Abstract
BACKGROUND:
The signaling adapter protein CrkL plays vital roles in multiple cancers. However, the expression pattern of CrkL protein and its clinical significance have not been well characterized in human gastric cancer (GC) so far.
OBJECTIVE:
To investigate the association of tissue-based CrkL protein expression level with the clinicopathological characteristics and prognosis of GC patients.
METHODS:
The expression level of CrkL protein in 380 GC patients was analyzed by immunohistochemistry. The associations of CrkL protein expression level with clinicopathologicalal characteristics and clinical outcome were evaluated.
RESULTS:
Compared with the matched adjacent non-tumor tissues, CrkL protein expression level was significantly up-regulated in tumor tissues. In addition, there was a positive correlation between CrkL and Ki67 expression levels in GC patients. An elevated CrkL level statistically correlated with aggressive clinicopathologicalal characteristics, such as larger tumor size, deeper local invasion, more lymph node metastasis, advanced TNM stage, and poorer prognosis. Notably, multivariate analysis identified tissue-based CrkL level as an independent predictor for the unfavorable prognosis of GC.
CONCLUSIONS:
These results indicate that CrkL protein may serve as a novel prognostic biomarker in GC.
Introduction
Gastric cancer (GC), although declining in incidence in the last decades, is still the fifth most common malignancy and the third leading cause of cancer-related death worldwide [1]. This disease affects nearly one million people per year, particularly in areas of Eastern Asian, Western Europe, and Southern and Central America [2]. In spite of the routine use of endoscopy combined with biopsy for diagnosis and health screening, due to the relatively asymptomatic nature of this disease in its early stages, the majority of GC patients are still diagnosed at advanced stages which negatively affect patient’s outcomes [3].
Nowadays, radical gastrectomy with D2 lympha- denectomy remains the best treatment in patients with resectable tumors [4], unfortunately, most GC patients are diagnosed in an advanced and unresectable stage; therefore, the best treatment for these patients includes chemotherapy, target therapies, and best supportive care, however, the clinical outcomes of these patients remain unsatisfactory with a 5-year overall survival rate of less than 30% [5].
Since no internationally standardized scheme of treatment could be unequivocally applied to GC patients, the identification of novel prognostic biomarkers which may provide important information on the likely course of disease, the prognosis as well as the suggestions for further treatment is urgently needed with the aim to improve the clinical outcomes of GC patients.
Our previous study showed that the expression of CrkL protein was remarkably up-regulated in GC tumor tissue compare to that in matched adjacent non-tumor tissue in a small set of GC patients. Moreover, a series of function assays in vitro, utilizing small-interfering RNA, were carried out. These assays included proliferation assay, cell cycle analysis, and apoptosis detection. Our finding revealed that knockdown of CrkL in SGC-7901 GC cells induced a suppression of cell proliferation along with a significant arrest of cell cycle in G0/G1 phase. Furthermore, we validated that CrkL was a direct target of miR-126, which functions as a tumor suppressor in GC, and over-expression of miR-126 inhibited the protein expression of CrkL significantly. Therefore, CrkL may contribute to GC progression through regulating proliferation, and miR-126 may function as a tumor suppressor in GC with CrkL as its direct target [6]. However, the prognostic value of CrkL protein in GC patients is yet to be elucidated. The aim of current study is thus to further analyze the relationship between tissue-based CrkL protein level and clinicopathological parameters, and its prognostic value in a larger set of GC patients. We expected that CrkL might become a novel prognostic biomarker of GC in the near future.
Materials and methods
Patients and clinical samples
Three hundred and eighty patients with primary GC who underwent radical gastrectomy with D2 lymphadenectomy at the Department of General Surgery, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine (Shanghai, China) from 2011 to 2012 were included in this study. The study protocol was approved by the Ethic Committee of Ruijin Hospital and signed informed consent was obtained from all study participants.
None of the subjects underwent chemotherapy or radiotherapy prior to surgery in order to eliminate potential treatment-induced alterations in gene expression profiles. Primary GC tumor tissues and matched adjacent non-tumor tissues were collected immediately following surgical resection. The pathologicalal stage was determined according to the TNM stage system of the eighth UICC/AJCC manual.
