Abstract
Introduction
Gastric cancer (GC) represented one of the most prevalent cancers at the recording of the latest global epidemiological data, ranking fifth in incidence (4.9%) and mortality (6.8%). 1 Chemotherapy represents the primary clinical intervention for GC. Cisplatin (CDDP) is a widely utilized chemotherapeutic agent. The platinum-based compounds induce oxidative stress damage in tumors by swiftly and transiently elevating the levels of reactive oxygen species (ROS). However, studies demonstrate that upon initial use of CDDP, tumor cells are effectively eliminated. Nevertheless, prolonged application leads to tumor cells adapting their antioxidant systems to withstand the cytotoxic effects of platinum-based chemotherapy drugs, ultimately resulting in the development of drug resistance. 2 Therefore, pinpointing how to lessen CDDP resistance has become a highlight in current research.
Studies have shown that there is a connection between cellular oxidative system imbalance and ferroptosis. Dysregulation and abnormal expression of various redox-active enzymes drive an imbalance in oxidative stress, leading to ferroptosis. 3 Ferroptosis is a form of cell demise that occurs when there is an excessive buildup of reactive oxygen species (ROS) reliant on iron ions within the cell. In the study, glutathione peroxidase 4 (GPX4) converted the substrate GSH to GSSG while simultaneously reducing toxic lipid peroxides in the cell to non-toxic lipid alcohols (L-OH) or converting free H2O2 to water, thereby repairing lipid cellular oxidative damage. Inactivation of GPX4 led to a decrease in the cell’s antioxidant capacity and an increase in lipid reactive oxygen species. With inadequate GPX4 to eliminate the excessive accumulation of ROS, an imbalance ensued in the homeostasis between ROS generation and breakdown. Reactive oxygen species (ROS) have the ability to engage with polyunsaturated fatty acids on the lipid membrane, resulting in the creation of a lipid radical L• and prompting lipid peroxidation, leading to the formation of Lipid-ROS. 4 When the cell’s intrinsic antioxidant capacity was insufficient to clear the excessive accumulation of lipid-ROS, it led to cell death through ferroptosis. 5 Malondialdehyde (MDA) is one of the products of lipid peroxidation and also one of the markers of free radical oxidation, associated with oxidative stress occurring in cancer cells as well as being related to ferroptosis. In previous colorectal cancer research, scholars found that Lysionotin could induce ferroptosis by promoting Nuclear respiratory factor 2 (Nrf2) degradation in the proteasome to regulate the accumulation of ROS, thereby inhibiting CRC cell proliferation, migration, and invasion. 6 In preclinical models of pancreatic cancer, the ferroptosis inducer MMRi62 inhibited tumor growth and prevented its metastasis by inducing ferroptosis. 7 However, at present, the approach of using increased ferroptosis to overcome resistance to platinum-based chemotherapy and to slow tumor progression has not been thoroughly investigated.
Curcumol (CUR), a sesquiterpene compound with anticancer pharmacological activity, is an effective ingredient in herbs of the Zingiberaceae family. 8 It is a commonly used traditional Chinese medicine, and one of the active components of turmeric and zedoary. 9 In liver fibrosis research, it was noteworthy that CUR alleviated the progression of liver fibrosis by inducing autophagy and ferroptosis in hepatic stellate cells. 10 In prostate cancer research, CUR could promote cancer cell ferroptosis by activating the antioxidant core NRF2/HO-1 signaling pathway. Subsequent to drug administration, levels of Nrf2 and Heme Oxygenase-1 (HO-1) decreased, leading to reduced cell proliferation, decreased levels of SLC7A11 and GPX4, increased Fe2+ content, and elevated ROS fluorescence intensity. Overexpression of Nrf2 increased the expression of downstream protein HO-1 and reversed the drug’s impact on relevant indicators, thereby inhibiting cancer progression. 11 Additionally, in previous in vivo and in vitro studies, CUR has been found to inhibit resistance to various chemotherapeutic agents, including cisplatin.12,13 For instance, in gastric cancer, CUR reduced the resistance of GC cells to cisplatin through NF-κB and decreased the resistance of the drug-resistant cell line BGC-823/DDP to cisplatin through PI3K/AKT signaling pathways. 14 However, it remained unclear whether CUR could overcome resistance to platinum-based chemotherapy by increasing ferroptosis.
Therefore, we utilized a cisplatin-resistant cell model in vitro to investigate the synergy between CUR and CDDP in inducing ferroptosis through the KEAP1/P62/NRF2 pathway, alleviating the proliferation, migration, and invasion of GC cells. Additionally, we conducted gastric gavage therapy with CUR and CDDP on subcutaneous tumor-bearing mice to assess the efficacy of the CUR and CDDP combination. This study aimed to provide more reliable evidence for the clinical application of curcumin as an adjuvant to cisplatin treatment for gastric cancer.
Materials and Methods
Moral and Ethical Statement
The study was conducted in strict accordance with the World Medical Association’s Code of Ethics (Declaration of Helsinki). This study has passed the ethical review of Zhejiang Chinese Medicine University (approval number: IACUC-20230410-08).
Cell Culture
From the cells of national certification warehouse (https://www.cellbank.org.cn/) for human gastric cancer cells MKN-45 (ATCC Cat#CRL-1852, RRID:CVCL_0434). AGS (ATCC Cat#CRL-1739, RRID:CVCL_0139) and AGS/DDP was purchased from China Yuchi Biology Co., LTD. MKN-45/DDP from our laboratory was interventional by low dose induction. The construction of MKN45/DDP cells using low-dose induction is consistent with previous descriptions. 15 Briefly, logarithmically growing MKN-45 cells were exposed to Cisplatin (CDDP), and the cells that survived after 96 hours were cultured. Once the cells continued to grow to 80% confluence, CDDP was added again to repeat the procedure. The research experiment was conducted once the RI index met the required standards. DMEM/F12K medium supplemented with 10% fetal bovine serum was used. Constant temperature 37°C, 5%CO2 incubator. In order to maintain the ability of cisplatin introduction, AGS/DDP and MKN-45/DDP cells were cultured with 1 and 1.5 μg/ml cisplatin, respectively. Curcumol (CUR, Cat#HY-N0104) and CDDP (Cat#HY-17394) were purchased from MedChemExpress (Monmouth Junction, NJ, USA).
