Abstract
In leather industries and tanneries, large amount of wastes has been disposed; which polluting water, soil, and atmosphere and causing serious human health problems. In particular, chemical dehairing process of leather industries produces fair amount of toxic wastes. It is, thus, urgently needed to use alternative processes free from pollution. As more than 90% of keratin is contained in feather, it is desirable to develop bioremediation process using keratinolytic microorganisms. In the present investigation, therefore, we first identified
Introduction
Although leather industries and tanneries involve highly polluting processes, they offer diverse amenities for manufacturing capacity as well as export potential, 1 which require several steps including dehairing, deliming, bating, degreasing, and finally pickling in a cascade manner for converting raw hide into long-lasting finished materials which are suitable for a variety of uses such as shoes, skirts, trousers, hats, jackets, belts, as well as book binding, furniture covering, and so on. Chemical dehairing process requires more than 75 chemicals including lime (CaO) and sulfide (Na2S/NaSH) to turn raw leathers into semi-fabricated leather, 2 which gives around 90% of total pollution in leather industries and also produces noxious gases and solid wastes. 3 Previous report estimated that nearly 850 kg out of 1000 solid wastes is generated during leather processing, and approximately 40 to 45 L of water is used per kilogram of raw hide/skin processing to get finished leathers. 4 This causes many difficulties including availability of good water, treatment of effluent, and sulfide emissions during dehairing. Along with solid wastes, biochemical oxygen demand (BOD) and chemical oxygen demand (COD) as well as total dissolved solid give almost 70% of pollution from pre-tanning, tanning, and re-tanning processes. 5 Thus, exposure of waste from tanneries not only pollutes water, soil, and air of respected area but also causes serious health difficulties including asthma, hepatic, dermatitis, various malignancies, and neurological disorders to the tannery workers as well as the people living nearby areas.
To overcome the above problems caused by chemicals and wastes in leather industry, keratinase shows promising nature due to significant advantages like reduced processing time, biodegradable action, better product quality, low energy input, lower cost, nontoxic and eco-friendly characteristics. 6 Extracellular keratinase, a versatile and often used enzyme, can be an alternative means of chemical dehairing for minimizing the toxic effects of chemicals in dehairing process of leather industries. Keratinase (EC 3.4.21/24/99.11) is classified as protease 7 and can be broadly used in several industries such as leather and detergent, textiles, medicine, cosmetics, fertilizers, waste bioconversion, as well as drug delivery 8 by hydrolyzing keratins.
Keratin is an insoluble (in nature) and structural protein in epithelial cells of vertebrates and represents the major constituents of skin and its attachments such as hair, feathers, nail, wool, hooves, beaks, and stratum corneum,
9
which have high mechanical stability and resistance to proteolytic degradation.
10
Feather is constituted of 91% of keratin protein, 8% water, and 1% of lipid,
11
and it is generated in large amounts as waste by-product in poultry processing industries, propagating millions of tons worldwide. As poultry feathers are rich in keratin protein (90%< of crude protein),
12
and they are not easily broken down by usual proteolytic enzymes like trypsin, pepsin, and so on,
13
keratin-rich feather waste degradation by keratinolytic microorganism is important, not only for removing feather wastes but also for providing the worthy protein, which can be used in animal feed, agriculture, and cosmetic industries as nutrient additives,
14
as well as nitrogen fertilizer for organic farming.
15
Keratin-degrading bacteria include the genus
As chemical treatment of keratin waste is regarded as an eco-destructive means, 20 playing negative roles on environment, it is highly desired to consider the industrial application of keratinase. 21 In the present investigation, therefore, environment friendly enzymatic dehairing by crude keratinase was considered in comparison with 2 traditionally used chemicals (CaO, Na2S). The reaction condition was optimized with respect to various operating parameters in shake-flask culture. Moreover, an attempt was made for partial purification of crude keratinase activity. In addition, fermentor-scale production of crude keratinase through batch culture with optimized condition was performed in 1 L fermentor to show the possible industrial applications.
Material and Methods
Sampling, isolation, and screening of keratinase-producing bacteria
A total of 50 soil samples were collected aseptically into sterile container from 15 different poultry farms of Chittagong city (Supplementary File 1) and preserved them into the refrigerator at 4°C until analysis.
Serial dilution (10−1-10−9) was conducted by using spread plate techniques, 22 and then bacterial pure culture was isolated by streak plate method on nutrient agar media (pH 7.0). Selected pure isolates were then subcultured on their respective media, purified, and stored in the laboratory (−80°C) in glycerol stock (50%) solution for further studies.
Isolates were then cultured and incubated at 37°C for 48 hours on skim milk agar for primary screening, 23 where clear zone–forming isolates were selected as proteolytic bacteria. 24 Then the isolates were further identified as keratinolytic bacteria by using feather meal at 37°C for 24 to 48 hours according to Raju and Divakar. 25
Phenotypic and biochemical characterization of keratinolytic bacteria
Selected keratinolytic bacteria were then characterized by gram staining based on cultural, morphological, as well as biochemical characteristics. 26 For the activities of oxidase, catalase, citrate utilization, indole production, methyl-red (MR), Voges-Proskauer (VP), urease production, and carbohydrate utilization, isolates were biochemically analyzed27,28 and provisionally identified according to Bergey’s Manual of Systemic Bacteriology. 29
Molecular identification of bacteria
Genomic DNA was extracted 30 and stored at −20°C. DNA concentration was measured by Thermo Scientific, NanoDrop 2000 spectrophotometer (Thermo Scientific, USA) following standard protocol. Polymerase chain reaction (PCR) detection of bacteria was performed using previously published primers and targeted gene.31,32 Primer specificity was determined by searching for similar sequences in microbial genome using the Basic Local Alignment Search Tool (BLAST). In each experiment, positive and negative control was carried out. A total composition of target gene, primer sequences, cycling parameters, PCR master mixture, and amplicon size (bp) used for PCR amplifications in a thermal cycler (Nyx Technik) are shown in Table 1.
Target genes, primer sequences, cyclic condition, PCR master mixture composition, and amplicon size.