Follow-up of all patients was carried out for 5 years (or until death) according to our standard protocol (every 3 months in the first two years, every 6 months in the third year, and every 12 months afterwards or until death). No subject was lost to follow-up. The routine check-up during follow-up included physical examination, laboratory tests, chest X-ray, computed tomography (CT) and endoscopy. Disease-specific survival was used for evaluating the associations of CrkL protein expression level with the prognosis in the GC patients since it allowed controlling for unrelated causes of death.
Immunohistochemistry
Surgically excised tissue specimens were fixed in 10% neutralized formalin and embedded in paraffin blocks. Sections (4
Representative photographs of immunohistochemical analysis of CrkL (A, B, E, F) and Ki67 (C, D) in two paired tumor/non-tumor tissues. CrkL was detected in the cytoplasm of cancer cells, as well as in adjacent normal epithelial cells with different staining intensity (magnification, 100 
All the stained sections were evaluated independently by two pathologists. Five random fields were examined per slide, and 100 cells were evaluated per field under 100 magnification. Immunostaining of CrkL was scored following a semiquantitative scale by evaluating representative areas for staining intensity and percentage of positive cells. Cytoplasmic immunostaining was considered to be positive. The staining intensity was categorized as follows: 0, negative; 1, weak; 2, strong. The percentage of stained cells was scored as 0, 0%; 1, 1–25%; 2, 26–50%; 3, 51–75%; and 4, 76–100%. The scores of each sample were multiplied to give a final score of 0–8, and the samples with a final score
The chi-square test was used to evaluate the association between CrkL expression and clinicopathological factors. The cumulative probability of disease-specific survival was estimated by means of the Kaplan-Meier method. Univariate analyses of prognostic factors associated with disease-specific survival were performed using log-rank tests. Factors with significant effects on disease-specific survival in the univariate analyses were then subjected to multivariate analysis using a Cox proportional hazards model, using the forward stepwise procedure for variable selection, to identify independent prognostic factors. A two-tailed value of
Results
CrkL protein expression was up-regulated in GC
We used immunohistochemistry to investigate the expression of CrkL in the tumor tissues and matched adjacent non-tumor tissues obtained from 380 patients with primary GC. The immunohistochemical results showed clearly that CrkL expressed more strongly in tumor tissues than matched adjacent non-tumor tissues (mean
Up-regulation of CrkL protein was associated with adverse clinicopathological parameters of GC patients
