Abstract
Circular RNAs (circRNAs) have been revealed to involve in the chemoresistance of various cancers, including non-small cell lung cancer (NSCLC). Here, we further investigate the role of circRNA_100565 in NSCLC cisplatin (DDP) resistance. The expression of circRNA_100565 and microRNA (miR)-337-3p, and ADAM metallopeptidase domain 28 (ADAM28) mRNA was detected using quantitative real-time polymerase chain reaction. Cell viability and apoptosis were measured by cell counting kit-8 assay and flow cytometry, respectively. Western blot was used to detect the level of ADAM28 and autophagy-related protein. The interaction between miR-337-3p and circRNA_100565 or ADAM28 was confirmed by dual-luciferase reporter assay or pull-down assay. In vivo experiments were conducted via the murine xenograft model. We found CircRNA_100565 was up-regulated in NSCLC DDP-resistant tissues and cell lines, and its high expression was associated with shorter overall survival of NSCLC patients. CircRNA_100565 deletion mitigated DDP resistance, reflected by the suppression of proliferation and autophagy, the reduction of IC50 value, as well as enhancement of apoptosis in DDP-resistant NSCLC cells. MiR-377-3p was confirmed to directly bind to circRNA_100565 or ADAM28 3’-UTR. Moreover, circRNA_100565 indirectly regulated ADAM28 expression by sponging miR-377-3p in NSCLC cells. Additionally, circRNA_100565 deletion-induced sensitivity of NSCLC resistant cells to DDP could be remarkably attenuated by miR-377-3p inhibition or ADAM28 re-expression. Meanwhile, circRNA_100565 knockdown contributed to the anti-tumor effects of DDP on NSCLC in vivo.
CONCLUSION:
CircRNA_100565 was an independent prognostic factor for NSCLC patient survival, and enhanced the resistance of NSCLC cells to cisplatin by regulating cell proliferation, apoptosis and autophagy via miR-337-3p/ADAM28 axis, shedding light on the development of a novel therapeutic strategy to boost the effectiveness of NSCLC chemotherapy.
Introduction
Lung cancer is counted as the leading cause of cancer-induced mortality worldwide. Non-small cell lung cancer (NSCLC) is one of the main sub-types of lung cancer, represents approximately 85% of all lung cancer cases, and has a high incidence and low five-year survival rate of approximately 17.1% [1, 2]. Currently, platinum-based chemotherapy is a well-recognized standard adjunctive treatment strategy in patients with advanced NSCLC, among these, cisplatin (DDP)-based chemotherapy is one of the most widely used therapeutic strategies in NSCLC [3]. Unfortunately, obvious resistance to DDP has been emerged in a large proportion of NSCLC patients receiving chemotherapy, and limits the clinical application of DDP in NSCLC treatment [4]. Thus, extensive efforts on developing low-toxic treatment strategies are necessary to manage the survival of NSCLC patients.
Circular RNAs (circRNAs) are a class of highly stable RNA molecules with the covalently closed-loop structures, which result in resistance to regular mechanisms of linear RNAs decay [5]. Currently, growing investigations have indicated that circRNAs act as critical functional modulators of physiological or pathological cellular processes implicated in cell cycle, growth, differentiation, apoptosis, and metastasis in tumors [6, 7]. Besides that, many circRNAs have also been exhibited to involve in the chemoresistance of various cancers by regulating cellular apoptosis and autophagy. For example, circRNA AKT3 inhibited gastric cancer apoptosis to promote DDP resistance by up-regulating PIK3R1 through miR-198 [8]. Overexpressed circ-Cdr1as sensitized ovarian cancer to DDP by mediating the DDP-induced cell apoptosis via the regulation of miR-1270/SCAI axis [9]. Circ_0035483 enhanced the resistance of renal cancer cells to gemcitabine by regulating autophagy through sponging miR-335 [10]. CircPAN3 facilitated doxorubicin resistance in acute myeloid leukemia by influencing autophagy and apoptosis [11]. In the previous research of our team, it was demonstrated that circ_100565 promoted NACLC cell proliferation, migration and invasion via increasing HMGA2 expression through absorbing miR-506-3p, indicating the oncogenic role of circ_100565 in NSCLC [12]. However, despite some advanced findings, the expression and possible carcinogenic involvement of circ_100565 in NSCLC chemoresistance remain largely unclear.
MiRNAs are small non-protein coding RNAs that modulate gene expression at post-transcriptional level [13]. MiRNAs have been reported to participate in many pathological processes of cancers by involving in cell inflammation, metabolism, metastasis, autophagy, apoptosis and drug resistance [14, 15, 16]. MiR-337-3p is a well-characterized tumor suppressor in many cancers, such as hepatocellular carcinoma, clear cell renal cell carcinoma, and cervical cancer, and have been found to inhibit tumor progression by regulating tumor cell proliferation, apoptosis, and metastasis [17, 18, 19]. Besides that, Du et al. revealed that miR-337-3p sensitized NSCLC cells to paclitaxel by regulating STAT3 and RAP1A expression, indicating the role of miR-337-3p in chemoresistance. ADAM metallopeptidase domain 28 (ADAM28) is a member of the ADAM family, which are found to implicate in a variety of biological events, such as cell-cell and cell-matrix interactions [20]. ADAM28 was found to be elevated in NSCLC, and was associated with cell proliferation, lymph node metastasis and tumor size [21]. However, the role of ADAM28 in chemoresistance of NSCLC remains unclear.
In this study, we attempted to investigate the expression pattern and effects of circRNA_100565 in DDP-resistant NSCLC, and evaluated the regulatory effects of circRNA_100565, miR-337-3p and ADAM28 in NSCLC with sDDP-resistance.