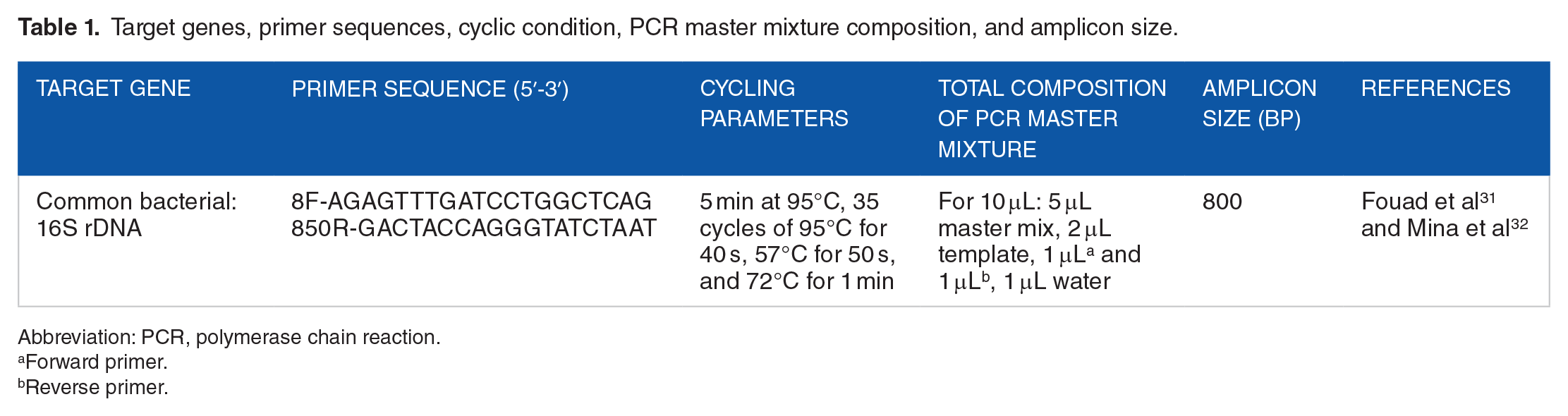
Abbreviation: PCR, polymerase chain reaction.
Forward primer.
Reverse primer.
Amplified PCR products were then analyzed by electrophoresis (Micro-Bio-Tech Brand) in 1% (w/v) agarose gel in 1×TAE buffer, stained with ethidium bromide (1%) 30 and compared with marker DNA (GeneRuler 1 kb DNA Ladder), during visualized under ultraviolet (UV) trans-illuminator (Benda company) and then photographed. Then PCR products were purified by ATPTM Gel/PCR Fragment DNA Extraction Kit (Catalog No. ADF100/ADF300).
Two (SJ, SA) biochemically identified
29
bacterial isolates were sent for sequencing (Macrogen, South Korea). The results were then analyzed by BLAST program of National Center for Biotechnology Information (NCBI) (https://blast.ncbi.nlm.nih.gov)
33
and submitted to GenBank by using Bankit submission tool. The phylogenetic tree was deduced by using the Neighbor-Joining method
34
and this tree was drawn with the sum of branch length (0.30277880). Replicates (500) were used in the bootstrap test and have been shown to the branches.
35
The branch lengths in the tree is drawn by the same units as those of the evolutionary distances used to infer the phylogenetic tree. The
Keratinase production in shake flask culture and keratinase assay
Identified two isolates (SJ and SA) were then transferred aseptically to sterile 50 mL of LB (Luria-Bertani) broth (pH 7.0 ± 0.2) and incubated overnight at 37°C in a rotary shaker incubator (HandyLab), at 150 r/min.
After that, a 250-mL Erlenmeyer flask containing 50 mL (w/v) sterile basal medium was prepared according to Williams et al 38 and incubated with 1.5 mL of bacterial inoculum which previously cultured for 24 hours at 37°C and 150 r/min for 96 hours in a shaker incubator.
Keratinase activity was assayed according to Cai et al
39
where 1.0 mL of crude enzyme properly diluted in Tris-HCl buffer (0.05 mol/L), followed by the incubation with 1.0 mL keratin solution prepared from chicken feathers
40
at 50°C for 10 minutes. Then enzyme-substrate reaction was stopped by adding 2.0 mL of 0.4 mol/L trichloroacetic acid (TCA). After centrifugation at 4000 r/min for 30 minutes, the absorbance of the supernatant was determined at 280 nm by UV-Spectrophotometer (Shimadzu, Japan) compared with control that was prepared by incubating the enzyme solution with 2.0 mL TCA without the addition of keratin solution, where 1 unit (U/mL) of keratinase activity was defined as an increase of corrected absorbance of 280 nm (
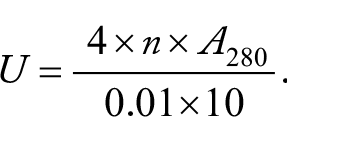
where
Optimization of culture conditions for maximum keratinase production
The keratinase production was optimized with respect to different culture conditions such as temperature, pH, agitation speed, incubation period, inoculum volume, substrates, C and N sources.24,42 The effects of media temperature, pH, and agitation speed (100-250 r/min) on the cell growth and keratinase activity were studied by examining bacterial growth at different temperatures such as 35°C, 37°C, 39°C, 40°C, and 43°C and various ranges of pH (6.5, 7.5, 8.5, 9.5, and 10.5), during 72, 84, and 96 hours of incubation. The culture extract was centrifuged at 4000 r/min for 30 minutes at 4°C, to be used to estimate keratinase production. To compare relative keratinase production, the control was also measured in each case.
To investigate the effect of inoculum volume on enzyme production, bacteria were cultured with 1%, 2%, 3%, 4%, and 5% of inoculum using different substrates such as chicken feather, human hair, and feather meal separately at previously optimized conditions.
Carbon sources (glucose, fructose, maltose, starch, and lactose) and nitrogen sources (peptone, tryptone, yeast extract, sodium molybdate, and ammonium sulfate) were used separately at 0.1% concentration to observe their effects on keratinase production at the optimized conditions. Controls were performed without addition of carbon and nitrogen sources to compare the production.
Partial characterization of crude keratinase activity (enzyme-substrate reaction)
Effects of various enzyme reaction conditions such as pH, temperature, reaction time, substrates, and metal ions were studied for optimum crude keratinase activity.24,43 The effect of pH on keratinase activity was determined at pH (4.0-11.0) by preparing substrate with acetate buffer (pH 4.0-5.5), phosphate buffer (pH 6.0-7.5), Tris-HCl buffer (pH 8.0-10.0), and sodium bicarbonate-sodium hydroxide (10.0-11.0). The keratinase activity was measured (λ = 280) using standard assay condition.
Besides, the enzymatic reaction was performed at different temperatures (30°C, 40°C, 50°C, 60°C, 70°C) in water bath and assayed (λ = 280) to find out the optimum temperature of the enzyme-substrate reaction.
Reaction mixtures were prepared with phosphate buffer (pH 6.0-7.5) and Tris-HCl buffer (pH 8.0-10.0), and enzymatic reaction was performed at different reaction times (10, 20, 30, 40, and 50 minutes) at the optimized temperature and pH. To study the substrates specificity, human scalp hair, casein, chicken feathers, and gelatin were used. The impact of enzyme inhibitor was also studied with different metal ions (Na+, K+, Co2+, Ca2+, Mg2+, Fe2+, Hg2+, Zn2+).
Protein estimation
Protein content of culture broth in shake-flask culture was assessed by Lowry method 44 using BSA as standard during optimized culture conditions.
Dehairing assay (laboratory trial)
Fresh raw skins of domestic animal (cow) were collected from local slaughter houses of Dewanhat, Chittagong. To remove impurities, they were washed with sodium chloride and water, and then dried at hot air oven (50°C). The skins were then cut into small pieces (5 cm 2 ), kept with 40 mL crude enzyme solution in a petri dish, and incubated at 37°C for 16 hours. Besides, chemical agents like Na2S and CaO were also used to compare dehairing efficiency. Skins treated with distilled water was considered as negative control. 45 By gently scraping with blunt scalpel, skins were then withdrawn at regular intervals at 4, 8, 12, and 16 hours to examine better dehairing results.