The association between CrkL expression and various clinicopathological parameters was investigated. As shown in Table 1, no statistical difference was found between the CrkL expression and such clinicopathological parameters as sex (
Clinicopathological associations of CrkL protein expression in GC
Clinicopathological associations of CrkL protein expression in GC
Univariate analyses of prognostic factors in GC
Kaplan-Meier curves of disease-specific survival according to the expression level of CrkL.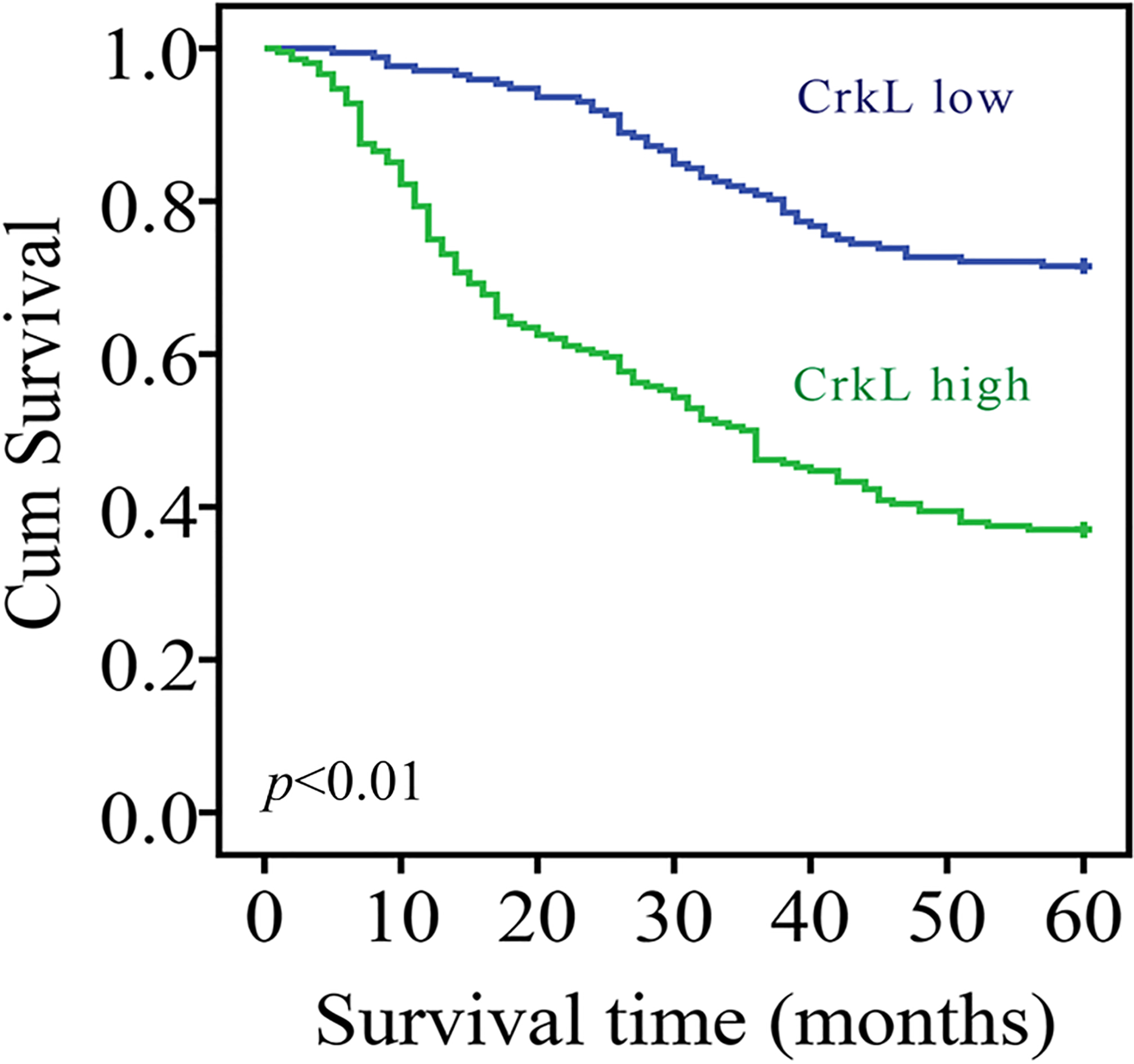
Multivariate analyses of prognostic factors in GC
We further investigated the factors that could predicate the prognosis of GC patients by using the univariate and multivariate analyses. An analysis of the disease-specific survival was performed using the Kaplan-Meier approach, with the statistical analysis performed using the log-rank test. The results demonstrated clearly that there was a significant difference in the survival between the CrkL high-expression group and low-expression group (Fig. 2). The 5-year disease-specific survival rate in the patients with high CrkL level was 37.0%, whereas that in the patients with low CrkL level was 71.5% (
Kaplan-Meier curves of disease-specific survival according to the expression level of CrkL, stratified by TNM stage. No statistically significant difference in the disease-specific survival was observed between low and high CrkL expression in stage I/II GC patients (A). The prognosis was significantly worse in the high CrkL expression population than that in the low CrkL expression population in stage III/IV GC patients (B).
To further evaluate the relationship between CrkL expression and prognosis in patients with GC, subgroup analysis based on the different TNM stage category or tumor size category was performed subsequently. No statistically significant difference in the disease-specific survival was observed between low and high CrkL expression groups in stage I/II GC patients (Fig. 3A). However, there was statistically significant difference in the disease-specific survival between low and high CrkL expression groups in stage III/IV GC patients. The prognosis was significantly poorer in the high CrkL expression group than that in the low CrkL expression group (Fig. 3B). We next analyzed the prognostic significance of CrkL expression with respect to tumor size. We found that the prognosis for high CrkL expression group was significantly poorer in both tumor size
Kaplan-Meier curves of disease-specific survival according to the expression level of CrkL, stratified by tumor size. The prognosis was significantly worse in the high CrkL expression population than that in the low CrkL expression population in both small tumor size subgroup (A) and big tumor size subgroup (B).
