Abstract
OBJECTIVE:
To investigate the role of miR-337-3p targeting Rap1A in modulating proliferation, invasion, migration and apoptosis of cervical cancer cells.
METHODS:
The expression levels of miR-337-3p and Rap1A in cervical cancer tissues and normal tissues were evaluated through quantitative Real-time PCR (qRT-PCR) and Western blotting; and correlations of miR-337-3p with clinicopathological characteristics and prognosis of patients were also analyzed. Besides, human cervical cancer cell line HeLa cells were randomly divided into five groups (Mock, NC, miR-337-3p mimic, Rap1A, and miR-337-3p mimic
RESULTS:
Cervical cancer tissues presented a significant decrease in miR-337-3p and a remarkable increase in Rap1A protein. Besides, the expression levels of miR-337-3p and Rap1A were closely related to the major clinicopathological characteristics of cervical cancer; and patients with high-miR-337-3p-expression had the higher 5-year survival rate (all
CONCLUSION:
MiR-337-3p was discovered to be decreased in cervical cancer, and miR-337-3p up-regulation may inhibit the proliferation, migration and invasion and promote the apoptosis of cervical cancer cells via down-regulating Rap1A.
Introduction
Cervical cancer, as a malignant tumor found in the cervical epithelium, is one of the most common gynecologic malignancies with very high incidence [1]. According to the statistics, there are over 500,000 newly diagnosed cervical cancer patients and nearly 250,000 deaths caused by cervical cancer each year, posing a serious threat to the health of women worldwide [2, 3]. Unfortunately, the current major therapies for cervical cancer including surgery, radiotherapy, chemotherapy or other comprehensive treatments, are very limited in the curative effect for patients with advanced cervical cancer [4], which had an important bearing on the invasion and metastasis of cervical cancer [5]. As documented previously, cervical cancer is suggested to be an extremely complex process, including the hyperplasia of normal cervical epithelium, the metaplasia to cervical intraepithelial neoplasia (CIN) I-III, and eventually the carcinogenesis, which is a multi-step process regulated by multiple genes [6, 7]. Besides, high-risk human papillomavirus (HR-HPV) infection has been recognized as one of the most important risk factors of cervical cancer [8]. Currently, the HPV-vaccines available protect against the two high-risk HPV types (including 16 and 18) that induce most cervical cancer [9], and cervical cancer caused by HPV might be successfully prevented by HPV vaccination and screening to some extent [10]. However, HPV vaccines are designed for prophylactic use only, which must be given before the infection occurs [11]. They do not clear existing HPV infection or treat HPV-related disease such as cervical cancer [12]. Therefore, we try best to explore in-depth the pathogenesis of cervical cancer in this study, hoping to lay a theoretical foundation for the early diagnosis and effective treatment of cervical cancer.
MicroRNAs (miRNAs), a class of non-coding endogenous single-strand RNA molecules composed of approximately 20–24 nucleotides, are widely distribu- ted in eukaryotes, which could control tumors’ formation and progression at different time points via regulating various cellular activities, such as proliferation, metastasis, and angiogenesis [13, 14]. In recent years, accumulating evidence has shown that many miRNAs were abnormally expressed in cervical cancer, such as miR-101 [15], miR-497 [16], and miR-372 [17], demonstrating significant effects of miRNAs on the early diagnosis and therapy selection for cervical cancer patients. Hsa-miR-337 (miR-337), a human miRNA locus located at chromosome 14q32.2 [18], has been reported to suppress the proliferation and invasion of pancreatic cancer through down-regulation of HOXB7 in the study by Zhang et al. [19], which could also inhibit the development of neuroblastoma by specifically reducing the expression of MMP-14 [20]. Of note, miR-337-3p could target both STAT3 and Rap1A, to modulate the progression and drug resistance of non-small cell lung cancer [18]. As for Rap1A, it is a member of the Ras-associated proteins (Raps) with the highest resemblance to Ras [21] and its function was realized by modulating endothelial cell junctions through E-cadherin, which was thought to be associated with the pathogenesis and development of tumors [22]. More importantly, the over-expression of RAP1 has an important bearing on the cervical intraepithelial neoplasia [23]. By referring to the target gene prediction website, we predicted Rap1A to be the target gene of miR-337-3p. However, no relevant report has elucidated clearly whether miR-337-3p can affect the development of cervical cancer through the modulation of Rap1A. Therefore, in this study, we investigated the effect of miR-337-3p and Rap1A on the proliferation, invasion, migration and apoptosis of cervical cancer cells, for the purpose of providing some new therapeutic targets for the diagnosis and treatment of cervical cancer.