Partial purification of crude enzyme
The growth extract from shake flasks was partially purified by acetone precipitation method, where keratinase was precipitated by prechilled acetone at 30%, 40%, 50%, 60%, 70%, and 80% fractionation. The acetone was added in ratio 3:1 to the cell free extract and incubated for 60 minutes at −20°C. The mixture was then subjected to centrifugation at 10 000 r/min for 10 minutes. After discarding the supernatant carefully, pellet was dissolved in Tris-acetate buffer (pH 7.0) and used for enzyme assay. 43
Keratinase production by the fermentor
Batch cultivations were performed using 1 L of filter sterilized basal medium composed of the following components (per liter of final volume): 0.5 g NH4Cl, 0.5 g NaCl, 0.3 g K2HPO4, 0.4 g KH2PO4, 0.1 g MgCl2.6H2O, 0.1 g yeast extract, including 1.0 g raw chicken feather and made up to 1000 mL with distilled water 38 in a 2-L fermenter (Hanil, South Korea) maintained at previously optimized conditions. The pH of the medium was maintained with a pH controller by automatic addition of 2.0 M HCl or 2.0 M NaOH. The aerobic condition was ascertained by controlling the stirring speed with the constant air flow rate of 1 L/min. Triplicate samples were taken after 96 hours to get maximum enzyme production. 46
Statistical analysis
Triplicate experiments were done in all the cases during isolation, biochemical analysis, optimization of growth conditions, and partial characterization of keratinase enzyme by the selected isolates.
Results and Discussion
Among 50 soil samples, 7 isolates (SA, SH(W), SH(Y), SJ, SK, SN, and SP) were found proteolytic on skim milk agar based on clear zone production (Figure 1A). Then, 2 of them were selected as best keratinolytic isolates (Figure 1B). This observation may be due to the fact that keratinase-producing bacteria can also produce one or more other specific bacterial proteases such as alcalase, pronase, and trypsin. 47 This indicates that the isolates which are able to hydrolyze casein might have also the ability to synthesize proteolytic enzymes along with keratinase enzyme.
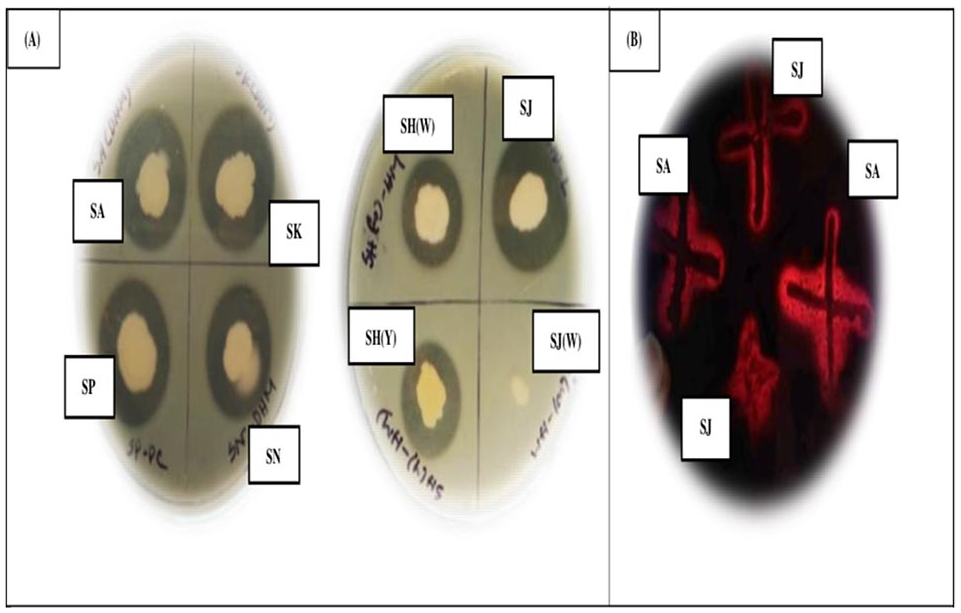
(A) Plate assays of 7 pure isolates. (B) Growth of isolates (SA, SJ) on FMA (Feather Meal Agar).
After two keratinolytic isolates were selected, they were subjected to different biochemical tests. One isolate was identified as gram positive by detecting the presence of peptidoglycan as a thick layer in that bacteria, whereas the other isolate was gram negative.
48
Then based on biochemical characterizations, they were provisionally identified26,29 as
Morphological and biochemical characterization of bacterial isolates.
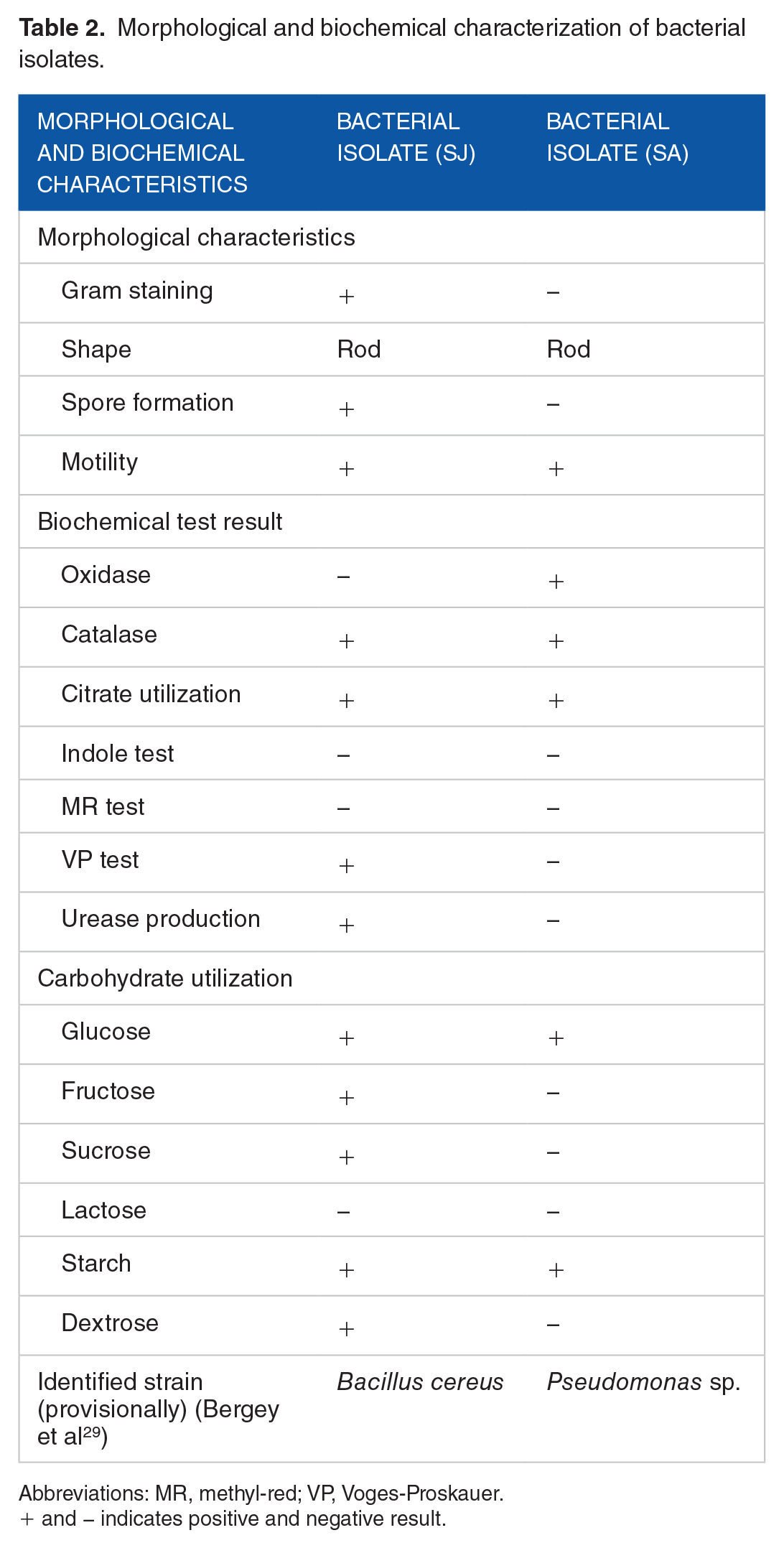
Abbreviations: MR, methyl-red; VP, Voges-Proskauer.
