Abstract
Neuroblastoma represents a malignancy of the sympathetic nervous system characteristic by biological heterogeneity. Thus, chemotherapy exhibits only low effectivity in curing high-risk forms. Previous studies revealed the cytotoxic potential of valproate on neuroblastoma cells. Nevertheless, these studies omitted effects of hypoxia, despite its undeniable tumorigenic role. In this study, we addressed the question whether valproate promotes binding of platinum-based anti-cancer drugs (cisplatin, carboplatin and oxaliplatin) to DNA and role of hypoxia, cellular antioxidant capacity and cisplatin resistance in this process. Following parameters differed significantly when cells were exposed to treatment with platinum-based drugs: elevation of platinum content bound to DNA, elevation of total thiol content, GSH/GSSG ratio, glutathione reductase and peroxidase, superoxide dismutase and elevation of antioxidant capacity. Hypoxia caused a decrease in cytosine/adenine peak, and no changes in platinum–DNA binding properties were observed. After valproate co-treatment, oxidative stress–related parameters and cytosine/adenine peak were only elevated. The amount of platinum bound to DNA was not changed significantly. Valproate is not able to enhance platinum binding to DNA in neuroblastoma cells, neither in case of intrinsic resistance (UKF-NB-4) nor in case of acquired resistance (UKF-NB-4CDDP). Therefore, another mechanism different from increase in platinum binding to DNA should be considered as a synergistic effect of valproate by cisplatin treatment.
Introduction
Neuroblastoma (NB) is the most common extracranial solid tumour in children. NB represents a malignancy of the sympathetic nervous system emerging from pluripotent neuroblasts and is special compared to other children solid tumours because of its huge biological heterogeneity. Consequently, the differences in outcome among patients with NB are remarkable. Survival from NB is influenced by tumour stage, age at diagnosis, and by chromosomal changes particularly by MYCN status. 1 MYCN oncogene amplification occurs in 20–25% of NB and is an indicator of unfavourable prognosis for all stages.1,2 Patients with low- and intermediate-risk NB have good prognosis, but approximately 40% of patients with NB are in the high-risk group (HR-NB) and have poor outcomes.3–5 Chemotherapy has low efficacy in curing HR-NB. This treatment failure is largely caused by multidrug resistance of cancer cells. 6
The most common combination of chemotherapeutics used to treat NB consists of platinum cytostatics (CDDP or carboplatin), anthracyclines, cyclophosphamide, vincristine and etoposide. 7 Inasmuch as CDDP-resistant cells may have cross-resistance to other drugs such as temozolomide, etoposide and irinotecan, 7 we focussed on CDDP resistance in this study. CDDP mechanism of action has been linked to crosslinks with the purine bases of DNA, which caused severe DNA damage, and subsequently triggered apoptosis. 8 Other platinum-based anti-cancer drugs such as carboplatin and oxaliplatin are also widely used. Combination therapies of CDDP with other drugs are highly considered to overcome resistance and simultaneously reduce toxicity. 8
It is well known that a compact chromatin structure may prevent CDDP from DNA binding. However, relaxation of chromatin by histone deacetylase inhibitors (HDACi) may increase the accessibility of DNA to DNA damaging agents.9,10 Consequently, HDACi seems to be a promising class of anti-cancer agents. Valproic acid (VPA), a short-chained fatty acid, is an HDACi with a well-established function in psychiatry and with a possible potential in cancer treatment. 11 Previous studies revealed the cytotoxic potential of VPA on NB cells.11,12 Nevertheless, these studies omitted effects of hypoxia, although there is undeniable clinical evidence across a range of malignancies to implicate hypoxia as a key player in treatment outcome.13,14 Hypoxic conditions in tumour (i.e. areas with O2 tensions (pO2 values) ≤2.5 mmHg) are commonly associated with resistance to radiotherapy, but have also been shown to reduce the efficacy of several forms of chemotherapy, immunotherapy and photodynamic therapy.13,15 Furthermore, hypoxia induces histone modifications associated with gene repression and heterochromatin16–18 and therefore potentially could decrease the accessibility of DNA to DNA damaging agents.
In this study, we addressed the question whether VPA could promote binding of platinum-based anti-cancer drugs (CDDP, carboplatin and oxaliplatin) to DNA and a role of hypoxia, cellular antioxidant capacity and CDDP resistance during this process.