Materials and methods
Ethics statement
All patients in the present study were informed of the experimental content and signed the written informed consent form. And the study was approved by the Ethics Committee for Clinical Experiments in The First People’s Hospital of Jingzhou City.
Subjects of the study
From January 2015 to January 2017, 86 cases of cervical cancer patients were enrolled as Case group of the study, who were 31–62 years old with the mean age of 48.7
Follow-up
The initial time point of follow-up was the date of diagnosis and the survival period was calculated on a monthly basis. The follow-up endpoint was patient death. Besides, follow-up examination was usually carried out every 3 months for the first year after treatment, every 3 to 6 months for the 2nd year after treatment, every 6 months for the 3rd year after treatment, and once a year since the 5th year after treatment.
Culture and transfection of cells
HeLa cells (the human cervical cancer cell line) were purchased from the American Type Culture Collection (ATCC) and then cultured in the RPMI-1640 medium (Hyclon) supplemented with 10% fetal bovine serum (FBS, Gibco), 100 units/ml penicillin (Hyclon) and 100 mg/ml streptomycin (Hyclon), in an incubator with 5% CO
Dual-luciferase reporter gene assay
In order to explore the impact of miR-337-3p on the expression of Rap1A, human Rap1A 3’-UTR fragment located at 3’-UTR and with the binding site of miR-337-3p was amplified to construct the wild-type plasmid Rap1A WT and the mutant-type plasmid Rap1A MUT. Cells were transfected with Lipofectamine 2000 Kit (Invitrogen) and the transcriptional activity was determined by the dual-luciferase reporter gene assay kit (Promega) according to the manufacturer’s instructions. The co-transfection systems were as follows: miR-337-3p mimic
Quantitative Real-time PCR (qRT-PCR)
Total RNA was extracted from tissues and cells with TRIzol Kit (Invitrogen), determined for concentration with NanoDrop2000 reagent (Thermo), and preserved at
Western blotting
Proteins extracted from tissues and cells were quantified by the BCA kit (Thermo Fisher, Rockford, IL). Then, proteins were heated for 10 min at 95
Serum-free cell survival assay
2
Cell proliferation by CCK-8 assay
HeLa cells were plated into the 96-well plate, with 100
Cell apoptosis by flow cytometry
HeLa cells were digested by trypsin, centrifuged, and collected, which were washed with ice-cold PBS buffer and re-suspended into single cell suspension (with 1
Wound-healing assay
Transfected HeLa cells were seeded into 6-well plates for incubation at 37
Transwell invasion assay
After 48 h transfection, matrigel (3.9 mg/ml, 60–80
Statistical method
Statistical analysis was performed using SPSS 21.0 software. Measurement data were presented by mean
Results
Expression of miR-337-3p and Rap1A in cervical tissues