Cell Viability and Drug Resistance Index Were Measured
The cells were treated with CUR or CDDP. Cell Counting Kit-8 (Epizyme Biotech Cat#CX001L, RRID: SCR_019090) was used to detect cell viability. According to the instructions, 10 µl CCK-8 reagent was added to each well and the cells were incubated at 37°C for 30 minutes. Absorbance was measured at 450 nm to determine cell viability. OD values were used to calculate cell viability and Graph pad 8.0 software was used to calculate cell IC50. The resistance index (RI) is calculated as follows: RI = IC50 of drug-resistant cell line/IC50 of parental cell line.
Cell Proliferation Capacity
The 5-ethyl-2′-deoxyuridine (EdU, Beyotime Biotechnology, Cat#ST067, RRID: SCR_003160) assay first inoculated the cells into a 6-well plate, followed by either CUR or CDDP intervention. After intervention, plasma cells were fixed and incubated with EdU dye for 1 hour. After cleaning, DAPI was added for nuclear staining, and the proliferation of cells was observed under fluorescence microscope.
Transwell
In the migration experiment, the cells were first implanted in 6-well plates with CUR (50 μM) or CDDP (75 μM) added to each group, respectively. Cells were collected after 24 hours. The cells are digested and centrifuged, discarding the old medium. Suspension and counting were performed using an FBS-free medium. Six hundred microliters of fetal bovine serum was added to the lower cavity. Two hundred microliters cell suspension was added to the upper chamber. The count concentration was 3 × 105 cells/ml. Cell culture for 24 hours. Subsequently, the upper chamber was separated, cleaned, fixed in 4% paraformaldehyde for 10 minutes, and then stained in 0.1% crystal violet dyeing solution for 30 minutes. Subsequently, the membrane was removed and photographed under a microscope. In the invasion experiment, substrate glue was added prior to the above steps and the incubator was hydrated for 24 hours.
ROS
Reactive oxygen species (ROS) content was determined by ROS Assay Kit (Beyotime Biotechnology, Cat#S0033S). The cells were cultured in 6-well plates. After the cell density healed to 60% to 70%, CUR (50 μM) or CDDP (75 μM) was added for 24 hours. In the cell experiment, the treated cells were added into the 1:1000 diluted DCFH-DA probe according to the instructions, incubated for 20 minutes in the dark, and then observed and photographed under the confocal laser microscope.
MDA
For malondialdehyde analysis (MDA, Beyotime Biotechnology, Cat#S0131S), a 12-well plate was implanted with 3 × 104 cells per well. The microscopic density was fused to about 70%. Then the CUR (50 μM) or CDDP (75 μM) was added for 24 hours. Cells and tissues are collected and cleaved in a liquid homogenate for detection. Each sample was added with 0.2 ml of MDA detection working fluid. Mix well and heat in a boiling water bath at 100°C for 15 minutes. After cooling, centrifuge at 1000g room temperature for 10 minutes. Finally, 200 μl supernatant was taken and implanted into the 96-well plate. The concentration of MDA was calculated using the absorbance obtained at 532 nm.
GSH and GSSG
Reduced glutathione (GSH) and oxidized glutathione disulfide (GSSG) concentrations were determined using the GSH/GSSG Ratio Fluorimetric Detection Assay Kit (YESEN, Cat#50120ES70). The cells were implanted in 12-well plates in a number of 3 × 104 cells. When the cell density under the microscope reaches about 70%. Add CUR (50 μM) or CDDP (75 μM) for 24 hours. Cells were collected after treatment. A working solution for glutathione detection was added to each sample. The concentrations of total glutathione and GSSG were measured by the fluorescence reading value under the fluorescence enzyme label, and the absorbance was calculated at Ex/Em = 490/520 nm. Formula: GSH = total glutathione − GSSG × 2.
BODIPY™ 581/591 C11
Lipid peroxidation levels are the same as before. The cells were incubated in a 6-well dish with 5 μM BODIPY 581/591C11 dye (Invitrogen, Cat#D3861, RRID: SCR_020123) at 37°C for 30 minutes. Subsequently, the cells were washed with PBS and stained with PI for 5 minutes. Flow cytometry was performed on the cells using Agilent NovoCyte flow cytometer (RRID: SCR_020330). Flowjo software (version 10.5.3, RRID: SCR_008520) was used to export and draw the results.
Protein Extraction and Western Blot
Equal amounts of RIPA (Thermo Fisher Scientific, Cat#89900) and the mixture of protease inhibitors and phosphatase inhibitors were added into the cleaned cells, and the supernatant was collected by centrifugation after complete lysis. BCA (Beyotime Biotechnology, Cat#P0010) was quantified according to instructions, adding 5X loading buffer (Beyotime Biotechnology, Cat#P0015) and boiling in water at 100°C to achieve denaturation. The protein molecular weight marker and the prepared sample were added to the prepared SDS-PAGE plate. The target protein was isolated at 80 V voltage. Under cooling conditions, the proteins were transferred to NC (Millipore, Cat#IPVH00010) membranes at constant pressure of 100 V using a conventional Tris-Glycine transfer buffer for 2 hours. Then it was sealed with 5%BSA (Sangon biotech, Cat#A600903) at room temperature for 1 hour. Finally, the NC membrane is placed into the target antibody. Overnight treatment at 4°C. On the second day, the antibodies were removed, TBST was washed, and the anti-rabbit IgG was incubated at room temperature for 1 hour. Finally, ELC (GE HealthCare, Cat#RPN810) was used for imaging on the instrument. Results image J software was used for quantitative analysis. The primary antibody was removed by stripping method, and the other antibodies were incubated. Table 1 describes the antibodies in detail.