Materials and methods
Chemical compounds
All reagents, standards and other chemicals were purchased from Sigma-Aldrich (St. Louis, MO, USA) in American Chemical Society (ACS) reagent-grade purity, unless noted otherwise.
Culture conditions and treatment conditions
The human NB cell lines used in the study are as follows: (1) the UKF-NB4 human cell line established from recurrent bone marrow metastases from patient with high-risk NB (stage IV, MYCN amplification, 7q21 gain 5 ) and (2) the CDDP-resistant UKF-NB4 designated UKF-NB4CDDP was established from parental cell line UKF-NB4 in the laboratory of Prof. Eckschlager by incubating the UKF-NB-4 cells with increasing concentrations of CDDP. 19 The UKF-NB4 cell line was a gift of Professor J. Cinatl, Jr. (J.W. Goethe University, Frankfurt, Germany). Cells were grown at 37°C and 5% CO2 and cultivated in Iscove’s modified Dulbecco’s medium (IMDM) with 10% fetal bovine serum (Life Technologies, Carlsbad, CA, USA); UKF-NB-4CDDP cells were cultivated in medium with CDDP at a concentration of 100 ng/mL, but prior to this cultivation, cells were cultivated in medium without CDDP for five passages. In all, 4 × 106 NB UKF-NB-4 or UKF-NB-4CDDP cells were seeded 24 h prior to treatment in 100-mm dishes and pre-treated with VPA at a final concentration of 1 mM for 24 h and then with platinum derivative for 24 h (for details of experimental protocol, see Figure 1). Appropriate controls and cells were cultivated either continuously in normoxia or in hypoxic conditions (1% O2) using Proox model C21 (BioSpheric, Lacoma, NY, USA).
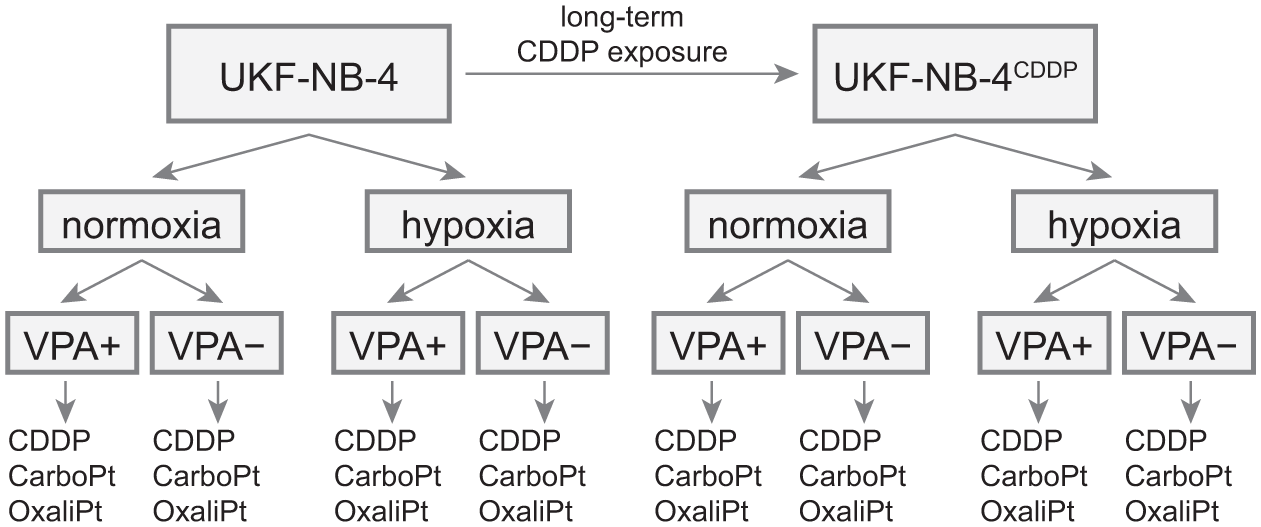
Experimental design. UKF-NB-4CDDP denotes a cisplatin-resistant variant of original neuroblastoma cell line established by a long-term cisplatin exposure (see experimental for details).
Measurements of cell viability by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay
The suspensions of 10,000 cells were added to each well of standard microtiter plates (E-plates 16) and incubated for 2 days to ensure cell growth. After treatment, plates were incubated for 24 h; then, media were removed and replaced by a fresh medium. Furthermore, a medium was replaced by 200 µL of fresh medium containing 50 µL of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT; 5 mg/mL in phosphate-buffered saline (PBS)), incubated in a humidified atmosphere for 4 h at 37°C and wrapped in aluminium foil. After the incubation, MTT-containing medium was replaced by 200 µL of 99.9% dimethyl sulphoxide. Then, 25 µL of glycine buffer (pH 10.5) was added to all wells and absorbance at 570 nm was determined using Infinite 200 PRO reader (Tecan, Maennedorf, Switzerland).
