Abstract
OBJECTIVE:
Renal cancer accounts for about 3% of human cancer, and clear cell renal cell carcinoma (ccRCC) is the most common subtype of renal cancer. Recently, microRNAs (miRNAs) are found to be the biomarkers for cancer diagnosis, prognosis and the targets for tumor management. This study aimed to examine the expression of miR-337-3p in ccRCC and to elucidate the molecular mechanisms underlying miR-337-3p-mediated ccRCC progression.
METHODS:
The miRNA and mRNA expression levels in ccRCC cells and tissues were measured by qRT-PCR. Cell proliferation, cell adhesion, colony growth and cell invasion were examined by CCK-8 assay, cell adhesion assay, colony formation assay and Transwell invasion assay, respectively. The protein levels were detected by western blot assay. The effects of miR-337-3p on tumor growth in vivo was assessed in a nude mice xenograft model.
RESULTS:
MiR-337-3p was down-regulated in ccRCC cell lines, and miR-337-3p overexpression suppressed cell proliferation, colony growth and invasion, but enhanced cell adhesion in ccRCC; while knockdown of miR-337-3p exerted the opposite effects on ccRCC. Bioinformatics analysis and luciferase reporter assay showed that Calpain small subunit 1 (Capn4) was negatively regulated by miR-337-3p, and overexpression of miR-337-3p attenuated the miR-337-3p-mediated effects on ccRCC cellular functions. In addition, miR-337-3p also suppressed epithelial-mesenchymal transition in ccRCC. The in vivo tumor growth was markedly suppressed after miR-337-3p overexpression. Data from clinical data showed that down-regulation of miR-337-3p and up-regulation of Capn4 mRNA and protein were identified in the ccRCC tissues, and miR-337-3p expression level was inversely correlated with Capn4 mRNA expression level in ccRCC tissues.
CONCLUSIONS:
Collectively, these data suggested the tumor suppressive role of miR-337-3p in ccRCC. MiR-337-3p suppressed cell proliferation and metastasis in ccRCC partially via targeting Capn4.
Introduction
Renal cancer accounts for about 2–3% of all types of human cancer worldwide, and clear cell renal cell carcinoma (ccRCC) accounts for more than 70% of all renal malignancies [1]. Among all the ccRCC patients, there were about half of the patients having tumor metastasis, which markedly affected the treatment outcome of radiotherapy and chemotherapy [2, 3, 4]. Recently, new therapeutic approaches including tyrosine kinase inhibitors sunitinib and sorafenib, and the mTOR inhibitors everolimus and temsirolimus, have improved the clinical management of ccRCC [5, 6, 7]. However, the 5-year survival rates for the patients with metastasis are still very low. In this regard, it is necessary to search for novel molecular targets to improve the therapeutic cues of ccRCC.
Recently, microRNAs (miRNAs) have come into attention because of their diverse biological functions [8, 9]. MiRNAs belong to a class of endogenous short non-coding RNAs with 21–23 nt in length, and they negatively regulate mRNA expression at the post-transcriptional level via binding to the 3’ untranslated region (3’UTR) of the target mRNA [8]. Up to date, studies reported that miRNAs could function as either oncogenes or tumor suppressors [10, 11]. In the ccRCC, a large number of miRNAs has been identified for their role in ccRCC progression. Chen et al. reported that miR-141 served as a potential biomarker for discriminating ccRCC from normal tissues and a crucial suppressor of ccRCC cell proliferation and metastasis by modulating EphA2 expression [12]. Jingushi et al. showed that miR-629 promoted TGF
In the present study, we aimed to characterize the role of miR-337-3p in the regulation of ccRCC cell proliferation and metastasis by performing corresponding in vitro and in vivo functional assays. In addition, the expression of miR-337-3p in clinical samples and its relevance to the clinicopathological parameters were also determined to validate the role ofmiR-337-3p in ccRCC progression.
The relationship between miR-337-3p expression level and clinicopathological parameters in patients with ccRCC
The relationship between miR-337-3p expression level and clinicopathological parameters in patients with ccRCC
The low expression and high expression groups were defined by median value of miR-337-3p expression.
