Abstract
BACKGROUND:
MicroRNAs (miRNAs) have been reported to serve as potential biomarkers in various cancer and play important roles in tumor progression.
OBJECTIVE:
The aim of this study was to investigate the prognostic significance and functional role of miR-383-5p in breast cancer.
METHODS:
The expression levels of miR-383-5p in breast cancer tissues and cell lines were measured using quantitative real-time PCR analysis. Kaplan-Meier curve and Cox regression analysis were used to explore the prognostic significance of miR-383-5p in breast cancer. The CCK-8 assay was used to assess cell proliferation ability. Transwell assays were used to assess cell migration and invasion abilities of breast cancer cells.
RESULTS:
The expression of miR-383-5p was significantly downregulated in breast cancer tissues and cell lines, compared with that in normal tissues and normal epithelial MCF-10A cells, respectively. The expression of miR-383-5p was associated with differentiation, lymph node metastasis, and TNM stage. Patients with low miR-383-5p expression had shorter overall survival than those with high miR-383-5p expression. Overexpression of miR-383-5p significantly inhibited cell proliferation, migration, and invasion, while downregulation of miR-383-5p promoted cell proliferation, migration, and invasion in vitro. LDHA was a direct target of miR-383-5p.
CONCLUSIONS:
Taken together, miR-383-5p was downregulated in breast cancer tissues and cell lines, and overexpression of miR-383-5p inhibited cell proliferation, migration, and invasion in breast cancer cells by targeting LDHA. Based on our findings, miR-383-5p may be a prognostic biomarker and therapeutic target for breast cancer.
Introduction
Breast cancer, one of the most frequently diagnosed malignancy with high prevalence in women worldwide, is the second and fifth leading cause of cancer death in the USA and China, respectively [1, 2]. Because breast cancer is a highly heterogeneous and complex malignancy, despite improvement in diverse therapeutic strategies, the overall survival rate of patients with advanced stages of breast cancer is still not ideal [3, 4]. Importantly, patients at advanced stage usually with metastatic lesions to the lymph nodes or other organs, such as bone marrow, lung, and liver leads to death. Therefore, it is essential to identify novel factors for the prediction and effective treatment of breast cancer and design more novel treatment strategies.
MicroRNAs (miRNAs) are a group of endogenous small noncoding RNAs (approximately 22 nucleotides in length), which regulate gene expression by binding the 3’-UTRs of the target genes, resulting in degradation or translation inhibition of target mRNAs [5, 6, 7]. Numerous studies have demonstrated that the function of miRNAs is widely involved in diverse biological processes, such as cell growth, proliferation, differentiation, migration, and invasion [8, 9]. Biological studies showed that miRNAs play an oncogenic or tumor suppressor role in the initiation and progression of human cancers [10, 11]. Recent studies also showed numerous miRNAs are aberrantly expressed in breast cancer, such as miR-329 [12], miR-27a [13], and miR-128-3p [14], indicating miRNAs play important roles in breast cancer. miR-383-5p, located in chromosome 8p22, acts as a tumor suppressor in many cancers, such as gastric cancer, colorectal cancer, and lung adenocarcinoma [15, 16, 17]. A previous study reported some breast cancer-associated miRNAs, including miR-383-5p [18]. Thus, it would be of interest to investigate the clinical significance and functional roles of miR-383-5p involved in breast cancer carcinogenesis.
Association between miR-383-5p expression and different clinical characteristics of patients with breast cancer
Association between miR-383-5p expression and different clinical characteristics of patients with breast cancer
In the present study, we investigated the expression levels of miR-383-5p in breast cancer tissues and cell lines, as well as the association between the expression profile of miR-383-5p and clinicopathological characteristics of patients with breast cancer. The prognostic significance of miR-383-5p and the effects of miR-383-5p on cellular behavior were also evaluated in breast cancer patients.
Patients and tissue samples
Paired human breast cancer tissue samples and adjacent normal tissue samples were obtained from 124 patients who received surgical resection between January 2011 and June 2013 at Shanxian Central Hospital. All surgical specimens were snap-frozen in liquid nitrogen and stored at
RNAseq analysis
RNAseq was performed to validate the expression levels of miR-383-5p in breast cancer based on the TCGA database. TCGA RNAseq data contains 1104 primary tumors and 105 solid tissue normal samples.