Catalytic electrochemical analysis of platinum bound to DNA of NB cells
DNA was isolated from NB cells using Wizard Genomic DNA Purification Kit (Promega, Madison, WI, USA) according to manufacturer’s instructions. The amount and purity of isolated DNA were checked by ultraviolet–visible (UV-Vis) spectrophotometry. Platinum was measured by differential pulse anodic stripping voltammetry (DPASV) in the presence of 2 mL of 0.36 M sulphuric acid, containing 0.24 mL of hydrazine (10 mM) and 0.01 mL of formaldehyde, following the conditions optimized in our previous study. 20
Electrochemistry of nucleotides – CA peak
The electrochemical responses of cytosine (C peak) and adenine (A peak) were studied by AdTS DPV in the presence of 0.2 M acetate buffer (pH 5.0). The detailed experimental parameters for both methods are described by Tmejova et al. 21
Preparation of cells for further analyses
The cells were frozen in liquid nitrogen and homogenized using ultrasonic homogenizer SONOPLUS mini20 (BANDELIN electronic, Berlin, Germany). Subsequently, 1 mL of 0.2 M phosphate buffer (pH 7.0) was added and the sample was homogenized for 5 min. The homogenates were further centrifuged using Microcentrifuge 5417R (Eppendorf, Hamburg, Germany) at 4°C at 15,000g for 15 min. Finally, the supernatant was filtered through a membrane filter (0.45 µm nylon filter disk; Millipore, Billerica, MA, USA) and analysed.
Estimation of total thiols
Total thiols were estimated by Ellman’s spectrophotometric method, using the Ellman’s reagent (5,5′-dithiobis(2-nitrobenzoic) acid). Mixture of reagent and tested sample was incubated for 10 min at 37°C. Absorbance was measured at λ = 405 nm at laboratory temperature.
Determination of metallothionein (MT)
MT was quantified using differential pulse voltammetry (747 VA Stand, connected to the 693 VA processor and 695 Autosampler; Metrohm, Herissau, Switzerland) following conditions used in our previous study. 22
Analysis of reduced and oxidized glutathiones
Contents of reduced (GSH) and oxidized glutathione (GSSG) were determined using high-performance liquid chromatography coupled with an electrochemical detector (HPLC-ED) system under the conditions described in our previous study. 23
Measurements of antioxidant activities
The antioxidant activities of cells were estimated using four different methods: (1) 2,2-diphenyl-1-picrylhydrazyl (DPPH), (2) 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid; ABTS), (3) free radicals (FR) and (4) ferric reducing antioxidant power (FRAP). The conditions of measurements are described in our previous study. 24
Measurements of superoxide dismutase, glutathione reductase and glutathione peroxidase activities
The enzyme activities were analysed using automated spectrophotometer BS-400 (Mindray, Shenzhen, China). Superoxide dismutase (SOD), glutathione reductase (GR) and glutathione peroxidase (GPx) activities were estimated using commercial kits (Kit 19160 SOD, GR Assay Kit and CGP1 Kit (Sigma-Aldrich)) according to manufacturer’s instructions.
Quantitation of total proteins in cell lines
For normalization of results, total proteins were determined using the SKALAB CBT 600T kit (Skalab, Svitavy, Czech Republic), on automatic spectrophotometer BS-400 (Mindray), following the manufacturer’s instructions.
Statistical analyses
Comparisons were performed using factorial analysis of variance (ANOVA) followed by planed comparisons. Multivariate tests were used. To reveal associations between possibly correlated variables and cases and thus to reduce dimensionality, principal component analysis (PCA) was performed. This analysis transforms variables into linearly uncorrelated ‘principal components’, whose number corresponds to the number of variables. The first component explains the largest variability of data; this variance decreases with every other component. Number of principal components was reduced based on this variance reflected by ‘eigenvalues’. Unless noted otherwise, p value <0.05 was considered significant. All analyses were performed in Statistica 12 (Dell, Tulsa, OK, USA).