Clinical samples
Sixty-eight ccRCC clinical samples and matched normal adjacent renal tissues (
Cell culture and cell transfection
The human kidney proximal tubular cell line (HK-2) and the ccRCC cell line including 786-O, Caik-2, ACHN and Caki-1 were obtained from ATCC (Manassas, USA) and cultured in RMPI 1640 medium (Sigma, St. Louis, MO, USA) containing 10% fetal bovine serum (FBS, Sigma) at 37
The miR-337-3p mimic, miR-337-3p inhibitor and the corresponding controls (mimic ctrl and inhibitor ctrl) were purchased from Ribobio (Guangzhou, China). For Capn4 overexpression, the Capn4-overexpressing vector, pcDNA3.1-Capn4, or the control vector, pcDNA3.1 were purchased from GeneChem company (Shanghai, China). Cell transfection was performed by using the Lipofectamine 2000 reagent (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions. The cells were collected at 24 h post transfection.
RNA isolation and quantitative real-time PCR (qRT-PCR)
Total RNA was extracted from tissues and cells by using TRIzol reagent (Takara, Dalian, China) by following the manufacturer’s instructions. Total RNA was reverse-transcribed into cDNA by using the commercial kit (PrimeScript™ 1st strand cDNA Synthesis Kit; Takara). qRT-PCR was performed with SYBR Green Real-Time PCR Master Mixes (Takara) on an ABI 7900 PCR system (Applied Biosystems, Foster City, CA, USA). U6 was used as the internal control for miR-337-3p expression, and GAPDH was used as the internal control for mRNA expression.
Cell proliferation assay
Cell counting kit-8 (CCK-8; Beyotime, Beijing, China) was used to determine cell proliferation. Briefly, cells were seeded at a density of 2
Cell adhesion assay
Cells were seeded at a density of 4
Colony formation assay
Cells were plated in a 6-well plate with complete medium, and then cells were grown in a humidified incubator at 37
Transwell invasion assay
Cells were digested and seeded in the upper Transwell chambers with 8
