Abstract
Intraoperative radiotherapy differs from the more commonly used external beam radiation with respect to fractionation, radiation energy, dose rate, and target volume, which may influence the irradiated cells in a complex manner. However, experimental studies of intraoperative radiotherapy are limited. Intrabeam is a frequently used intraoperative radiotherapy device; we evaluated its effects on the proliferation, apoptosis, migration, and invasion of MCF-7 human breast cancer cells. We performed colony formation assays for cells irradiated with single radiation doses of 0 to 16 Gy. Other cells were irradiated with single radiation doses of 0 to 6 Gy and then continued to be cultured. We measured cell-cycle distributions and apoptosis rates 24 hours later, using flow cytometry, and performed wound-healing assays, Transwell tests, and terminal deoxynucleotidyl transferase–mediated 2′-deoxyuridine 5′-triphosphate nick-end labeling staining 4 weeks later. Colony formation assays showed no positive colonies from cells irradiated with doses of ≥6 Gy. In flow cytometry, the experimental groups had higher late-apoptosis/necrosis rates (
Introduction
Breast cancer is a common malignancy in women. The American Cancer Society estimates that approximately 266 000 new breast cancer diagnoses and 40 000 deaths from breast cancer will occur in the United States in 2018. 1 Radiotherapy is widely used as local treatment for breast cancer, and radiotherapy modalities for breast cancer include external beam radiotherapy (EBRT), brachytherapy, and intraoperative radiotherapy (IORT). In contrast to EBRT and brachytherapy, IORT only uses a single large irradiation dose. Because the effective radiation target area of IORT is small and the dose attenuation is rapid, the effect on nonirradiated areas are minimized. 2,3
The species and other characteristics of radioactive rays vary in different IORT equipment. Intrabeam (Carl Zeiss, Oberkochen, Germany) is a small, mobile IORT device with adjustable source applicator direction. Its low-energy X-ray (50 kV) can generate high-ionization density radiation for local radiotherapy, and the spatial irradiation dose attenuates rapidly. 4 Intrabeam was first and most widely used in breast-conserving surgery. It is also used for gastrointestinal cancer, brain cancer, skin cancer, spinal metastasis, and head and neck cancer. 2,5 -9
Experimental researches on IORT remains limited. Belletti
Materials and Methods
Cell Culture
Human breast cancer MCF-7 cells (Breast Tumor Centre of SunYat-Sen Memorial Hospital, Guangzhou, China) were grown in 10% fetal bovine serum (FBS; Gibco, Thermo Fisher, Waltham, Massachusetts) and 90% Dulbecco’s Modified Eagle Medium (DMEM, Gibco), at 37°C in an atmosphere containing 5% CO2. The culture medium was free of insulin and was changed every 2 or 3 days. The cells were subcultured in trypsin EDTA solution (Gibco). Prior to the experiments, the cells were subcultured approximately 5 to 10 times in our laboratory.
Radiation Device and Radiation Parameter Setting
Flat application of the Intrabeam device (PRS 500; Carl Zeiss) could decrease radiation doses according to flat distance. To ensure all cells received uniform radiation doses, flat applicators of 60-mm diameter and T-25 cell culture flasks (Thermo Fisher Scientific) were used in this study. When applying radiation, the flat applicator was placed upside down and the cell culture flask was placed on the surface of applicator, making the bottom of the flask overlay with the surface of applicator to the extent possible. The plastic-shell thickness at the bottom of the flask was 1.2 mm (measured by a Vernier caliper); the density of the plastic was close to human tissue density and the MCF-7 cells were grown in monolayers in the culture medium. Thus, the target depth was set as 1.2 mm from the flat applicator surface. Before the experiment, the transformation output factor between bare source and target radiation dose (at the adherent cell site) was measured using a water tank with an ionization chamber. The calculated result was 1.31, and the corresponding target radiation dose rate was 14.8 Gy/h.
