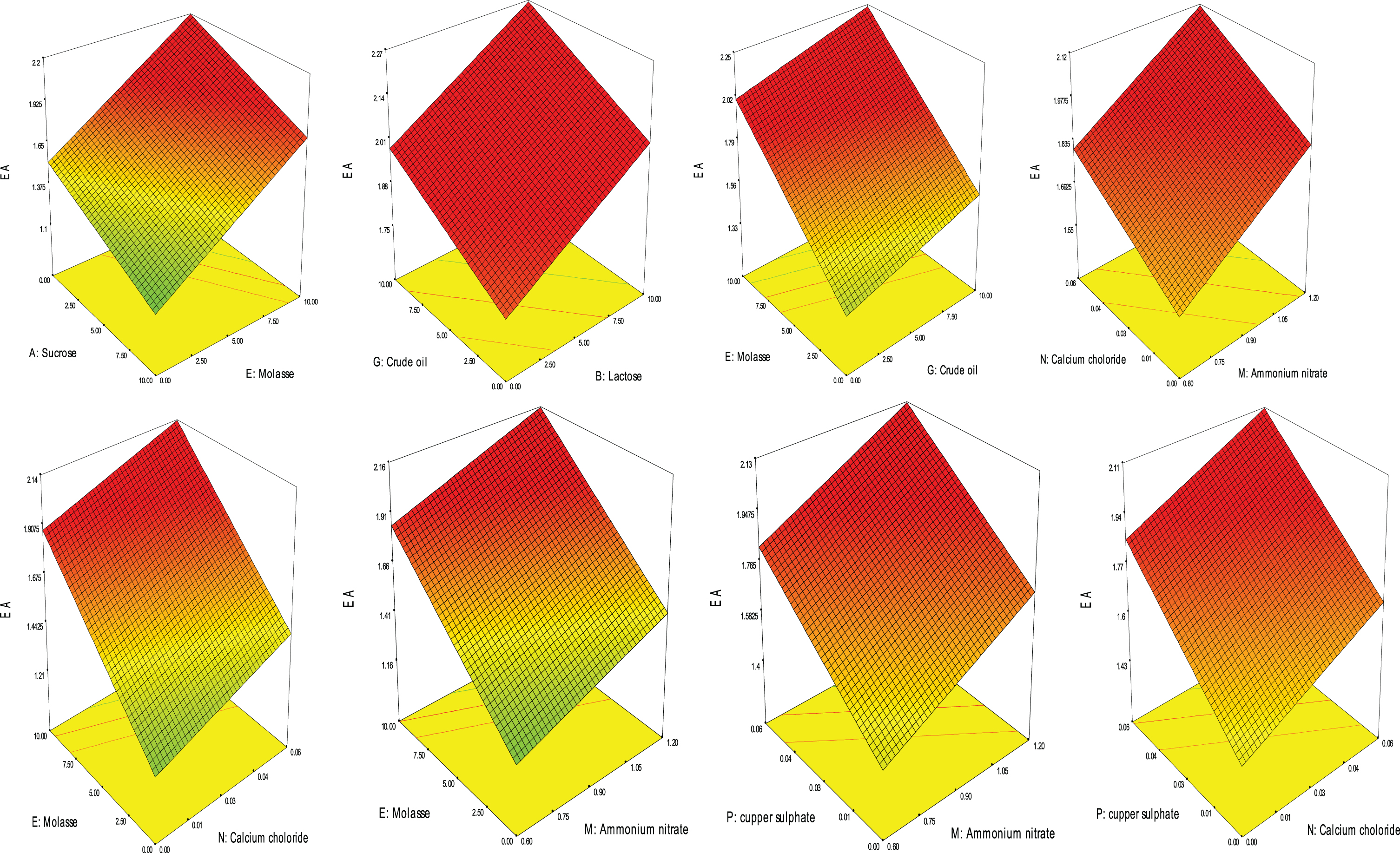
3D plotting of biosurfactant production based on PB Design.
Analysis of variance (ANOVA) of the obtained data in Table 4 showed that the model (with “F-value” of 20.08) and the factors coded: E, G, H, J, M, N, P, AB, AD were all significant model. The model “Lack of Fit” was insignificant with “F-value” of 0.11. The model “Adequate Precision”; which measured the signal to noise ratio, was 19.408 which indicates an adequate signal. These results showed that the model could be used for the navigation of BS model space. The following equation represented BS production using the significant factors according to PB factorial design.
ANOVA analysis for Plackett Burman Design based on Emulsification Activity (EA)
Where; A: Sucrose, B: Lactose, D: Glycerol, E: Molasse, G: Crude oil, H: Ammonium sulphate, J: Sodium nitrate, K: Peptone, M: Ammonium nitrate, N: Calcium chloride, O: Zinc sulphate, P: copper sulphate.
The significant factors obtained from PB Design were used for formulation of Box-Behnken design. Implementation of Box-Behnken design resulted in the data in Table 5 and Fig. 4. The results showed an excellent correlation among the predicted and actual data R2 of 0.6289 and adjusted-R2 of 0.3712.
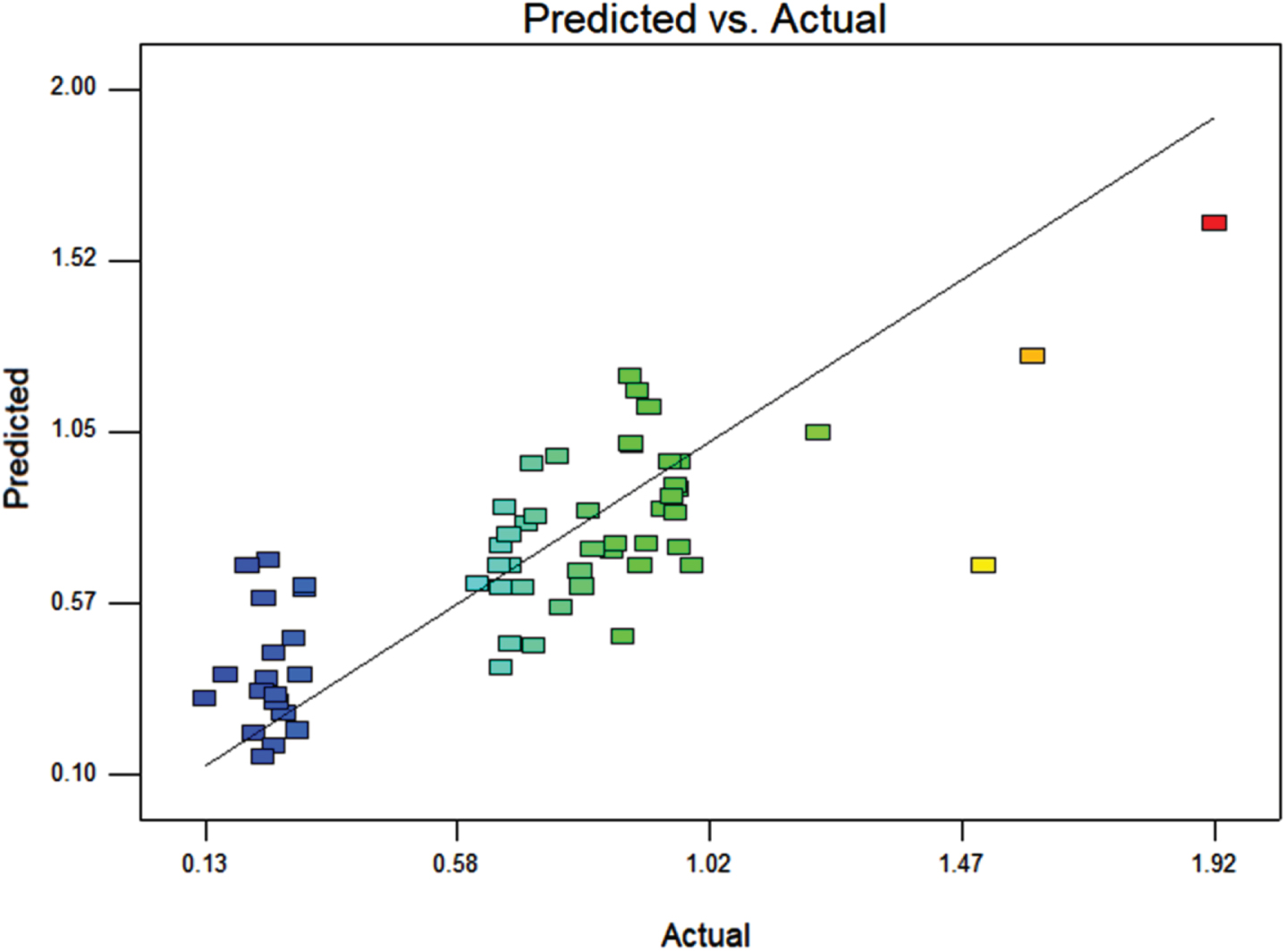
Correlation between the actual and predicted results obtained from Box-Behnken Design.
Experimental and predicted results of Box-Behnken design
ANOVA of the data obtained from Box-Behnken design in Table 6 showed that the model is significant with p-value of 0.0071. The model “Lack of Fit was insignificant with p-value of 0.9648. These results show that the model could be used for the navigation of BS model space.
Results of ANOVA for data obtained from Box-Behnken design
The following equation represented BS production using the significant factors according to PB Design.