The effects of miR-337-3p on cell proliferation, adhesion and invasion in ccRCC cells. (A) The expression of miR-337-3p in HK-2 and ccRCC cells was measured by qRT-PCR, and miR-337-3p was down-regulated in ccRCC cells when compared with HK-2. The expression of miR-337-3p (B) in Caki-1 cells transfected with mimic ctrl or miR-337-3p mimic, and (C) in 786-O cells transfected with inhibitor ctrl or miR-337-3p inhibitor was determined by qRT-PCR. Cell proliferation (D) in Caki-1 cells transfected with mimic ctrl or miR-337-3p mimic and (E) in 786-O cells transfected with inhibitor ctrl or miR-337-3p inhibitor was determined by CCK-8 assay. Cell adhesive activity (F) in Caki-1 cells transfected with mimic ctrl or miR-337-3p mimic, and (G) in 786-O cells transfected with inhibitor ctrl or miR-337-3p inhibitor was determined by cell adhesion assay. Colony growth (H) in Caki-1 cells transfected with mimic ctrl or miR-337-3p mimic and (I) in 786-O cells transfected with inhibitor ctrl or miR-337-3p inhibitor was determined by colony formation assay. Cell invasive ability (J) in Caki-1 cells transfected with mimic ctrl or miR-337-3p mimic, and (K) in 786-O cells transfected with inhibitor ctrl or miR-337-3p inhibitor was determined by Transwell invasion assay. 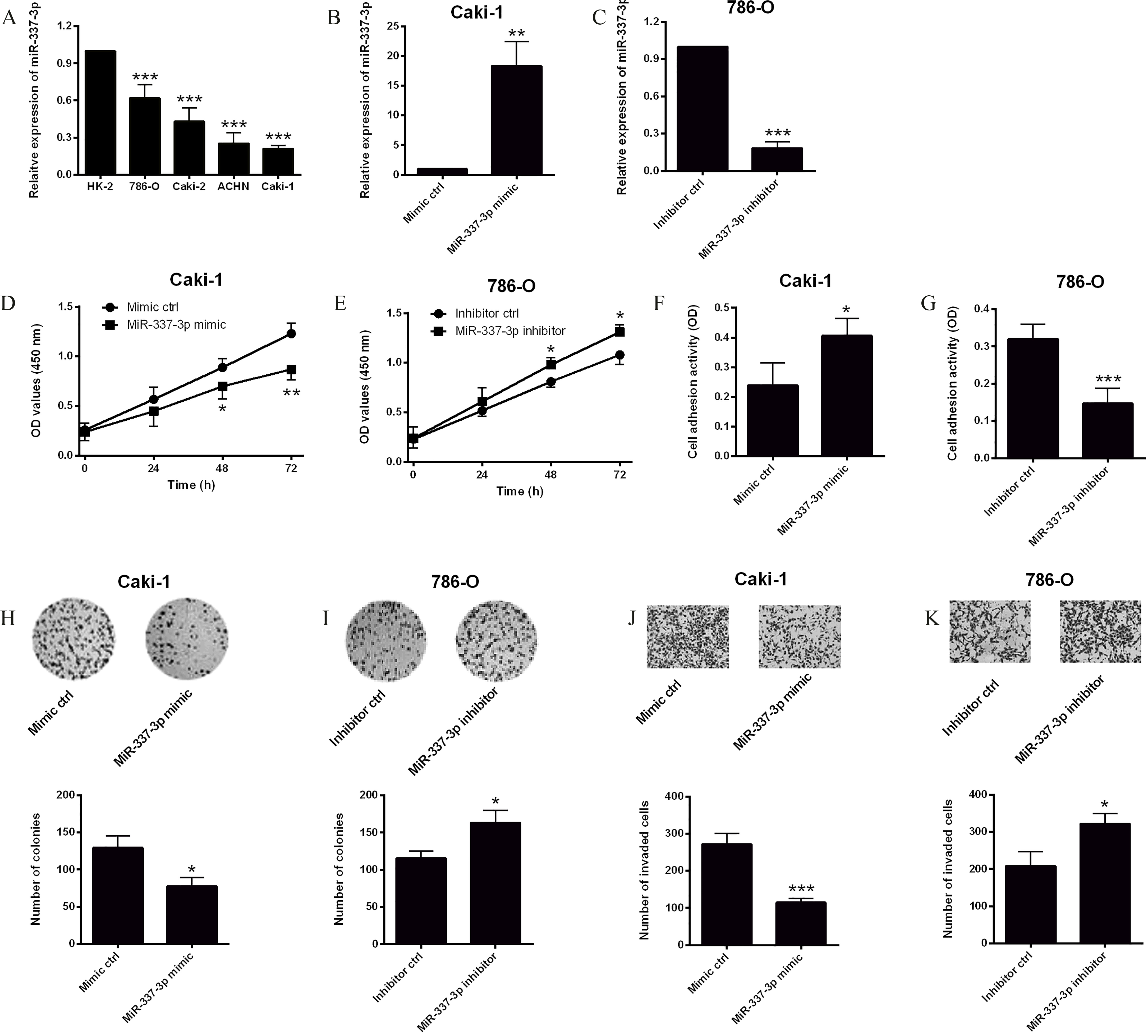
MiR-337-3p suppresses Capn4 expression by directly targeting the 3’UTR of Capn4. (A) Putative miR-337-3p binding sites in the 3’UTR of Capn4, and red letters highlighted in yellow background were indicated as mutated sites. Luciferase activity in Caki-1 cells (B) co-transfected with luciferase reporter vector containing wild-type 3’UTR of Capn4 and miR-337-3p mimic (or mimic ctrl), and (C) co-transfected with luciferase reporter vector containing mutant 3’UTR of Capn4 and miR-337-3p (or mimic ctrl). (D) qRT-PCR and (E) western blot analysis of Capn4 mRNA and protein levels, respectively, in Caki-1 cells transfected with mimic ctrl or miR-337-3p mimic. 
