Abstract
BACKGROUND:
MicroRNAs have been proven to be key molecules in human malignancy. However, to our knowledge, there is no study reporting miR-383-5p expression level and the role it plays in bladder cancer (BC).
METHODS:
We identified miR-383-5p to be one of the tumor-suppressing genes through using data from The Cancer Genome Atlas (TCGA) and GEO database. We evaluate the expression and activity of miR-383-5p in both BC tissue and cell lines. The impacts of miR-383-5p on proliferative, migratory ability and apoptotic rate in BC cell were evaluated by utilizing CCK-8 kits, flow cytometry, and Transwell assays. qRT-PCR, western blot, and luciferase reporter assays have been adopted to investigate the underlying mechanisms. In vivo tumorigenicity testing was conducted to determine the impact of miR-383-5p on BC cellular proliferative capacity.
RESULTS:
Reduced miR-383-5p expression has been determined in BC tissue than in normal bladder tissue. Furthermore, BC cell proliferative, migratory ability was inhibited while apoptosis enhanced in vitro and in vivo by miR-383-5p up-regulation. In vitro and in vivo, silencing miR-383-5p considerably improved the growth and invasive capacity of cell, while decreased the apoptotic rates of BC cells.
CONCLUSION:
miR-383-5p plays its role as a tumor-suppressing gene by suppressing the PI3K/AKT signaling, hence preventing the development of BC.
Abbreviations
Introduction
Bladder cancer (BC) is amongst the leading prevalent urogenital malignancies globally [1]. It is the tenth most prevalent cancer among all malignancies in the world. Its recurrence rate and mortality are increasing over time [2, 3]. The development and advancement of bladder cancer are influenced by several factors, such as smoking, exposure to chemicals, and the aberrant expression of specific genes [4]. Despite the effectiveness of surgery and targeted drugs in treating bladder cancer, patients who experience recurrence or metastasis still face a poor prognosis, with recurrence rates as high as 20.6% [5]. So a better understanding of the relevant mechanisms of bladder cancer is necessary to develop effective and novel treatments for BC patients, especially in the field of targeted therapy [6].
MicroRNAs (miRNAs) are short RNA molecules consisting of about 20 nucleotides that can bind to specific sites on the 3’ untranslated regions (UTRs) of target mRNAs, causing either direct mRNA degradation or inhibition of mRNA translation [7, 8]. Numerous studies have shown that miRNA dysregulation is one of the primary drivers of tumor development and that many miRNAs function as biomarkers for tumor diagnosis [9]. MiRNAs exert both promoting and inhibitory effects on cancer development [10]. Previous research has shown that miR-383-5p is situated on chromosome 8p22 and function like a tumor-suppressing gene in a wide range of malignancies, such as oral squamous cell carcinoma [11], breast cancer [12], gastric cancer [13], ovarian cancer [14] and lung adenocarcinoma [15]. Nevertheless, the related mechanism and function of miR-383-5p in BC have never been investigated.
The progress made in whole-genome sequencing has validated PI3K/AKT signaling pathways are key driver genetic pathways of bladder carcinoma [16]. The PI3K/AKT pathway has been known among the most prevailing and widespread mechanisms associated with BC. According to The Cancer Genome Atlas (TCGA) records, 72% of bladder cancer cases are due to changes in the RTK/RAS/PI3K/AKT/mTOR signaling pathway [17]. The PI3K signaling pathway could be affected by many genes and miRNAs which are dysregulated in many tumors; thus, it is considered as an essential anticancer target [18]. Therefore, in our research, we tried to study the role of miR-383-5p in regulating cell proliferation and apoptosis through the PI3K/AKT pathway in BC.
In this study, miR-383-5p downregulation was a characteristic molecular change among BC patients, providing further evidence to elucidate the biological mechanism by which miR-383 functions as a tumor suppressor of BC progression.