Colony Formation Assay
A total of 1000 cells were seeded separately in each flask (T-25) and radiation with 50 kV X-rays from the Intrabeam were administered approximately 4 hours after the cells were seeded. At this time, cells were attached to the bottom of flask but had not divided. For the experimental groups, the single radiation doses were 2 Gy, 4 Gy, 6 Gy, 8 Gy, 12 Gy, or 16 Gy. Cells were further incubated for 12 days, with medium changed only on day 7. The colonies were then fixed with methanol (15 minutes) and stained with 0.5% crystal violet (15 minutes). Colonies (>50 cells) were scored under the microscope. Each experiment was repeated 3 times. Surviving fraction (SF) was calculated as follows: SF = [average number of colonies per flask in the irradiated group/average number of colonies per flask in the control group × 100%]. Plating efficiency (PE) was calculated as follows: PE = [average number of colonies per flask in the control group/number of seeded cells per flask × 100%].
Cell Preparations for the Following Experiments
The medium was changed when the cells had reached approximately 70% confluency in the T-25 flask, and the cells were then subjected them to radiation approximately 12 hours later. For the experimental groups, the single radiation doses were 2 Gy, 4 Gy, or 6 Gy. Three independent repeat experiments were performed for each experimental group and the control group. Some cells were incubated for another 24 hours and then were harvested for flow cytometry. The other cells were cultured and subcultured for about 4 weeks and were then harvested for wound healing assays, Transwell assays, and terminal deoxynucleotidyl transferase mediated 2′-deoxyuridine 5′-triphosphate nick-end labeling (TUNEL) staining.
Flow Cytometry for Cell Apoptosis and Cell Cycle
For apoptosis detection, 1 × 106 cells in 100 µL stain buffer were added with 5 µL Annexin V-FITC (BD Pharmingen, San Diego, California) and 5 µL propidium iodide (PI; BD Pharmingen, San Diego, California). After incubation for 15 minutes away from light, the cells were evaluated by flow cytometry.
For cell-cycle detection, 1 × 105 cells in 100 µL phosphate-buffered saline (PBS) were fixed in 75% ethanol at −20°C overnight. The cells were centrifuged, washed with PBS twice, and then incubated in 100 µL PBS containing RNase A (1 mg/mL, Sigma, St. Louis, Missouri) for 30 minutes at room temperature. Finally, the samples were stained with 0.5 mL PI (50 µg/mL) for 30 minutes away from light at room temperature, followed by detection by flow cytometry.
Wound Healing Assay
We seeded MCF-7 cells in 12-well plates (5 × 104 cells per well) and incubated them for 24 hours. Scratches were introduced into the monolayers of confluent adherent cells in each well using a sterile pipette tip. Cells were washed twice with PBS and were then incubated in serum-free medium. Images were taken by microscopy (Olympus, Tokyo, Japan) 48 and 72 hours later. Wound areas were observed and calculated using the software tool ImageJ (version 1.51). Average scratch widths for each sample were analyzed. Scratch repair rate (%) = [(original scratch width − scratch width at 48 [or 72] hours)/original scratch width ×100%].
Transwell Assay
We plated MCF-7 cells in serum-free media on the inserts of a Corning BioCoat Matrigel Invasion Chamber (1 × 105 cells/well; Corning, Tewksbury, Massachusetts) in 24-well plates. Medium containing 20% FBS plus 80% DMEM was added to the lower chamber. After a 16-hour incubation, invasive cells were fixed with 4% paraformaldehyde at 4°C for 10 minutes and stained with 0.1% crystal violet for 20 minutes. Cells on the upper surface of the membrane were removed with a cotton swab and air-dried. We took images at ×100 magnification from each membrane and counted the invasive cells.
TUNEL Staining for Apoptosis Assessment
Following washing of MCF-7 cells in PBS, cells were fixed in 4% paraformaldehyde for 25 minutes and then treated with 0.2% Triton X-100 solution for 5 minutes. We assessed apoptosis using TUNEL (DeadEnd Fluorometric TUNEL System, Promega, Madison, Wisconsin) according to the manufacturer’s instructions. Samples were then evaluated under a fluorescence microscope (DMI 6000 B; Leica, Stuttgart, Germany) at ×100 magnification, and percentages of TUNEL-positive cells were calculated.