Cell culture and transfection
Breast cancer cell lines (MDA-MB-231, MDA-MB-453, MCF-7, ZR-75-1) and human normal epithelial cells MCF-10A were all purchased from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China). All breast cancer cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM; Thermo Fisher Scientific, Waltham, USA) supplemented with 10% fetal bovine serum (FBS; Thermo Fisher Scientific, Waltham, USA) and 1% penicillin-streptomycin mixture (Gibco; Thermo Fisher Scientific, Waltham, USA) at 37
The miR-383-5p mimic (5’-AGAUCAGAAGGUG AUUGUGGCU-3’), mimic negative control (mimic NC; 5’-ACUACUGAGUGACAGUAGA-3’), miR-383-5p inhibitor (5’-AGCCACAAUCACCUUCUGA UCU-3’), and inhibitor NC (5’-UUCUCCGAACGU GUCACGUTT-3’) were purchased from GenePharma Co., Ltd (Shanghai, China). 1
RNA extraction and quantitative real-time PCR
Total RNA was extracted from all breast cancer tissues or cells using TRIzol reagent (Invitrogen; Thermo Fisher Scientific, Waltham, USA) following the manufacturer’s protocol. After quantifying the RNA by using NanoDrop 2000 (Thermo Fisher Scientific, Waltham, USA), extracted RNA was used to synthesize complementary DNA (cDNA) using a PrimeScript RT reagent kit (Takara, Otsu, Japan). Reverse transcription primer for miR-383-5p was 5’-CTC AACTGGTGTCGTGGAGTCGGCAATTCAGTTGAGAGCCACAA-3’. The following temperature was used: 37