Methods
Data collection
To acquire RNA sequencing profiles and clinical data pertaining to bladder cancer in March 2022, the Cancer Genome Atlas (TCGA) data portal (
Cell culture
The Shanghai Institute of Biological Sciences was the supplier for both normal bladder cells (SV-HUC-1) and human bladder cancer cells (UMUC3, T24, and J82) (Shanghai, China). RPMI-1640 and DMEM supplemented with 10% fetal bovine serum (Hyclone, Shanghai, China) and 1% penicillin/streptomycin (Gibco, Shanghai, China) were used to culture T24, J82 and UMUC3 cells. All the cells were kept at 37 degrees Celsius in an incubator with humidified environment HUC-1 cultures were grown in Ham F12 (Gibco, Shanghai, China) medium that was augmented with 7% fetal bovine serum (FBS), 4 mM L-glutamine, 1% penicillin/streptomycin All the cells were kept at 37 degrees Celsius in an incubator with humidified environment. For PI3K/Akt inhibition or activation BC cells were administered with 10
RNA extraction and qRT-PCR
By utilizing TRIzol Reagent, total RNA was extracted from BC cell lines (Takara, Dalian, China) A miRNA reverse transcription PCR kit (RiboBio, China) was utilized to transcribe obtained RNA to its complementary DNA (cDNA). In the SYBR Green mix system Applied Biosystems 7500 equipment was adopted to run the qRT-PCR. The miR-383-5p primer sequences utilized in this investigation are F: 5’-GGGAGACAGAAGGTGATTGTGGCT-3’; R: 5’ CAGTGCGTGTCGTGGAGT-3’; U6 F: 5’-CTCGCTTCGGCAGCACA-3’; R 5’-AACGCTTCACGAATTTGCGT-3’; U6 was utilized as the internal control.
Lentivirus packaging and cell transfection
Lentiviruses encoding miR-383-5p or anti-miR-383-5p were procured from GenePharma, China. Meanwhile empty vectors and anti-miR-control were utilized as negative controls. RiboBio (Guangzhou, China) supplied us with the mimics and inhibitors of miR-383-5p, along with their pairing control oligonucleotides, miR-NC