Where; A: Molasse, B: Crude oil, C: Ammonium nitrate, D: Calcium chloride, E: Copper sulphate, F: Incubation period, G: pH
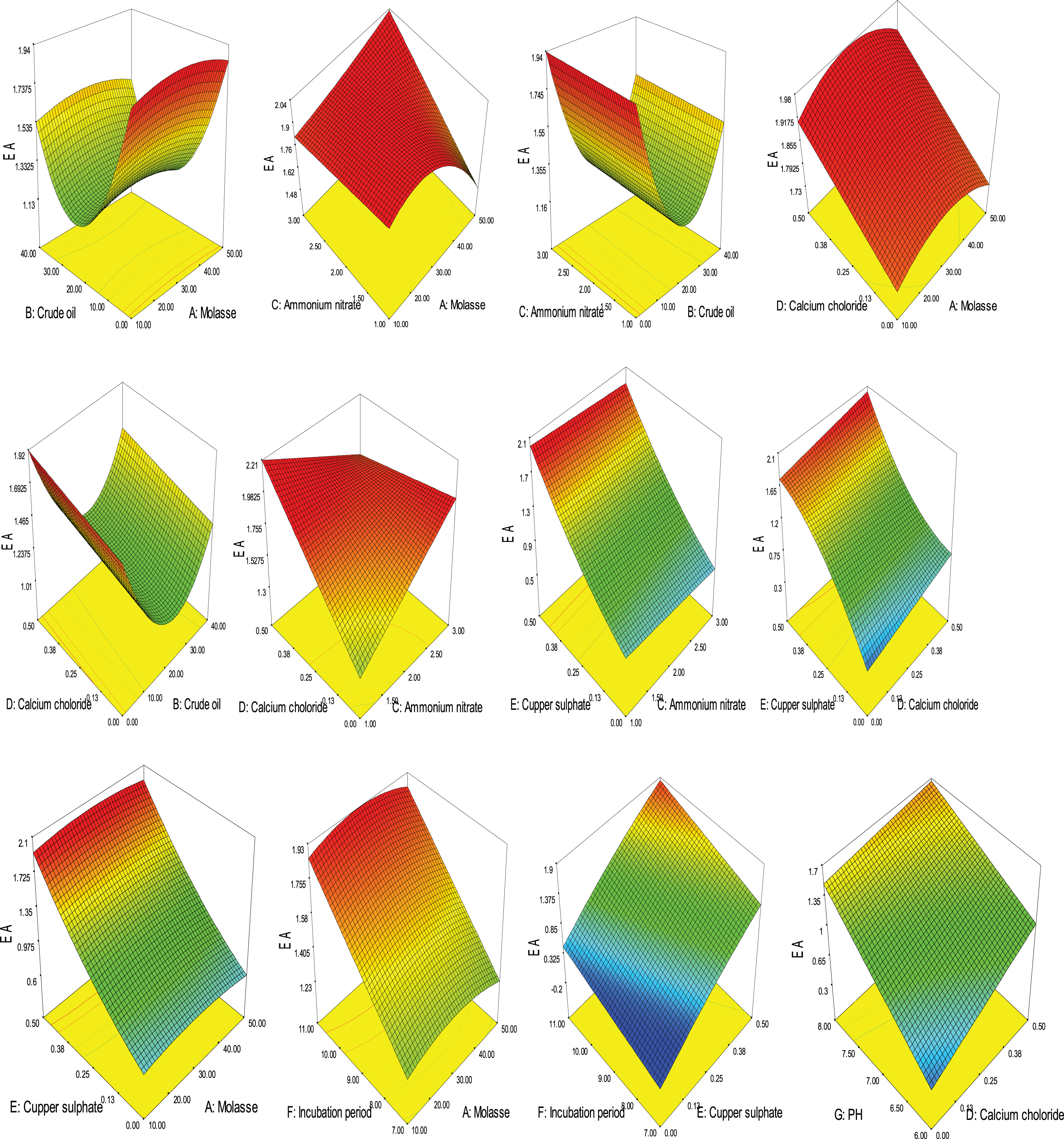
3D plotting showing the effect of different variable and factor-factor interactions on BS production based on Box-Behnken design.
The optimum conditions for maximum BS production by Bacillus cereus AHMNAZ1 were predicted from the produced Box-Behnken model as follows: molasse, 33.86 ml/l; crude oil, 0.11 ml/l; ammonium nitrate, 2.35 g/l; calcium chloride, 0.35 g/l; copper sulphate, 0.48 g/l; incubation period, 10.98 days and pH 8 to obtain maximum emulsification activity of 1.92. The data showed significant increase in emulsification activity (1.92) compared with that obtained at the original conditions (1.18).
Extraction of BS produced from Bacillus cereus AHMNAZ1 under the optimum conditions resulted in 2.23 g/L. TLC of the produced BS revealed that it was lipopeptide with RF value of 0.36 which is a member of surfactin family [41]. The silica gel column chromatography purification of the BS resulted in the fractions activities shown in Fig. 6.
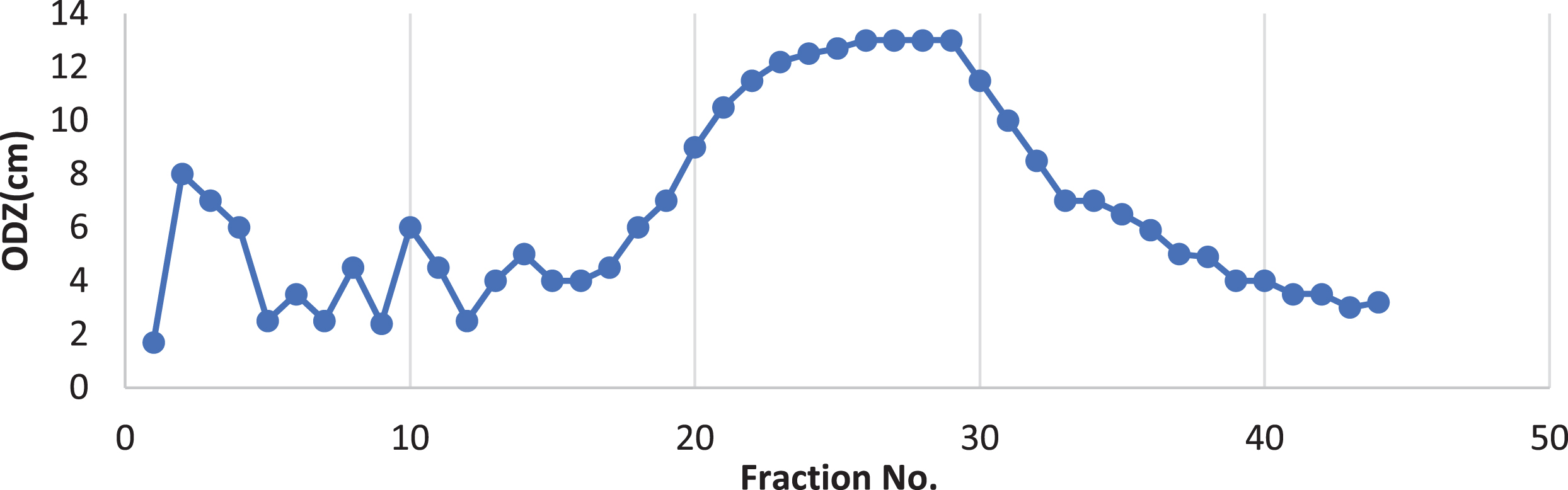
Typical elution profile for the chromatography of Bacillus cereus lipopeptide biosurfactant on silica gel (60) column chromatography.
The fractions (20 : 31) (peak: 1) were collected together in a single vial which demonstrate 12.03±0.057 cm in oil displacement zone, 60.06±0.11% in emulsification index and reduction in surface tension of 36.53±0.057 mN/m when compared with fractions (2 : 4) (peak:2) which demonstrated 7.03±0.057 cm in oil displacement zone, 50.1±2.15% in emulsification index and reduction in surface tension of 40.73±0.64 mN/m.
As a result of C–H stretching vibrations, N–H stretching vibrations and hydrogen bonded –OH functional groups, a strong broad absorbance peak (centred around 3442.36 cm–1) with wave numbers ranging from 3600 cm–1 to 3100 cm–1 was observed (Fig. 7a). This is typical of carbon-containing compounds with amino groups. The FTIR spectra showed a peak at 2359.579 which was characteristic of alkyl chains, peak at 2100 which represented aliphatic group and a sharp peak at 1015.07 cm–1 that corresponded to the presence of C–N aliphatic amines. A strong band was also observed at 1635.06 cm–1. This is due to ester carbonyl group of peptide components.