Statistical Analysis
Normally distributed data are shown as mean ± standard deviation. We used SPSS version 19.0 software (IBM Corp, Armonk, New York) to perform statistical analysis. Statistical comparisons were made using 1-way analysis of variance (ANOVA). Value of
Results
Single-Dose Irradiation With the Intrabeam Inhibited Cell Proliferation
Colony formation assays indicated that the MCF-7 cells in the control group were in logarithmic division, with colony-like distribution. In the experimental groups (especially those that had received doses of ≥6 Gy), cell proliferation became much slower, most of the cells swelled, fewer mitotic cells were seen, and cell communities were unevenly distributed. In the control samples, the average number of positive clones was 246 (PE = 24.6%). As the radiation dose increased, the numbers of positive clones greatly decreased; no positive clone was found in the groups with doses of ≥6 Gy. The cell survival curve fitted by positive clone numbers is shown in Figure 1.

Cell survival curve for MCF-7 cells irradiated by the Intrabeam device (50 kV X-ray source) with a flat applicator. Adherent cells in T-25 flasks were irradiated at a constant dose rate of 14.8 Gy/h. Results show the mean of 3 independent experiments.
Single-Dose Irradiation With the Intrabeam Induced Apoptosis/Necrosis and G1 Phase Arrest at 24 Hours After Treatment
MCF-7 apoptosis was detected by Annexin V-FITC/PI staining (Figure 2A and B). One-way ANOVA indicated that the late apoptosis/necrosis ratios were greater in the 2 Gy (

Flow cytometry Annexin V-FITC/PI apoptosis analyses and cell-cycle analyses of MCF-7 cells 24 hours after single-dose irradiation with the Intrabeam device. A, Intact viable cells (lower left), early apoptotic cells (lower right), and late apoptotic or necrotic cells (upper right). B and D, Data are expressed as mean ± SD; *
The flow cytometry results for cell-cycle distribution are shown in Figure 2C and D. Greater numbers of cells were stagnated in G1 phase in each experimental group compared with the control group (all
Single-Dose Irradiation With the Intrabeam Inhibited Migration and Invasion and Promoted Apoptosis 4 Weeks After Treatment