Antibody Information.

In table, we explained the detailed information of the target used in the Western blot experiment, including brand, article number and dilution ratio.
Subcutaneous Tumor Formation in Nude Mice
BALB/c nude mice (male, SPF grade, 4-6 weeks old, 18-22 g, RRID:IMSR_JAX:000011) were purchased from Animal Experimental Center of Zhejiang Chinese Medicine University. The mice were placed in a light-dark cycle environment for 12 hours at a temperature of 23 ± 2°C, humidity of 55 ± 5%, and fed standard laboratory food and water. 5 × 106 AGS/DDP cells were injected subcutaneously into the left axilla of each mouse to establish a xenograft tumor model. Matrix adhesive was used to assist (Corning Incorporated, Cat#354248). The tumor volume of 100 mm3 suggests that xenotransplantation mouse model can be effectively established. Administration was performed by intragastric administration and was grouped as follows: control (saline 0.2 ml/10 g/day), CUR (30 mg/kg once a day), CDDP (6 mg/kg once a week) and CUR (30 mg/kg daily) + CDDP (6 mg/kg once a week). Tumor volume and weight were assessed at 3-day intervals. After 21 days, the mice were killed with an overdose of barbiturates (150 mg/kg, intraperitoneal injection), and the tumors were obtained in later experiments.
Evaluation of Combined Drug Use
Refer to the previous experiment. Cells cultured with CUR or CDDP alone or in combination with CUR + CDDP were cultured using the OD values obtained by CCK8 to calculate cell viability, and the data were inserted into SynergyFinder3.0 (http://synergyfinder.org) to obtain the drug combination index.
Statistics
Statistical analyses were performed using Student’s t-test for two groups and a one-way analysis of variance (ANOVA) with post hoc analysis for multiple comparisons. Data analyses were conducted using GraphPad Prism software version 8.0.2. P < .05 as the statistical difference was considered significant.
Results
CUR Synergizes With CDDP to Enhance Sensitivity in Resistant Gastric Cancer Cells
The resistance index (RI) is commonly used to determine whether cells exhibit drug-resistant characteristics by comparing the half-maximal inhibitory concentration (IC50) of resistant cells to that of parental cells. Cisplatin (CDDP) (0, 8, 16, 32, 64, 128, 256 μM) was administered to parental gastric cancer (GC) cells (AGS, MKN-45) and resistant cells (AGS/DDP, MKN-45/DDP) for 24 hours. The IC50 values for AGS and MKN45 cells were 10.46 and 10.07 μM, while for AGS/DDP and MKN45/DDP cells, they were 69.24 and 54.96 μM, respectively. The results showed the RI for AGS/DDP cells was 6.62 and for MKN45/DDP was 5.46 (Figure 1B). The research findings unequivocally demonstrate that both AGS/DDP and MKN45/DDP exhibit resistance to CDDP. This indicates a consistent and robust resistance pattern across both cell lines, laying a foundation for subsequent experiments involving curcumol (CUR) alone and in combination with CDDP. To confirm the inhibitory effect of CUR on GC cells, we intervened with CUR at concentrations ranging from 0 to 150 μM on AGS/DDP and MKN-45/DDP cells. For 24 and 48 hours, the IC50 values for AGS cells were 111.8 and 75.94 μM, while for AGS/DDP cells, they were 76.40 and 61.15 μM. Similarly, the IC50 values for MKN45 cells were 127.4 and 107.6 μM at the same time points, and for MKN-45/DDP cells, they were 103.2 and 86.57 μM (Figure 1C and D). The inhibitory effect of CUR on resistant cells was evident in the results, with the IC50 values of resistant cells being lower than those of parental cells at the same intervention time. To further validate the better therapeutic effect of the combined use of CUR + CDDP on resistant cells, we employed SynergyFinder3.0 for interactive analysis and visualization of multiple drug combination response data. The use of tools and the acceptance of results are the same as before. 16 The results indicated a synergistic effect of combined treatment with CUR + CDDP on AGS/DDP and MKN-45/DDP cells (Figure 1E). In conclusion, the above results demonstrate that the combined use of CUR + CDDP can enhance the sensitivity of resistant GC cell lines to cisplatin.

Inhibition of Curcumol on drug-resistant cells and synergistic effect on CDDP. (A) The chemical formula and chemical construction of Curcumol. (B) The IC50 index of AGS, MKN45, AGS/DDP, and MKN45/DDP. (C) Curcumol interfered with AGS/DDP and MKN45/DDP for 24 and 48 hours. (D) CDDP and CUR + CDDP act on AGS/DDP and MKN45/DDP, (E) and (F) Synergies 3.0 software calculator simulates the synergies between CUR and CDDP. **P < .01; N.S. P > .05 NC versus CDDP or CUR + CDDP group (X ± SE, cell experiments were repeated at least three times). One-way ANOVA with Dunnett’s post hoc test.