Cell proliferation assay
Cell proliferation abilities were measured using the Cell counting kit-8 (CCK-8; Dojindo, Kumamoto, Japan) assay. The breast cancer cells were transfected with miR-383-5p mimic, miR-383-5p inhibitor, or NCs as aforementioned. After 48 h of transfection, the transfected cells were seeded in 96-well plates at a density of 3
Cell migration and invasion assays
Cell migratory and invasive abilities were assessed using a 24-well Transwell chamber (Corning, NY, USA) with 8
The expression of miR-383-5p was decreased in breast cancer tissues and cell lines. (A) Expression levels of miR-383-5p were decreased in breast cancer tissues compared with adjacent normal tissues. (B) Validation expression of miR-383-5p in breast cancer using TCGA RNAseq data. (C) Expression levels of miR-383-5p were downregulated in breast cancer cell lines (MDA-MB-231, MDA-MB-453, MCF-7, and ZR-75-1) compared with normal epithelial cells MCF-10A. ***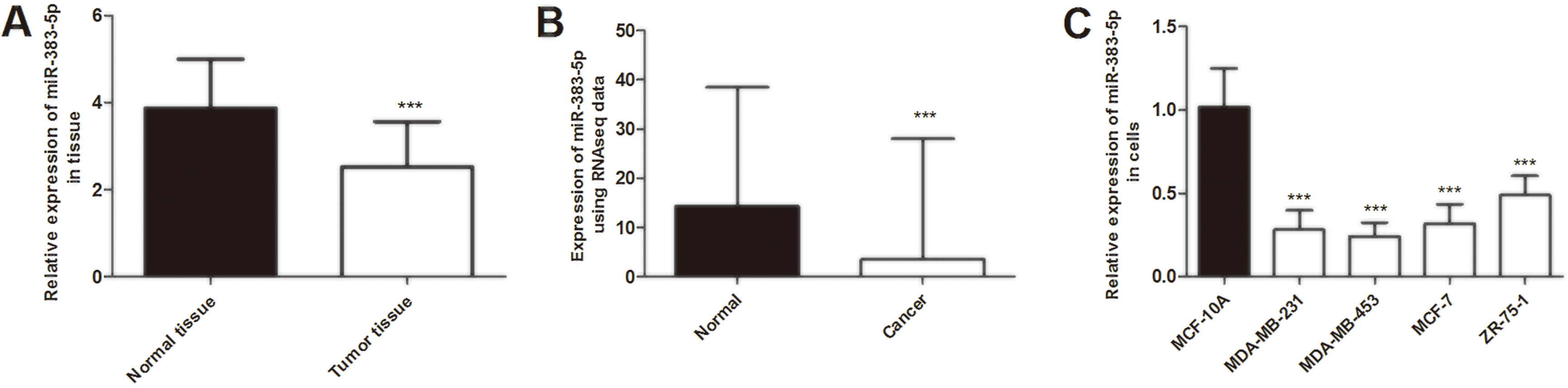
Putative miR-383-5p targets were predicted using online algorithms TargetScan (
Statistical analysis
All assays were performed at least three times. Data were analyzed using SPSS 20.0 software (SPSS, Inc., Chicago, IL, USA) and GraphPad Prism 5.0 software (GraphPad, Inc., La Jolla, CA, USA) and presented as mean