Proteins from tissues and cells were extracted and protein concentration were measured by a BCA protein assay kit (Bio-Rad, Hercules, CA, USA). The proteins were separated by SDS-PAGE and transferred onto polyvinylidene difluoride membranes. The membranes were blocked with 5% skim milk for 1 h at room temperature, and then incubated with corresponding primary antibodies: rabbit Capn4 polyclonal antibody (1:1000), rabbit vimentin polyclonal antibody (1:1500), rabbit snail polyclonal antibody (1:1000), rabbit E-cadherin polyclonal antibody (1:1500), rabbit N-cadherin polyclonal antibody (1:2000) and rabbit
In vivo tumor growth assay
All the animal experiments were carried out in accordance with the animal experimental protocol of the Third Affiliated Hospital of Soochow University, and all the animal experiments were approved by the Animal Ethics Committee of the Third Affiliated Hospital of Soochow University (Soochow, China). The female nude mice were purchased from Shanghai SLAC Laboratory Animal (Shanghai, China). Caki-1 cells with miR-337-3p overexpression or control Caki-1 cells were injected subcutaneously into the back flank of six nude mice. The tumor volume was evaluated every 7 d for 35 d, and the tumor volumes were calculated with the following formula: V
Statistical analysis
All the data analysis was performed by using the GraphPad Prism Software (Version 5.0, La Jolla, USA). All the data are presented as mean
Capn4 involves in the miR-337-3p-mediated effects on cell proliferation, adhesion and invasion in ccRCC cells. (A) qRT-PCR and(B) western blot analysis of Capn4 mRNA and protein levels, respectively, in Caki-1 cells transfected with pcDNA3.1 or pcDNA3.1-Capn4.(C) CCK-8 proliferative ability, (D) cell adhesive activity, (E) colony growth ability and (F) cell invasive ability in Caki-1 cells co-transfected with mimic ctrl 
The effects of miR-337-3p on EMT in ccRCC cells. (A) qRT-PCR and (B) western blot analysis of vimentin, snail, N-cadherin or E-cadherin, respectively, in Caki-1 cells transfected with mimic ctrl and miR-337-3p mimic. 
The effects of miR-337-3p on the tumor growth in vivo. (A) Overexpression of miR-337-3p suppressed in vivo tumor growth over time in the nude mice. (B) The tumor weight at 35 d after inoculation was reduced after miR-337-3p overexpression. The expression level of(C) miR-337-3p, (D) Capn4 mRNA and (E) Capn4 protein in tumor tissues. 
The effects of miR-337-3p on cell proliferation, adhesion and invasion in ccRCC cells
The expression levels of miR-337-3p in the normal cells (HK-2) and ccRCC cells were determined by qRT-PCR. All the four carcinoma cell lines (786-O, Caki-2, ACHN and Caki-1) had significantly lower expression levels of miR-337-3p compared to HK-2 cells (Fig. 1A), and among the ccRCC cell lines, Caki-1 presented the lowest miR-337-3p expression level, while 786-O had the highest expression level of miR-337-3p (Fig. 1A). Next, up-regulation of miR-337-3p in Caki-1 cells was achieved by transiently transfecting the cells with miR-337-3p mimic, and the down-regulation of miR-337-3p in 786-O cells was carried out by transfecting the cells with miR-337-3p inhibitor. Results from qRT-PCR assay demonstrated that miR-337-3p expression level was significantly up-regulated in Caki-1 cells transfected with miR-337-3p mimic when compared with cells transfected with mimic ctrl (Fig. 1B), and miR-337-3p inhibitor transfection significantly suppressed the expression level of miR-337-3p in 786-O cells when compared with inhibitor ctrl transfection (Fig. 1C).
To test the effects of miR-337-3p on the cell proliferation, cell adhesion, colony growth and cell invasion, CCK-8 assay, cell adhesion assay, colony formation assay and Transwell invasion assay were performed accordingly. CCK-8 assay revealed an apparent decrease in the cell proliferative ability of Caki-1 cells transected with miR-337-3p mimic when compared with mimic ctrl group (Fig. 1D); while miR-337-3p inhibitor transfection promoted cell proliferation of 786-O cells (Fig. 1E). Cell adhesion assay showed that miR-337-3p mimic transfection promoted cell adhesion in Caki-1 cells (Fig. 1F), and miR-337-3p inhibitor suppressed cell adhesion in 786-O cells (Fig. 1G). Colony formation results demonstrated that overexpression of miR-337-3p reduced the number of colonies in Caki-1 cells (Fig. 1H), and knockdown of miR-337-3p increased the number of colonies in 786-O cells (Fig. 1I). In addition, Transwell invasion assay results showed that the number of invaded cells was significantly reduced in Caki-1 cells transfected with miR-337-3p mimic (Fig. 1J), and was significantly increased in 786-O cells transfected with miR-337-3p inhibitor (Fig. 1K).