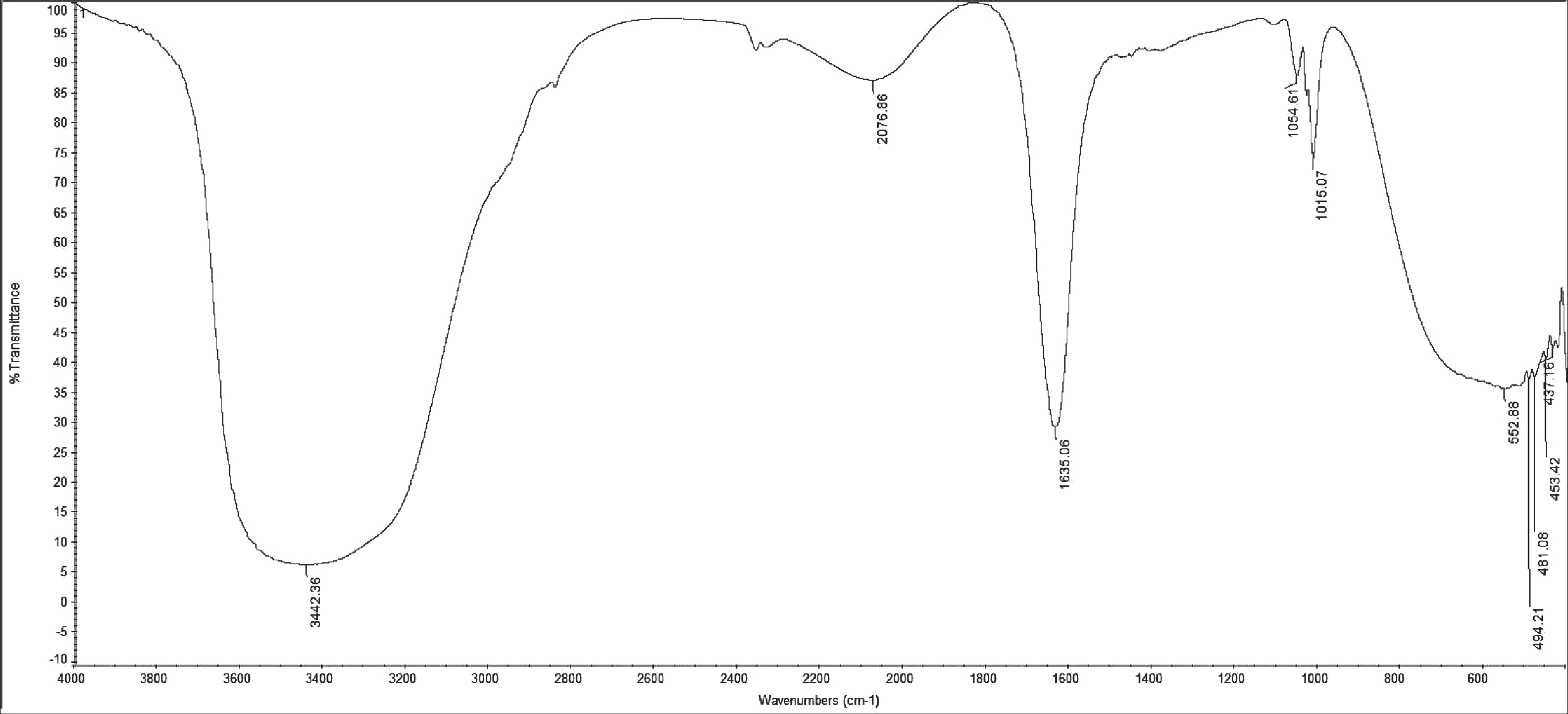
Fourier transform infrared spectra (FTIR) spectra of the silica gel (60) column liquid chromatography - purified bioactive fraction of the BS produced by Bacillus cereus AHMNAZ1.
The identification of fatty acids components was done in scan mode and the target mass spectra obtained from sample were compared with mass spectra obtained from the library. The GC-MS analysis showed that the compound produced by Bacillus cereus AHMNAZ1 was a lipopeptide derivative with hydrophobic moiety investigated to be a Hexadecanoic acid, methyl ester (31.47%) as the predominant major compound. Other major compounds included: 9-Octadecenoic acid (Z)-, methyl ester (8.78%), 1-{2-[(2-isopropyl-5-methylcyclohexyl)Oxy]-2-Oxoethyl}-2-(4-Methoxyphenyl)-3-Methyl-3H-Benzimidazol-1-IUM (8.94%), Heptadecanoic acid, 16- methyl-, methyl ester (5.99%). Esters and alkanol groups were also eluted. Total ion chromatogram of biosurfactant produced by Bacillus cereus AHMNAZ1 showed the peaks of the individual compounds is presented in Fig. 8.
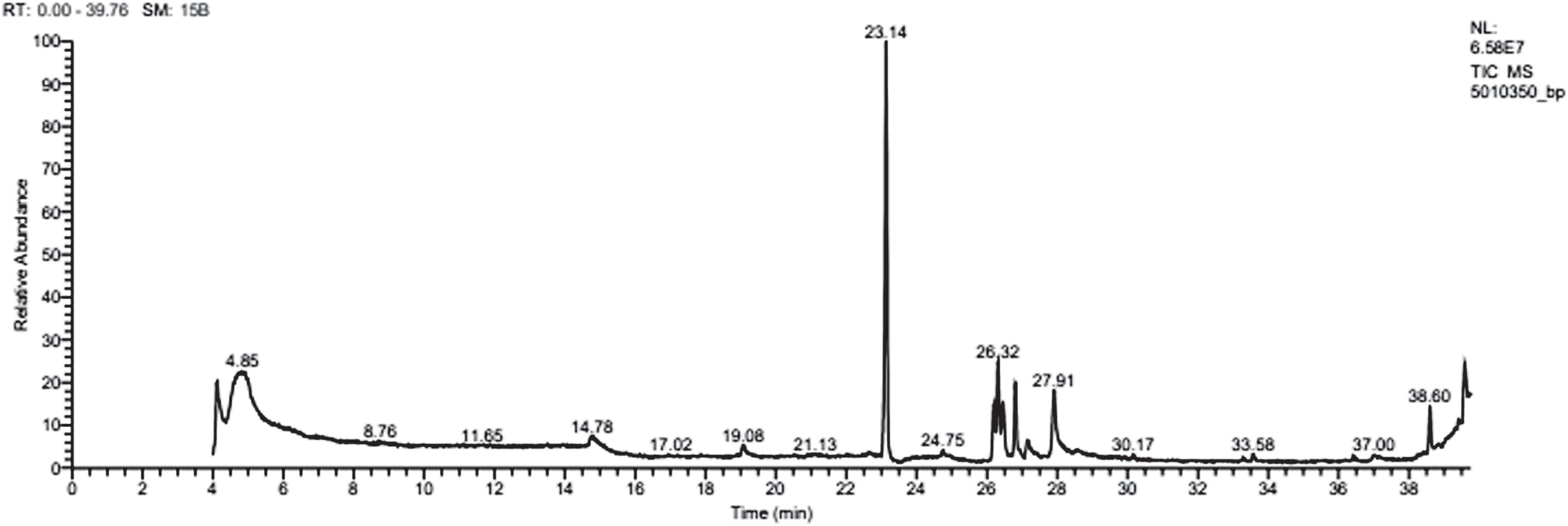
GC-MS of biosurfactant produced by Bacillus cereus AHMNAZ1.
In order to verify the lipopeptide structure, the sample was submitted to LC–ESI–MS/MS [42]. Tandem-MS used both ionization modes and various cations or anions typically yielded useful complementary information that is relevant for structural investigation [43,44, 43,44]. Mass spectrum analyses of the purified BS fractions of Bacillus cereus AHMNAZ1 are presented in Fig. 9. They demonstrated one well-resolved cluster of peaks matched compounds from the surfactin family and represented homologues of surfactins, which included the acyl chains C12, C13, C14, C15, and C16 [45,46,47,48,49,50,51,52, 45,46,47,48,49,50,51,52] Table 7 and Fig. 9 which corresponded to the molecular weights of 1008, 1022, and 1036, 1050, 1064, 1072, 1086 Da in positive ionization mode and 992, 1006, 1020, 1034, 1048, 1062 Da in (M-H) negative ionization mode.
Cyclic lipopeptides produced by Bacillus cereus AHMNAZ1 detected by ESI-MS
Cyclic lipopeptides produced by Bacillus cereus AHMNAZ1 detected by ESI-MS
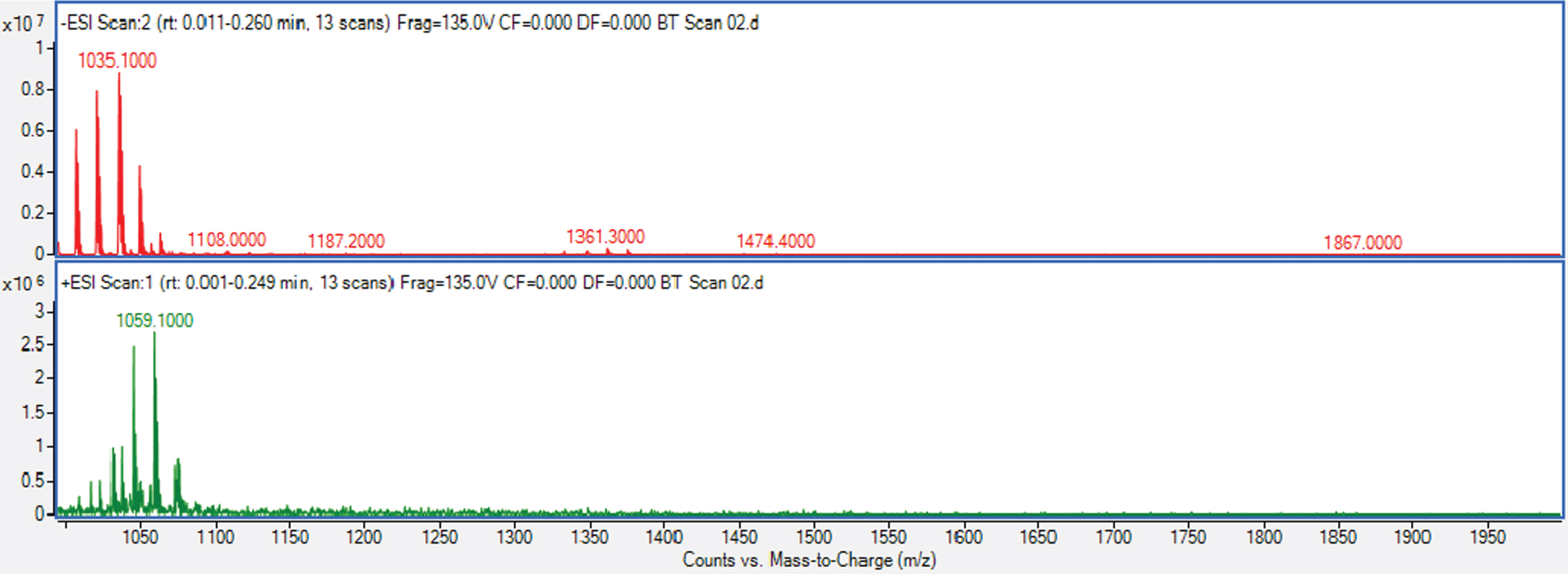
ESI–MS spectra of purified BS of Bacillus cereus.
By contrasting the actual masses and observed peaks with those from the literature, cyclic lipopeptides were identified [53, 54].