The results of qRT-PCR demonstrated that miR-337-3p was markedly down-regulated in cervical cancer tissues when compared with normal tissues (
The association of miR-337-3p expression with cervical cancer patients’ clinicopathological features
The association of miR-337-3p expression with cervical cancer patients’ clinicopathological features
Expression levels of miR-337-3p and Rap1A in cervical cancer tissues and normal tissues. Note: A: The expression of miR-337-3p in cervical cancer tissue, cervical intraepithelial neoplasia (CIN) tissues and normal tissues determined by qRT-PCR; B, C: The expression of Rap1A protein in cervical cancer tissues and normal tissues evaluated by Western blotting; D: The effect of miR-337-3p expression on the 5-year survival rate of cervical cancer patients.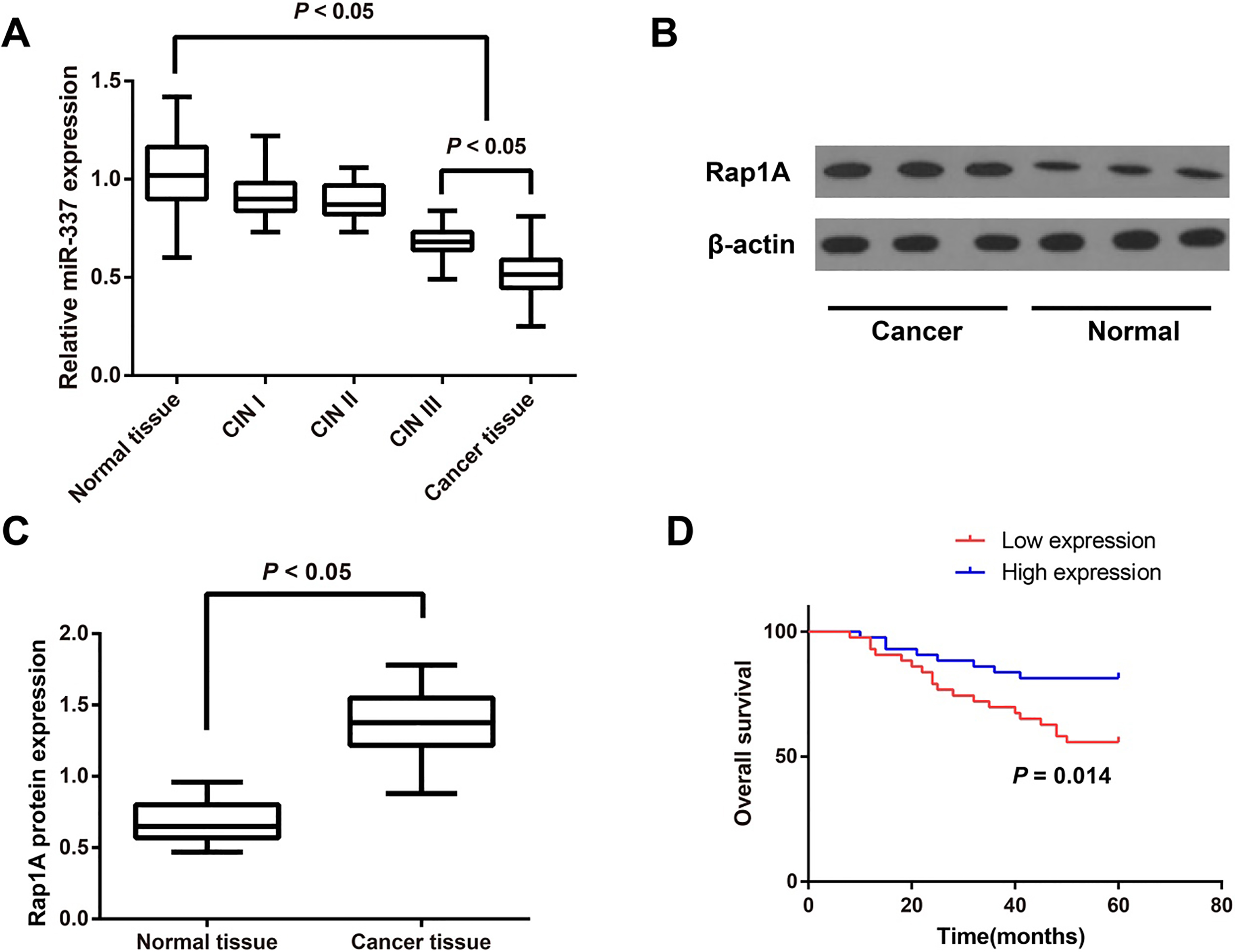
The targeting relationship between miR-337-3p and Rap1A. Note: A, The binding site of miR-337-3p to Rap1A predicted by using a public database and the results of dual-luciferase reporter gene assay; B, The expression of miR-337-3p in each group detected by qRT-PCR; C, D: The expression of Rap1A protein determined by Western blotting; *, 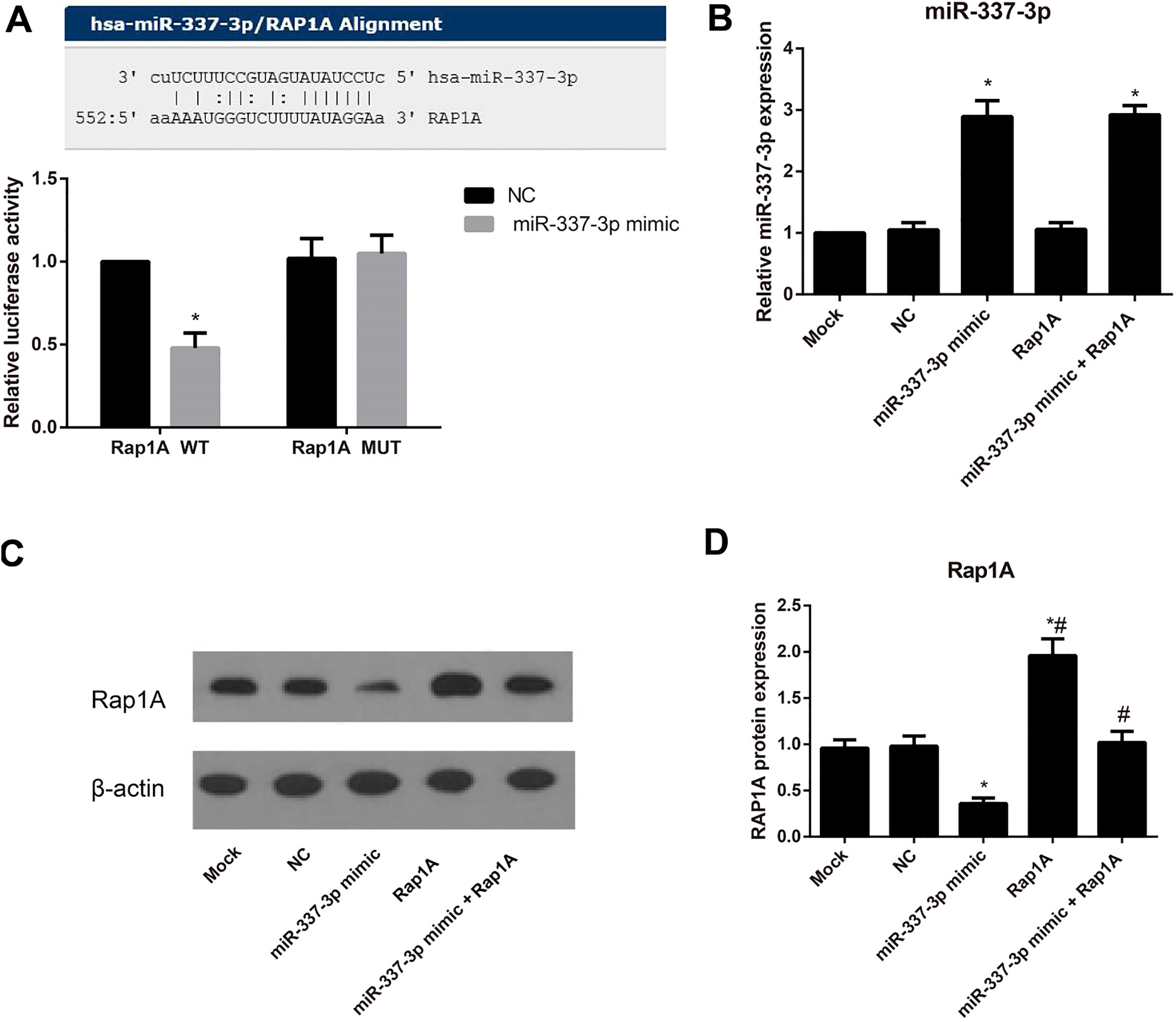
As shown in Table 1, the expression level of miR-337-3p in cervical cancer tissues had no correlation to the age and tumor size of patients (both