Results
Expression of miR-383-5p in breast cancer tissues and cell lines
First, qRT-PCR was used to identify the expression levels of miR-383-5p in 124 pairs of breast cancer tissues and normal tissues. The results showed that miR-383-5p expression in breast cancer tissues was significantly lower than that in paired normal tissues (
Then we detected the expression levels of miR-383-5p in breast cancer cells. We observed that the relative expression of miR-383-5p was downregulated in breast cancer cells compared with human normal epithelial MCF-10A cells (
Association between miR-383-5p expression and clinical characteristics of breast cancer patients
Next, we investigated the relationships between miR-383-5p expression and clinical factors of breast cancer patients. According to the mean level of miR-383-5p expression, breast cancer patients were divided into two groups, with 64 in the low miR-383-5p expression group and 60 in the high miR-383-5p expression group. As shown in Table 1, miR-383-5p expression was significantly associated with differentiation (
The miR-383-5p expression is correlated with survival outcomes of breast cancer patients
Considering miR-383-5p expression was associated with lymph node metastasis and TNM stage of breast cancer patients, we wonder whether miR-383-5p was correlated with the survival of patients. In order to explore the prognostic significance of miR-383-5p in breast cancer, Kaplan-Meier analysis, and log-rank tests were used to analyze the association between miR-383-5p expression and 5-year overall survival information. As expected, Kaplan-Meier survival analysis indicated that low miR-383-5p expression predicted poorer overall survival (
Multivariate Cox analysis of factors for survival of breast cancer patients
Multivariate Cox analysis of factors for survival of breast cancer patients
Kaplan-Meier curve of survival time in patients with breast cancer. Patients with low miR-383-5p expression levels exhibited a shorter overall survival rate (log-rank test 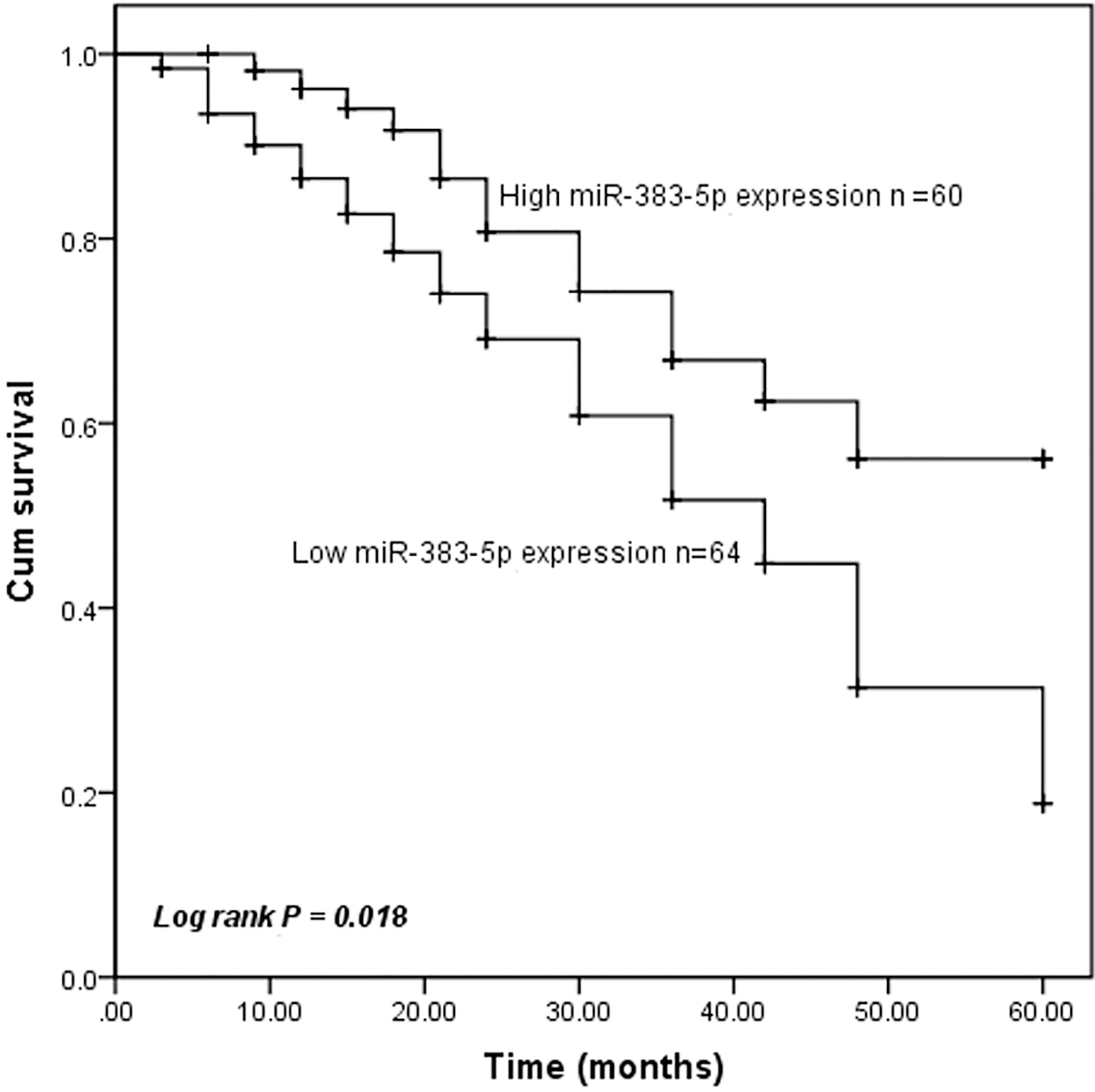
To further investigate the role of miR-383-5p in breast cancer tumorigenesis, miR-383-5p mimic, mimic NC, miR-383-5p inhibitor, or inhibitor NC was transfected into MDA-MB-231 or MDA-MB-453 cells, respectively, to conduct gain-of-function and loss-of-function assays. The qRT-PCR results showed that miR-383-5p mimic significantly upregulated miR-383-5p expression, while miR-383-5p inhibitor downregulated miR-383-5p expression, compared with control (