CUR Combined With CDDP Inhibited the Proliferation, Migration and Invasion of Drug-Resistant GC Cells
Based on confirming the synergistic action of CUR and CDDP, we conducted proliferation, migration, and invasion experiments on cisplatin-resistant cells. Upon separately adding CUR (50 μM) or CDDP (75 μM), the proliferation capacity of AGS and MKN-45 cells swiftly decreased (P < .05). When CUR was combined with CDDP, it further suppressed cell proliferation, both compared to the Control group and to the CUR or CDDP groups (P < .05). This effect was also observed in drug-resistant cells. The proliferation capacity of drug-resistant cells was inhibited upon the addition of CUR or CDDP separately (P < .05). When CUR and CDDP were used in combination, the proliferation capacity of drug-resistant cells was further inhibited compared to any single treatment. However, there was no superior inhibition of proliferation demonstrated in the experimental results when comparing the individual use of the CUR group to the CDDP group, whether in drug-resistant cells or parental cells (Figures 2A, 3B and C).

Inhibitory effects of CUR or CDDP on proliferation of AGS and AGS/DDP. (A) Proliferation inhibition effects of CUR or CDDP alone and in combination on AGS and AGS/DDP.

Inhibitory effects of CUR or CDDP on proliferation of MKN-45 and MKN45/DDP. (B) Proliferation inhibition effects of CUR or CDDP alone and in combination on MKN45 and MKN45/DDP. (C) Quantitative and statistical analysis of pictures Figure 2 and B. *P < .05 **P < .01 NC versus CUR or CDDP or CUR + CDDP group; N.S. P > .05 CDDP versus CUR; ## P < .01 CUR or CDDP versus CUR + CDDP (X ± SE, cell experiments were repeated at least three times). One-way ANOVA with Dunnett’s post hoc test.
Through migration and invasion assays on parental and cisplatin-resistant GC cell lines, the migratory and invasive potential of GC cells, particularly drug-resistant cells, was evaluated. The experimental results indicate that in parental cells, the addition of CUR (50 μM) or CDDP (75 μM) led to a significant reduction in the number of migratory cells compared to the control group (P < .05). Specifically, in AGS cells, the reductions were 40.2% and 58.9%, while in MKN-45 cells, the reductions were 55.8% and 66.4%. When CUR and CDDP were used in combination, the number of migrating cells decreased further compared to the separate use of CUR or CDDP (P < .05), with AGS showing an 81.7% reduction and MKN-45 an 84.2% reduction. In cisplatin-resistant cells, after CDDP treatment, the number of migrating cells in AGS/DDP decreased by 6% and in MKN45/DDP by 1.2% compared to the control group, but these differences were not statistically significant (P > .05). However, treatment with CUR after CDDP resulted in a reduction in the number of migratory cells in resistant cell lines, specifically a decrease of 19.4% in AGS/DDP and 18% in MKN45/DDP. The combination of CUR and CDDP further inhibited cell migration, with AGS/DDP reducing by 85.1%, 84.2%, and 79.6%, and MKN45/DDP by 48.1%, 40.9%, and 36.9%. In parental cells, the CDDP group exhibited stronger inhibition compared to the CUR group (P < .05), whereas in resistant cells, the CUR group had fewer migrating cells than the CDDP group (Figure 4A and B). Similar results were observed in invasion assays. In parental cells, both CUR and CDDP groups had a lower number of invasive cells than the control group (P < .05), with AGS showing 78.4% and 55.6% reductions, and MKN45 showing 37.3% and 43.6%. In AGS, the CUR group had 52.48% fewer invasive cells compared to the CDDP group (P < .05), while in MKN-45, there was a reduction of −11.1% with no statistical difference (P > .05). When CDDP and CUR were used together, the number of invasive cells gradually decreased compared to the control group or when CDDP or CUR were used separately (P < .05), showing reductions of 86.5%, 69.6%, and 36.7% in AGS and 85.5%, 74.4%, and 76.9% in MKN-45. In resistant cells, CDDP treatment resulted in a slight decrease in invasive cells compared to the control group, with AGS/DDP decreasing by 10.1% and MKN45/DDP by 7.1%, but these results lacked statistical significance (P > .05). Compared to the control or CDDP groups, CUR effectively reduced the number of invasive cells (P < .05), showing reductions of 68.5% and 60.5% in AGS/DDP and 37% and 33.2% in MKN45/DDP. When comparing the combination of CDDP and CUR to the control, CDDP, or CUR groups, further inhibition of invasion was observed in AGS/DDP (reductions of 75.7%, 73%, and 22.4%) and MKN45/DDP (76.5%, 74.4%, and 62.2%). The occurrence of migration and invasion suggests further tumor deterioration, highlighting the importance of inhibiting these processes. This study clearly demonstrates that both CUR and CDDP, whether used alone or in combination, effectively suppress the proliferation, migration, and invasion of both parental and drug-resistant cells (Figure 5A and B).

Effects of CUR or CDDP alone and in combination on the migration ability of GC cells. (A) Effects of CUR or CDDP alone and in combination on AGS, AGS/DDP, MKN45 and MKN45/DDP migration capacity. (B) Statistical analysis of migration interventions. *P < .05 **P < .01 N.S. P > .05 NC versus CUR or CDDP or CUR + CDDP group; N.S. P > .05 $ P < .05 $ P < .01 CUR versus CDDP; ## P < .01 CUR or CDDP versus CUR + CDDP (X ± SE, cell experiments were repeated at least three times). One-way ANOVA with Dunnett’s post hoc test.