or NC inhibitor. For the purpose of determining the effectiveness of the transfection, the proportion of fluorescein-positive cells was examined. For the aim of transfection, the BC cells were implanted into plates with a confluence level of between 80 and 90%. Following the procedures outlined by the manufacturer of Lipofectamine 2000 (Invitrogen), mimics and inhibitors were introduced into the cells by transfection.
Luciferase reporter assay
The pmirGLO vector from Promega (Michigan, United States) was utilized, and it included either wild-type PI3KR1 or mutant PI3KR1 certain binding location of miR-383-5p We transfected 293T cells with the luciferase reporter containing negative control or miR-383-5p mimics by using Lipofectamine 2000 reagent. We utilized the Lipofectamine 2000 reagent to transfect the luciferase reporter containing negative control or mimics of miR-383-5p into 293T cells. Afterwards we determined the luciferase activity by following the instruction of a Luciferase Reporter Assay System (Promega).
Cell proliferation assays
The density of BC cells which were seeded into 96-well plates was 2
Tumorigenicity assays in nude mice
The Chongqing Medical University Experimental Animal Center was the supplier of the total of 30 female athymic BALB/c nude mice which are four weeks old used in this experiment (Chongqing, China). The nude mice were divided into six groups according to a random selection. Briefly, in the right flank of nude mice, 5
Transwell invasion assay
Transwell chambers were purchased from BD Biosciences, USA. In the top chamber, BC cells at the concentration of 1.0
Flow cytometry analysis
An Annexin V-FITC and PI Detection Kit (BD Biosciences, NJ, USA) was applied in order to conduct an analysis of apoptosis in the BC cells. BC cells (1