Effects of miR-383-5p expression levels on proliferation in MDA-MB-231, MDA-MB-453, and MCF-7 cells. (A) Validation of miR-383-5p expression levels using qRT-PCR analysis after transient transfection. The expression of miR-383-5p-5p was significantly higher following transfection with miR-383-5p mimic and lower following transfection with miR-383-5p inhibitor compared with control (B) Proliferation of breast cancer cells was inhibited following transfection with miR-383-5p mimic and promoted following transfection with miR-383-5p inhibitor. *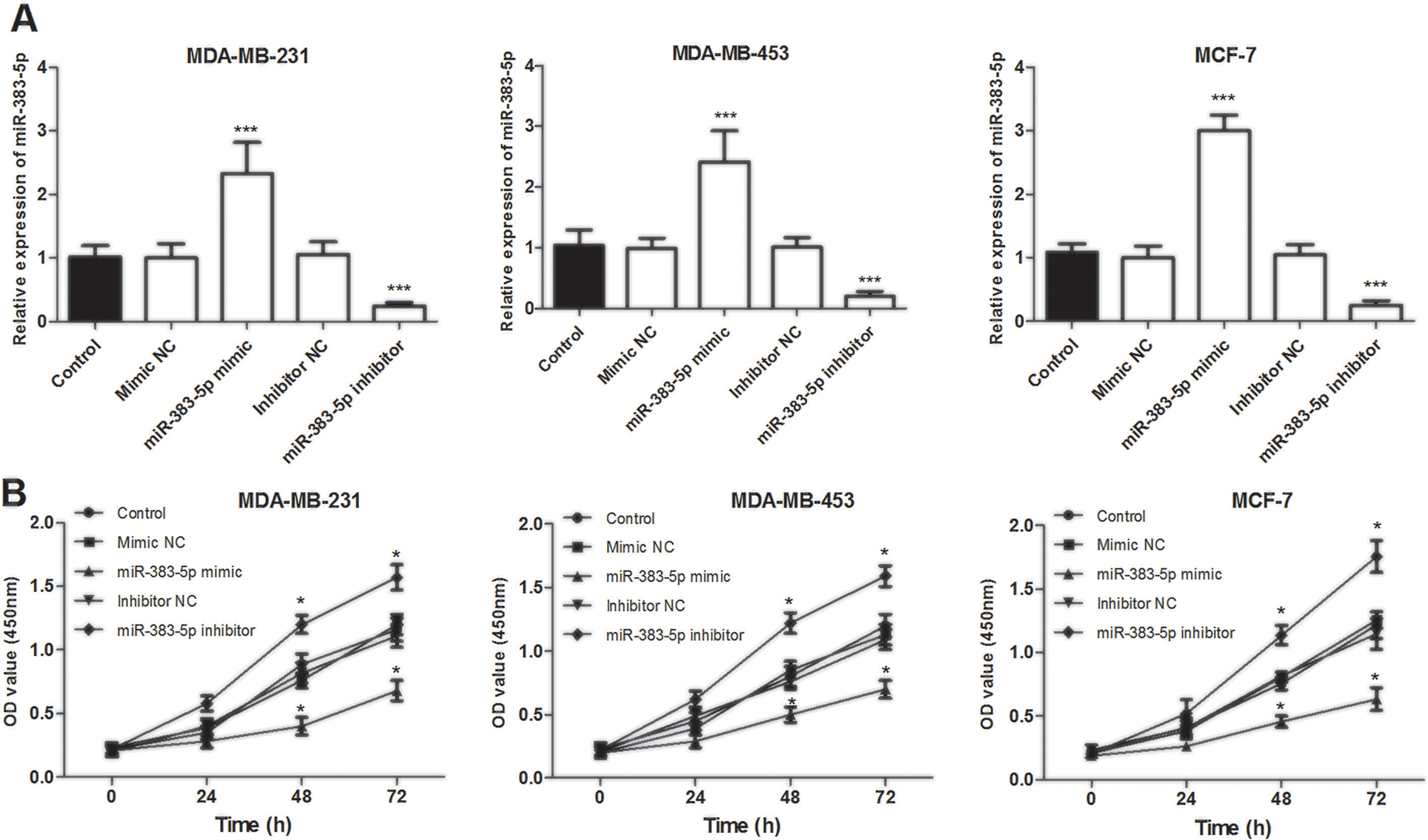
Effects of miR-383-5p on cell migration and invasion abilities in MDA-MB-231, MDA-MB-453, and MCF-7 cells. (A) Representative images of Transwell migration assay (magnification, 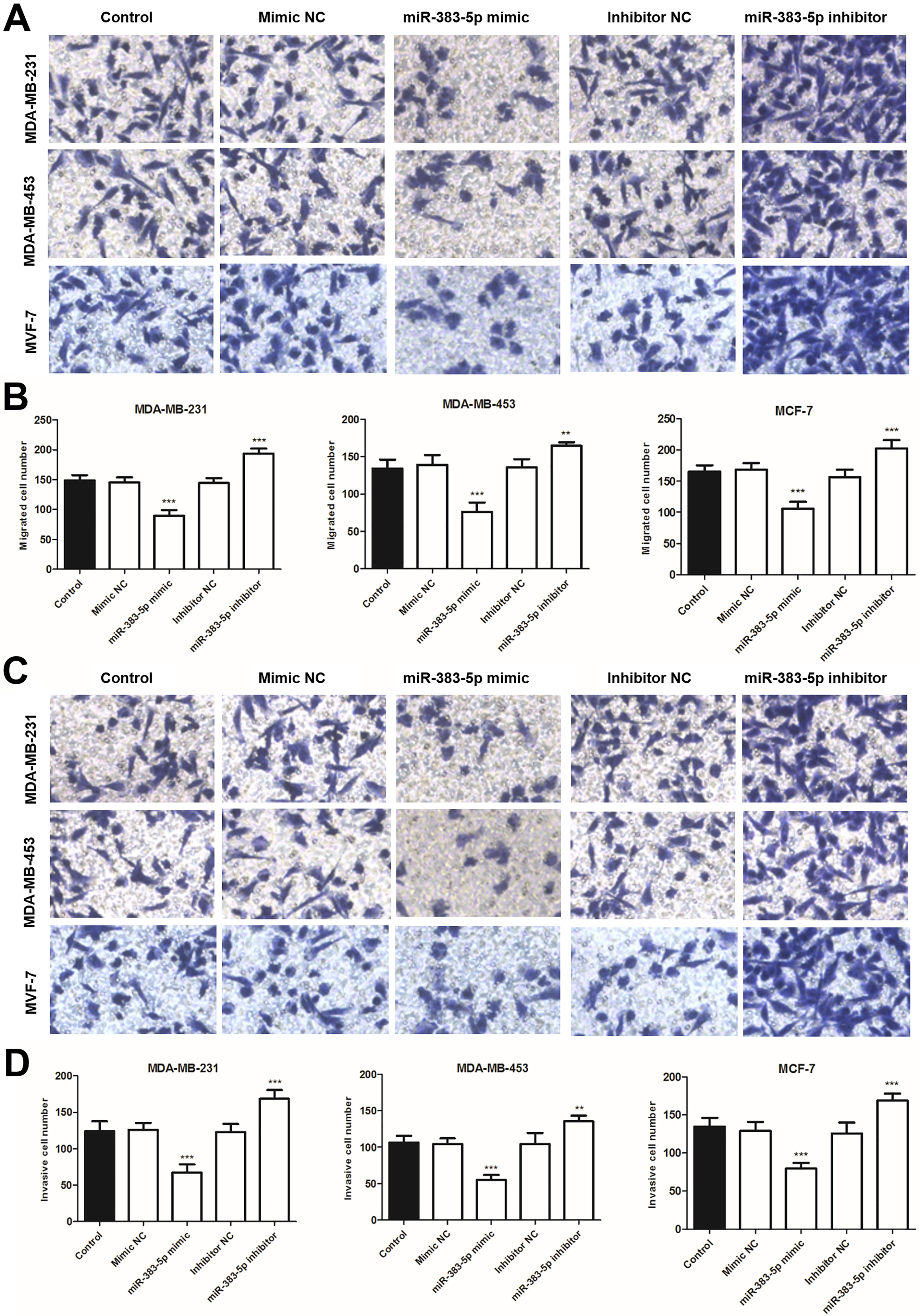
The potential targets of miR-383-5p were investigated by bioinformatics analysis using the website TargetScan. Among these predicted targets, LDHA was selected as a potential target gene of miR-383-5p. The putative binding sites between miR-383-5p and LDHA 3’-UTR were presented in Fig. 5A. Then, luciferase reporter assay was performed to validate this interaction. The reporter plasmids were co-transfected with miR-383-5p mimic, mimic NC, miR-383-5p inhibitor, or inhibitor NC in MCF-7 cells. The luciferase reporter assay results indicated that miR-383-5p mimic transfection significantly decreased the luciferase activity of the WT 3’-UTR of LDHA, while without effects on the Mut plasmids in MCF-7 cells (