Materials and methods
Patients and specimens
Tumor tissues and matched normal tissues were obtained from 50 NSCLC patients who underwent surgical resection at The First Affiliated Hospital of Hainan Medical University and immediately stored in liquid nitrogen until used. All patients were diagnosed by histopathological examination and only received DDP-based neo-adjuvant chemotherapy before surgery. The patients were divided into two groups depending on the sensitivity to DDP: DDP-sensitive group (
Cell culture and transfection
NSCLC cell lines (A549 and H1299) were purchased from Shanghai Academy of Life Science (Shanghai, China). The stable DDP-resistant lines A549/DDP and H1299/DDP were established by exposing stepwise increasing concentrations of DDP (Sigma, St. Louis, MO, USA) over 6 months. Parental cells were grown in the Dulbecco’s modifed Eagle’s medium (DMEM; Gibco, Carlsbad, CA, USA) harboring with 10% fetal bovine serum and 1% penicillin/streptomycin (Gibco) with 5% CO
The miR-337-3p mimic (miR-337-3p), miR-337-3p inhibitor (anti-miR-337-3p), and their corresponding negative control (miR-NC and anti-NC) were purchased from RIBOBIO (Guangzhou, China). The short hairpin RNA (shRNA) targeting circRNA_100565 covalent closed junction (sh-circRNA_100565), shRNA scramble control (sh-NC), empty vector (vector), pcDNA-ADAM28 overexpression vector (ADAM28) were synthesized by Genepharma (Shanghai, China). The transfection of oligonucleotides was performed using Lipofectamine
Quantitative real-time polymerase chain reaction (qRT-PCR)
Total RNA was extracted using TRIzol reagent (Invitrogen) according to the standard procedure, and then was interacted with Rnase R (Epicentre, Madison, WI, USA), followed by incubation with RNeasy MinElute Cleanup Kit (Qiagen, Valencia, CA, USA). Subsequently, complementary DNA (cDNA) was synthesized using a High Capacity cDNA Reverse Transcription Kit (Qiagen), and then quantitative PCR was conducted using SYBR Green methods on the ABI7500 system. Glyceraldehyde 3-phosphate dehydrogenase (GADPH) and U6 small nuclear B noncoding RNA (U6) were used as internal references to normalize the relative transcription expression using 2
Cell viability assay
The DDP-sensitivity of cells was analyzed by cell counting kit- 8 (CCK-8) assay. Transfected DDP-resistant cells (5000 per well) were seeded into 96-well plates overnight and then were incubated with different concentrations of DDP (1, 10, 20, 40, 80, 160
Cell apoptosis assay
Transfected DDP-resistant cells were incubated with DDP for 48 h in six-well plates. Subsequently, cells were resuspended with binding buffer, followed by staining with 10
CircRNA_100565 expression in DDP-resistant NSCLC tissues and cells and its correlation with overall survival. (A) The expression of circRNA_100565 was detected using qRT-PCR in NSCLC tumor tissues (DDP-sensitive group (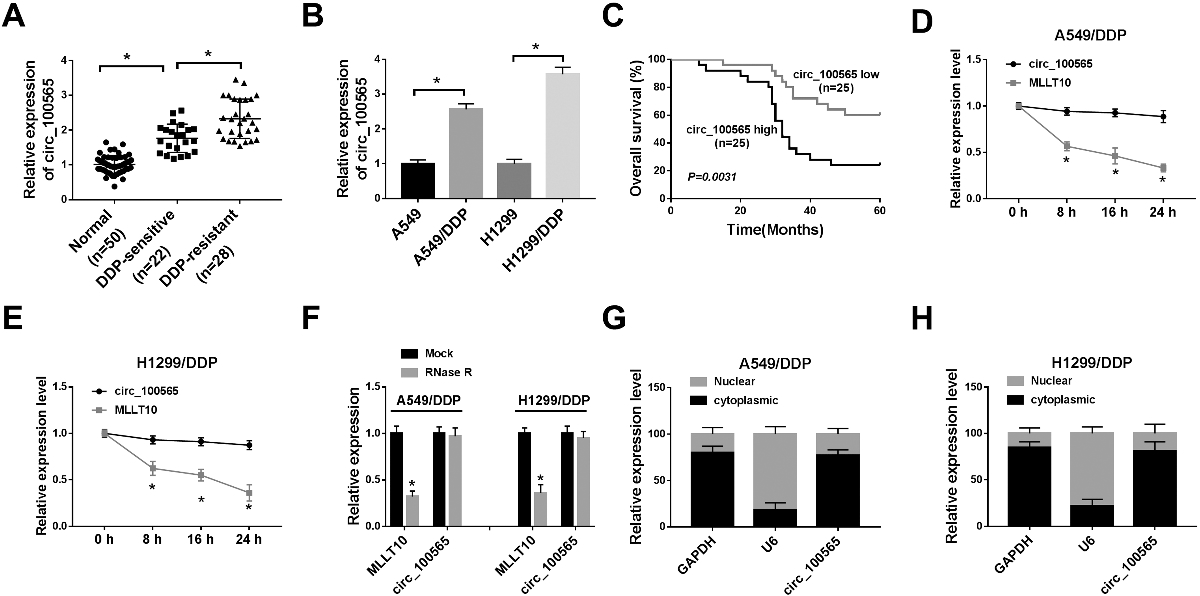
Proteins were extracted using RIPA lysis buffer (Beyotime, Beijing, China), and then were quantified by the bicinchoninic acid method according to the standard protocol. After that, the equal amount of protein was separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis, shifted onto a polyvinylidene fluoride membrane, and blocked with 5% non-milk. Subsequently, the membrane was probed with primary antibodies against light chain 3B (LC3B) (1:3000, ab51520, Abcam, Cambridge, MA, USA), Beclin 1 (1:3000, ab62557, Abcam), p62 (1:10000, ab109012, Abcam), Ki-67 (1:5000, ab92742, Abcam), ADAM28 (1: 5000, ab28292, Abcam), and GAPDH (1:10000, ab181602, Abcam). After interaction with HRP-conjugated secondary antibody (1:1000, ab9482, Abcam), protein bands were detected using the chemiluminescence chromogenic substrate (Beyotime).