The differential expression of miR-383-5p in BC patients and BC cell lines. A. Down-regulated microRNAs in BC patients from the GSE39093 cohort. B. Down-regulated microRNAs in BC patients from the GSE68594 cohort. C. Profile of down-regulated microRNA expression in TCGA cohort BC patients D. The number of genes upregulated or downregulated in BC patients from GSE39093, GSE68594 and TCGA cohorts. E. Scatter plot from the GSE39093 cohort demonstrating miR-383-5p expression in BC tumor tissue is down-regulated than in matching heathy tissue. F. Scatter plot from the GSE68594 cohort revealing miR-383-5p expression is reduced in BC tumor tissue in comparison to normal tissue. G. BC tumor tissues express lower miR-383-5p according to a scatter plot from the TCGA cohort. H. miR-383-5p in HUC-1, UMUC3, T24 and J82 cell lines was examined by qPCR. The error bars in the graphs represent SD. Each experiment was repeated three times. 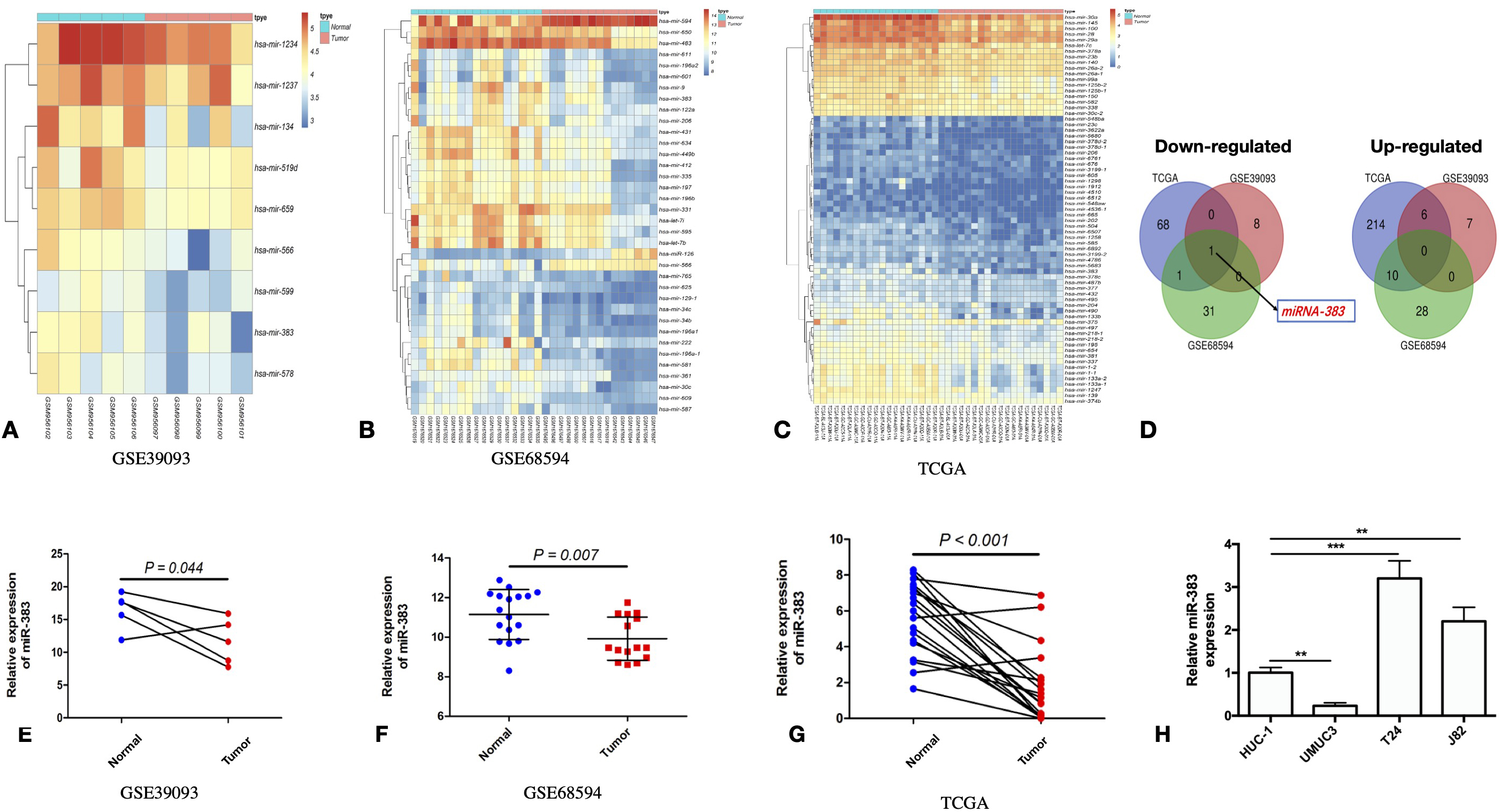
Increasing miR-383-5p expression promoted the proliferative and invasive capacity of UMUC while inhibited its apoptosis in vitro. Three groups of UMUC3 cells were involved, including: NC, NC-mimic, miR-383-mimic. A. Growth curves representing cell proliferation ability of UMUC3. B. Apoptotic rate of BC cells after treatment with different mimics were detected by FACS analysis. C. Quantification of apoptotic cells detected by FACS analysis. D. Cleaved caspase-3 expression involved in cell apoptosis was detected by WB. E. Cell invasive ability was determined by Transwell assay. magnification:200X. F. Quantification of invading cells detected by Transwell assays. The error bars in the graphs represent SD. Each experiment was repeated thrice. 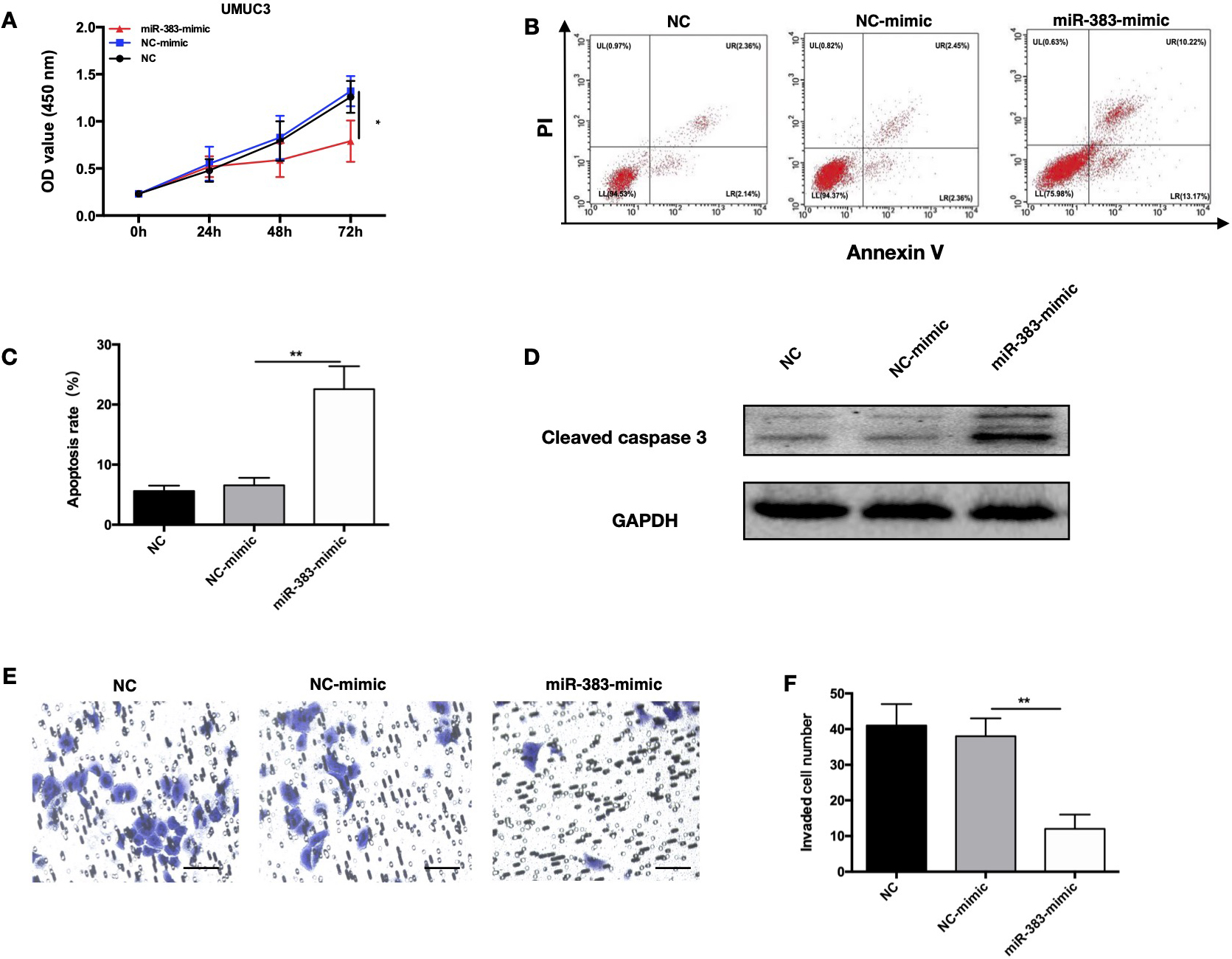