Results
Cytotoxicity analysis
In the first step, we aimed to elucidate the IC50 of tested cell lines for tested compounds and thus to verify the acquired resistant status and benefit of co-treatment with valproate (Table 1). Using MTT test, it was revealed that CDDP-resistant cell lines show up to 12-fold and 4-fold increase in IC50 under normoxic and hypoxic conditions compared to wild-type (WT) counterparts. Moreover, we have also demonstrated that valproate is relatively non-toxic to cells with IC50 values higher than 3.7 mM. The co-treatment of cells with sub-IC50 concentration of valproate caused a significant increase in CDDP toxicity. This trend was observed either in resistant and in wild-type cells. In addition, hypoxic environment even increased the additive effect of valproate with subsequent 4-fold reduction of IC50 compared to treatment without valproate.
Half-maximal inhibition concentrations (IC50).

IC50 values for either cisplatin or valproate or for cisplatin co-treated with 1 mM valproate are listed.
IC50 values for cisplatin are in micromole, while IC50 values for valproate are in milimole.
In sum, we verified the resistant status of the cells, provided evidence that valproate is relatively non-toxic and its addition increases the sensitivity of cells to CDDP.
Effect of platinum compounds on DNA damage– and oxidative stress– related parameters of the tested cells
Next, we aimed to elucidate whether various forms of platinum drugs (CDDP, oxaliplatin and carboplatin) affect the levels of measured DNA damage– and oxidative stress– related parameters. Using multivariate ANOVA followed by planned comparisons, it was revealed that distinct types of platinum drugs used in this study affected only a minority of parameters measured; total thiol content and GSH/GSSG ratio were significantly higher when treated with novel analogues (oxaliplatin and carboplatin) compared to CDDP. Moreover, the level of metallothionein was significantly higher after carboplatin treatment (compared to CDDP), and CA peak was significantly higher after oxaliplatin treatment (compared to CDDP; Figure 2).

Levels of DNA damage– and oxidative stress–related parameters determined in samples. (a) Levels of measured parameters displayed as a heatmap of standardized z-scores with mean = 0 and standard deviation = 1 clustered according to similarity in values. (b) Absolute values of platinum content bound to DNA in cell lines. (c) Results of planned comparisons displayed as p values of subsequent tests, sorted according to the levels of significance.
These findings are in accordance with general characteristics of oxaliplatin and carboplatin, namely, lower cytotoxicity compared to its parental drug. Importantly, no significant difference in platinum-DNA-binding properties was observed between CDDP and its analogues. In accordance with these findings, these minor differences between platinum forms were not taken into account and thus ‘platinum drug treatment’ was used in following analyses.
Following parameters differed significantly when cells were exposed to Pt treatment (general effects of platinum drug compared to control): elevation of Pt content bound to DNA, elevation of total thiol content, a GSH/GSSG ratio, GR, GPx, SOD and antioxidant capacity determined by FR and by a FRAP.
Effect of CDDP resistance on cellular parameters
Previous analysis did not take into account the differential contribution of other factors including the resistance of NB cells to the CDDP treatment. As expected, resistant status of the cells affected measured parameters significantly, F(6, 2) = 49.07, p = 0.02. Because different mechanisms of the response to Pt drugs in control- and platinum-treated cells were expected, the effect of resistance was analysed in control- and treated cells separately.
In the control group (without platinum treatment), the effect of resistance caused an increase in thiol compounds’ content, a decrease in metallothionein, an increase in antioxidant capacity determined by ABTS and FR, an increase in SOD and decrease in glutathione reductase and peroxidase compared to wild-type counterparts.
In the platinum-treated cells, the platinum resistance was confirmed by significantly lower amount of platinum bound to DNA (2-fold, Figure 2(b)), higher total thiols and antioxidant capacity (DPPH, FRAP, FR and ABTS methods) and SOD levels and lower glutathione-related enzymes reductase and peroxidase. For details, see Figure 2(a) and (c) and Supplementary Table 1.
Effect of VPA
After VPA treatment, the following parameters were elevated: CA peak, a total thiol content, a GSH/GSSG ratio and antioxidant capacity (FR method). These parameters were elevated in both a group of platinum-treated cells and in untreated cells. Moreover, VPA treatment also caused an increase in antioxidant capacity determined by ABTS, GR and GPx in a platinum-treated subgroup of samples (Figure 2(a) and (c), Supplementary Table 1).