Dual-luciferase reporter assay
The circRNA_100565 and 3’ UTR of ADAM28 containing the wild-type (WT) or mutant (MUT) binding sites of miR-337-3p were cloned into the pmiR-RB-Report (Promega, Shanghai, China), respectively. Subsequently, cells were co-transfected with constructed luciferase reporter vectors and miR-337-3p or miR-NC using Lipofectamine
Pull-down assay
MiR-337-3p and miR-NC were biotinylated to generate Bio-miR-337-3p and Bio-miR-NC by GenePharma Company (Shanghai, China), and then these biotinylated oligonucleotides were transfected into NSCLC-resistant cells for 48 h. After that, cells were collected and lysed, and the lysate was incubated with streptavidin-coated magnetic beads. After elution, the biotin-coupled RNA complex was pulled down and then analyzed by qRT-PCR.
Xenograft experiments in vivo
BALB/c athymic mice (
Statistical analysis
Data from at least three independent experiments were expressed as the mean
Results
CircRNA_100565 expression in DDP-resistant NSCLC tissues and cells and its correlation with overall survival
Correlation between circ_100565 expression and the clinical pathological features of 50 NSCLC patients
Correlation between circ_100565 expression and the clinical pathological features of 50 NSCLC patients
*
The expression of circRNA_100565 in NSCLC tumor tissues which were divided into DDP-sensitive group (
Univariate analysis for factors related to overall survival using the COX proportional hazard model
*
CircRNA_100565 deletion mitigates DDP resistance by regulating resistant cell proliferation, apoptosis and autophagy in NSCLC. CircRNA_100565 was silenced in A549/DDP and H1299/DDP cells using sh-circRNA_100565. (A) The relative expression of circRNA_100565 was measured using qRT-PCR. (B, C) The CCK-8 assay was performed to detect cell proliferation. (D-F) The IC50 value for DDP was assessed by CCK-8 assay. (G) Cell apoptosis was analyzed using flow cytometric analysis. (H-J) The expression of autophagy-associated protein LC3II/I, Beclin-1 and p62 was measured using qRT-PCR and western blot. *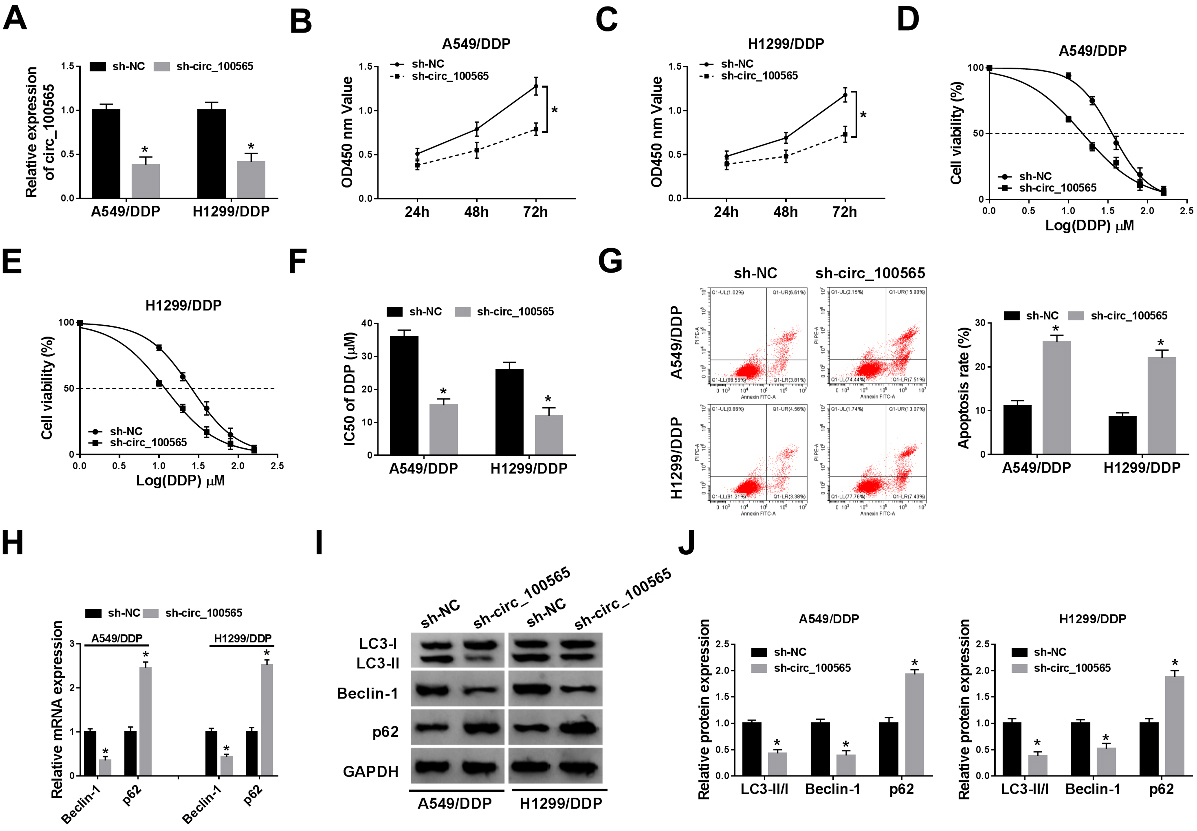
To explore the biological effects of circRNA_100565 on DDP resistance of NSCLC, circRNA_100565 was silenced in DDP-resistant NSCLC cells using sh-circRNA_100565. As expected, the level of circRNA_100565 was greatly down-regulated in A549/ DDP and H1299/DDP cells (Fig. 2A). Subsequently, CCK-8 assay indicated that circRNA_100565 silence inhibited the proliferation of A549/DDP and H1299/DDP cells (Fig. 2B and C). Furthermore, we analyzed the IC50 of A549/DDP and H1299/DDP cells to DDP, which was more than 2-fold lower in circRNA_100565-decreased cells than that of sh-NC transfected-A549/DDP and H1299/DDP cells (35.8 vs. 15.2 in A549/DDP cells, 25.8 vs. 11.9 in H1299/DDP cells), suggesting that circRNA_100565 deletion led A549/DDP and H1299/DDP cells sensitive to DDP (Fig. 2D–F). Immediately, flow cytometry analysis showed the apoptotic A549/DDP and H1299/DDP cells, reflected by the lower and upper right quadrants, were increased by circRNA_100565 silencing (Fig. 2G). Besides that, we also found knockdown of circRNA_100565 down-regulated the ratio of LC3II/I and the expression of Beclin-1, while up-regulated p62 expression in A549/DDP and H1299/DDP cells, suggesting circRNA_100565 knockdown inhibited resistant cell autophagy in NSCLC (Fig. 2H–J). Taken together, knockdown of circRNA_100565 sensitized NSCLC resistant cell to DDP by inducing cell apoptosis, and inhibiting cell proliferation and autophagy.