Knockdown of miR-383-5p inhibited proliferative and invasive capacity of T24 cell while promoted its apoptosis in vitro. Three groups of T24 cells were involved, including: NC, NC-inhibitor, miR-383- inhibitor. A. Growth curves representing cell proliferation ability of T24 cells was determined via CCK-8 assays. B and C. Apoptotic rate of BC cell and its quantification detected by FACS analysis. D. Cleaved caspase-3 expression involved in cell apoptosis was detected by WB. E. Cell invasive ability was determined by Transwell assay. Magnification:200X. F. Quantification of invading cells detected by Transwell assays. The error bars in the graphs represent SD. Each experiment was repeated thrice. 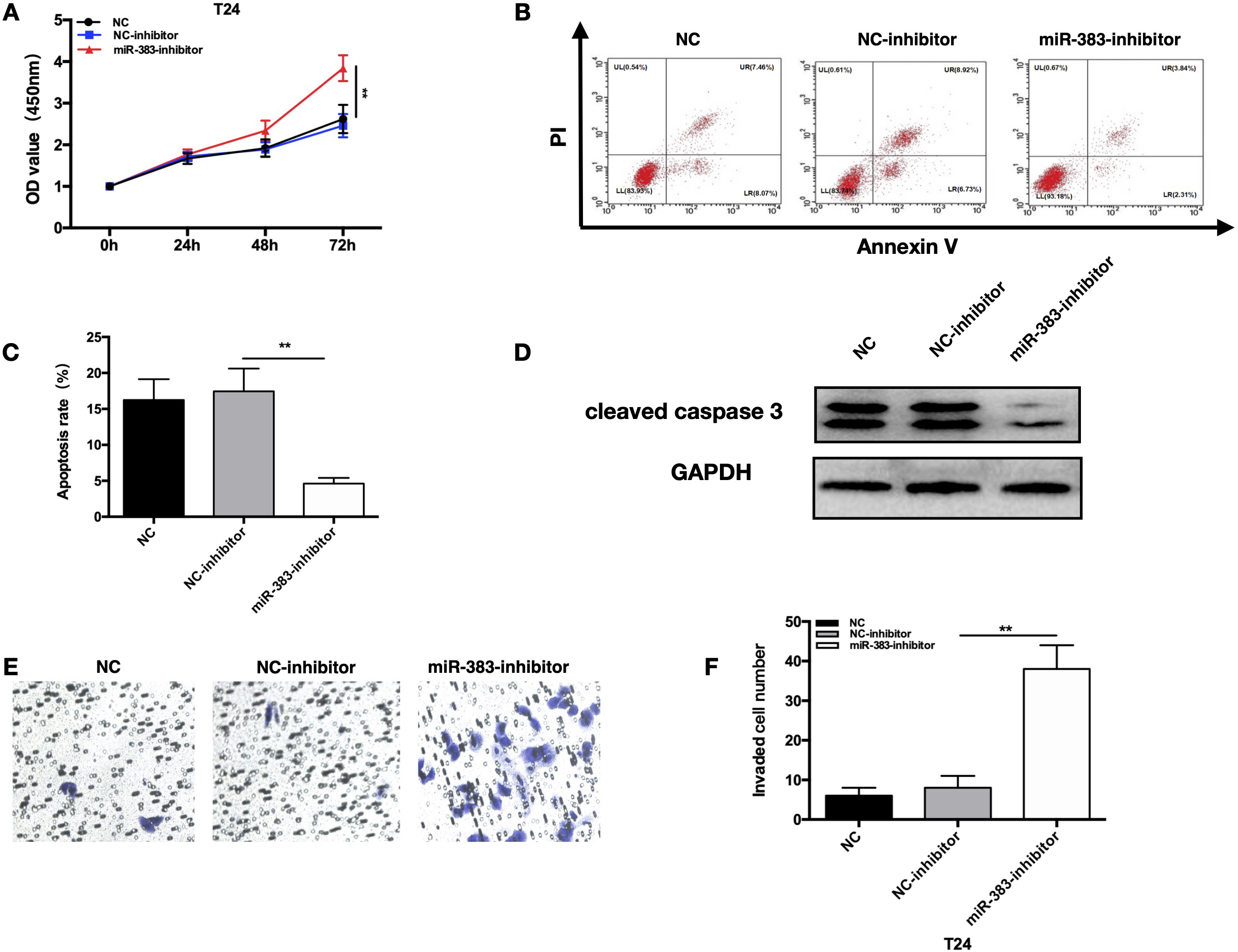
In vivo research demonstrated that miR-383-5p mediates BC. A. Tumors collected from nude mice 4 weeks after the inoculation of stably transduced vector or Lv-miR-383-5p UMUC3 cells. Tumor weight (B) and growth curve (C) of mouse xenografts subcutaneously injected with UMUC3 cells with stable miR-383-5p overexpression. D. Ki-67 was stained by immunohistochemistry in xenograft tissues formed by UMUC3 cells. magnification:200X. E. Quantification of proliferative cells detected by Ki-67 staining. F. Tumors collected from nude mice 4 weeks after inoculation of stably transduced shscr or anti-miR-383-5p T24 cells. Tumor weight (G) and growth curve (H) of mouse xenografts subcutaneously injected with T24 cells with stable miR-383-5p knockdown. I. Ki-67 was stained by immunohistochemistry in xenograft tissues formed by T24 cells. Magnification:200X. J. Quantification of proliferative cells detected by Ki-67 staining. The error bars in the graphs represent SD. Each experiment was repeated thrice. 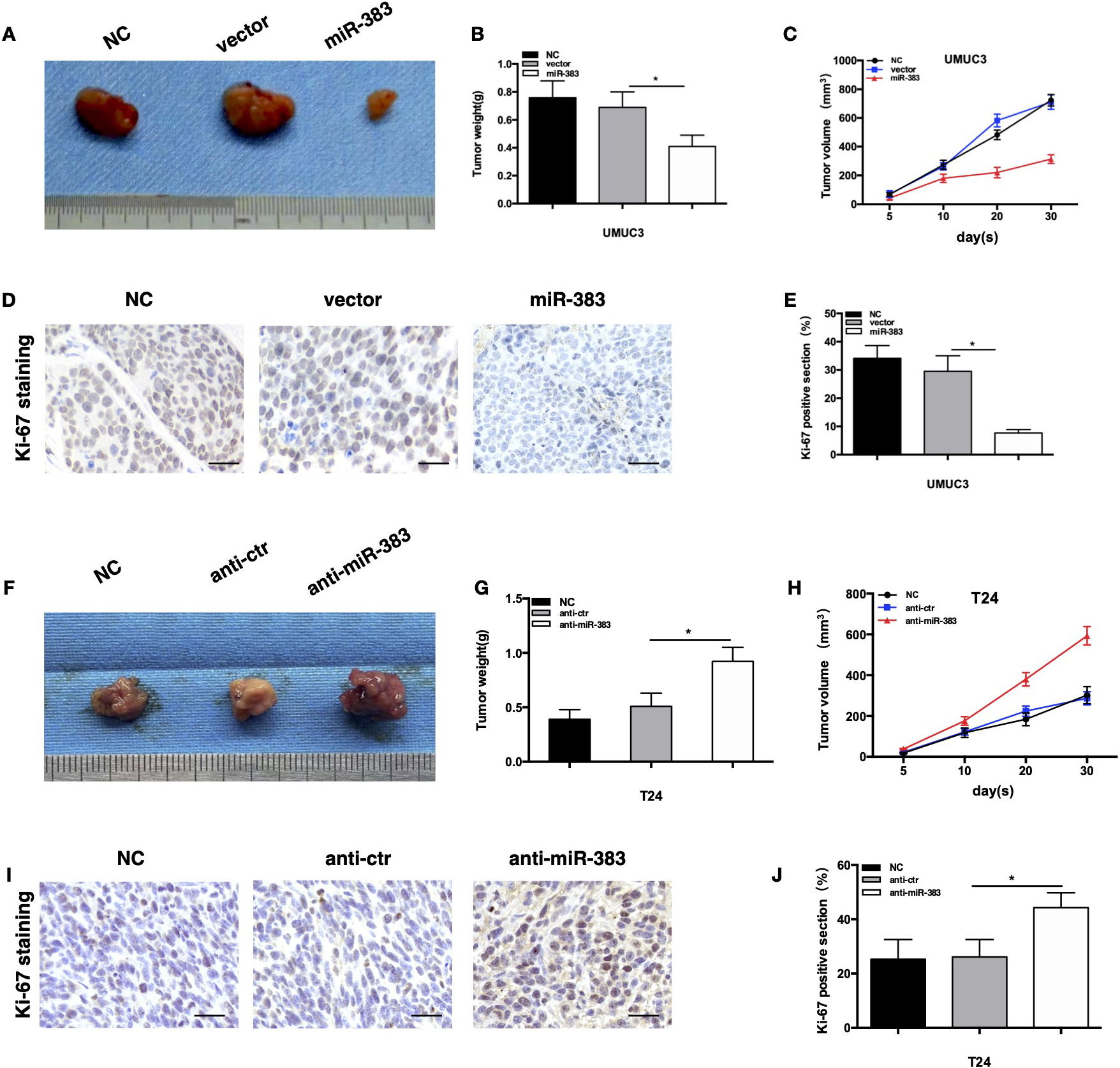
miR-383-5p interact with PI3KR1 protein directly in BC cells. A. Diagram showing the anticipated binding site for PI3KR1 on miR-383-5p. B. Following treatment with different mimics, PI3KR1-WT or PI3KR1-MUT luciferase activity was observed in 293T cells. C. PI3KR1 protein level determined by WB in UMUC3 cells. D. Expression of PI3KR1 was detected by WB in T24 cells. The error bars in the graphs represent SD. Each experiment was repeated thrice. 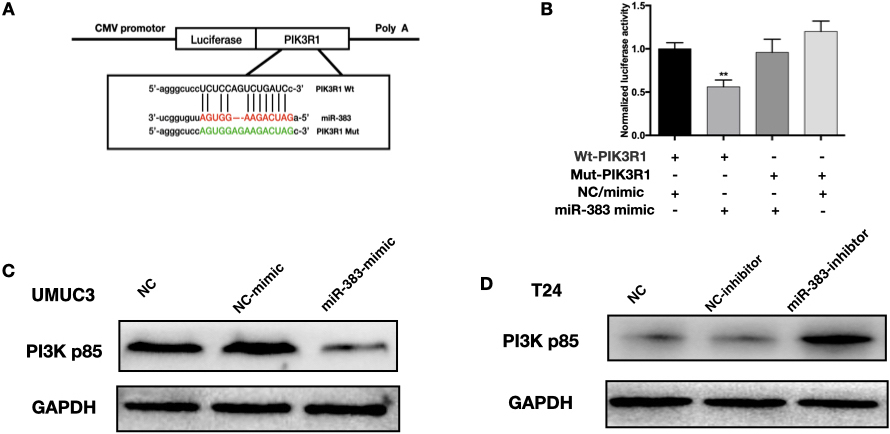