Critical micelle concentration (CMC) for surfactin produced from B. cereus AHMNAZ1
The extracted surfactin reduced the surface tension of water from 71.5±0 to 36±0.1 mN/m as shown in Fig. 10. Upon reaching the CMC (60 mg/L) the surface tension became stable, and no significant reductions were detected even at the highest concentrations tested.
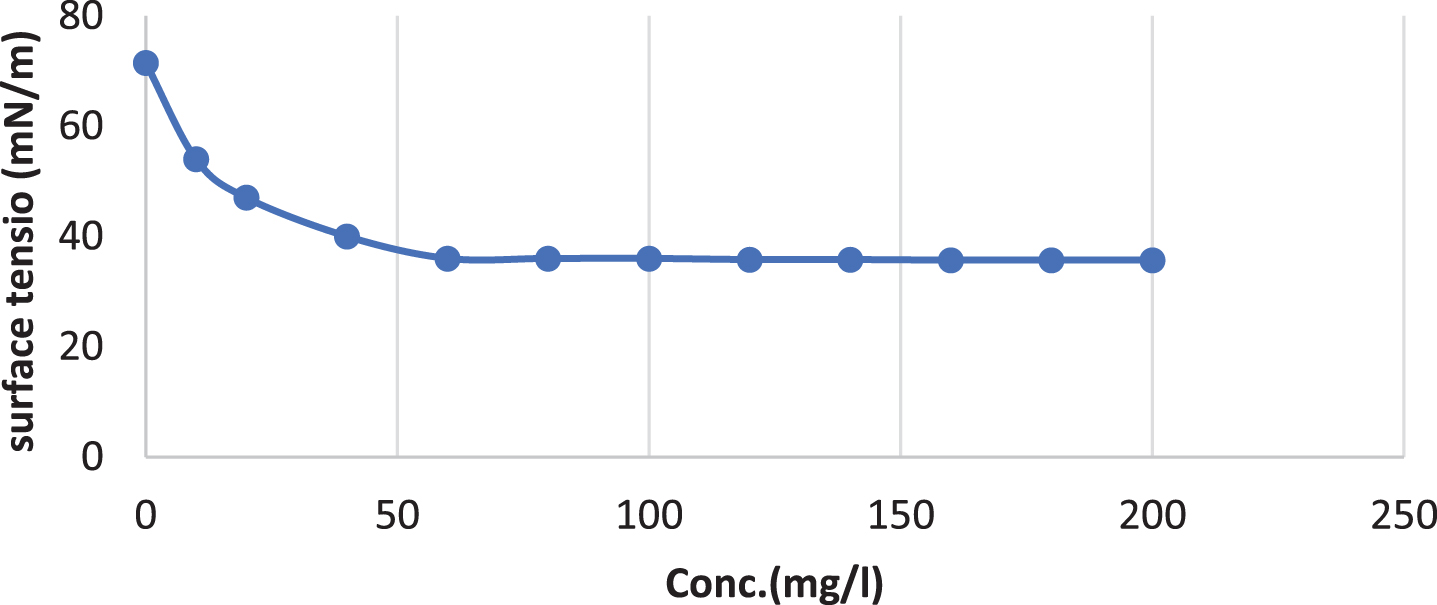
Critical Micelle concentration of surfactin produced by B. cereus.
The surfactin produced by Bacillus cereus AHMNAZ1 showed strong thermostability. Heating of the surfactin to 100°C caused no significant effect on its performance. The emulsification activity was quite stable at temperature 100°C Fig. 11. There was no considerable change in emulsification activity even at low temperature 4°C. It also retained 55–61.11% surface activity at pH 4.0–8.0 for the lipopeptide surfactin extracted from B. cereus AHMNAZ1 Fig. 12. The surface activity was reduced to 40% at pH 2.0 and it started precipitating. There is slightly decrease in activity at pH 10–12. The emulsification activities of the lipopeptide surfactin were stable above pH 7.0. Higher stability was observed under alkaline (pH 8) compared to acidic conditions. Under an acidic pH (pH 2.0) the lipopeptide surfactin showed much less activity; emulsification index decreased, due to precipitation of the lipopeptide surfactin. Whereas, it retained 60.86% surface activity even at 12% salt (NaCl), for B. cereus AHMNAZ1 Fig. 13. Thus the lipopeptide surfactin produced by B. cereus AHMNAZ1 was found to be stable up to 100°C, pH range of 4.0–12.0, and 12% salt, respectively.
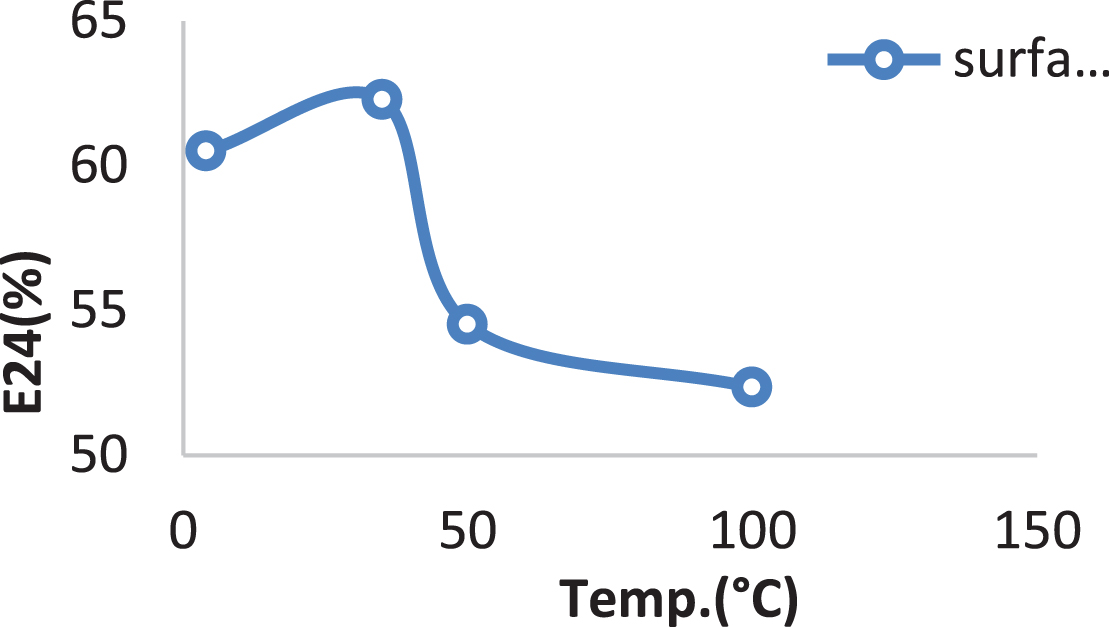
Influence of temperature on stability of the surfactin.
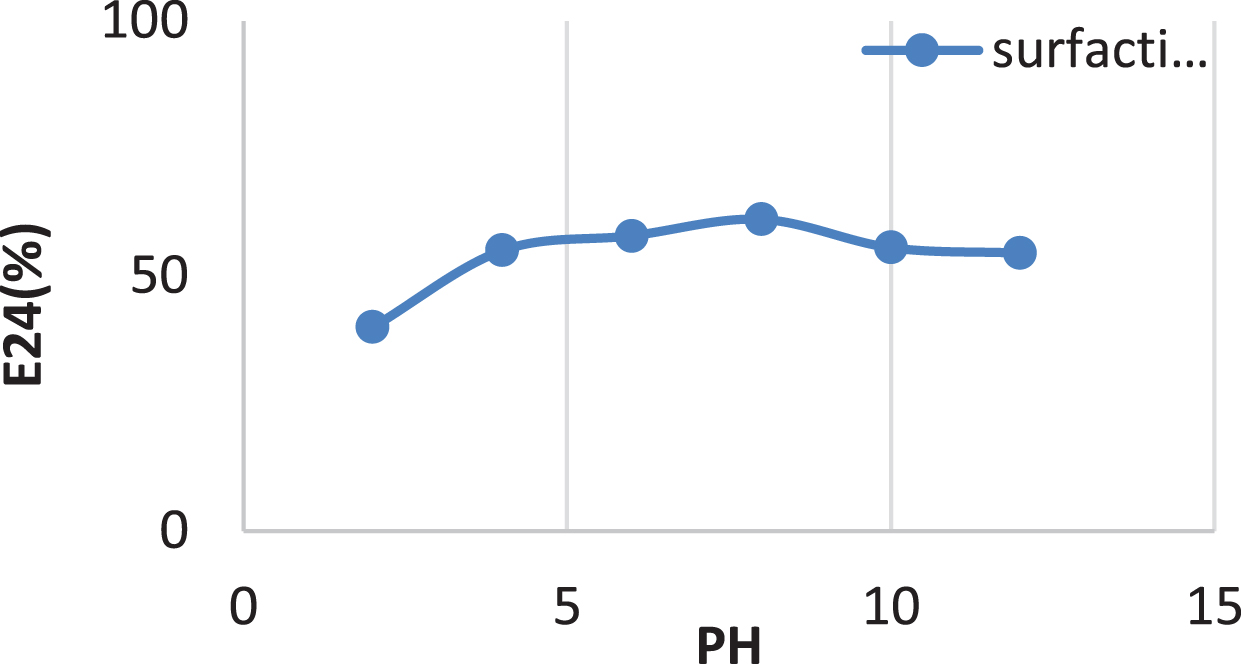
Influence of PH on stability of the surfactin.