CircRNA_100565 is a sponge of miR-337-3p and negatively regulates its expression. (A) The putative binding site between circRNA_100565 and miR-337-3p was presented. (B-D) The interaction between circRNA_100565 and miR-337-3p was confirmed by the dual-luciferase reporter assay and pull-down assay in A549/DDP and H1299/DDP cells, respectively. (E) The expression of miR-337-3p in A549/DDP and H1299/DDP cells transfected with sh-NC or sh-circRNA_100565 was detected by qRT-PCR. (F, G) The expression of miR-337-3p in DDP-resistant NSCLC tissues and cell lines was measured by qRT-PCR. (H) The correlation between circRNA_100565 and miR-337-3p was analyzed using Spearman’s correlation test. *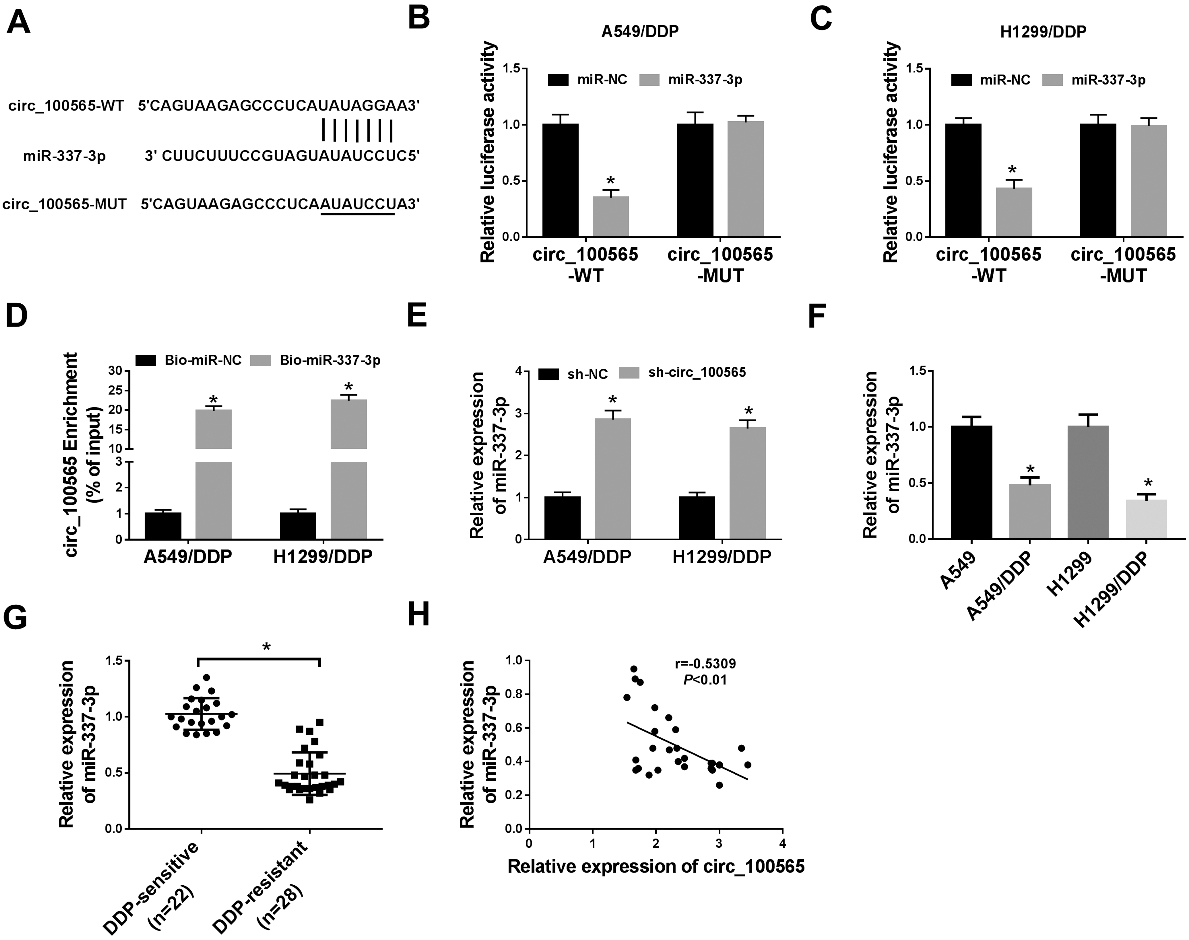
To detect the potential pathways in which circRNA_100565 involved in NSCLC DDP resistance, we used the Circinteractome database to search the potential miRNA targets of circRNA_100565, and miR-337-3p had putative binding sites of circRNA_ 100565 (Fig. 3A). Then the dual-luciferase reporter assay was performed and results showed overexpressed miR-337-3p reduced the luciferase activity of the circRNA_100565-WT reporter vector but not circRNA_100565-MUT reporter vector in A549/DDP and H1299/DDP cells (Fig. 3B and C). Moreover, RNA pull-down assay displayed that circRNA_100565 enrichment in Bio-miR-337-3p-probe group was markedly higher than the negative control group (Fig. 3D). These results indicated the direct interaction between circRNA_100565 and miR-337-3p. After that, we found miR-337-3p was increased by the knockdown of circRNA_100565 in A549/DDP and H1299/DDP cells (Fig. 3E). Additionally, miR-337-3p was down-regulated in DDP-resistant NSCLC tissues and cells compared with the controls (Fig. 3F and G), and also was negatively correlated with circRNA_100565 expression in tissues (Fig. 3H). In all, we confirmed that circRNA_100565 directly interacted with miR-337-3p and negatively regulated its expression in NSCLC cells.