miR-383-5p serves as a cancer negative regulator inhibiting BC by suppressing the PI3K/AKT signaling pathway. A. Following treatment with different mimics or pathway activators, CCK-8 tests determined the proliferative capacity of UMUC3 cells. B and C. Following treatment with different mimics or pathway activators, FACS analysis revealed apoptosis rates of UMUC3 cells. D. Protein level of targets involved in the related pathway in UMUC3 cells identified by WB. E. Following treatment with different inhibitor or pathway inhibitor, CCK-8 tests showed proliferation of T24 cells. F and G. Following treatment with different inhibitor or pathway inhibitor, FACS analysis revealed apoptosis rates of T24 cells. H. Protein level of targets involved in the related pathway in T24 cells identified by WB. The error bars in the graphs represent SD. Each experiment was repeated thrice. 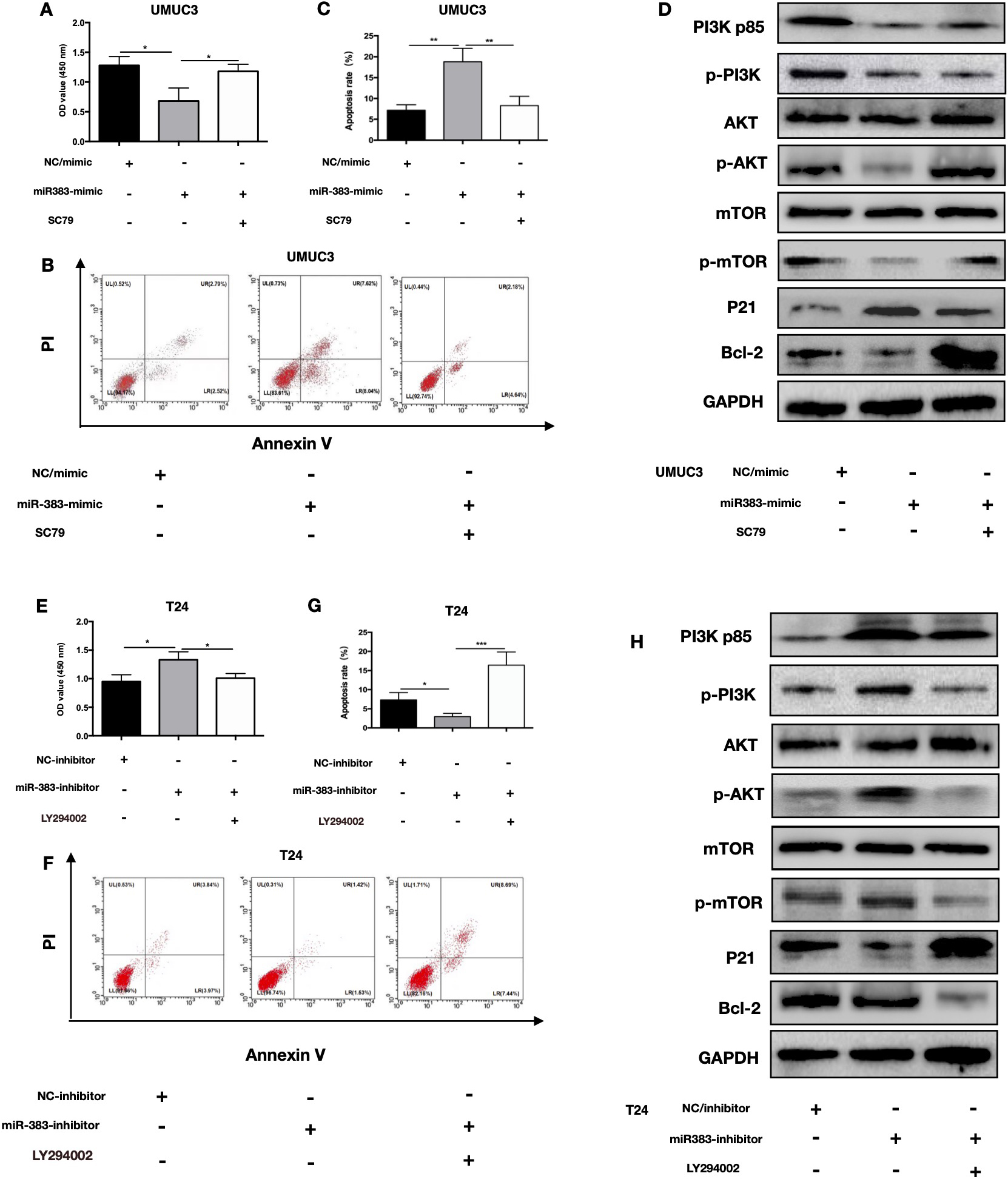
Protein samples were isolated using a 1x RIPA buffer that was purchased from Beyotime (Shanghai, China). The proteins were then denatured by heating at a temperature of 100
Statistical procedures
The data are presented as the mean
Results
miR-383-5p is downregulated in BC tissues
To gain a deeper understanding of the role of miR-383-5p in bladder cancer, we analyzed data from the TCGA and GEO cohorts. To conduct an analysis in the different miR-383-5p expression between healthy and malignant bladder tissue, our study investigated RNA-Seq data from the TCGA-based BLCA cohort and the GEO datasets GSE39093 and GSE68594. In the GSE39093 cohort, 15 microRNAs have been found that expressed differently in cancerous bladder tissue compared to healthy bladder tissue, 9 of which have found to be down-regulated (Fig. 1A). We found 59 miRNAs expressed differentially in cancerous bladder tissue versus healthy bladder tissue in the GSE68594 cohort, 33 of which have found to be down-regulated (Fig. 1B). 