GC is a leading cause of cancer-related mortality, and a major health problem worldwide [7]. Despite improvements in surgical and adjuvant treatment approaches, the clinical outcome for patients with GC remains worse than that for patients with other common malignancies such as colon, breast, and prostate cancer. The TNM staging (tumor, lymph nodes and metastasis) is the most important and widely used tool to predict prognosis of GC, and the main therapeutic reference and guidelines in current clinical practice as well [8]. However, the fact that prognosis often varies between GC patients even at the same TNM staging manifests clearly that there must be molecular factors responsible for these clinical differences [9].
It is generally believed that GC results from the combined effects of environmental factors and genetic alterations, including tumor suppressor genes, oncogenes, growth factors, and cell adhesion molecules [10]. Although previous studies have reported that multiple aberrantly expressed genes could contribute to the malignant behavior of GC cells, biomarkers that are able to identify tumor progression and predict the aggressive phenotype are still lacking in GC. Thus, identifying novel prognostic biomarkers of GC in addition to TNM staging will be definitely helpful to refine the accuracy of prognostic stratification, and henceforth design more reasonable treatment and surveillance strategy for this devastating illness [11].
The signaling adaptor protein Crk was originally isolated as an oncogene fusion product of an avian sarcoma CT10 retrovirus (v-Crk) [12]. Its mammalian homologs including Crk I, Crk II and CrkL proteins are the first identified adaptor proteins which connect with upstream proteins through their SH2 (Src homology 2) domain and with downstream proteins through their NH2-terminal SH3 (Src homoloyg 3) domain [13]. Crk proteins and their effectors are highly conserved throughout evolution. Several binding partners have been identified for the SH2 and SH3 domains of Crk proteins so far. The SH2 domain binds a specific phosphorylated tyrosine motif found in proteins involved in cell spreading, actin reorganization, and cell migration. These SH2 domain binding partners include p130Cas and paxillin [14, 15], which are effectors downstream of integrin activation, as well as the Gab1 and Gab2, which are effectors downstream from many tyrosine kinases [16]. The NH2-terminal SH3 domain binds proline-rich motifs present within several proteins which include Abl tyrosine kinase, Arg, C3G (a nucleotide exchange factor for Rap1), DOCK180 (an exchange factor for Rac1), etc. [17, 18, 19, 20]. However, the binding proteins for the COOH-terminal SH3 domain are still poorly understood. It’s now clear that Crk proteins play central roles in signaling cascades that regulate a variety of cellular and biological processes such as proliferation, differentiation, migration and apoptosis through i) protein complex formation downstream from receptor activation, ii) regulation of cellular tyrosine kinase activity, and iii) upstream roles in signal initiation [21].
Although early studies focused on Crk proteins in the transformation of fibroblasts [22], the studies of Crk proteins in epithelial cell models has further elucidated the role of Crk proteins in the amplification and/or initiation of oncogenic signals, suggesting that Crk proteins dysregulation can cooperate with a variety of oncogenic signals to promote tumorigenesis. For example, overexpression of Crk II and CrkL in breast cancer epithelial cells enhances epithelial-to-mesenchymal transitions (EMT), characterized by the breakdown of adherens junctions, enhanced cell dispersal, and remodeling of cortical actin [23]. Whereas, siRNA-mediated knockdown of Crk significantly reduced invasion and migration of breast cancer cells [24], implicating Crk proteins as critical signal transducers for cell invasion in breast cancer. Similar conclusions have been demonstrated in lung, oral, head, prostate, and ovarian cancer. For example, Crk II knockdown could down-regulate IGF-IR levels and suppressed PI3K/Akt signaling pathway in prostate cancer cells, and further inhibited their migration and invasion [25].