The impact of miR-337-3p on the proliferation and apoptosis of HeLa cells in each transfection group. Note: A, Survival of HeLa cells in serum-free or 10% fetal bovine serum medium; B, The impact of miR-337-3p expression on the HeLa cell proliferation in each group detected by CCK-8 assay; C, The impact of miR-337-3p expression on the HeLa cell apoptosis detected by flow cytometry; *, 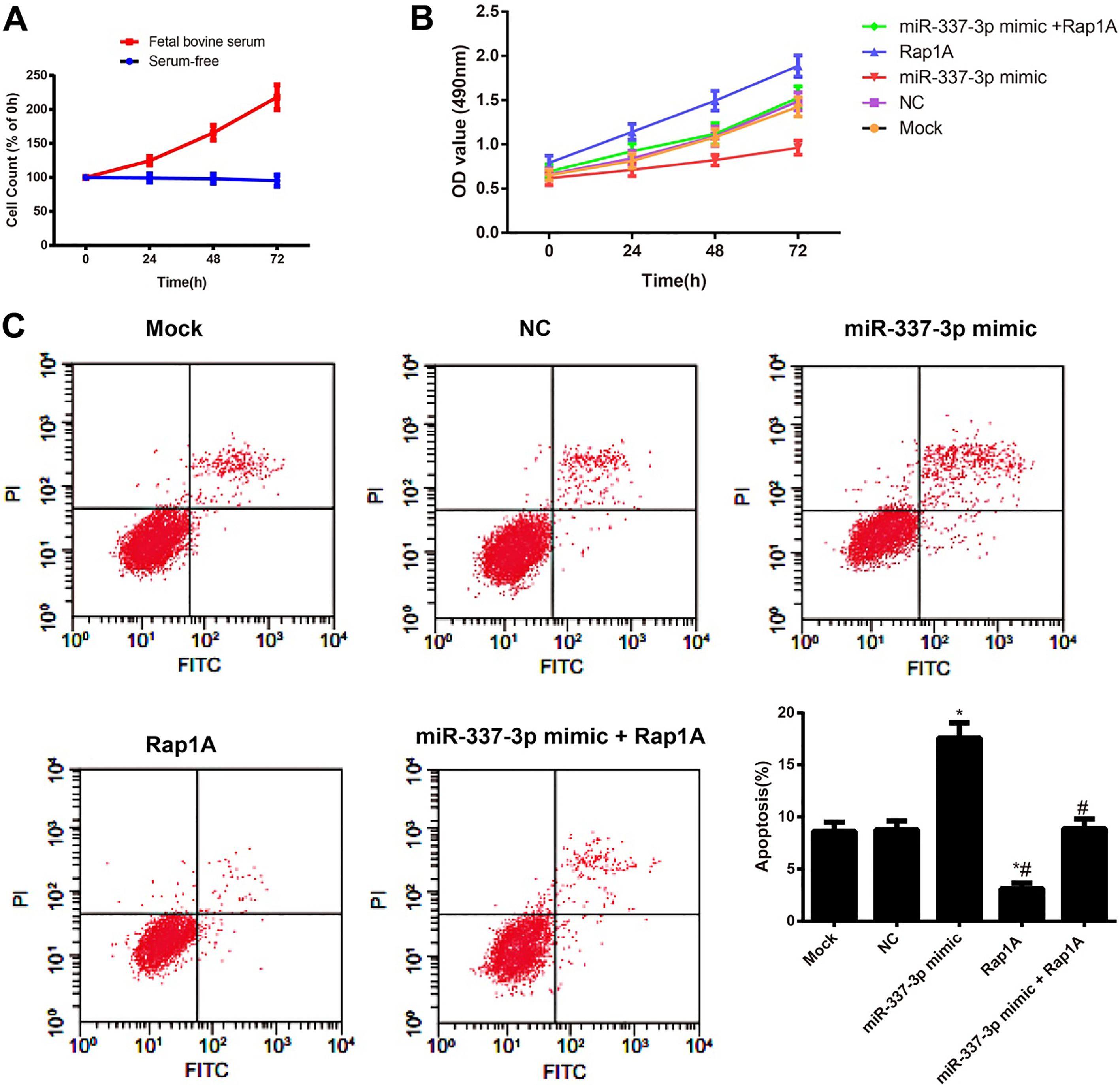
According to the prediction on the microRNA.org, the putative binding sequence of miR-337-3p was located on the 3’-UTR of Rap1A (Fig. 2A). The luciferase reporter gene assay showed that the luciferase activity was significantly decreased in cells co-transfected with the Rap1A WT and miR-337-3p mimic (all
The impact of miR-337-3p on the migration and invasion of HeLa cells. Note: A, The migration ability of HeLa cells in each group evaluated by the wound-healing assay; B, The invasion ability of HeLa cells in each group measured by Transwell Invasion assay; *, 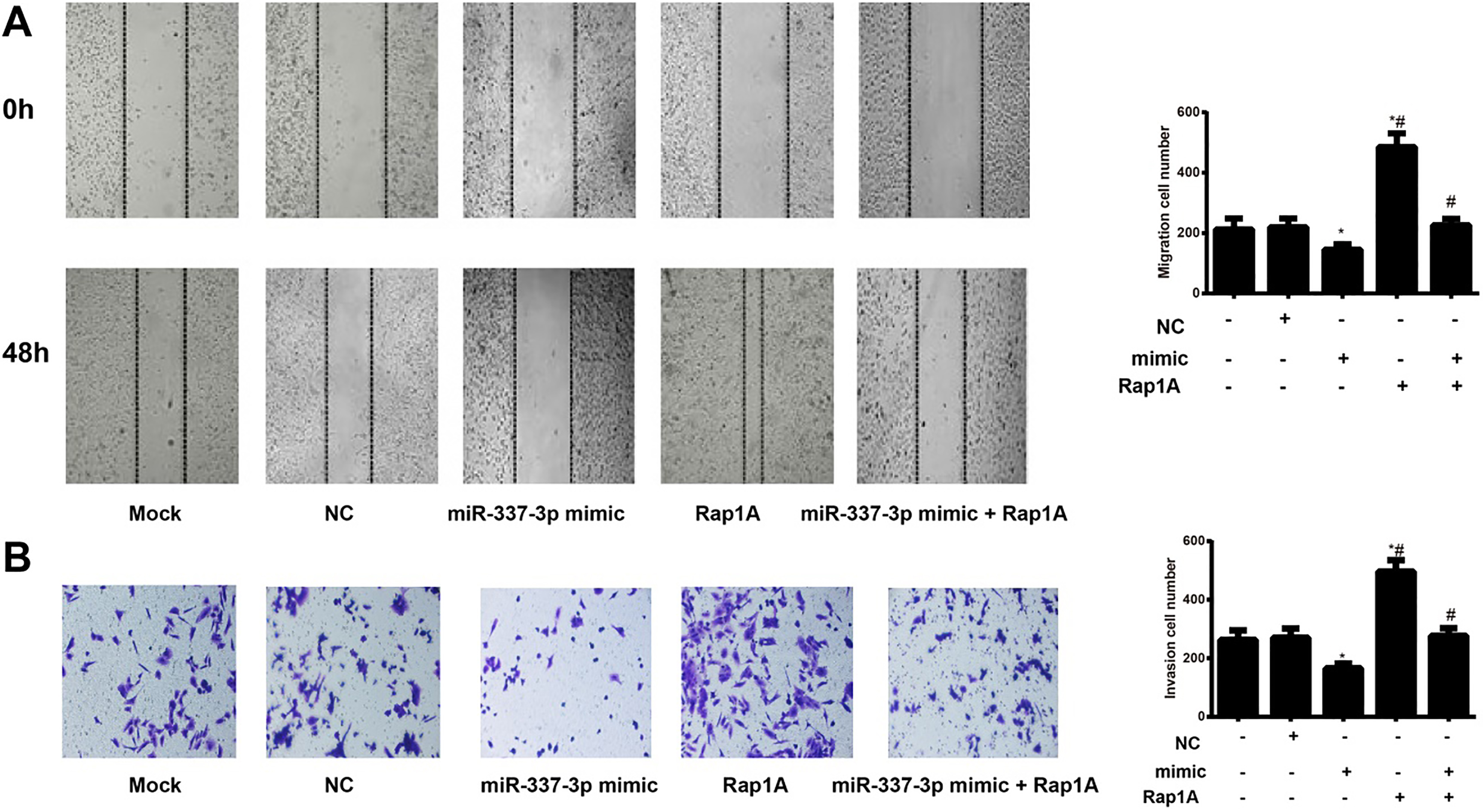
To investigate the effects of serum-free environment on the proliferation of HeLa cells, the cells were assessed cell proliferation over a three-day period. Although 10% fetal bovine serum-treated cells showed an exponential increase in cell count, cells treated with serum-free environment did not show much change in the number of viable cells, even after 3 days of treatment (Fig. 3A). Our data indicated that serum-free environment was able to rapidly inhibit proliferation of HeLa cells. Meanwhile, CCK-8 assay and flow cytometry were employed to evaluate the proliferation and apoptosis of cells, respectively (Fig. 3B and C). Compared with cells in the Mock group, those in the miR-337-3p mimic group were decreased in proliferation and elevated in apoptosis rate (both