+ and − indicates positive and negative result.
The 16S rDNA amplification was conducted through PCR for two keratinolytic
49
and biochemically identified bacteria, where extracted genomic DNA showed positive bands in 1% agarose gel (~800 bp), compared with the DNA 1 kb marker (Figure 2). Homology analysis inferred from 16S rDNA sequence comparison clearly shows that the two strains clustered with
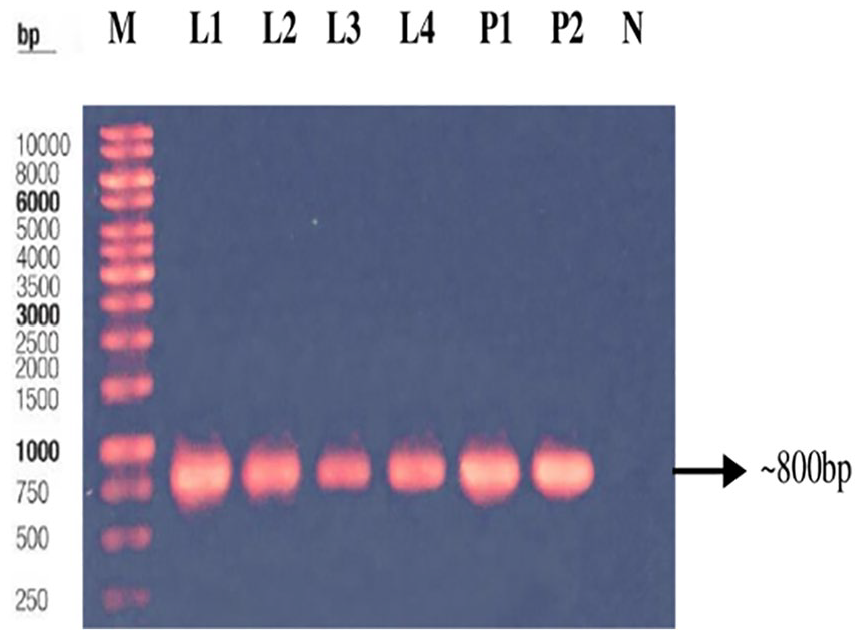
Electrophoretic (1% agarose) separation of 16S rDNA gene of different isolates. M indicates 1 kb DNA marker; L1-L2, L3-L4 show bands of SJ and SA, respectively; P1, P2 for positive control (SJ and SA); and N for negative control, respectively.

Phylogenetic tree construction. Phylogenetic tree showing relationship between 

The optimal growth rates of
In general, higher agitation rates (200-250 r/min) provide good growth of bacteria with possibly low keratinase production due to high dissolved oxygen concentration. On the other hand, Pissuwan and Suntornsuk
51
reported that, at low agitation rate (100 r/min), substrates and bacterial cells were not mixed properly and produced heterogeneous formation, thus lowering dissolved oxygen and leading to lower keratinase production. In our study, both isolates produced maximum keratinase (57.33 ± 2.08) U/mL for