The role for Crk proteins in promoting gastric tumorigenesis is suggested by multiple studies. The phosphorylated CagA binds Crk proteins (Crk I, Crk II, and CrkL), and that interaction is important for the CagA-mediated host responses during H. pylori infection which may further cause GC [26]. Crk genetic variants can be susceptible genetic factors for the development of GC by controlling signals through the CagA transduction pathways, and an interaction between Crk genetic polymorphism and phytoestrogen biomarkers appear to play a role as risk modifiers in gastric carcinogenesis [27]. Knockdown of CrkL abrogates CCL20-induced Erk1/2 signaling and the progression of EMT which plays an important role in the initiation of pathogenesis of normal epithelial cells, migration, invasion as well as metastasis in GC [28]. Of note, miR-126 which was found to be down-regulated and function as a tumor suppressor in a variety of cancers also affects the proliferation and metastatic potential of GC cells, partly through regulating Crk I, Crk II and CrkL in our previous studies [6, 29].
Pathophysiologically, Crk proteins are overex- pressed in a wide array of human cancers, including but not limited to ovarian cancer, lung cancer, and breast cancer; whereby the level of expression often correlates with the tumor aggressiveness, disease progression, and inversely segregates with prognosis, indicating that Crk proteins may be used as potential prognostic biomarkers. For example, Miller et al. demonstrated that CRK I/II mRNA expression in lung cancer was increased in more advanced, larger and poorly differentiated tumors and was associated with poor survival [30]. Wang et al. showed that overexpression of CrkL correlated with poor prognosis in lung cancer patients [31]. Analogous to overexpression of Crk proteins in aggressive lung cancer, analysis of CrkL in ovarian cancer also revealed strong correlations between CrkL expression and poor prognosis [32]. CrkL overexpression was also found to be a significant, independent poor prognostic indicator for hepatocellular carcinoma. Results from IHC assay indicated that the high expressions of CrkL strongly correlated with reduced disease-free and overall survival of hepatocellular carcinoma patients [33]. Glioblastoma patients with high Crk II and Crk phosphoY251 expression had distinctly lower overall survival rates than those with low Crk II and Crk phosphoY251 expression [34]. In addition, our previous study demonstrated clearly that up-regulation of Crk I/II proteins was associated with poor prognosis of GC patients [35].
Despite growing evidence highlighting its pivotal roles in the progression of GC, no study has systematically explored the prognostic value of CrkL in GC. In the current study, we evaluated the clinical and prognostic significance of CrkL expression in GC using immunohistochemistry. We observed the overexpression of CrkL in GC tumor tissues comparing to that in adjacent non-tumor tissues. We further examined the correlation between CrkL level and the progression and prognosis of GC. Our results demonstrated that the overexpression level CrkL was associated with various clinicopathological factors related with poor prognosis, such as tumor size, local invasion, lymph nodes metastasis and TNM staging, suggesting that CrkL expression may influence biological features of GC and might play a pivotal role in tumorigenesis and development of GC. Moreover, GC patients with higher CrkL level had worse prognosis compared with those with lower level. Cox proportional hazards regression model analysis revealed that high expression level of CrkL was an independent factor predicting poor outcome of GC patients, which was in accordance with the results of lung, ovarian, and liver cancer. In this respect, CrkL might possess the same potential in prognosis prediction of GC as found with the classical prognostic factors such as lymph node metastasis or TNM stage. To the best of our knowledge, this is the first study to investigate the clinical significance of CrkL expression in a large set of GC patients.
In conclusion, our data offer the convincing evidence for the first time that the CrkL expression was markedly and consistently up-regulated in GC patients, and high CrkL level was associated with poor prognosis. CrkL thus has potential clinical utility as a promising biomarker for molecularly monitoring the progression and predicting the clinical outcome of patients with GC. This discovery may help to better identify GC patients with poor prognostic potentials and facilitate personalized treatment of GC patients.
Footnotes
Acknowledgments
We appreciate all subjects that participated in this study. This project was supported, in part, by the grant from Shanghai Municipal Education Commission (12zz102), Yi Gong Jiao Cha Foundation of Shanghai Jiao Tong University (YG2016MS65) and Shanghai Pujiang Program (17PJ1406000).
Conflict of interest
The authors declare no conflict of interest.