282 genes were found differently expressed between cancerous bladder tissue versus healthy bladder tissue in the TCGA cohort, 70 of which have found to be down-regulated (Fig. 1C). MiR-383-5p was the only miRNA found to be downregulated in all three cohorts (Fig. 1D). MiR-383-5p expression was considerably reduced in all three cohorts in malignant bladder tissue compared to normal bladder tissue (Fig. 1E–G). Different miR-383-5p expressions has also been detected in the BC cell lines when normal epithelial cell SV-HUC-1 was used as control (Fig. 1H).
Overexpression of miR-383-5p inhibits proliferative and invasive property of UMUC3 cell while inducing cellular apoptosis in vitro
To study the impact of miR-383-5p up-regulation in the progression of BC, we chose UMUC3 cell line which had the lowest miR-383-5p level among other cells and transfected with miRNA mimics. Following treatment with a miR-383-5p-mimic in UMUC3 cells, there was a discernible increase in miR-383-5p (Fig. S1A). The result of CCK-8 assay verified that proliferation of UMUC3 cells at 72 hours after treatment with mimic was dramatically inhibited (Fig. 2A). Significantly more apoptotic cells were detected in UMUC3 cells after treatment with a miR-383-5p-mimic (Fig. 2B and C). WB revealed a significantly higher levels of cleaved caspase-3 in the cells in UMUC3 cells after treatment with a miR-383-5p-mimic (Fig. 2D). In transwell test, the invasive capability of UMUC3 cells was found effectively decreased in UMUC3 cells after treatment with a miR-383-5p-mimic (Fig. 2E and F). All these findings indicate that miR-383-5p overexpression in BC cells inhibits their proliferative and metastatic ability and promoted their apoptosis.