Impact of miR-337-3p on the migration and invasion of HeLa cells
The migration and invasion abilities of HeLa cells were measured by wound-healing and Transwell assays separately. Obviously, miR-337-3p mimic group was statistically lower than the Mock group concerning cell migration and invasion abilities (both
Discussion
In this study, we detected the expression levels of miR-337-3p and Rap1A in cervical cancer tissues and normal tissues, and found the remarkable down-expression of miR-337-3p and the appreciable up-expression of Rap1A in cervical cancer tissues, which suggested that miR-337-3p may serve as a tumor suppressor gene while Rap1A as an oncogene in cervical cancer. What’s more, miR-337-3p was found to be down-regulated in a variety of tumors, including gastric cancer [24], non-small cell lung cancer [18], and pancreatic cancer [25]. On the other hand, Rap1, classified into two subtypes, namely Rap1A and Rap1B, was widely distributed in cells, which can be converted to inactive GDP-bound form or active GTP-bound form in a dynamic process [26]. Rap1-GTP as the active form was found abnormally expressed in many malignant tumor tissues [22, 27]. Evidence has pointed out that Rap1A expression was highly elevated in gynecologic malignancies, such as breast cancer and ovarian cancer [28, 29]. Worth mentioning, the persistent human papillomavirus (HPV) infection was the major risk factor of cervical cancer [21], and the HR-HPV E6 on its early protein encoding region can specifically degrade E6TP1 (which was one type of GTPase-activator proteins, RapGAPs) [30], to enhance the GTP hydrolysis activity of Rap1, thereby leading to the over-expression of Rap1 [30]. Recently, another study reported that Rap1-GTPase expression was significantly increased in cervical intraepithelial neoplasia and cervical cancer tissues with the severity of cervical lesions [23], providing the possibility that the increase of Rap1A expression in cervical cancer may result from its activation by HPV in the process of cervical carcinogenesis. Moreover, our study also found that miR-337-3p was lower in cervical cancer patients with poor differentiation, higher FIGO staging, and lymph node metastasis, as well as a poorer prognosis. Similarly, Xiang et al. also observed neuroblastoma patients had a better prognosis with a relatively higher expression of miR-337-3p, which could inhibit the disease progression in vitro and in vivo through down-regulation of MMP-14 [20]. The afore-mentioned findings all added weight to the hypothesis that miR-337-3p was closely associated with the pathogenesis and development of cervical cancer, and it was expected to become a biomarker in the prediction of patients’ prognosis.