Wound healing assays indicated that the width of scratches were gradually reduced (Figure 3A). Multiple comparisons of the 72 hour-scratch repair rates (Figure 3C) indicated that the rates in the experimental groups were significantly lower than in the control group (all
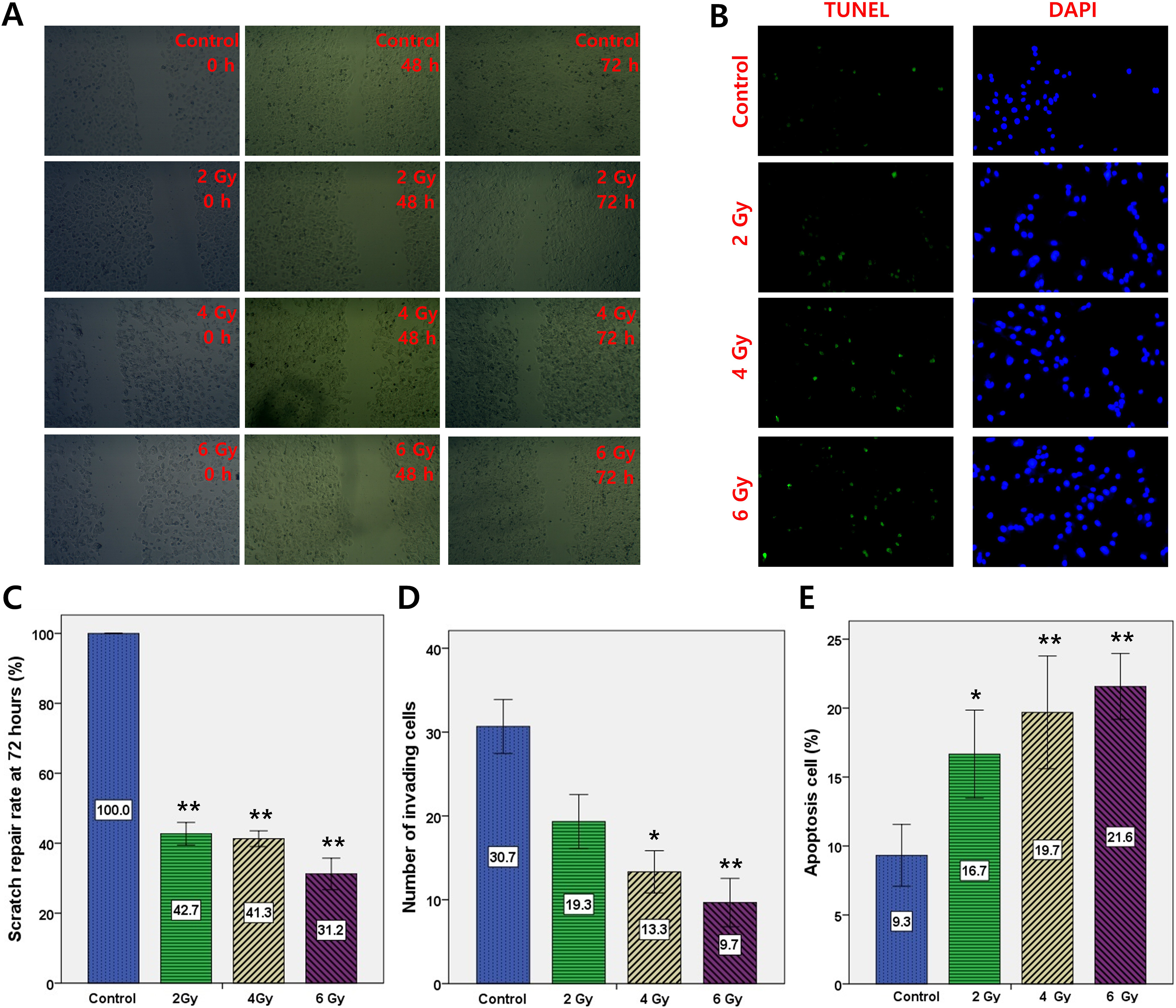
MCF-7 cell migration, invasion, and apoptosis assessments 4 weeks after single-dose irradiation by the Intrabeam device. Each experiment was repeated 3 times. A and C, Wound healing assays. Images of cells taken at 0, 48, and 72 hours after scratching in wound healing assays; quantitative analysis indicated that a single irradiation dose of 2, 4, or 6 Gy inhibited cell migration. D, Transwell assay indicated that a single irradiation dose of 4 or 6 Gy inhibited cell invasion. B and E, TUNEL apoptosis assay indicated that a single irradiation dose of 2, 4, or 6 Gy promoted cell apoptosis. Green: TUNEL-positive apoptotic cells; blue: nuclei stained with 4′,6-diamidino-2-phenylindole (DAPI); magnification ×100. C to E, Data are expressed as mean ± SD; *
As shown in Figure 3D, through multiple comparisons (1-way ANOVA), compared with the control group, the number of invading cells in the 2 Gy group exhibited a downward trend (
As shown in Figure 3B and E, multiple comparisons (1-way ANOVA) revealed that the apoptosis rates in the experimental groups were significantly higher than in the control group (all
Discussion
Radiotherapy is a local treatment for cancer and affects the radiation target area by killing or inhibiting tumor cells. Radioactive rays include α/β/γ/X-rays, electron beam, proton beam, and other proton beams, and ionizing radiation can generate biological effects including physical stage, chemical stage, and biological stage. 14 -16 In some studies, Intrabeam IORT delivers a single-radiation dose of 20 Gy to the tumor bed in breast-conserving surgery 2 or 16 Gy to the nipple-areola complex flap in nipple-sparing mastectomy. 3 Relative to the radiation energies of most EBRT devices (∼MeV), the Intrabeam device only produces 50 kV X-rays. Here, we explored the functional changes in breast cancer cells after single-dose irradiation with the Intrabeam.