LDHA was targeted by miR-383-5p in breast cancer. (A) Putative binding sites of miR-383-5p and LDHA were predicted by TragetScan. (B) The luciferase activity was investigated in MCF-7 cells co-transfected with WT or Mut luciferase vectors and miR-383-5p mimic, mimic NC, miR-383-5p inhibitor, or inhibitor NC. Overexpression of miR-383-5p decreased the luciferase activity of the WT 3’-UTR of LDHA, while without effects on the Mut plasmids in MCF-7 cells *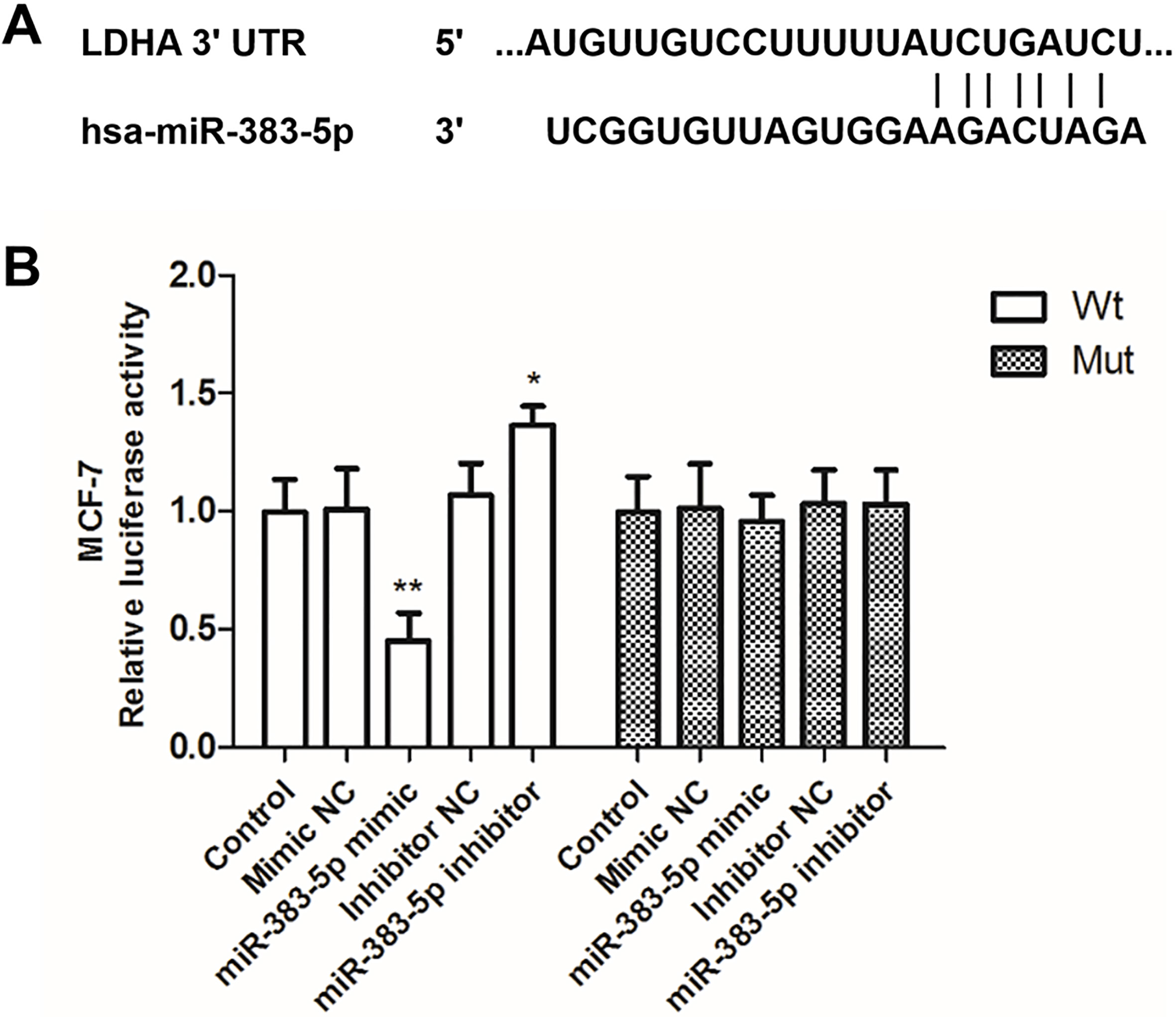
Accumulating evidence has demonstrated that miRNAs involved in tumor initiation and progression of cancers, which are still under investigation and act as a new class of promising cancer biomarkers [20, 21]. Numerous miRNAs were also reported to be associated with breast cancer [22, 23]. For instance, miR-876-5p was downregulated in breast cancer cells and suppressed breast cancer cell proliferation, migration, and invasion by targeting TFAP2A [22]. Serum miR-21 and miR-125b were upregulated in breast cancer patients and both of them were identified as the novel, noninvasive predictive markers for neoadjuvant chemotherapy response and prognosis in breast cancer [23]. However, there is still lacking molecular markers available for clinical application in breast cancer.