Effect of hypoxia on cellular parameters
Planned comparisons revealed that only CA peak was decreased when cultivated under the hypoxic conditions (compared to normoxic conditions). Moreover, cells exposed to platinum drugs demonstrated a decrease in metallothionein, a GSH/GSSG ratio, and an increase in antioxidant capacity (ABTS and FR method) following cultivation in hypoxic environment (combined effect of hypoxia and platinum treatment; Figure 2(a) and (c), Supplementary Table 1).
Mutual interactions between VPA and hypoxia on resistance of cells to CDDP
Looking at the individual effects separately, that is, effect of resistance, VPA and hypoxia, most noticeable trends were elucidated. Nevertheless, more complex analyses are needed to reveal trends occurring only in certain cells or only after certain treatment. Using series of measured parameters, we were able to describe the whole path and mechanism of platinum–cell interaction – from its binding to DNA, through the effect of co-treatment on DNA tertiary structure (determined by CA peak) to platinum-stress buffering (markers of antioxidant capacity, metallothionein, etc.).
To reveal complex interactions between variables and cases, PCA together with cluster analysis was employed. PCA combines the advantages of correlation analysis with clustering by transforming the potentially correlated variables into uncorrelated ‘principal components’. This analysis also helps us to orient in a larger number of variables due to reduction into a small number of principal components. A two-principal-component model was created with eigenvalues 6.01 and 2.57 for factors 1 and 2, respectively (Figure 3(a)). Thus, quality of representation gained 71.5% (50.09 and 21.41 for principal components 1 and 2, respectively). Based on a similarity, measured parameters are grouped into three major groups marked as ‘cluster 1–3’ one two-dimensional plane formed by two principal components. First, such cluster consists of markers of antioxidant capacity, and thiols were characterized by positive values of principal component 1. However, DNA-bound platinum, metallothionein contents, and GSH-related enzymes formed a second cluster characterized by negative values of principal components. GSH/GSSH ratio and CA peak are not clustered with previous ones and form a third cluster characteristic by negative values of principal component 2.

Mutual interaction between measured parameters and samples. (a) Principal component analysis: projection of variables (measured parameters) on a two-factor plane. Note that markers of antioxidant capacity are clustered together, while Pt bound to DNA is clustered together with glutathione-related enzymes and metallothionein across Factor 1. CA peak and GSH/GSSG ratio forms the ‘Factor 2’ variable. (b) Projection of cases (samples) on an identical factor-plane formed in (a). Clear differentiation of resistant and wild-type cells is evident, and untreated cells are clustered together by positive values of factor 2. Thus, cisplatin-sensitive and -resistant and untreated cells can be clearly differentiated based on measured parameters.
In order to demonstrate whether measured parameters describe shifts in resistance after VPA or hypoxia treatment, cases (individual cells with individual treatments) were projected into this two-dimensional plane (Figure 3(b)). Based on principal component 1 (see previous paragraph), resistant and WT cells can be distinguished (WT cells characteristic by negative values and resistant cells by positive values). Platinum-untreated samples (in chart depicted as outlined points) were characterized by most positive values of principal component 2. Thus, the upper part of PCA plot was designated as ‘viability’, the lower left as ‘sensitivity’ and lower right as ‘resistance’. With regard to VPA treatment, a shift across the principal component 2 away from the ‘viability’ part was observed; no shift towards ‘sensitivity’ zone was observed, that is, VPA does not sensitize the cells.