Although feather degradation can be completely made between 3 to 5 days, it also takes more than 5 days to be degraded. Agrahari and Wadhwa
52
as well as Yadav et al
53
reported the maximum keratinase production on days 5 and 7 by
Several inoculum volumes were also tested, where 3% of inoculum volume was found responsible for the highest keratinase production for both
Carbon and nitrogen sources also affect keratinase production, where
Overall optimized conditions for maximum keratinase production by
Optimization of culture conditions for production of extracellular keratinase from
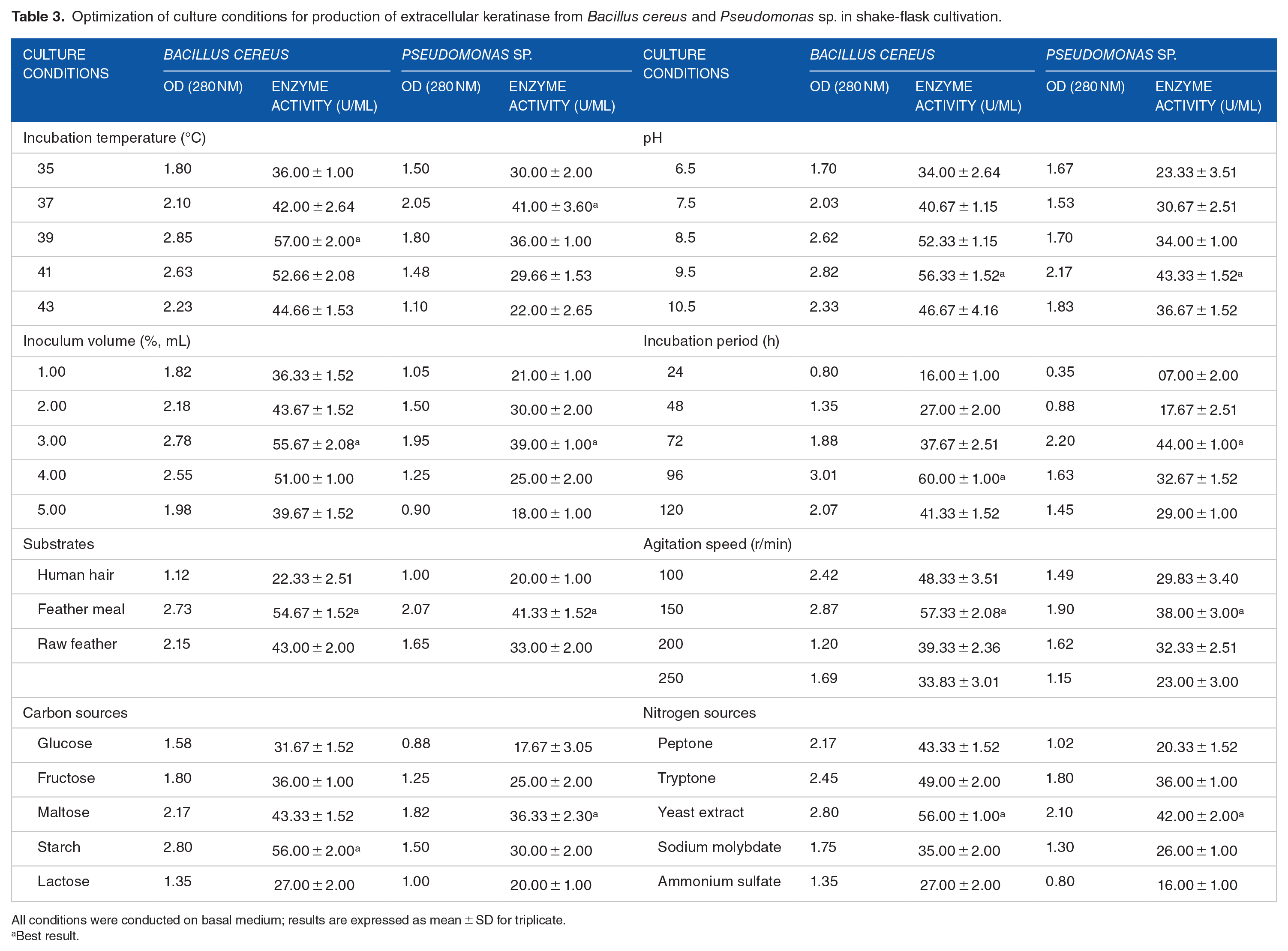
All conditions were conducted on basal medium; results are expressed as mean ± SD for triplicate.
Best result.
Figure 4 shows the effects of culture condition on the keratinase activity, where Figure 4A indicates that the optimal temperature of keratinase from
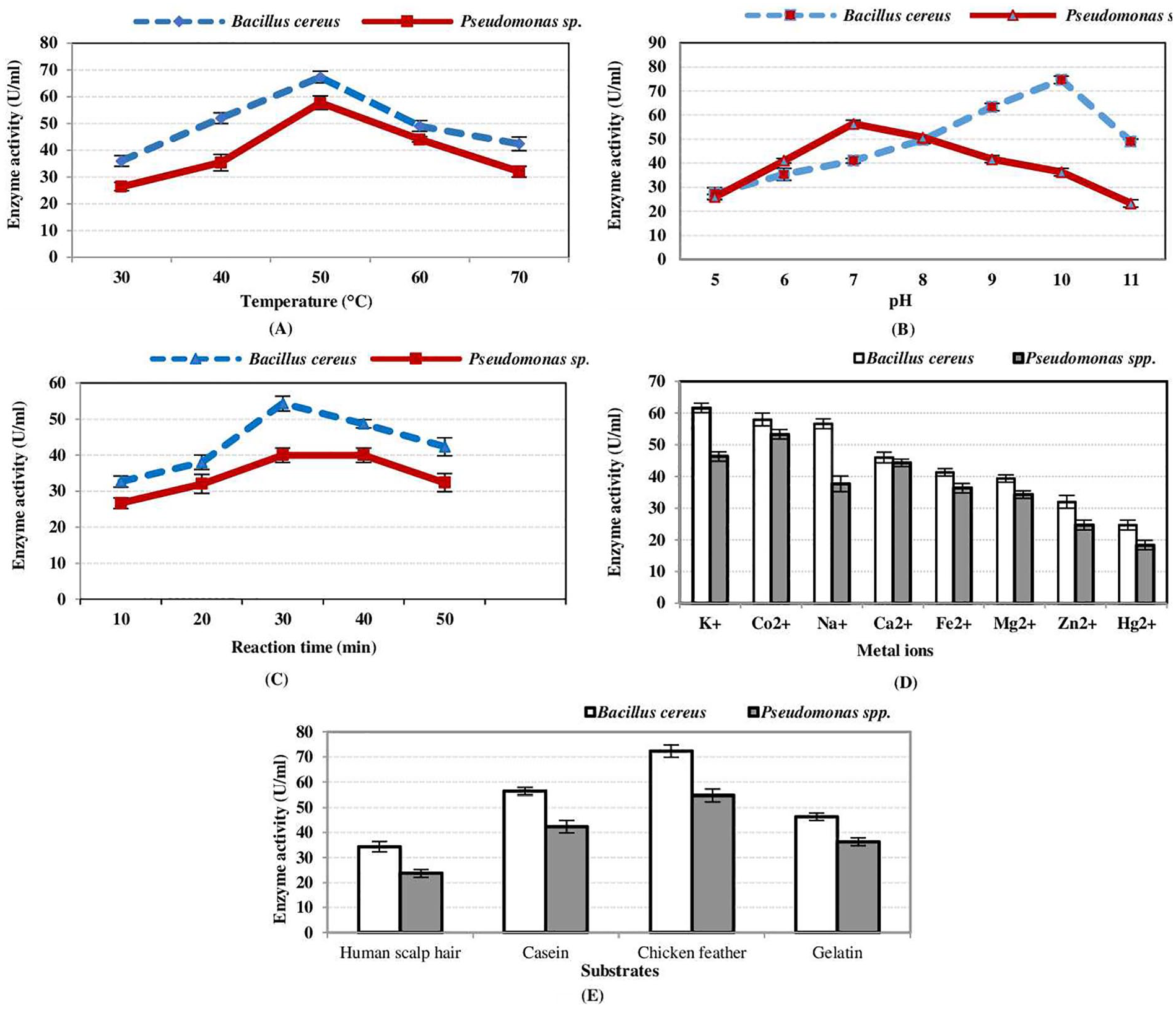
Partial characterization of crude keratinase activity (enzyme-substrate reaction). Effects of (A) temperature, (B) pH, (C) reaction time, (D) metal ions, and (E) substrates.