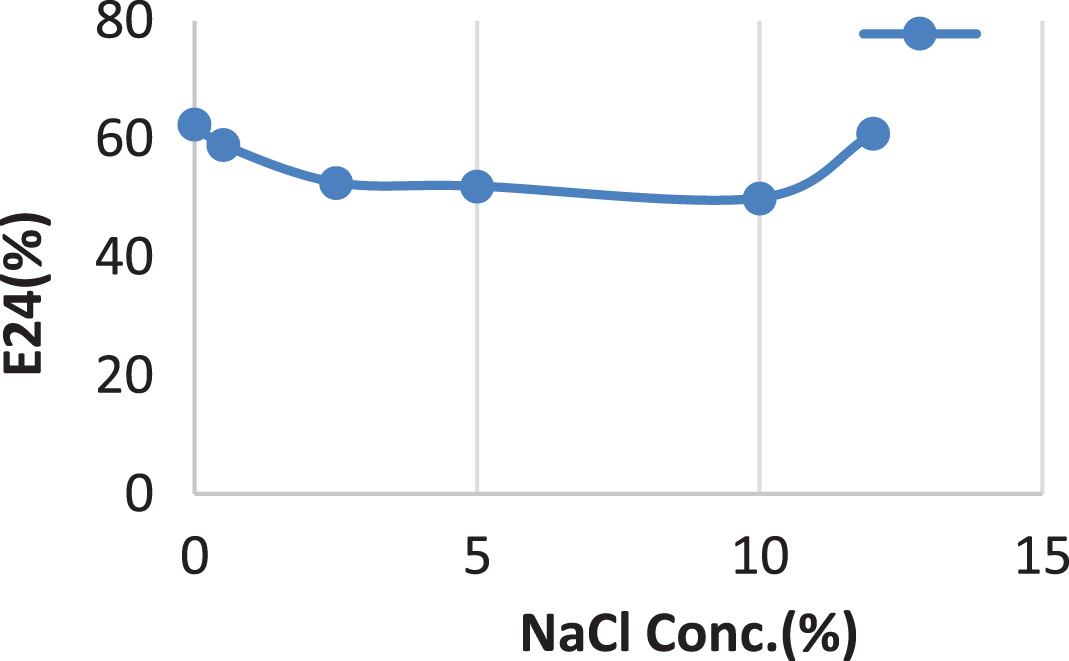
Influence of NaCl conc. (%) on stability of the surfactin.
Removal of crude oil from clothes
The extracted Surfactin in the present study was able to remove the majority of crude oil adsorbed on clothes Table 8. The maximum removal efficiency from cloth was for 4 days by the supernatant of B. cereus AHMNAZ1 was 72.5%; that is, 5.8-fold that of the control.
Crude oil removal of extracted surfactin
Crude oil removal of extracted surfactin
Concentration of 10 mg/ml of surfactin was tested against some pathogenic microorganisms Table 9, it affected fungi as Candida albicans RCMB 05031 with inhibition zone diameter of 7 mm. While inhibition zone diameter of Gram-positive bacteria as Staphylococcus aureus ATCC 25923 was 16 mm. While inhibition zone diameter of Gram-positive bacteria as Bacillus subtilis NRRL B-543 was 19 mm. Whereas inhibition zone diameter of Staphylococcus epidermidis RCMB 009 (2) for surfactin was 10 mm whereas for Enterococcus faecalis (ATCC 29212) for surfactin was 9 mm respectively. While inhibition zone diameter of Gram-negative bacteria as Escherichia coli ATCC 25922 for surfactin was 10 mm. While inhibition zone diameter of Gram-negative bacteria as Proteus vulgaris ATCC 13315 for surfactin was 7 mm, suggesting the broad spectrum of its antimicrobial activity. On the other hand, surfactin had no any inhibitory activity to Bacillus cereus RCMB 027 (1), Aspergillus fumigatus (RCMB 002008) and Methicillin-Resistant Staphylococcus aureus (MRSA 2658RCMB).
Biological activity of surfactin against some tested microorganisms
Biological activity of surfactin against some tested microorganisms
MIC of surfactin was different between tested microorganisms, it was 5000μg/ml for each of the Candida albicans RCMB 05031, Staphylococcus epidermidis RCMB 009, Enterococcus faecalis (ATCC 29212) and Proteus vulgaris ATCC 13315, also MIC was 312.5μg/ml for Staphylococcus aureus ATCC 25923, 625μg/ml for Bacillus subtilis NRRL B-543 and it was 2500μg/ml for Escherichia coli ATCC 25922. On the other hand, Bacillus cereus RCMB 027 (1), Aspergillus fumigatus (RCMB 002008) and Methicillin-Resistant Staphylococcus aureus (MRSA 2658RCMB) were not affected at all with surfactin Table 10.
Minimum inhibitory concentrations (MIC) (μg/ml) of surfactin against tested
microorganisms
Minimum inhibitory concentrations (MIC) (μg/ml) of surfactin against tested microorganisms
The cytotoxic activity of Surfactin against normal Mammalian cells African Green Monkey Kidney (VERO) cells was detected under these experimental conditions with 50% cell cytotoxic concentration (CC50)=61.01±1.78μg/ml, as Fig. 14.
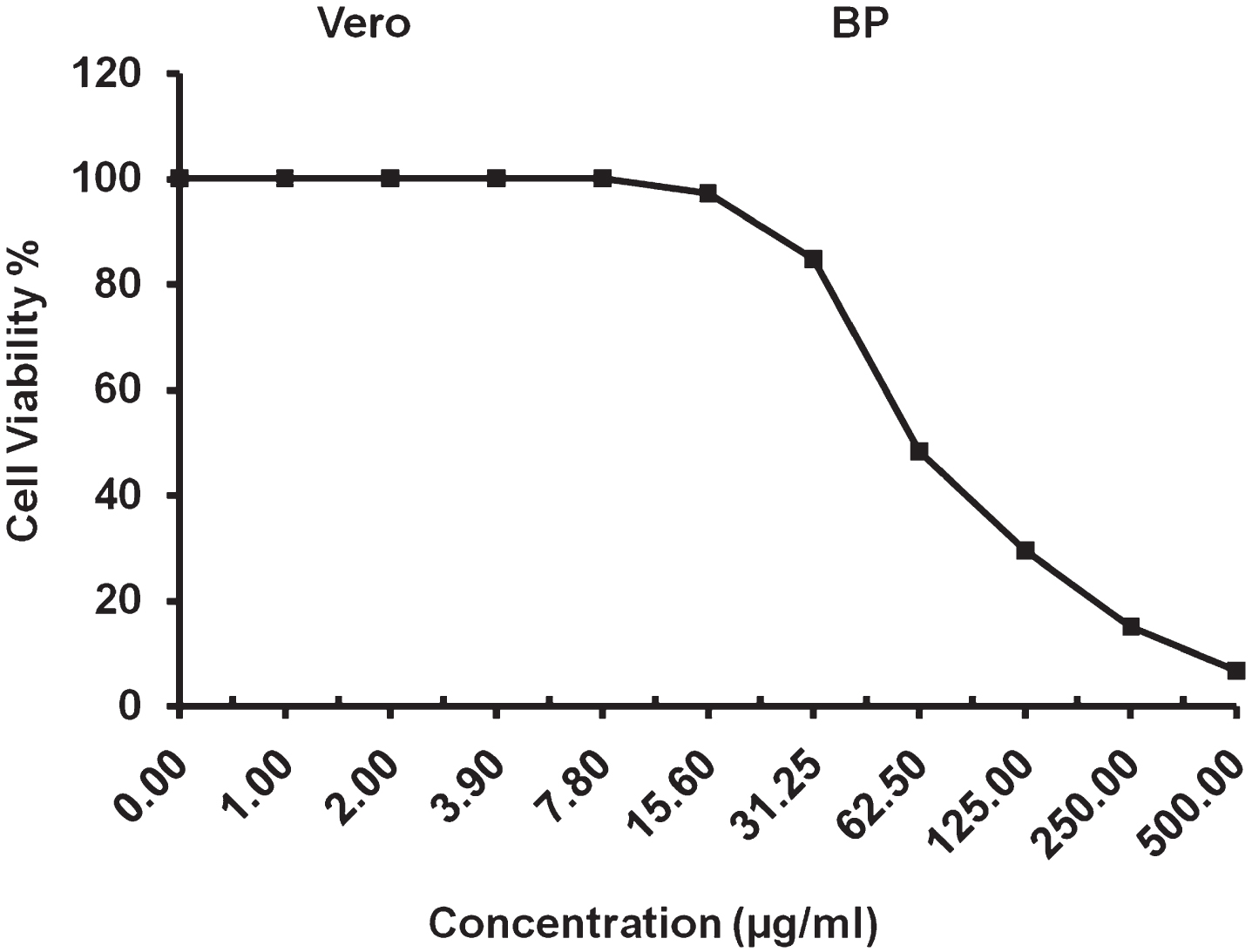
CC50 analysis of surfactin against (VERO) cell line.
The present study reveals that IC50 value of 20.77±1.05μg/ml for surfactin against colon carcinoma cells and a maximum inhibitory suppression was observed up to 98.31% for surfactin at a concentration of 500μg/ml as shown in Fig. 15. The results reveal that the surfactin showed significant anti-proliferation activity against colon carcinoma cell line.