Multivariate analysis for factors related to overall survival using the COX proportional hazard model
Multivariate analysis for factors related to overall survival using the COX proportional hazard model
*
Knockdown of circRNA_100565 sensitizes NSCLC resistant cells to DDP by sponging miR-337-3p. A549/DDP and H1299/DDP cells were transfected with sh-NC, sh-circRNA_100565, sh-circRNA_100565 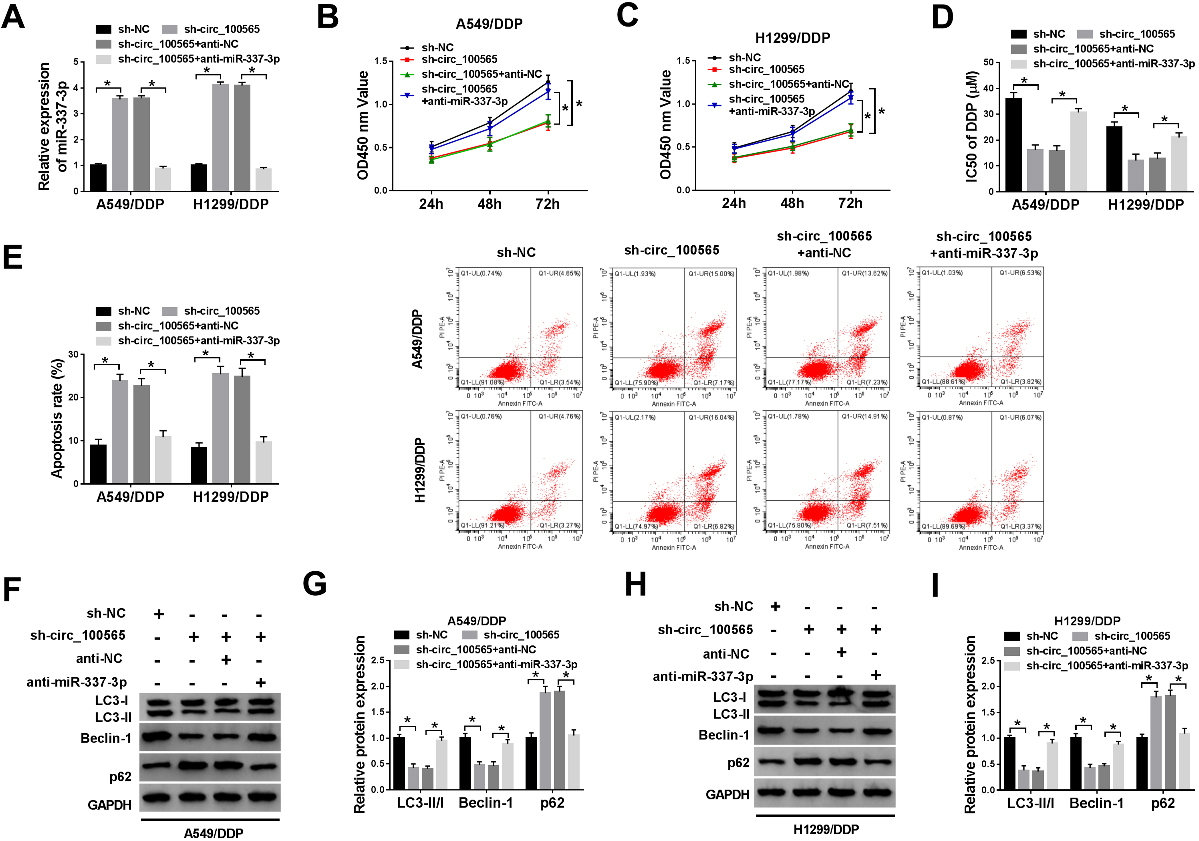
ADAM28 is a target of miR-337-3p and circRNA_100565 regulates ADAM28 by sponging miR-337-3p. (A) The putative binding site between ADAM28 and miR-337-3p was listed. (B, C) The interaction between ADAM28 and miR-337-3p was confirmed by the dual-luciferase reporter assay in A549/DDP and H1299/DDP cells. (D) The expression of ADAM28 in NSCLC tissues was measured using qRT-PCR and western blot. (E, G) The correlation between miR-337-3p and circRNA_100565 or ADAM28 was analyzed using Spearman’s correlation test. (F) ADAM28 expression in A549/DDP and H1299/DDP cells transfected with miR-NC or miR-337-3p was detected using qRT-PCR. (H, I) ADAM28 protein expression was determined using western blot in A549/DDP and H1299/DDP cells transfected with sh-NC, sh-circRNA_100565, sh-circRNA_100565 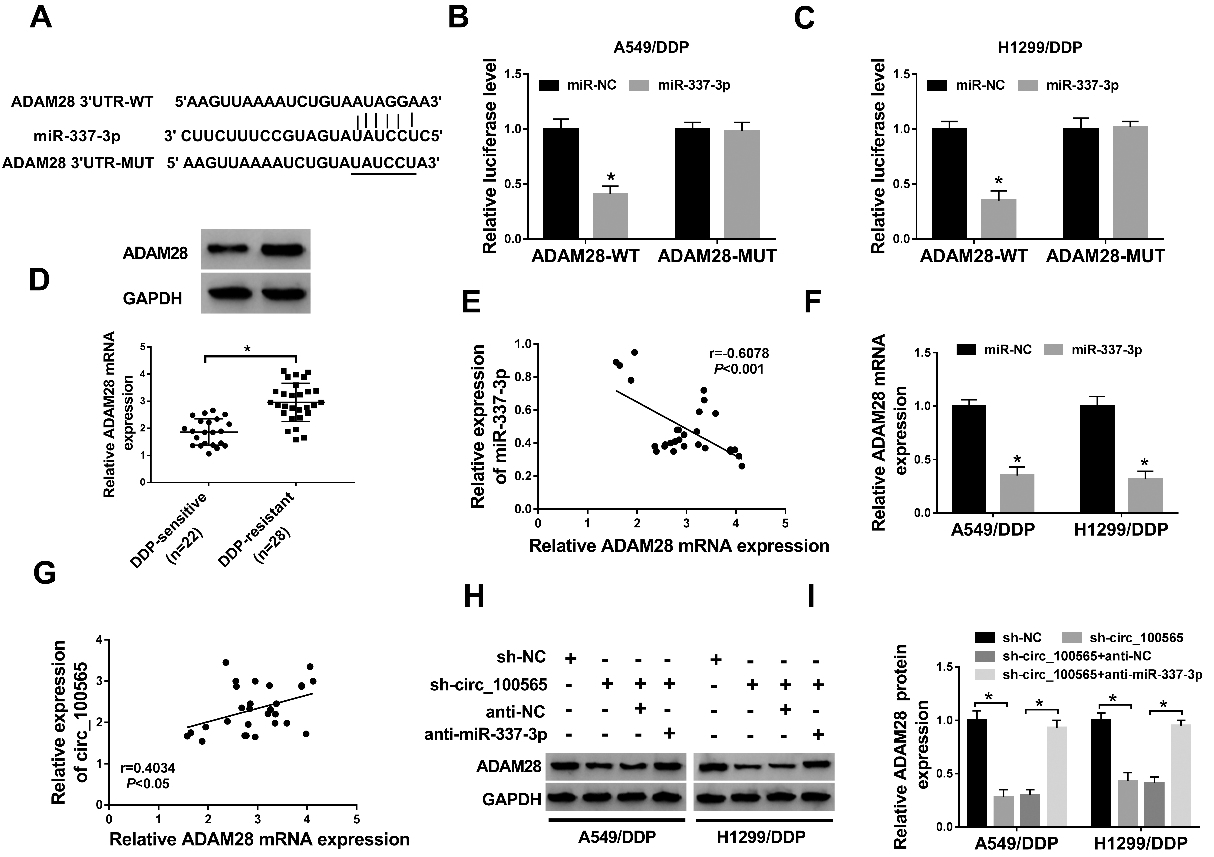
Based on the relationship between circRNA_100565 and miR-337-3p, we further gained insight into whether miR-337-3p involved in circRNA_100565 deletion-induced inhibition of DDP resistance in NSCLC. A549/DDP and H1299/DDP cells were transfected with sh-NC, sh-circRNA_100565, sh-circRNA_100565
Knockdown of circRNA_100565 promotes the sensitivity of resistant cells to DDP in NSCLC by regulating ADAM28 inhibition. A549/DDP and H1299/DDP cells were transfected with sh-NC, sh-circRNA_100565, sh-circRNA_100565 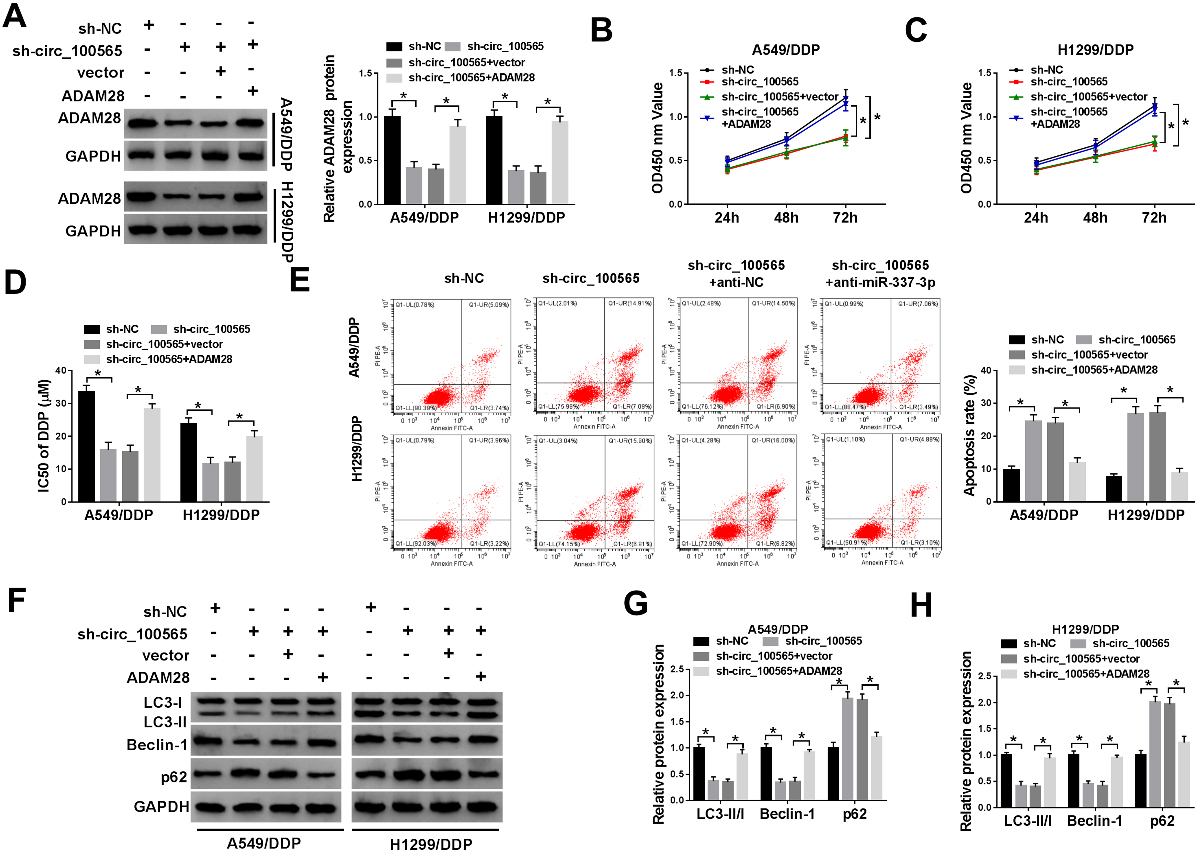