Furthermore, it has been well-recognized that Rap1 can act as the target protein of some miRNAs to participate in the regulation of tumor progression. For example, a previous study reported that Rap1 was regulated by miR-518b in esophageal squamous cell carcinoma (ESCC) and the exogenous over-expressed Rap1 can reverse the regulatory role of miRNA [31]. In colorectal cancer, silencing miR-139 could suppress the tumor growth and carcinogenicity by up-regulating the expression of its target gene RAP1B [32]. Besides, Lin et al. [33] observed lowly-expressed miR-708 and highly-expressed Rap1B in ovarian tumors, and they also proved that miR-708 could down-regulate Rap1B in ovarian cancer cell lines to modulate the invasion and migration of cancer cells. Another important finding of our study was that Rap1A was the target gene of miR-337-3p, which was confirmed by the dual-luciferase reporter gene assay and further proved that miR-337-3p could specifically act on Rap1A in cervical cancer to affect the biological characteristics of cancer cells. On the other hand, in vitro cell transfection experiments in this study further proved that over-expressed miR-337-3p could restrain the proliferation, migration and invasion of cervical cancer cells, and enhance cell apoptosis rate at the same time. Meanwhile, the over-expression of Rap1A resulted in the completely opposite results. Consistent with our results, miR-337-3p could specifically down-regulate the expression of Rap1A to affect the development of non-small cell lung cancer, as indicated by Du et al. [18]. Another important finding of this study was that over-expressed Rap1A could reverse the inhibitory role of miR-337-3p in cell growth, which further added weight to the hypothesis that miR-337-3p played its roles in cervical cancer by targeting Rap1A. More similar examples have been presented in recent studies. For instance, it has been reported that Rap1 can promote the invasion and distant metastasis of human pancreatic cancer and breast cancer by mediating integrin and regulating actin reorganization [34, 35]. Besides, Rap1A could activate the transcription of MMP-7 in squamous cell carcinoma of the head and neck via the mediation of integrin and MMP-7 acted as an important biomarker for the invasion and migration of malignant cells [36]. Additionally, Rap1 could not only promote cell migration and inhibit cell differentiation by activating the MAPK/ERK signaling pathway, but also promote integrin-dependent cell adhesion and change cell invasion and metastasis [37, 38]. Moreover, Rap1-GTP could induce Rac1 to convert to active GTP-binding form, which can interact with the downstream effector PAK1 and activate the Rac1/PAK1 signaling pathway that was associated with the migration, invasion and other cellular processes [39]. Based on those findings, we hypothesized that miR-337-3p may target Rap1A to modulate the expression of its downstream genes and pathways, and thereby regulate the proliferation, migration and invasion of cervical cancer cells.
Taken together, our study found decreased miR-337-3p expression in cervical cancer tissues. Besides, over-expressed miR-337-3p may inhibit the proliferation, migration and invasion of cervical cancer cells and promote cell apoptosis by reducing the expression of Rap1A. Therefore, miR-337-3p may become a new diagnostic and prognostic biomarker for cervical cancer, and it also provides a novel perspective for the finding of therapeutic targets.
Footnotes
Acknowledgments
The authors appreciate the reviewers for their useful comments in this study.
Conflict of interest
None of the authors have any competing interests.