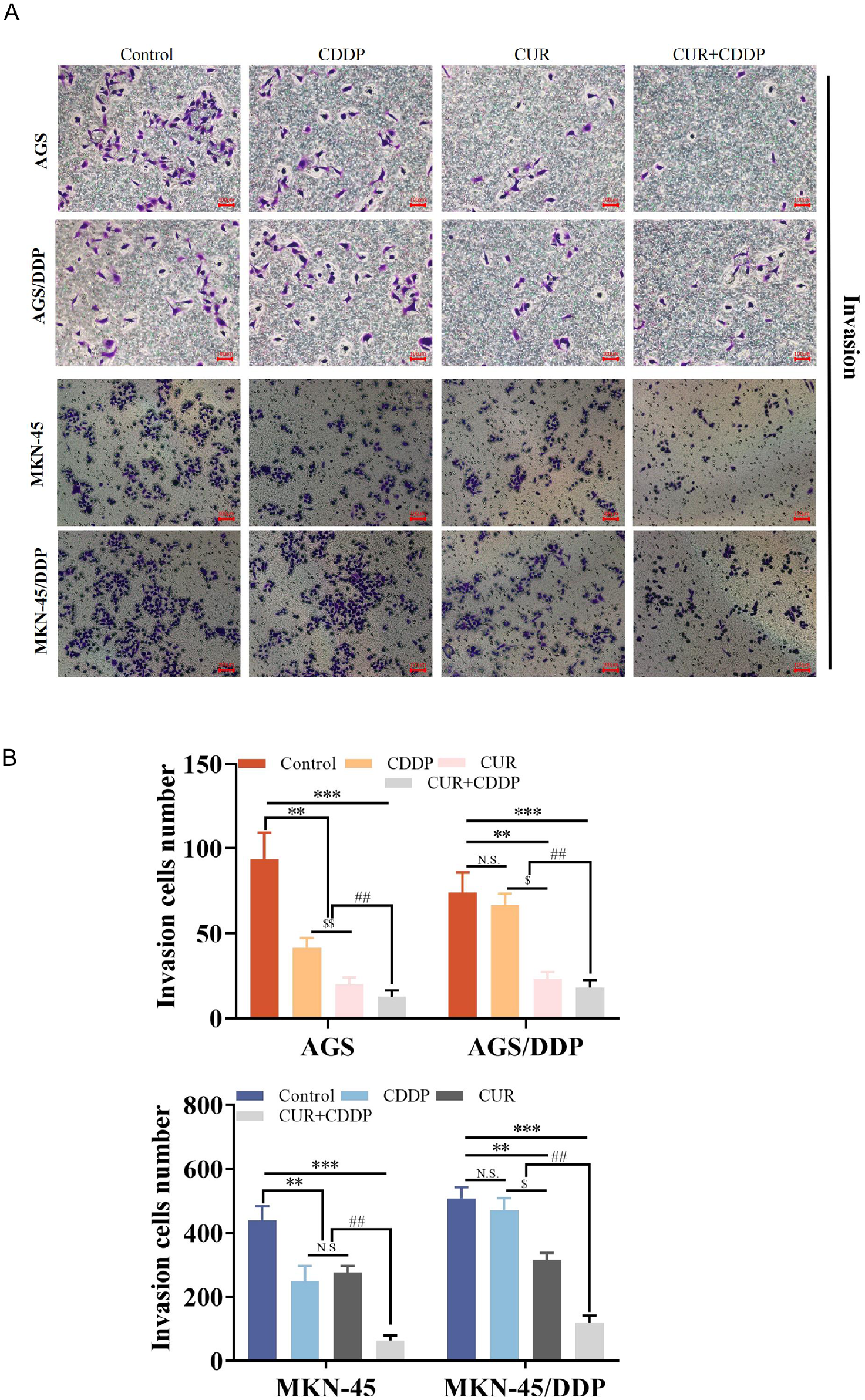
Effects of CUR or CDDP alone and in combination on the invasion ability of GC cells. (A) Effects of CUR or CDDP alone and in combination on the invasion capacity of AGS, AGS/DDP, MKN45, and MKN45/DDP. (B) Statistical analysis of invasion interventions. *P < 0.05 **P < .01 N.S. P > .05 NC versus CUR or CDDP or CUR + CDDP group; N.S. P > .05 $ P < .05 $ P < .01 CUR versus CDDP; ## P < .01 CUR or CDDP versus CUR + CDDP (X ± SE, cell experiments were repeated at least three times). One-way ANOVA with Dunnett’s post hoc test.
CUR and CDDP Synergistically Induce GC Cell Ferroptosis
To validate the effects of CUR and CDDP on the antioxidant levels and ferroptosis of cisplatin-resistant GC cells, we conducted validation in AGS/DDP and MKN-45/DDP cells. In both AGS/DDP and MKN45/DDP cells, CUR and CDDP alone could induce cellular ROS to a certain extent (Figure 6A). However, when CUR and CDDP were used in combination, the ROS levels significantly increased. We further verified the expression of MDA levels, and found that CUR or CDDP could induce MDA expression in AGS/DDP cells, but this induction did not have statistical significance in MKN-45/DDP cells (P > .05; Figure 6B). CUR + CDDP can further induce the expression of MDA (P < .05). When CDDP was used alone, it could induce an increase in iron content, while CUR alone, the iron content in AGS/DDP increased, but the increase in iron content in MKN45/DDP compared to the NC group was not statistically significant. In the Fe content determination, CUR and CDDP alone can increase the Fe content of AGS/DDP. However, when CUR was used alone, MKN45/DDP did not increase significantly compared with NC (P > .05). CDDP alone can increase the Fe content of MKN45/DDP. When using CUR + CDDP, Fe content was further increased in both AGS/DD and MKN45/DDP (Figure 6C). In terms of the core antioxidant indicator GSH/GSSG, CUR, or CDDP used alone could both inhibit the ratio. However, CUR + CDDP can further inhibit this ratio (Figure 6D). In addition, we used the C11 probe to detect Lipid-ROS (Figure 6E). The test results showed that CUR and CDDP alone could increase the C11 probe fluorescence content in AGS/DDP, while CUR + CDDP could further promote the expression of C11 fluorescence.