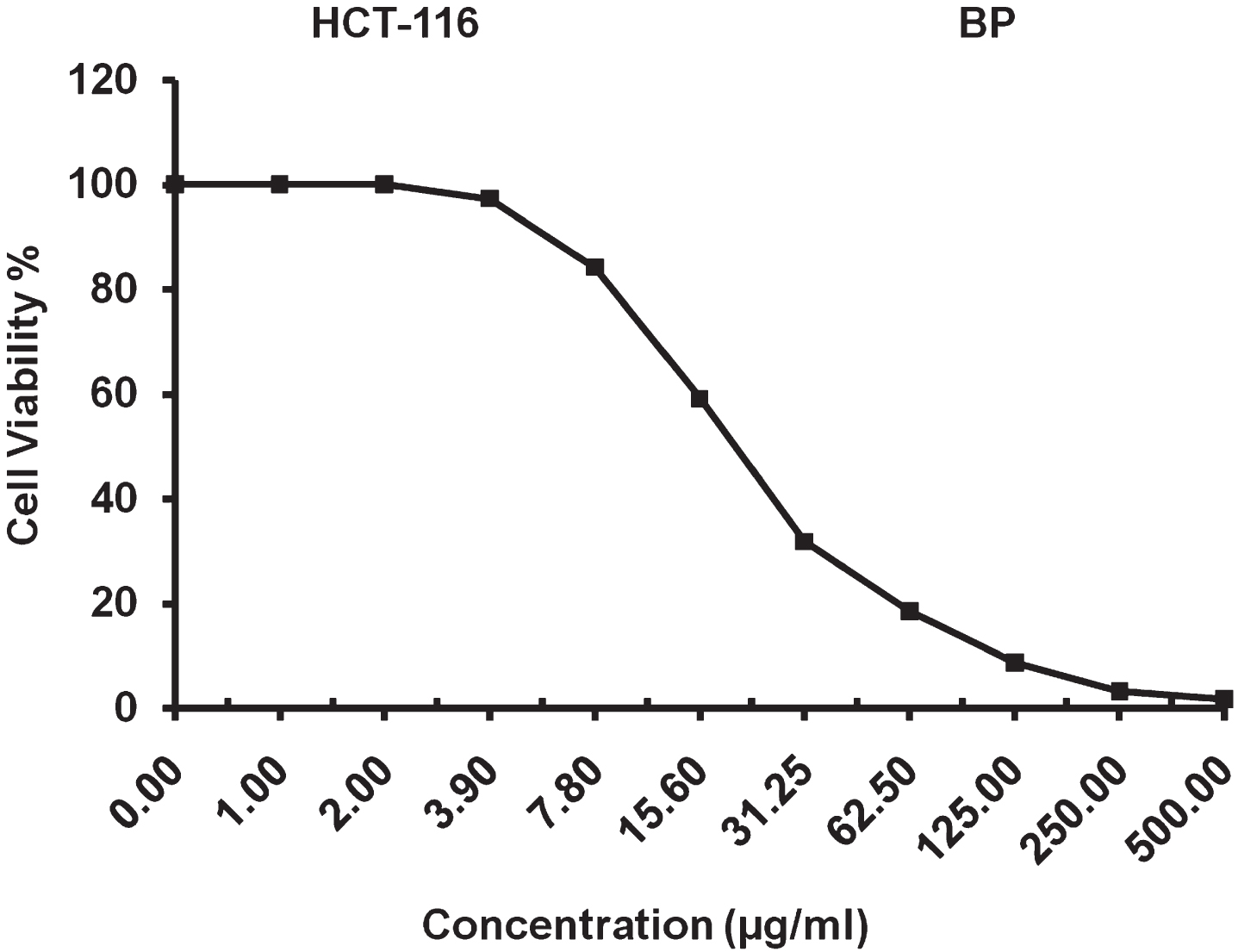
IC50 analysis surfactin against colon carcinoma (HCT-116) cell line.
The present study revealed that IC50 value of 30.87±1.53μg/ml for surfactin against breast carcinoma cells and a maximum inhibitory suppression was observed up to 97.63% for surfactin at a concentration of 500μg/ml as shown in Fig. 16. The results reveal that the biosurfactants surfactin showed significant anti-proliferation activity against breast carcinoma cell line.
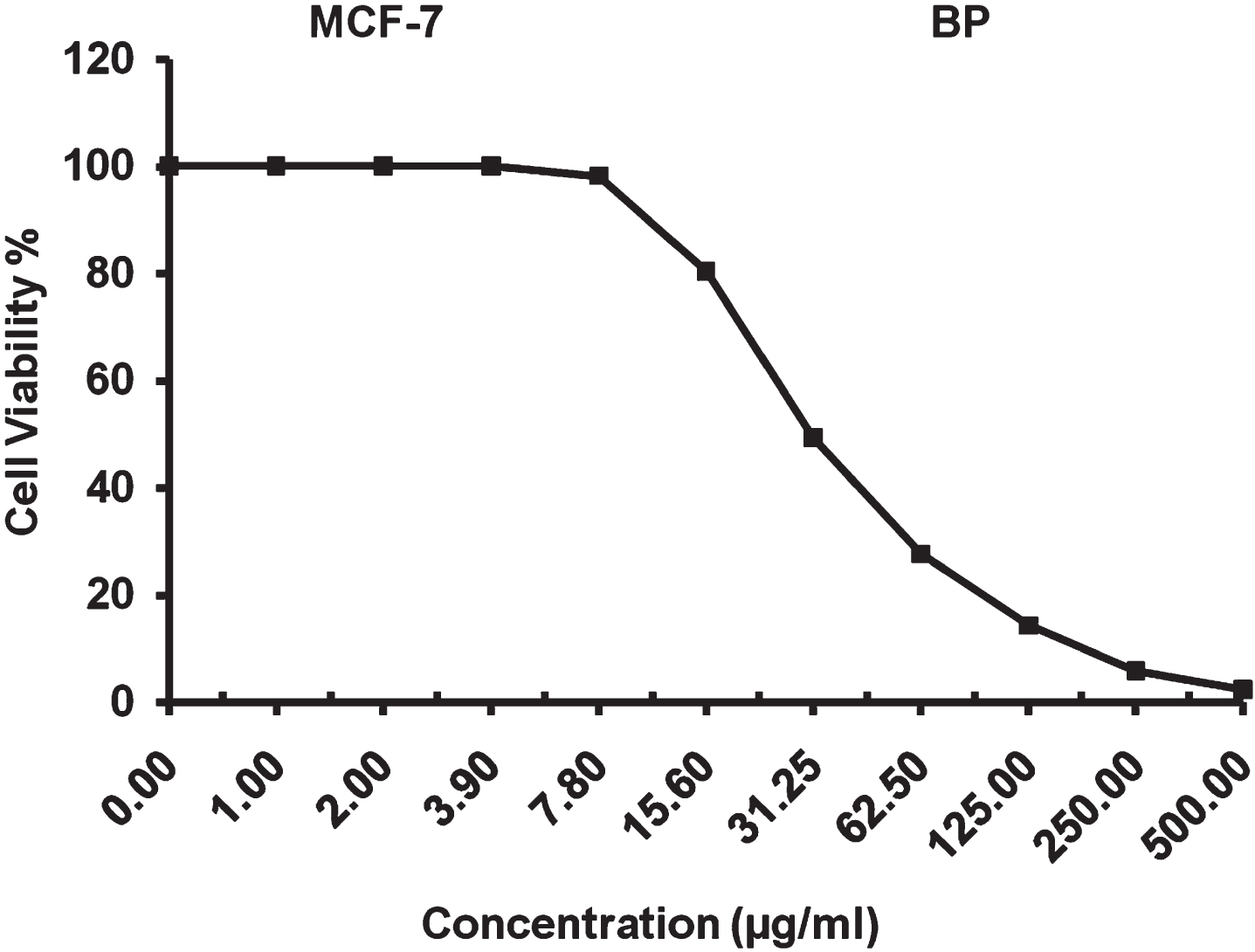
IC50 analysis of surfactin against breast carcinoma (MCV-7) cell line.
In this study, 63 microorganisms were isolated from different wastes and screened them for biosurfactant production Bhuvaneswari and Sivagurunathan [55] isolated ten bacteria from the marine sediments of the Cuddalore coastal region in India and streaked in Bushnell Haas agar medium supplemented with 1% crude oil over a period of time, only one bacterial isolate that was kept as a pure culture produced biosurfactant successfully. As well as Chittepu [56] isolated 30 bacteria from groundnut oil cake dumping locations in Hyderabad, India, the best ten isolates were chosen for biosurfactant production. Also, Phulpoto et al. [41] were isolated thirty-two different bacterial colonies from various Yanqi lake water layers, located in the southeast region of Huairou District, Beijing, but only one isolate produced the best biosurfactant.
BP bacterial isolate was selected as the most potent microbial isolate which exhibited the highest biosurfactant production in modified BHM with oil spreading zone 11.26, emulsification index 54.5±0.1, emulsification activity 1.18±0.035, positive drop collapse and blood hemolysis as well as 37.53 reduction of surface tension and has ability for solubilization of crystalline anthracene and penetration activity compared with control used. The BP isolate was identified by 16 S rRNA as Bacillus cereus AHMNAZ1 (with accession number OP714421.1).
Bacillus subtilis (BS3) and Pseudomonas aeruginosa (PS2) were identified as verified biosurfactant producers by Adejumo et al. [57] because they achieved oil displacement of 1.8 and 1.9 cm, respectively. The emulsification indexes for the bacterial isolates of Pseudomonas aeruginosa (PS2), Bacillus subtilis (BS1), and Bacillus subtilis (BS3) are 50.05%, 60.68%, and 55.18%, respectively. In the present investigation, BP was positive for drop collapse assay. Similarly, the bacterial isolate Bacillus nealsonii S2MT is able to collapse the oil drop [41].
Also, Olteanu et al. [58] showed that, BS from bacterial isolates OS17 and B2 had emulsification stability values (E24) of 51.25, 48.75% against kerosene [58]. Surprisingly, the biosurfactant from isolate BP in the current investigation had better emulsification activity (EA) than the biosurfactant from B. subtilis B6 and bacterial isolate OS17, which had EA at 540 nm of 0.30 and 0.441 against kerosene [59, 58].
The optimum conditions for maximum BS production by Bacillus cereus AHMNAZ1 were conducted from the produced Box-Behnken model as follows: molasse, 33.86 ml/l; crude oil, 0.11 ml/l; ammonium nitrate, 2.35 g/l; calcium chloride, 0.35 g/l; copper sulphate, 0.48 g/l; incubation period, 10.98 days and pH 8 to obtain maximum emulsification activity of 1.92. The data showed a significant increase in emulsification activity (1.92) compared with that obtained at the original conditions (1.18).