The target genes of miR-337-3p were further explored by using Targetscan online database, results showed that ADAM28 had putative binding sites in miR-337-3p (Fig. 5A). Then the declined luciferase activity in A549/DDP and H1299/DDP cells co-transfected with ADAM28-WT and miR-337-3p confirmed the interaction between miR-337-3p and ADAM28 (Fig. 5B and C). ADAM28 expression was found to be elevated in DDP-resistant NSCLC tissues (Fig. 5D), and was negatively correlated with miR-337-3p expression (Fig. 5E); importantly, ADAM28 expression in A549/DDP and H1299/DDP cells was reduced by miR-337-3p mimics (Fig. 5F). All these results suggested that miR-337-3p targeted ADAM28 and negatively regulated its expression in NSCLC cells. In addition, we also observed a positive correlation between ADAM28 and circRNA_100565 expression in NSCLC (Fig. 5G); synchronously, it was found that ADAM28 expression was inhibited by circRNA_100565 deletion, but was up-regulated by following miR-337-3p inhibition (Fig. 5H and I). Altogether, we confirmed that circRNA_100565 could indirectly regulate ADAM28 expression by serving as a sponge of miR-337-3p in NSCLC cells.
CircRNA_100565 deletion contributes to DDP-mediated repression on NSCLC tumor growth in vivo. (A) Tumor volumes were calculated every 7 days. (B) Mice were killed on day 35 days, and then tumor masses were excised and weighed. (C, D) The expression of circRNA_100565 and miR-337-3p were determined using qRT-PCR. (E) Western blot was used to detect the level of ADAM28 and autophagy-associated protein. (F) The expression levels of Ki-67, ADAM28 and LC3-II in tumors were analyzed using ICH. *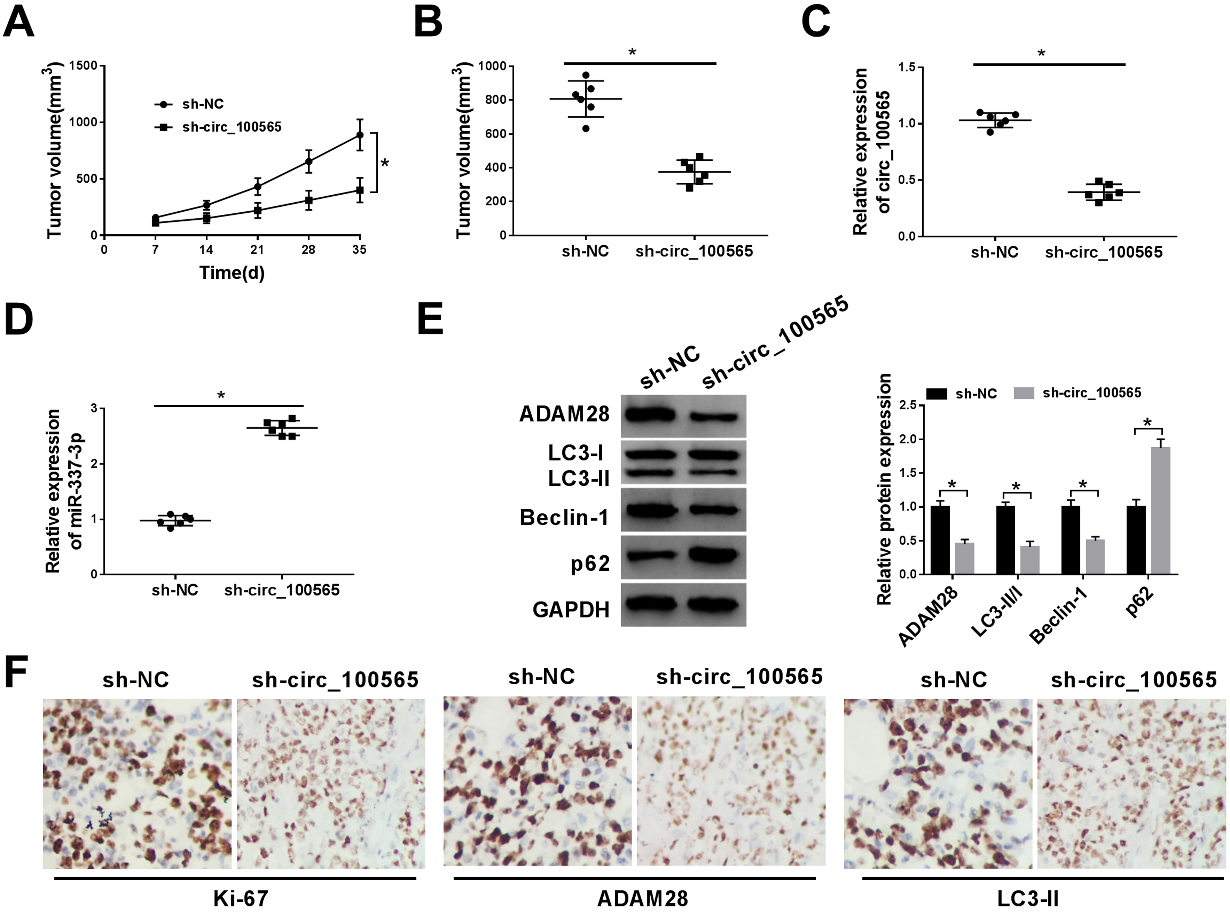
Due to the regulatory network of circRNA_100565/ miR-337-3p/ADAM28, we wanted to know whether ADAM28 participated in the regulation of circRNA_ 100565 deletion on DDP resistance in NSCLC cells. Firstly, A549/DDP and H1299/DDP cells were transfected with sh-NC, sh-circRNA_100565, sh-circRNA_ 100565
CircRNA_100565 deletion contributes to DDP-mediated repression on NSCLC tumor growth in vivo