The expression of miR-337-3p and Capn4 in clinical samples. (A) The expression of miR-337-3p in ccRCC tissues (
To explore the molecular mechanism by which miR-337-3p regulates ccRCC progression, the computational algorithm (TargetScan) was used to search for potential targets of miR-337-3p. Among these targets, Capn4 was predicted to be a target of miR-337-3p and was selected for further experiment. The predicted binding sites between miR-337-3p and 3’UTR of Capn4 were illustrated in Fig. 2A. The interaction between miR-337-3p and 3’UTR of Capn4 was confirmed by luciferase reporter assay. The wild-type or mutant 3’UTR of Capn4 was inserted into a luciferase reporter vector, and each construct was co-transfected with miR-337-3p mimic or mimic ctrl. The results showed that miR-337-3p mimic transfection significantly inhibited the luciferase reporter activity of the wild-type Capn4 3’UTR vector (Fig. 2B), but did not significantly change the luciferase activity of the mutant Capn4 3’UTR vector (Fig. 2C). Furthermore, overexpression of miR-337-3p in Caki-1 cells significantly suppressed the mRNA and protein expression levels of Capn4 (Fig. 2D and E).
Capn4 involves in the miR-337-3p-mediated effects on cell proliferation, adhesion and invasion in ccRCC cells
To explore the involvement of Capn4 in the miR-337-3p-mediated effects on ccRCC progression, we overexpressed the expression of Capn4 in Caki-1 cells, and pcDNA3.1-Capn4 transfection significantly increased the mRNA and protein expression levels of Capn4 in Caki-1 cells (Fig. 3A and B). The functional in vitro assays showed that co-transfection with miR-337-3p mimic and pcDNA3.1 significantly suppressed cell proliferation, colony growth and cell invasion, but increased cell adhesion of Caki-1 cells when compared to that co-transfected with mimic ctrl and pcDNA3.1 (Fig. 3C–F); while co-transfection with miR-337-3p mimic and pcDNA3.1-Capn4 attenuated the effects of miR-337-3p mimic and pcDNA3.1 co-transfection on cell proliferation, cell adhesion, colony growth and cell invasion of Caki-1 cells (Fig. 3C–F).
The effects of miR-337-3p on epithelial- mesenchymal transition (EMT) in ccRCC cells
As Capn4 was found to regulate the expression of EMT-related markers [21], we performed furtherqRT-PCR and western blot assays to determine the effects of miR-337-3p on the expression of vimentin, snail, N-cadherin and E-cadherin. Overexpression of miR-337-3p significantly suppressed the mRNA and protein levels of vimentin, snail and N-cadherin, but increased the mRNA and protein levels of E-cadherin (Fig. 4A and B).
The effects of miR-337-3p on the tumor growth in vivo
To test the effects of miR-337-3p on ccRCC tumor progression in vivo, we injected the Caki-1 cells with miR-337-3p overexpression or control Caki-1 cells subcutaneously into the nude mice. The results showed that the tumor volume on day 21, 28 and 35 in mice injected with Caki-1/miR-337-3p mimic were significantly reduced when compared to those in mice injected with Caki-1/mimic ctrl (Fig. 5A). Consistently, the tumor weight was significantly lower in the miR-337-3p mimic group than that in the mimic ctrl group (Fig. 5B). The expression of miR-337-3p and Capn4 in tumor tissues was determined by qRT-PCR, and miR-337-3p was up-regulated, and Capn4 was down-regulated in tumor tissues from miR-337-3p mimic group when compared to that from mimic ctrl group (Fig. 5C and D).Western blot analysis showed that Capn4 was also down-regulated in tumor tissues from miR-337-3p mimic group when compared with mimic ctrl group (Fig. 5E).