Curcumol or CDDP alone or in combination induce ferroptosis. (A) Curcumol or CDDP alone or in combination induce ROS. (B) Curcumol or CDDP alone or in combination intervened in MDA indicator trends. (C) Curcumol or CDDP alone or in combination intervened in Fe indicator trends. (D) Curcumol or CDDP alone or in combination intervened in GSH/GSSG ratio. (E) Curcumol or CDDP alone or in combination intervened in C11. *P < .05 **P < .01 N.S. P > .05 NC versus CUR or CDDP or CUR + CDDP group; # P < .05 CUR or CDDP versus CUR + CDDP (X ± SE, cell experiments were repeated at least three times). One-way ANOVA with Dunnett’s post hoc test.
CUR Collaborates With CDDP to Induce Ferroptosis in Resistant GC Cells Through P62/KEAP1/NRF2 Signal Pathway
To further validate the correlation between ferroptosis and cisplatin resistance, we first assessed the differential expression of NRF2 and GPX4 in CDDP-resistant cells compared to parental cells. Quantitative analysis revealed increased expression of NRF2 in both CDDP-resistant AGS/DDP and MKN45/DDP cells, with amplifications of 5.8 and 8.1 times, respectively, compared to AGS and MKN-45. Additionally, GPX4 expression in AGS/DDP and MKN45/DDP was amplified 1.7-fold and 2.9-fold compared to AGS and MKN-45 (Figure 7A-C). We further investigated the changes in the expression of key ferroptosis proteins, including GPX4, and the P62/KEAP1/NRF2 pathway along with the downstream protein NQO1, after interventions with CUR and CDDP in CDDP-resistant cells. Quantitative analysis indicated that after combined treatment with CUR and CDDP, NRF2 decreased by 4.3-fold, KEAP1 increased by 1.8-fold, p-p62 decreased by 0.9-fold, NQO1 decreased by 2.1-fold, and GPX4 decreased by 6.4-fold compared to the untreated control group. Moreover, when CDDP and CUR were used individually as controls, no significant modulation of NRF2, KEAP1, or NQO1 was observed with CDDP alone, while CUR demonstrated minor modulation across all proteins. Both CDDP and CUR alone downregulated the expression of the ferroptosis-related protein GPX4 by 0.8-fold and 1.6-fold, respectively, which was less effective than their combined use (Figure 7D and E). These findings collectively indicate that CUR in combination with CDDP effectively inhibits the P62/KEAP1/NRF2 signaling pathway and the activity of downstream proteins NQO1, while also suppressing the expression of the key ferroptosis protein GPX4 in CDDP-resistant cells in vitro.

CUR and CDDP interfere with the P62/KEAP1/NRF2 signaling pathway. (A) Differences in GPX4 and NRF2 expression between AGS and MKN45 and Cisplatin-resistant AGS/DDP and MKN45/DDP. (B) Statistical analysis of NRF2 protein expression. (C) Statistical analysis of GPX4 protein expression. (D) Differences in protein expression of P62/KEAP1/NRF2/NQO1 and GPX4 after CUR and CDDP intervention. (E) Figure (D) was quantitatively and statistically analyzed. *P < .05 **P < .01 ***P < .005 N.S. P > .05 NC versus CUR or CDDP or CUR + CDDP group (X ± SE, cell experiments were repeated at least three times). One-way ANOVA with Dunnett’s post hoc test.
CUR Collaborates With CDDP to Induce Ferroptosis In Vivo
To validate the synergistic effect of CUR and CDDP in vivo, we conducted subcutaneous tumor transplantation experiments using AGS/DDP in nude mice to evaluate the anticancer activity of CUR in combination with CDDP. The experimental results indicated that at the end of the 21-day experiment, the tumor weight in the control group receiving saline injections was 1361 ± 200 mg. The group receiving CDDP alone had a tumor weight of 1258 ± 492 mg. The CUR treatment group weighed 981 ± 322 mg, while the group receiving the combination of CUR and CDDP had a tumor weight of 709 ± 254 mg. The intervention of CDDP showed some individual differences in the subcutaneous tumor formation of cisplatin-resistant cell lines, but overall, the intervention of CDDP did not have a significant effect (P > .05; Figure 8A). After CUR intervention, there was a significant reduction in the size of the subcutaneous tumors in nude mice (P < .05). Furthermore, when CUR and CDDP were used in combination, the weight and size of the subcutaneous tumors in nude mice were further suppressed (P < .05; Figure 8B-D). Upon euthanasia of the nude mice, we obtained dissected subcutaneous tumors and performed protein extraction, followed by another validation of the expression of the antioxidant pathway p62/keap1/nrf2 and the ferroptosis-related marker GPX4. The results clearly indicated that both CUR and CDDP could inhibit the expression of NRF2 and GPX4 in vivo (P < .05). CDDP was unable to intervene in the expression of KEAP1 and phosphorylated p62 (P > .05), while CUR was able to do so (P < .05). CDDP activated the expression of NQO1 (P < .05), while CUR did not show significant differences. When CUR and CDDP were used in combination, the regulation of p62/keap1/nrf2 and GPX4 was further significantly adjusted (P < .05), but NQO1 did not exhibit significant differences (P > .05; Figure 8E and F).

The synergistic effect of CUR and CDDP was verified in vivo. (A) Tumor picture. (B) Tumor weight statistics. (C) Tumor volume statistics. (D) Mice body weight statistics. (E) Expression verification of P62/KEAP1/NRF2/NQO1 and GPX4 by tumor extracted proteins. (F) Figure (E) was quantitatively and statistically analyzed. *P < .05 **P < .01 N.S. P > .05 NC versus CUR or CDDP or CUR + CDDP group (X ± SE, n =5). One-way ANOVA with Dunnett’s post hoc test.