Discussion
NB is childhood malignancy, with high numbers of patients dying due to emergence of treatment resistance. Consequently, the identification of resistance mechanisms as well as new therapeutic targets is highly needed. There are three morphologic cell types in NB assigned as S (substrate adherent), N (neuroblastic) and I (intermediate). Amid them, S-type cells are most prone to develop resistance, 25 and therefore, we chose S-type cells into our study. The used UKF-NB-4 human cell line (S-type cells), established from recurrent bone marrow metastases from patient with high-risk NB (stage IV), owns the intrinsic multidrug resistance phenotype including 7q21 gain affecting MDR1 gene. 5 The highly CDDP-resistant counterpart of UKF-NB-4 designated UKF-NB-4CDDP was established from a parental cell line UKF-NB-4 by incubating in increasing concentrations of CDDP. 5 Because CDDP affects manifold cellular targets (mainly nucleophilic parts of nucleic acids, proteins and membrane phospholipids), development of resistance is a very intricate process, involving multiple pathways such as (1) modulation of drug uptake or efflux, (2) increased detoxification of CDDP by glutathione or metallothionein (MT) and increased antioxidant capacity in cells, (3) enhanced activity of detoxification enzymes, (4) more effective repair of DNA, (5) DNA methylation followed by gene activation and/or silencing and (6) attenuation of apoptosis. 26 In accordance, increased antioxidant capacity measured by ABTS, FR, DPPH and FRAP was recorded in resistant UKF-NB-4CDDP cells. Enhanced activity of the detoxification enzyme SOD and an increased total thiol concentration was also revealed in resistant cells. On the contrary, a GSH/GSSG ratio and activity of GPx and GR seems to be crucial for coping with platinum in wild-type UKF-NB-4 cells which is in accordance with literature.27,28 Overexpression of SOD was previously shown to be important in protection against CDDP-induced cellular damage. 29 Furthermore, in CDDP-resistant UKF-NB-4CDDP cells, a significant reduction in platinum-DNA adduct formation (2-fold) was observed. Simultaneously, there was no significant difference in platinum-DNA adduct formation due to CDDP, carboplatin and oxaliplatin treatment. Moreover, CDDP-resistant cells also showed cross-resistance to oxaliplatin and carboplatin. This finding is a possible consequence of the fact that the mentioned platinum derivatives form the same types of adducts at the same spots on DNA (approximately 60%–65% intra-strand GG, 25%–30% intra-strand AG, 5%–10% intra-strand GNG and 1%–3% inter-strand GG adducts).30,31 However, an unequal recognition of CDDP- and oxaliplatin-GG adducts and a consequent different activation of reparatory systems contribute to the differences in final cytotoxicity of CDDP and oxaliplatin. 30 Accordingly, the intracellular total thiol content was higher in cells treated by novel Pt analogues compared to CDDP.
In this study, we addressed the question of whether VPA could promote platinum-based anti-cancer drugs (CDDP, carboplatin and oxaliplatin) binding to DNA under hypoxia and normoxia. One of the hypotheses behind such experimental design was an anticipated relationship between the HDAC inhibitors and the DNA binding of platinum complexes as proposed by some authors.28,32 However, no statistical significant association between VPA treatment and DNA binding of platinum complexes was found under hypoxia or normoxia (some observable effect of VPA was shown only on UKF-NB-4CDDP cells in normoxia, but it was not significant; see Figure 2(b)). Inasmuch as previous studies revealed a synergistic cytotoxic effect between VPA and platinum,10,33 another mechanism different from enhanced platinum binding to DNA should be considered. After valproate or platinum treatment, a GSH/GSSG ratio was elevated. Consequently, VPA and CDDP can compete for cellular GSH resources and deplete cellular GSH by long-term exposure.34,35 A potential mechanism of GSH depletion by VPA could be an inhibition of the catalytic activity of glutathione synthetase and glutamate–cysteine ligase.28,34 Furthermore, we used free VPA in our experiments, which could be a reason for insufficient enhancement of platinum binding to DNA. It was shown that the Pt(IV)-VPA conjugates have higher effect on de-condensation of heterochromatin and consequently a higher amount of platinum from the Pt(IV)-VPA conjugates is bound to DNA. 28 Moreover, sensitivity of some NB cell lines (e.g. SK-N-AS and UKF-NB-4) to VPA is quite low, because of their intrinsic resistance phenotype.5,36
Hypoxic conditions in tumours are commonly associated with resistance to therapy.13,15 One of the resistance mechanisms could be hypoxia-induced histone modifications associated with gene repression and heterochromatin.16–18 The IC50 value for CDDP in a wild-type UKF-NB-4 cells and CDDP-resistant UKF-NB-4CDDP cells was genuinely higher in hypoxia, but no significant effect on platinum binding to DNA was observed. Nevertheless, height of CA peak (redox signal of adenine and cytosine 31 ) reflecting the accessibility of DNA for electrochemical reactions was significantly lower in hypoxic condition, anticipating lower accessibility of DNA in hypoxia.
In conclusion, VPA is not able to enhance platinum binding to DNA in NB cancer cells, neither in case of intrinsic resistance (UKF-NB-4) nor in case of acquired resistance (UKF-NB-4CDDP). Therefore, another mechanism different from platinum binding to DNA should be considered as a synergistic effect of VPA by CDDP treatment. This study proved the well-known fact that chemoresistance is caused by multiple mechanisms.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Health of the Czech Republic (grant no. 15-28334A) and by the Ministry of Health of the Czech Republic for conceptual development of research organization 00064203 (University Hospital Motol, Prague, Czech Republic).