The expression of miR-337-3p and Capn4 in clinical samples
To evaluate the expression of miR-337-3p and Capn4 in ccRCC tissues, qRT-PCR assay was performed in 68 patients with ccRCC and matched normal adjacent renal tissues. Results showed that the expression level of miR-337-3p in ccRCC tissues was significantly lower than that in the corresponding normal adjacent renal tissues (Fig. 6A), and the expression level of Capn4 mRNA in ccRCC tissues was significantly higher than that in the normal adjacent renal tissues (Fig. 6B). The expression of Capn4 protein in ccRCC tissues was also significantly upregulated compared to the normal adjacent renal tissues (see supplemental Fig. S1). Further Spearman’s correlation analysis showed that the expression level of miR-337-3p was inversely correlated with the mRNA expression level of Capn4 in ccRCC tissues (Fig. 6C,
Discussion
ccRCC exhibits as the most common histological types of renal malignancies and patients often suffered from the metastatic disease, which markedly affected the overall survival of these patients. Therefore, finding novel potential targets may aid us to develop new therapeutic approaches, which may improve the patients’ outcome. Recently, miRNAs have been suggested to play important roles in the regulation of ccRCC progression. Hence, in the present study, we identified the down-regulation of miR-337-3p in the ccRCC cell lines, and miR-337-3p suppressed ccRCC cell proliferation, colony growth, cell invasion and EMT, but promoted ccRCC cell adhesion; and further mechanistic data revealed that miR-337-3p repressed the expression of Capn4 via targeting the 3’UTR of Capn4, and overexpression of Capn4 attenuated the miR-337-3p mediated effects on ccRCC cellular functions. In vivo data showed that miR-337-3p suppressed tumor growth. In addition, down-regulation of miR-337-3p and up-regulation of Capn4 mRNA were identified in the ccRCC tissues, and miR-337-3p expression level was inversely correlated with Capn4 mRNA expression level in ccRCC tissues. Collectively, these data suggested the tumor suppressive role of miR-337-3p in ccRCC.
The miR-337-3p is located at chromosome 14q32.2. Previous studies have reported that miR-337-3p was dysregulated and functioned as a tumor suppressor in various types of human cancers. Wang et al., reported that loss of miR-337-3p expression was associated with lymph node metastasis of human gastric cancer [22]. MiR-337-3p was also found to suppress the proliferation and invasion in pancreatic ductal adenocarcinoma by targeting homeobox protein Hox-B7 [19], and miR-337-3p targeted the transcription of matrix metalloproteinase 14 to suppress neuroblastoma progression [16]. In addition, Cui et al. reported that miR-337-3p regulated the PI3K/AKT and Wnt/
In the present study, the further mechanistic study revealed that Capn4 was a direct target of miR-337-3p. Capn4 belongs to the calpain family, which consist of a large subunit (Capn1 and Capn2 encoded) and a small one (Capn4 encoded). As a subunit of calpain, Capn4 plays an essential role in the tumor metastasis. Previous studies showed that Capn4 was up-regulated in intrahepatic cholangiocarcinoma and ccRCC, and was positively correlated with lymphatic metastasis and tumor node metastasis stage [24, 25]. Our previous mechanistic study further showed that Capn4 promoted tumor invasion and metastasis in ccRCC cells via modulating talin-focal adhesion kinase signaling pathway [21]. As Capn4 also regulated the metastasis-related markers in ccRCC [21], we examined the effects of miR-337-3p on EMT in ccRCC and found that miR-337-3p suppressed EMT in ccRCC. In the present study, Capn4 was negatively regulated by miR-337-3p in ccRCC cells, and overexpression of Capn4 attenuated the effects of miR-337-3p on ccRCC cell proliferation and metastasis. More importantly, the mRNA expression of Capn4 was also down-regulated in mice tumor tissues with miR-337-3p overexpression. In clinical samples, the mRNA expression level of Capn4 was inversely correlated with miR-337-3p expression level in ccRCC tissues. Collectively, these data suggested that miR-337-3p suppressed the progression of ccRCC possibly via targeting Capn4. As miR-337-3p may target other genes to regulate ccRCC progression, it is necessary for us to explore other targets of miR-337-3p in the ccRCC progression in the future studies, which may enable us to have a better understating of the pathophysiology of ccRCC.
In summary, we found that miR-337-3p suppressed in vitro cell proliferation and metastasis, and in vivo tumor growth in ccRCC. In addition, mechanistic study revealed that the effects of miR-337-3p on ccRCC cellular functions were partially mediated via Capn4. The present study provided the novel insights into the role of miR-337-3p in ccRCC progression.
Footnotes
Acknowledgments
This project was supported by the National Science Foundation of Jiangsu Province (no. BK20150251), the Youth Medical Talent Project of Jiangsu Province (no. QNRC2016292), and the China Postdoctoral Science Foundation (no. 2018M632371).
Supplementary data
Expression of Capn4 protein was up-regulated in ccRCC tissues. Expression of Capn4 protein in normal adjacent renal tissues (NC1, NC2, NC3 and NC4) and ccRCC tissues (C1, C2, C3 and C4) from patients with ccRCC. NC 