MCF-7 cells are invasive ductal carcinoma and luminal subtype cells. 17 -21 Reportedly, 22 -24 the luminal subtype accounts for approximately 60% of the 4 subtypes of breast cancer, and invasive ductal carcinoma is the major pathological type of breast cancer. The effective radiation volume of IORT is much smaller than EBRT. Whether it is employed in breast-conserving surgery or nipple-sparing mastectomy, IORT is more appropriate for early- and middle-stage breast cancer during which tumor development is not fast. Compared to other commonly used invasive breast cancer cell strains (MDA-MB-231, MDA-MB-157, and T-47D), MCF-7 cells are weakly invasive. 17,25
In our preliminary study of Intrabeam IORT for nipple-sparing mastectomy,
3
the prescribed radiation dose was 16 Gy. In the colony formation assays of the present study, 0 to 16 Gy were used as the radiation doses. We found that although few MCF-7 cells survived in the groups dosed with ≥6 Gy, no positive clone was identified. The results of our study (using a flat Intrabeam applicator) were consistent with those of Liu
In this study, at 24 hours after Intrabeam irradiation, the percentages of normal cells in the 4/6 Gy groups were reduced, and the percentages of cells in late apoptosis/necrosis in the experimental groups (2/4/6 Gy) were increased, but the percentages of early apoptotic cells in the experimental groups were similar to that in the control group. This may be because some radiation-sensitive cells immediately initiated apoptosis after Intrabeam radiation, and the ionizing radiation thermal effect generated by Intrabeam radiation promoted early-stage necrosis. Percentages of cells arrested in G1 phase in the experimental groups were increased, but those in S phase and G2 phase were decreased. This is consistent with the slow proliferation rates in the experimental groups after radiotherapy. We also found that, at day 2 and 3 after radiotherapy, the number of new dead cells gradually began to increase. This may be because some cells repaired themselves partially after Intrabeam radiation, finally leading to limited mitosis.
Radiotherapy leads to apoptosis and necrosis in MCF-7 cells and can also induce cell senescence and other biological changes. 26 -29 In this study, we found that even at 4 weeks after single-dose irradiation with the Intrabeam, the proliferation rates of the cells in the experimental groups were slower than that in the control group. In the experimental groups, more granule-like substances could be observed in most of the cells, and more cells became swollen. In most studies, functional experiments with tumor cells were performed shortly after radiotherapy. 26,29 -34 However, after radiotherapy, the characteristics of sublethal tumor cells may change over time. In the study, at 4 weeks after single-dose irradiation with the Intrabeam, we found the following: (a) the migratory abilities of the experimental groups decreased in a dose-dependent manner; (b) invasiveness in the 4/6 Gy groups was lower than that in the control group; and (c) apoptosis rates of the experimental groups were all higher than that of the control group. Thus, we deduced that single-dose Intrabeam radiation might have a long-term effect on the inhibition of migration and invasion and promotion of apoptosis, with higher doses leading to more significant effects.
In conclusion, single-dose irradiation of 6 Gy using an Intrabeam device can effectively inhibit proliferation, migration, and invasion and promote apoptosis in MCF-7 cells with long-lasting effects.
Footnotes
Authors’ Note
Lingxiao Pan, Minghui Wan, and Wenbo Zheng, contributed equally. The current study was approved by the Ethics Committee of the First Affiliated Hospital of Guangzhou Medical University (registering order 201750).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Medical and Health Technology Project of Guangzhou Municipal Health Bureau (No. 20161A010078); the Health and Family Planning Technology Project of Guangzhou Municipal Health and Family Planning Bureau (No. 20181A011063).