We further investigated the effects of circRNA_ 100565 on DDP-mediated tumor growth in vivo. As shown in Fig. 7A and B, circRNA_100565 silencing reinforced DDP-induced repression on the tumor growth, reflected by the decreased tumor volume and lowered tumor weight in sh-circRNA_100565-transfected H1299/DDP cells group. Moreover, molecular analyses displayed circRNA_100565 deletion reduced the level of circRNA_100565 and ADAM28, but promoted the level of miR-337-3p in excised tumor masses from sh-circRNA_100565-transfected group (Fig. 7C–E). Meanwhile, western blot also showed circRNA_100565 deletion decreased the LC3II/I ratio and Beclin-1 level, while increased p62 expression in tumor masses from the sh-circRNA_100565-transfected group (Fig. 7E). Besides that, the ICH assay indicated circRNA_100565 knockdown decreased the level of Ki-67, ADAM28 and LC3-II in tumors (Fig. 7F). Collectively, all these results implicated circRNA_100565 deletion reinforced DDP-mediated repression on NSCLC tumor growth in vivo through suppressing autophagy by regulating ADAM28 and miR-337-3p.
Discussion
Although recent chemotherapeutics have markedly improved the outcome of patients with advanced NSCLC, invariably, almost all NSCLC patients become chemoresistance accompanied by distant metastasis in the end recurrence [4, 22]. Thus, clarifications of the mechanism of chemoresistance in NSCLC and identifications of new therapeutic markers might provide novel strategies to improve the prognosis of NSCLC. In this study, we found circRNA_100565 was up-regulated in NSCLC tissues, especially in DDP-resistant tissues, its high expression was associated with shorter overall survival in patients; what’s more, univariate and multivariate analysis revealed that circRNA_100565 expression represented a significant independent prognostic marker in NSCLC. Furthermore, circRNA_100565 was also elevated in DDP-resistant NSCLC cell lines, then knockdown of circRNA_100565 mitigated DDP resistance in NSCLC by inducing apoptosis and inhibiting proliferation and autophagy in cells. Besides that, the murine xenograft model analysis also showed that circRNA_100565 knockdown contributed to the anti-tumor effects of DDP on NSCLC in vivo.