The effects of reaction time (10-50 minutes) on extracellular keratinase enzyme activity shows that the highest activity was attained at 30 minutes (Figure 4C) for both species as Gupta et al,
57
stated that incubation at 70°C for 30 minutes enhanced keratinase activity (21%) of
The effects of metal ions (Na+, K+, Co2+, Ca2+, Mg2+, Fe2+, Hg2+, and Zn2+) were also studied (Figure 4D) by preincubating them with enzymes. Inhibitory effects of Hg2+ and Zn2+ were reported by Kainoor and Naik
56
as well as Lin and Yin
58
in case of
Hydrolysis of complex protein substrate responds to keratinase activity efficiently, which had a broad spectrum of substrate specificity for soluble and insoluble substrates. Practically, keratinase can hydrolyze feather keratin with maximum activity, followed by casein, gelatin, and human scalp hair (Figure 4E) in our study. Similar findings were obtained by Gupta et al
57
and Rajput et al,
59
where the highest keratinase activity was found on chicken feather substrate by both
Crude proteins were estimated approximately to be 0.73 and 0.57 mg/mL (Figure 5) from keratinases of
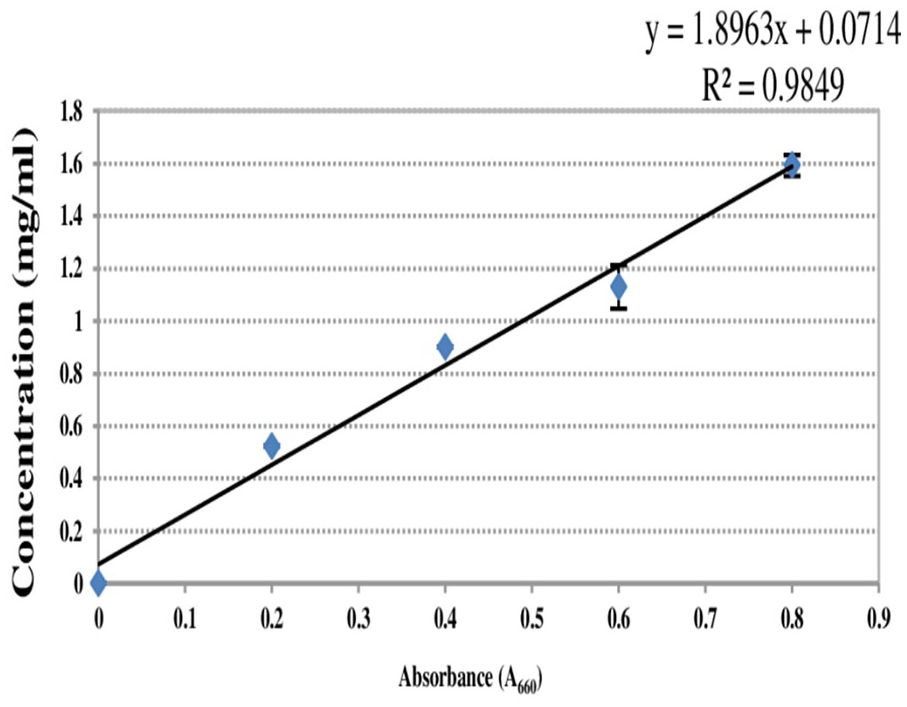
Standard graph of Folin-Lowry assay.
Folin-Lowry standard assay for BSA.
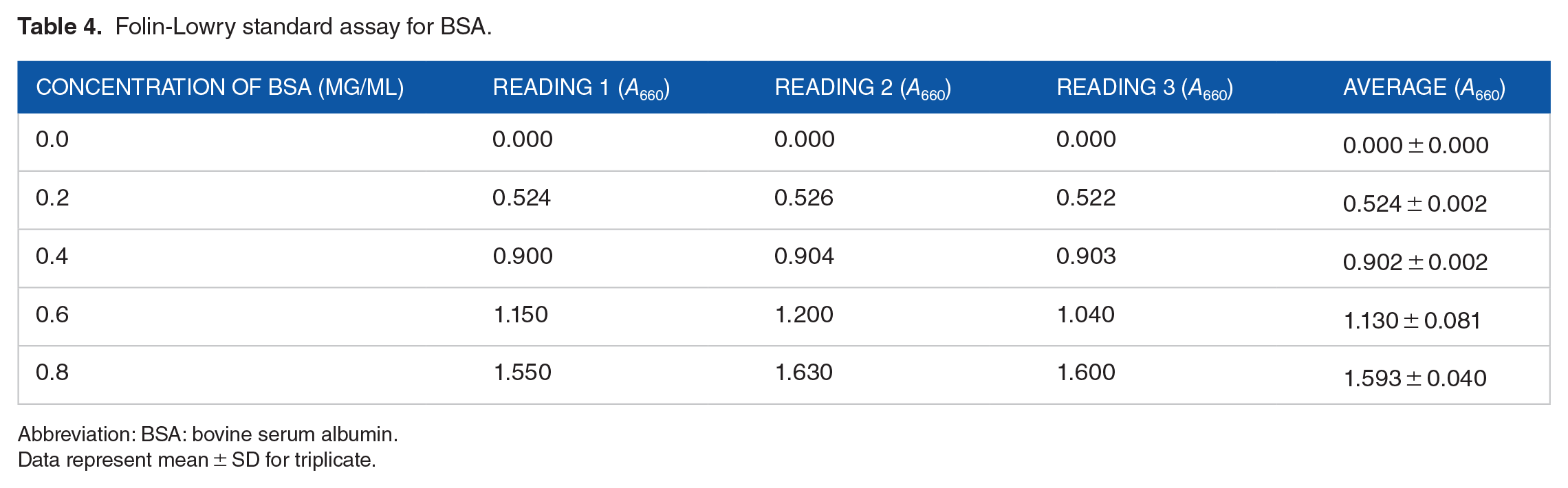
Abbreviation: BSA: bovine serum albumin.
Data represent mean ± SD for triplicate.
To study the chicken feather degradation rate (%), 96 hours of incubation was performed for
Visual observation of chicken feather degradation (%) by

Data represent mean ± SD for triplicate.
Best result.