According to Alyousif et al. [60], olive oil, sunflower oil, mannitol, and glucose were the best carbon sources for P. aeruginosa A2 isolate biosurfactant synthesis, with emulsification activity values of 1.678, 1.535, 1.515, and 1.440, respectively. Lactose was shown to be the least effective carbon source for the formation of biosurfactant, with zero values for emulsification activity 0.054. The best carbon source concentration was found at 2%, with an emulsification activity of 1.648. The best nitrogen source for the formation of biosurfactant was urea, with an emulsification activity of 1.744, followed by NH4NO3, with a value of 1.638. The best nitrogen concentration had an emulsification activity of 1.834 and was 0.2% urea.
However, the production of biosurfactants from Bacillus licheniformis TR7 and Bacillus subtilis SA9, which were isolated from mangrove sediment in the southern part of Thailand, was carried out using only molasses as a whole medium for growth and production. Under optimal conditions, the yields of TR7 and SA9 BS have been shown to be 3.30 and 3.78 g/l, respectively [61]. S. marcescens produced the greatest amount of biosurfactant at pH 8 (0.85 g/L output and 31.9 mN/m surface tension) [62]. Additionally, it was reported that S. marcescens strain DT-IP produced biosurfactant at pH 8.[63]. Moreover, Bacillus mycoides produced the highest amount of biosurfactant and reduced surface tension when the pH level was between 7–8 [64]. Utilizing S. marcescens DT-IP, the yield of biosurfactant increased over the course of cultivation, beginning in the early stationary phase and peaking in 8 days [65]. It was discovered that S. marcescens NSK-1 produced large levels of biosurfactants and low surface tension when grown on glycerol as the only carbon source [66]. An earlier investigation revealed that using glucose at 3% to raise biosurfactant output to 600 mg/L with a surface tension of 27 mN/m resulted in the maximum biosurfactant production [67].
Additionally, the beef extract 1% (w/v) as a complex organic nitrogen source was discovered to be the greatest component for the formation of LP by B. subtilis KLP2015 [68].
Bacillus licheniformis R2 has been shown to produce surfactin at its highest levels on a mineral medium containing NH4NO3 (1 g/l) as a nitrogen source [69]. The maximum amount of surfactin (439.0 mg/l) that B. subtilis ATCC 21332 could make was in a medium with glucose (10.0 g/l) and ammonium nitrate (4.0 g/l) [70]. According to previous studies, the highest lipopeptides production (530 mg/l) in the fermentation broth containing B. subtilis KLP2015 was also attained after 72 hours of shaking incubation at 30°C (545 mg/l) [68]. Medeot et al. [71] succeeded in getting Bacillus amyloliquefaciens MEP218 to produce the highest biosurfactant concentration (1.7 mg/mL) upon using glucose and NH4NO3. Similar to this, according to Fernandes et al. [72], Bacillus subtilis RI4914 produced the maximum concentration of biosurfactant (0. 2 g/L) when NH4NO3 and sucrose were mixed. Additionally, Abdel-Mawgoud et al. [73] examined surfactin production by Bacillus subtilis BS5 utilizing various carbon and nitrogen sources and came to the conclusion that NaNO3 and NH4NO3 presence resulted in the maximum biosurfactant production. According to Silva et al. [74], the formation of biosurfactants clearly depends on the combination of carbon and nitrogen substrates. Ammonium salt was found to be a preferred nitrogen source for Arthrobacter paraffinus when producing biosurfactants, according to Fagade et al. [75]. In media containing olive oil and NH4NO3 as carbon and nitrogen sources, respectively, Pseudomonas fluorescens’ greatest emulsifying activity and lowest surface tension were achieved [76]. Trace ions are involved in the production of biosurfactants and act as coenzymes for important enzymes in the production process. The entire biosurfactant production system will be destroyed if a particular ion or ion(s) are missing, or if their proportions are inappropriate [77].
Lactose, yeast extract, and Na2HPO4 were shown to be the best sources of carbon, nitrogen, and metal ions, respectively, for the synthesis of biosurfactant from marine Vibrio sp. strain 3B-2, according to Hu et al. [77].
According to Dominguez [78], calcium served as a common mediator in processes that carry signals from the cell surface into the intracellular of microorganisms Calcium was typically supplied in the production of biosurfactants as chloride or hydrated chloride salts, typically in concentrations of 0.1 g/L for biosurfactant production from P. aeruginosa and less than 0.02 g/L for glycolipid production from B. megatarium, demonstrated that very small quantities of Ca+2 ions were still needed in the production medium. Both potassium and calcium ions are essential for maintaining the proper level of osmotic pressure and managing the membrane potential of the cell, which could prevent the lysis of the cell in the medium [80]. According to Umar et al. [81], Bacillus subtilis SNW3 produced 1.17 g/L of crude lipopeptide when grown on optimum conditions. Adejumo et al. [57] found that mutant strains of BS3 and PS2 produced dry weight biosurfactants of 1112 mg/L and 992 mg/L, respectively, compared to wild strains of BS3 and PS2, which only produced 905 mg/L and 878 mg/L, respectively, of these secondary metabolites. Other investigations had used a mineral medium supplemented with date molasses and NH4NO3 as carbon and nitrogen sources to produce crude biosurfactant yields of 0.30–2.3 g L–1 [82, 83]. According to Sousa et al. [84], B. subtilis LAMI005 produced 0.44 g L1 of biosurfactant using a mineral medium containing raw glycerol and (NH4)2SO4. In the current work, preliminary characterization of our extract using thin layer chromatography revealed red color spot with an RF value of 0.36 that is related to the surfactin family. Priya and Usharani [85], Phulpoto et al. [41], and Ramyabharathi et al. [86] reported the same results during the production of surfactin biosurfactant by Bacillus subtilis, Bacillus nealsonii strain S2MT, and Bacillus subtilis Bbv57, respectively which was confirmed on TLC with an Rf value of 0.3 for the surfactin family when compared with the standard.
The FTIR spectrum indicated the presence of peptide moiety and aliphatic groups, a distinguishing characteristic of the lipopeptide nature of the biosurfactant produced by Bacillus cereus. The current study was in agreement with previous FTIR investigations according to Das et al. [87], Varadavenkatesan and Murty [26], John et al. [88], and Umar et al. [81] for lipopeptide biosurfactant produced from Bacillus subtilis SNW3, Bacillus siamensis, Lysinibacillus fusiformis MK559526, a marine Bacillus circulans, respectively.
Hexadecanoic acid, methyl ester was the predominant compound in the biosurfactant. Due to variations in the fatty acid component’s nature, such as chain length (C6–C18), the frequency of hydroxyl groups and/or iso- or anteiso-methyl branches, and the type, number, and configuration of the amino acids in the peptide chain, there was a significant structural diversity in the lipopeptides produced by Bacillus species [89]. Qiao and Shao [90] also stated the presence of a mixture of hexadecanoic, pentadecanoic, dodecanoic, tetradecanoic, octadecanoic, (9)-octadecenoic and (9,12)-octadecadienoic β-hydroxy fatty acids in the lipoprotein biosurfactant derived from Alcanivorax dieselolei B-5.
In the acid hydrolyzed solvent fraction for the fatty acid composition of the biosurfactant “Aneurinifactin” isolated from a marine Aneurinibacillus aneurinilyticus SBP-11, Balan et al. [91] found the presence of a single major peak at the retention time of 17.56, which was identified as stearic acid methyl ester with a molecular mass (m/z) of 298.3 based on the NIST mass spectrum in the GC–MS analysis. According to Balan et al. [92], palmitic acid made up the majority of the acid hydrolyzed solvent fraction for the biosurfactant “Pontifactin,” a linear lipopeptide biosurfactant isolated from the marine bacterium Pontibacter korlensis strain SBK-47. According to Peng et al. [93], the hydrophobic moiety in Rhodofactin, a linear lipopeptide biosurfactant from Rhodococcus sp. strain TW53, is made up of six fatty acids with chain lengths ranging from C14–C19.
One well-resolved cluster of peaks identified in mass spectral analysis of the pure biosurfactant fractions of Bacillus cereus related to compounds from the surfactin family. By comparing the mass with the mass numbers given for the lipopeptide complexes from other Bacillus strains, each group of peaks may be ascribed to different lipopeptide isoforms. Each isoform group may be a member of the same family and likely share the same amino acid sequence, while the fatty acid chain lengths might vary [94]. The current investigation was consistent with earlier findings [45–52]. Past studies have demonstrated that the growing medium affects the fatty acid’s length and composition, which might lead to a higher specific surfactant activity [95, 96].
The Critical micelle concentration)CMC(of purified biosurfactant from Bacillus cereus AHMNAZ1 was 60 mg/L. Hanano et al. [97] discovered that the biosurfactant produced by B. safensis PHA3 has a CMC of 3.5 mg/mL. According to Mnif et al. [98], lipopeptide biosurfactant made from Bacillus subtilis strain ZNI5 had a CMC value of 350 mg/L. According to Wan et al. [99], Bacillus flexus S1 produced lipopeptides with a CMC of 46.04 mg L–1. According to Umar et al. [81], the lipopeptide generated by Bacillus subtilis SNW3 had a CMC value of 0.5 mg/mL and lowered water surface tension from 72 to 36 mN/m. Bacillus nealsonii strain S2MT produced a biosurfactant product with a CMC value of 40 mg/L with the SFT reduction from 81.2 mN/m to 34.5±0.56 mN/m, and there were no subsequent significant changes in the SFT measurement [41]. According to Datta et al. [29], CMC for Bacillus subtilis MG495086 was 40 mg/L. Lower CMC values are crucial for industrial applications because a smaller amount of the product is needed to fulfill the application’s aim [42]. Biosurfactants are “green chemicals” utilized to improve oil recovery. Biosurfactants must be stable across a range of temperatures, pH levels, and salinities in order to be used for ex situ MEOR [30, 100]. The biosurfactant produced by Bacillus cereus in the present study was shown to be thermostable. Bacillus methylotrophicus produces biosurfactant that is thermostable and maintain its activity even when exposed to high temperatures (120°C for 15 min) [101]. Numerous authors have discussed the same results after heat treatment (102,30,103,83). Even at a low temperature of 4°C, the emulsification activity did not significantly change. This quality makes the Bacillus cereus lipopeptide appropriate for the ice cream and cosmetic sectors [104].