Discussion
The mortality rate of gastric cancer (GC) remains high and it continues to be one of the most common causes of cancer-related deaths worldwide. 1 Platinum-based drugs are one of the main treatment options for clinical GC. Therefore, platinum resistance poses a significant challenge in the treatment of GC. 17 Oxidative stress is one of the primary mechanisms by which cisplatin (CDDP) exerts its anti-tumor effects, and it is closely associated with the cellular ferroptosis process. 18
Many studies have indicated that chemical compounds extracted from natural herbs are ideal adjuvants for chemotherapy, and they may have the potential to reverse chemotherapy resistance in GC cells. 19 Curcumol (CUR) has been confirmed in previous research to increase the sensitivity of normal cells to cisplatin chemotherapy drugs through the PI3K/AKT signaling pathway or the microRNA-7/NF-κB/Snail axis.14,20 Furthermore, in the study of cisplatin resistance mechanisms, some scholars have proposed that activating oxidative stress through the AKT/GSK-3β/NRF2/GPX4 pathway can enhance the sensitivity of resistant GC cells to cisplatin, promoting ferroptosis in GC cells. 21 Interestingly, in liver cancer research, CUR has been found to alleviate the progression of liver fibrosis by inducing autophagy and ferroptosis in hepatic stellate cells or by regulating ferroptosis-induced hepatocyte senescence through the HIF-1α/NCOA4/FTH1 pathway. 22 However, no researchers have proposed that CUR enhances the sensitivity of cisplatin-resistant GC cells by inducing ferroptosis.
In this study, it was proposed that CUR can enhance the sensitivity of resistant GC cells to CDDP by inducing ferroptosis, potentially mediated through the P62/KEAP1/NRF2 pathway. Initially, we established cisplatin-resistant cells MKN45/DDP and obtained AGS/DDP from Shanghai Jinyuan Biotechnology Co., Ltd. to validate the cells’ resistance. CUR was used alone to induce growth inhibition in resistant cell lines to explore sensitivity. We observed that CUR effectively inhibited the growth of resistant cell lines in a dose-dependent manner, with no significant difference between 24 and 48 hours. Research by Wang et al indicated that CUR exhibited synergistic effects with CDDP in in vitro and in vivo studies of osteosarcoma. The combination of CUR and CDDP enhanced CDDP-induced apoptosis in osteosarcoma cells, inhibited M2-like macrophage activation, and enhanced anticancer effects. 13 To determine the ideal chemotherapeutic adjuvant role of CUR with CDDP, we analyzed the cell inhibition rates obtained from the CCK8 experiments using the synergy3.0 online tool and confirmed the synergistic effects of CUR and CDDP. Furthermore, we evaluated the changes in proliferation, migration, and invasion abilities between cisplatin-resistant GC cell lines and parental cell lines when CUR and CDDP were used alone or in combination. In parental cells, both CUR and CDDP alone effectively inhibited cell proliferation, migration, and invasion. When used in combination, these abilities were further suppressed. When experiments were conducted using drug-resistant cell lines, the group treated with CDDP alone showed relatively poor inhibitory capability compared to the control group. While CUR exhibited some inhibitory effects, its effectiveness was not ideal compared to the parental cell group. However, when CDDP was combined with CUR, the inhibition of proliferation, migration, and invasion significantly increased compared to using CUR or CDDP alone. In vitro studies have shown that inducing ferroptosis can inhibit cell proliferation, migration, and invasion. 23 Furthermore, research has found that cancers with high levels of metastasis and resistance are particularly susceptible to ferroptosis-inducing therapies. 24
Oxidative stress is one of the primary mechanisms by which cisplatin exerts its anti-tumor effects. Cisplatin interferes with mitochondrial homeostasis by forming complexes with mitochondrial DNA, inducing oxidative stress. When the abnormal increase of reactive oxygen species (ROS) exceeds the physiological and compensatory range, cell death occurs. 18 During long-term treatment with cisplatin, tumor cells gradually adapt and regulate the balance of ROS, ultimately leading to cisplatin resistance. 25 Therefore, targeting the antioxidant system, particularly the glutathione system, to maintain the dynamic balance of oxidation-reduction and regulate cisplatin resistance has been emphasized by many scholars. ent-kaurane diterpenoids, through the regulation of GSH and Prdx I/II to modulate ROS, exhibit a stronger anti-tumor effect in synergy with CDDP and overcome CDDP resistance in cancer. 26
The essence of ferroptosis is the depletion of intracellular GSH, reduced activity of GPX4 leading to the inability to catalyze redox metabolism, abnormal iron ion metabolism, accumulation of a large amount of ROS, and lipid peroxides triggering cell death. 27 Reversing chemotherapy resistance through the regulation of ferroptosis by the antioxidant system has gained attention. 28 The MKN45/DDP cell line, resistant to cisplatin, was found to be susceptible to the regulation of ferroptosis in GC chemotherapy resistance by Eremias multiocellata in combination with cisplatin. 29 Additionally, CUR was found to intervene in the progression of hepatic fibrosis by regulating ferroptosis. 10 Therefore, we further examined the molecular mechanism of ferroptosis in AGS/DDP and MKN45/DDP. The results clearly indicated that when CUR or CDDP was used alone in cisplatin-resistant GC cells, ROS showed a certain inductive capacity in AGS/DDP but did not exhibit significant effects in MKN45/DDP. However, when CUR and CDDP were used in combination, ROS induction was significant. The same scenario was observed in the MDA detection results as well. In the results of GSH/GSSG, both individual use of CUR or CDDP showed a downward trend, but this trend was further significantly enhanced when CUR and CDDP were used in combination. Just as in the C11 probe detection, CUR and CDDP exhibited induction effects individually, but when used together, the induction effects were further enhanced. These results collectively indicate that the combined use of CUR and CDDP can effectively induce ferroptosis in cisplatin-resistant cells.