An attempt was made to observe the effectiveness of produced keratinase enzyme over chemicals in dehairing process, as chemicals cause damage and reduce the quality of leather. Leather pieces were soaked with enzymes and chemicals (Na2S, CaO), separately for 16 hours in total. After 12 hours, 100% dehairing with enzyme showed effectiveness over chemicals without any damaging of leather (Figure 6). After 16 hours, Na2S caused damage to leather, while dehairing remain incomplete by CaO. Moreover, pulp of hair exhibited rough texture when chemical treatment was conducted (Supplementary File 3). In this study, the crude keratinase from
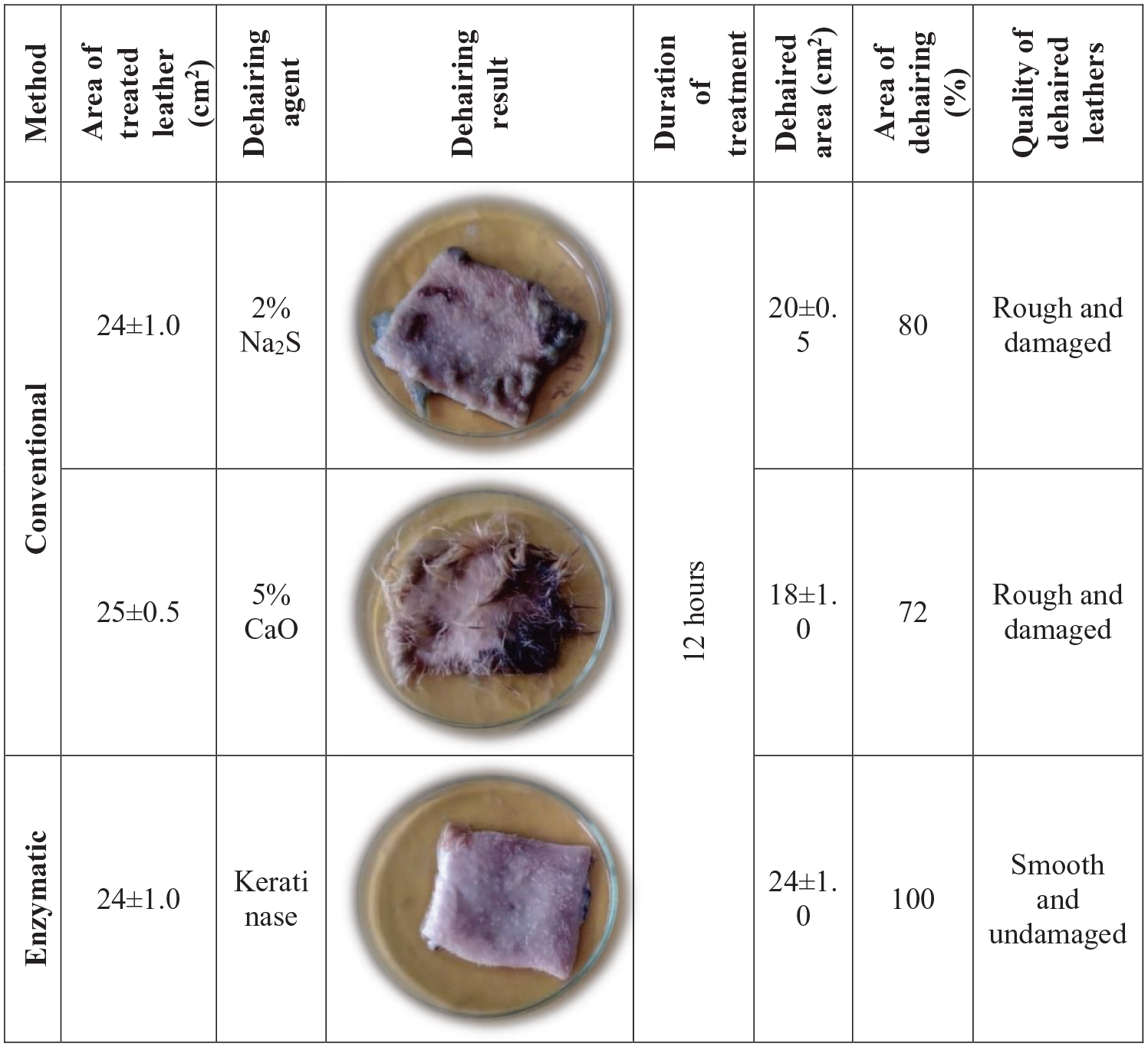
Comparative study of dehairing assay (field trial).
Partial purification of crude keratinase contained in the cell free supernatant by acetone precipitation eliminates interfering elements present in that supernatant. Although acetone is a good purifying agent for proteins, due to denaturing tendency, it is not commonly used in purification procedures. Maximum specific activity was observed for
Purification fold of enzymes at different saturation levels of acetone precipitation.
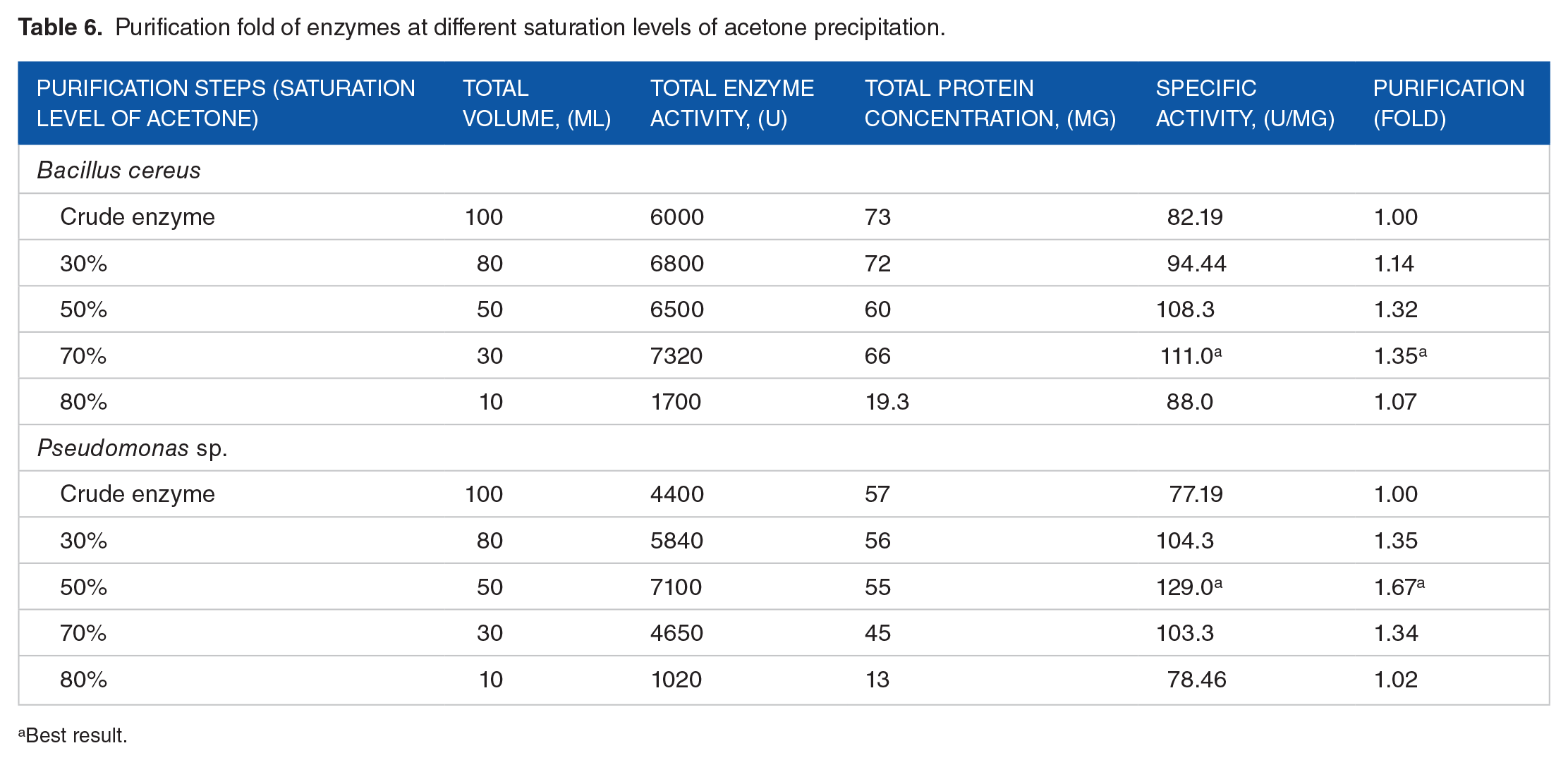
Best result.