In comparison to acidic settings, higher stability was demonstrated under alkaline (pH 8) conditions. These findings suggest that raising pH has a beneficial impact on the stability and surface activity of the biosurfactants [32]. According to some results, biosurfactants produced from some Bacillus strains were stable at various pH levels, but primarily in alkaline environments [83, 105]. Overall, relatively high salinity stability was found compared with other studies that used B. subtilis, Nocardiopsis sp. B4 and Serratia marscecens [83, 106]. The biosurfactants produced by B. atrophaeus 5-2a retained their surface activity and were stable under a variety of environmental conditions [32]. In contrast, Bacillus cereus in this study maintained 60.86% of its surface activity even at 12% salt (NaCl). Thus the biosurfactant derived from Bacillus cereus was stable up to 100°C, had a PH range of 4.0–12.0, and 12% salt. It is comparable to other results on biosurfactant stability produced by various microorganisms [83, 107–110]. The observed stability of the biosurfactant evaluated in this study for Bacillus cereus over the pH range of 4–12 and salinity concentrations of 0–12% indicates that it would be appropriate for oil recovery in the majority of reservoirs. Oil reservoirs are harsh environments with the potential for high salinity and a wide range of pH values. These findings demonstrated that Bacillus cereus biosurfactant is a strong candidate for use in MEOR.
One of the most promising strategies for recovering a significant amount of residual oil was the application of biosurfactants for MEOR, which has recently drawn increasing attention [111]. Bacillus cereus supernatant in this study had a maximum removal efficiency from cloth of 72.5% over a period of 4 days, which is 5.8 times better than the control. After 4 days of static incubation, the supernatant of B. atrophaeus 5-2a from BU medium had a 93.1 removal efficiency for crude oil from the filter paper. [32]. Pornsunthorntawee et al. [21] stated that 61.6% of crude residual oil was eliminated using a cell-free broth containing a biosurfactant produced from B. subtilis PT2. Three B. subtilis strains were effective at recovering oil from sand pores, according to Pereira et al. [112], who extracted crude oil from contaminated sands at rates between 19 and 22%. It is encouraging for MEOR that the fermentation broth used in the current study, which contains biosurfactant from Bacillus cereus, was so effective at removing crude oil. Similar to this work, a different study using Bacillus simplex SBN 19 lipopeptide biosurfactant demonstrated 87.4 1.7% crude oil recovery using 100 mg L–1 of pure biosurfactant [113]. These findings highlighted the importance of surfactin as a possible agent for recovering crude oil in the petroleum industry and as a bio-stimulation agent for the bioremediation of oil-polluted soils. The extracted lipopeptide used for antibacterial activity showed that it was more effective against E. coli (7 mm), K. pneumoniae (10 mm), and S. aureus (6 mm), respectively, at 5μg/mL concentration [56].A lipopeptide worked by attaching to the bacterial surface bilayer and changing the local lipid organizational linkage on negatively charged fatty acids, which causes the lipid bilayer to be restructured and stops cellular functions [114, 115]. According to past reports, surfactin had only been documented to exhibit antibacterial activity but not the antifungal activity [54, 116]. When diesel was employed as the biosurfactant source, the inhibition zone diameters for Staphylococcus aureus, Escherichia coli, Klebsiella sp., Salmonella typhi, and vibrio sp. were 7, 12, 18, 12, and 20, respectively [117]. In aqueous solutions, biosurfactants cluster and then show lytic action against a wide variety of bacteria, possibly via creating pores and rupturing membranes [118, 119]. Bacillus is generally effective against a variety of bacteria, and these compounds are also beneficial to the pharmaceutical, food, chemical, and agricultural industries [54, 120]. Additionally, biosurfactants were crucial tools in the petroleum sector to help in the biodegradation of oil spills in contaminated areas [119] and in EOR (enhanced oil recovery) or MEOR (Microbial EOR), which is a tertiary oil recovery strategy that increases petroleum yields by lowering the surface and interfacial tensions of the oil to enable oil flow [121]. Ghribi et al. [122] have reported that diameter of inhibition zone for 0.5 g/l biosurfactant obtained from B subtilis SPB1 were 19, 12, 8, 14, 3, 10 and 4 mm for Staphylococcus aureus, Escherichia coli, Salmonella typhimirium, Pseudomonas aeruginosa, Enterococcus faecalis, Aspergillus oryzae, Aspergillus niger and Rhizopus oryzae, respectively. Rhamnolipid extracted from Pseudomonas aeruginosa strain PAO1-B13 showed inhibition zone diameters for Gram positive bacteria like Staphylococcus aureus, Bacillus subtilis, and Enterococcus faecalis of 20.2, 23.4, and 16.3 mm, respectively, while Gram negative bacteria like Escherichia coli, Salmonella typhi, and Klebsiella pneumonia showed inhibition zone diameters of 18.2, 22.3, and 20.3 mm, respectively, and did not show any inhibitory effect on. Candida albicans (RCMB 05031) [123]. The biosurfactant isolated in the present investigation displayed a broad spectrum of action, including antimicrobial efficacy against microorganisms with multidrug-resistance profiles. In comparison to Gram-negative bacilli, the compound demonstrated greater effectiveness against Gram-positive cocci. Its efficacy against Enterococcus faecalis was crucial. These results are very interesting, as discussed by Furtado et al. [124] as these microorganisms exhibit limited sensitivity to aminoglycosides and penicillin G and natural resistance to aztreonam, co-trimoxazole, cephalosporins, chloramphenicol, and clindamycin [122, 124]. The compound had significant antimicrobial action against Staphylococcus aureus, a bacteria known to be resistant to at least two -lactams. It was also evident that action against Gram-negative bacteria was less than that against Gram-positive bacteria. The lipopeptide N1, produced from B. subtilis C1, was also found to be effective against several microorganisms, particularly S. aureus and Mycobacterium sp., according to Singh and Cameotra [125]. Balan et al. [91] demonstrated lowest minimum inhibitory concentration (MIC) of Aneurinifactin lipopeptide biosurfactant against six bacterial strains like Klebsiella pneumoniae (4 g mL–1), Escherichia coli (8 g mL–1), Staphylococcus aureus (8 g mL–1), Pseudomonas aeruginosa (16 g mL–1), Bacillus subtilis (16 g mL–1), and Vibrio cholerae (16 g mL–1). The effective anchoring of the biosurfactant on the bacterial cell surface, which resulted in pore formation in the cell membrane, uncoupling of oxidative phosphorylation, hyperactivation of the electron transport chain, and rapid depletion of reducing equivalents in superoxide production, is the proposed mechanism of action of the lipopeptide biosurfactants. The redox system’s iron-sulfur clusters are damaged by the superoxide radicals that generate, making them vulnerable to the Fenton reaction. Through the Fenton reaction, the ferrous ions are oxidized, producing hydroxyl radicals as a byproduct. These hydroxyl radicals promote lipid peroxidation in membranes and increased membrane permeability, which leads to protein leakage and eventual cell death [119, 127]. Furthermore, Brevifactin showed promising antimicrobial activity against Candida albicans and Klebsiella pneumonia [128] while Pontifactin demonstrated excellent antimicrobial activity against Streptococcus mutans, Micrococcus luteus, Salmonella typhi, and Klebsiella oxytoca [92]. However, against any of the studied bacterial strains, proline lipid exhibited no antibacterial action [90]. These findings suggest that surfactin had considerable potential as an antimicrobial in a variety of biomedical applications. According to Elkhouly et al. [123], the CC50 (cell cytotoxic concentration) value of rhamnolipid against the VERO cell line was 44.1μg/ml. According to Kim et al. [129], surfactin was important in preventing the growth of human colon cancer cells. According to Elkhouly et al. [123], the CC50 value of rhamnolipid against the VERO cell line was 44.1 g/ml. According to Kim et al. [2007], surfactin was essential in preventing the growth of human colon cancer cells. Additionally, lipopeptides had the ability to increase the production of reactive oxygen species (ROS) in K562 cells, which inhibits Bcl-2 expression and promotes the release of cytochrome-C (Cyto-C), leading to the induction of death. [130]. With an IC50 of 9.65μM after 24 hours, surfactin isolated from B. subtilis CSY 191 was reported to inhibit the growth of human MCV-7 breast cancer cells (Lee et al., 2012). T47D and MDAMB-231’s cell survival at 48 and 72 hours, respectively, could be significantly reduced by a concentration of 193μM surfactin purified from B. subtilis 573 [131]. Additionally, purified surfactin-like lipopeptides from B. subtilis Hs0121 showed strong cytotoxicity on human Bcap-37 breast cancer cells. According to these findings, after 24 hours, C15 surfactin-like lipopeptide showed an IC50 of 29±2.4μM [132]. MCF-7 cell proliferation was demonstrated to be inhibited by surfactin isolated from B. subtilis TK-1 strains with IC50 values of 86.2, 27.3, and 14.8μM at 24, 48, and 72 h, respectively. Furthermore, it was revealed by Cao et al. [133] that surfactin-induced apoptosis was mediated by the mitochondrial/caspase pathway via reactive oxygen species/c-Jun N-terminal kinase (ROS/JNK).
Conclusion
The study found a cheaper way of creating biosurfactants from agro-industrial wastes, delivering a twofold benefit of lowering environmental pollution and manufacturing useful biotechnological products (biosurfactants) with high activity and thermostability. Moreover, it can be used for the bioremediation of oil-polluted soils and in medical fields.
Conflict of interest
There are no conflicts to declare.