Many studies have shown miR-383-5p was abnormally regulated in various cancers [15, 16, 17]. As most studies have indicated, miR-383 acts as a tumor suppressor in inhibiting cell proliferation, migration, and enhancing chemo-sensitivity in multiple cancers [24]. However, the potential role of miR-383-5p in breast cancer is still unclear. In the present study, miR-383-5p was downregulated in breast cancer tissues and cells, compared with adjacent normal tissues and cells, respectively. Then, we validated the expression of miR-383-5p in TCGA RNAseq data and the results confirmed that miR-383-5p was downregulated in breast cancer compared with normal control. Furthermore, the majority of patients with low miR-383-5p expression exhibited poor differentiation, positive lymph node metastasis, and advanced TNM stage. The above results suggest that miR-383-5p may be involved in the development of breast cancer. Furthermore, given the dysregulated miR-383-5p expression in breast cancer tissues, we evaluated the prognostic significance of miR-383-5p in breast cancer. Kaplan-Meier results showed that patients with low miR-383-5p expression had shorter overall survival rates. The results of the present study suggested that miR-383-5p was an independent prognostic biomarker for predicting overall survival of patients with breast cancer. The above results of the present study were consistent with those of previous studies demonstrating that miR-383-5p was downregulated in cancer tissues and acted as a potential marker of the therapeutic target [17, 25]. For instance, miR-383 is downregulated in lung adenocarcinoma tissues and acts as a potential prognostic biomarker and therapeutic target for lung adenocarcinoma [17]. In glioma, downregulation of miR-383 promotes cell invasion by targeting IGF1R and miR-383 may act as a useful diagnostic marker or therapeutic target [25].
Numerous studies have demonstrated that aberrant expression of miRNAs is associated with diverse biological processes, such as proliferation, migration, and invasion [26, 27, 28]. In breast cancer, aberrant miRNAs expression has been reported involved in numerous physiological processes and these miRNAs may be potential therapeutic targets for the treatment of breast cancer, such as miR-206 [29], miR-211-5p [30], and miR-590-3p [31]. According to the expression levels of miR-383-5p in breast cancer tissues and cells, we speculate that miR-383-5p may have a functional role as a tumor suppressor in breast cancer. To further explore whether miR-383-5p functions as a suppressor in breast cancer progression, the effects of miR-383-5p on breast cancer cell proliferation, migration, and invasion were assessed. Ectopic expression of miR-383-5p significantly inhibited cell proliferation, migration, and invasion in transfected MDA-MB-231, MDA-MB-453, and MCF-7 cell lines, while inhibition of miR-383-5p promoted these cellular behaviors. Therefore, these results suggested that miR-383-5p may play an inhibitory role in breast cancer. In colon cancer, miR-383 was downregulated in cancer tissues and inhibited cell proliferation, migration, and invasion of colon cancer cells through the regulation of target gene APRIL [32]. In hepatocellular carcinoma (HCC), miR-383 was downregulated in HCC tissues and cell lines and might inhibit HCC cell proliferation, invasion and glycolysis by targeting LDHA [33]. LDHA has been reported upregulated in multiple types of cancers and played crucial role in cancer cell proliferation, invasion, and metastasis of various tumors [34, 35]. In breast cancer, LDHA was reported upregulated and knockdown of LDHA could inhibit the migration and invasion of breast cancer cells [36, 37, 38]. In the present study, bioinformatics analysis prediction showed there are putative binding sites between miR-383-5p and LDHA 3’-UTR. And dual-luciferase reporter assay results showed that LDHA was the direct target of miR-383-5p. Based on previous studies and the present study results, we speculate that miR-383-5p may inhibit breast cancer cell proliferation, migration, and invasion by targeting LDHA. However, the detailed molecular mechanisms underlying the role of miR-383-5p in breast cancer still need to be investigated in future researches.
Taken together, the results of the present study indicated that miR-383-5p is downregulated in breast cancer tissues and cell lines and downregulation of miR-383-5p is associated with poor differentiation, positive lymph node metastasis, advanced TNM stages, and poor survival in patients with breast cancer. The results of the present study revealed that overexpression of miR-383-5p may suppress breast cancer proliferation, migration, and invasion by targeting LDHA, suggesting that miR-383-5p/LDHA axis may be a novel therapeutic target and strategy for the treatment of patients with breast cancer.
Footnotes
Conflict of interest
The authors declare no conflict of interest.