In addition to flask culture, large-scale keratinase production was studied by batch cultivation, while it was found (136.0 ± 0.030) U/mL after 4 days (80 hours) of incubation, 2-fold more than in shake flask culture (60 ± 1.0) U/mL (Table 7).
Data from fermentor for keratinase production by
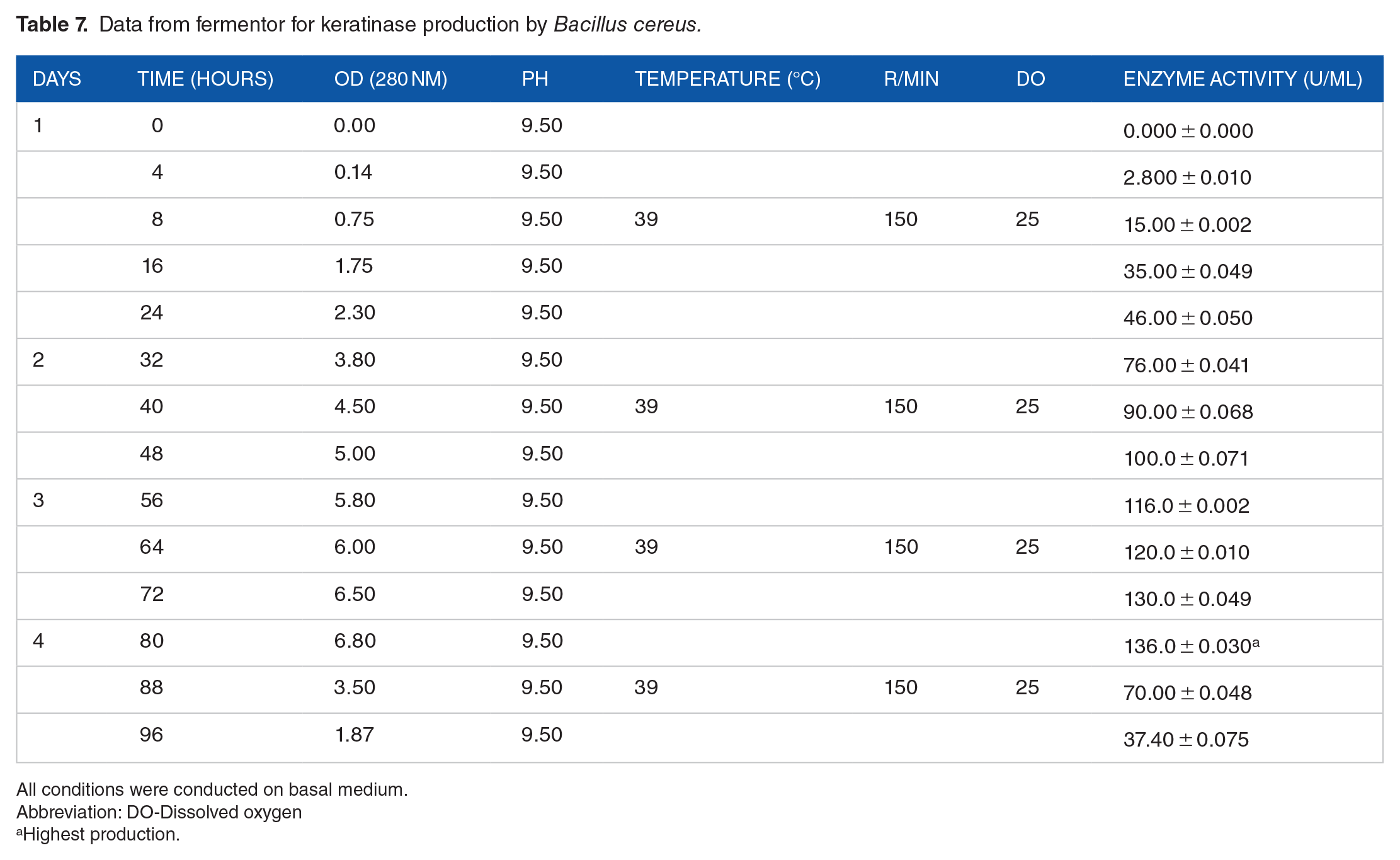
All conditions were conducted on basal medium.
Abbreviation: DO-Dissolved oxygen
Highest production.
Conclusions
In the present work, it was shown that the keratinase produced from
Supplemental Material
Supplementary_file_1_xyz33416b0980c9e – Supplemental material for Microbial Bioremediation of Feather Waste for Keratinase Production: An Outstanding Solution for Leather Dehairing in Tanneries
Supplemental material, Supplementary_file_1_xyz33416b0980c9e for Microbial Bioremediation of Feather Waste for Keratinase Production: An Outstanding Solution for Leather Dehairing in Tanneries by Mursheda Akhter, Lolo Wal Marzan, Yasmin Akter and Kazuyuki Shimizu in Microbiology Insights
Supplemental Material
Supplementary_file_2_xyz3341651296bf9 – Supplemental material for Microbial Bioremediation of Feather Waste for Keratinase Production: An Outstanding Solution for Leather Dehairing in Tanneries
Supplemental material, Supplementary_file_2_xyz3341651296bf9 for Microbial Bioremediation of Feather Waste for Keratinase Production: An Outstanding Solution for Leather Dehairing in Tanneries by Mursheda Akhter, Lolo Wal Marzan, Yasmin Akter and Kazuyuki Shimizu in Microbiology Insights
Supplemental Material
Supplementary_file_3_xyz334166ba60c36 – Supplemental material for Microbial Bioremediation of Feather Waste for Keratinase Production: An Outstanding Solution for Leather Dehairing in Tanneries
Supplemental material, Supplementary_file_3_xyz334166ba60c36 for Microbial Bioremediation of Feather Waste for Keratinase Production: An Outstanding Solution for Leather Dehairing in Tanneries by Mursheda Akhter, Lolo Wal Marzan, Yasmin Akter and Kazuyuki Shimizu in Microbiology Insights
Footnotes
Acknowledgements
The authors are pleased to mention about the fruitful discussions with Md. Mahbub Hasan, Assistant Professor of Department of Genetic Engineering and Biotechnology, University of Chittagong.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was done in the “Molecular Biology Laboratory” of the Department of Genetic Engineering and Biotechnology, University of Chittagong, Bangladesh and partially funded by the Research & Publication Office of the University of Chittagong (Ref. 6016/2017; fiscal year 2016-2017).
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
LWM and YA contributed to the conceptualization, acquired funding, and assisted in the supervision; MA performed formal analysis and investigation, and wrote the original draft of the manuscript; LWM, YA, and KS wrote, reviewed, and edited the manuscript; and all authors approved the final draft of the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