At present, stimulation of cell death and suppression of cell survival are the key principles of cancer therapy; most anticancer therapies, such as chemo-, radio- and immunotherapy primarily function by inducing cell death including apoptosis and autophagy in cancer cells [23, 24]. Currently, growing evidence has indicated that circRNAs are involved in cell viability, apoptosis and autophagy in NSCLC. For example, circ-HIPK3 induced cell proliferation and repressed cell apoptosis in NSCLC by interacting with miR-149 to promote tumor progression [25]. Circ-HIPK3 facilitated NSCLC development by impairing cell autophagy and induced cell proliferation and metastasis through miR-124-3p-STAT3-PRKAA/AMPK
It has been reported that circRNAs function as miRNA sponges to regulate gene expression [29]. Here, we further investigated the underlying mechanism of circRNA_100565 on the DDP-resistance of NSCLC. We found miR-377-3p directly bound to circRNA_100565 or ADAM28 3’-UTR. Moreover, circRNA_100565 could indirectly regulate ADAM28 expression by sponging miR-377-3p in NSCLC cells. Additionally, circRNA_100565 deletion-induced sensitivity of NSCLC resistant cells to DDP could be remarkably attenuated by miR-377-3p inhibition or ADAM28 re-expression.
Previous studies have clarified that miR-337-3p was a crucial biomarker in many cancers progression. For example, miR-337-3p inhibition inhibited CoCl
In conclusion, these findings demonstrated that circRNA_100565 might serve as a significant independent prognostic marker in NSCLC; besides, circRNA_100565 sensitized NSCLC cells to DDP by regulating cell proliferation, autophagy and apoptosis via miR-337-3p/ADAM28 axis, providing a novel insight into the development of the therapeutic strategy to boost the effectiveness of NSCLC chemotherapy.
Footnotes
Conflict of interest
The authors declare that they have no competing interests.
Funding
This study was supported by Natural Science Foundation of Hainan Province (Grant No. 818MS147).