Further investigation involved the detection of changes in the expression of relevant proteins, with GPX4 and NRF2 recognized as key targets of ferroptosis associated with chemotherapy resistance. Huang et al found that the expression of GPX4 in cisplatin-resistant GC cells was lower than that in the parent cells. 21 In contrast, Deng et al found that the expression of GPX4 in cisplatin-resistant NSCLC cell lines was higher than that in the parent cell lines. 30 In this study, the protein expression of GPX4 in AGS/DDP was higher than that in AGS cells, which is consistent with the findings of Deng et al. However, there was no significant difference in GPX4 expression between MKN45/DDP and MKN45. Chian et al found that in non-small cell lung cancer, the expression of NRF2 and NQO1 in cisplatin-resistant A549 cell lines was higher than in their parental cells. 31 We also observed a similar pattern in AGS/DDP and MKN45/DDP, where NRF2 was expressed at higher levels in cisplatin-resistant cells than in parental cells. Overall, in cisplatin-resistant cells, there is an imbalance in targets closely associated with antioxidants and ferroptosis. Additionally, Feng et al reported that isoorientin regulates the SIRT6/Nrf2/GPX4 signaling pathway to promote ferroptosis and reverse cisplatin resistance in lung cancer. 32 Triptolide induces ferroptosis in cisplatin-resistant human ovarian cancer cells by targeting the NRF2/GPX4 signaling axis to reverse chemotherapy resistance in ovarian cancer. 33 This extensive evidence highlights the critical roles of GPX4 and NRF2 in cisplatin resistance.
P62/KEAP1/NRF2 pathway is a classical antioxidant pathway, where activated P62 binds to KEAP1, releasing NRF2 into the nucleus to activate downstream antioxidant signaling molecules such as NQO1 and HO-1. In Xu et al ’s study, KEAP1 was competitively interacted by TNFAIP2 to stabilize NRF2, inhibiting cisplatin-induced oxidative stress and cell apoptosis, ultimately conferring cisplatin resistance in head and neck squamous cell carcinoma. 34 Wu et al indicated that NEDD4L inhibits the P62/KEAP1/NRF2 pathway to suppress cisplatin resistance and promote apoptosis in bladder cancer cells. 35 Additionally, P62 activates NRF2 to upregulate the expression of GPX4 and GSH, protecting cells from ferroptosis. 36 Based on previous research, we hypothesized that CUR, in combination with CDDP, promotes ferroptosis and induces apoptosis in resistant GC cells by modulating the P62/KEAP1/NRF2 pathway. Therefore, we selected the more pronounced imbalance in AGS/DDP for further protein experiments to validate the ferroptosis-related indicators GPX4 and the classical antioxidant pathway P62/KEAP1/NRF2/NQO1. The results showed that when CDDP was added alone, NRF2, KEAP1, and NQO1 did not exhibit significant changes, but GPX4 showed a downward trend after the addition of CDDP. However, p-p62 expression increased, possibly triggering other compensatory mechanisms. When CUR was added alone, NRF2, NQO1, and GPX4 all showed decreased expression, while KEAP1 exhibited increased expression. However, p-p62 did not show significant differences. When CUR and CDDP were used simultaneously, the expressions of NRF2, NQO1, and GPX4 were further significantly suppressed, while KEAP1 expression significantly increased. Although p-P62 expression showed further inhibition compared to using CUR or CDDP alone, there was only marginal statistical difference compared to the control group. The synergistic effect of CUR and CDDP on the regulation of GPX4 and the P62/KEAP1/NRF2/NQO1 signaling pathway can be reversed by the ferroptosis inhibitor Ferrostatin-1. In in vivo validation, we transplanted cisplatin-resistant GC cell line AGS/DDP into nude mice subcutaneously and treated them with CDDP alone, CUR alone, and in combination after tumor formation. The in vivo results were consistent with the in vitro findings. Unexpectedly, the NQO1 index in the tumor cell detection in the CDDP group showed an abnormal increase, possibly through other pathways, warranting further exploration. However, all of the above confirmed that the combined use of CUR and CDDP regulates the P62/KEAP1/NRF2 pathway, induces ferroptosis, and promotes the apoptosis of cisplatin-resistant GC cells.
The study has certain limitations. Currently, it is not clear by what compensatory pathway CDDP further activates NQO1 in subcutaneous tumors in vivo. Furthermore, we only used male nude mice in the animal studies and did not explore gender differences. These limitations need to be addressed in future research to elucidate more comprehensive implications.
Conclusion
Our study investigated the differences in proliferation, migration, and invasion between cisplatin (CDDP)-resistant cells and parental cells when curcumol (CUR) was used alone or in combination with cisplatin. Furthermore, we clarified that CUR and CDDP induced iron death in resistant GC cells by regulating the ROS and GSH systems, possibly through modulation of the P62/KEAP1/NRF2 signaling pathway. This study not only enriches previous research on the treatment of cisplatin-resistant gastric cancer with CUR and the combined intervention of CUR and CDDP in CDDP-resistant treatment but also provides a solid foundation for the clinical application of CUR as an ideal adjuvant for cisplatin in gastric cancer treatment.
Footnotes
Acknowledgements
Thanks to all authors for his review and comments on this paper.
Availability of Data and Materials
The data used to support the study are available from the corresponding author upon request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Zhejiang Provincial Natural Science Foundation, China (No. LGD21H290002) and (No. LQ20H030003).