In vitro, miR-383-5p knockdown enhances T24 cell growth and invasion, while suppressing apoptosis
To learn more about the impact of miR-383-5p knockdown during the advancement of BC, we used T24 cell line with substantially greater miR-383-5p expression. After transfection of T24 cells with a miRNA inhibitor, miR-383-5p expression decreased dramatically (Fig. S1B). The result of CCK-8 assay verified that proliferation of T24 cells transfected with miRNA inhibitor was significantly promoted at 72h after transfection (Fig. 3A). Apoptotic cells decreased significantly in T24 cells transfected with inhibitor (Fig. 3B and C). Significantly lower cleavedcaspase-3 was determined by WB in BC cells after treatment with miR-383-5p-inhibitor (Fig. 3D). The transwell test revealed that the invasive potential of T24 cells was much higher in T24 cells after treatment with miR-383-5p-inhibitor (Fig. 3E and F). These experiments verified that knocking down miR-383-5p in BC cells increased their proliferative and metastatic potential and reduced their apoptosis.
miR-383-5p mediates BC cell proliferation in vivo
To study further if miR-383-5p may influence BC proliferation in vivo, a tumor development experiment was done. After the transfection of UMUC3 cells with lentivirus stably upregulating miR-383-5p, qPCR determined that miR-383-5p expression was noticeably elevated (Fig. S1C and D). When T24 cells were treated with lentivirus with stable miR-383-5p knockdown, its expression declined dramatically (Fig. S1E and F). Right flanks of nude mice were implanted with UMUC3 cells persistently overexpressing miR-383-5p while the other half of mice were implanted with T24 cells with consistent miR-383-5p suppression. The size, weight, and volume of the neoplasm at the given time point during the in vivo experiment were then documented. The miR-383-5p group have much less tumor growth, weight, and volume than the NC or vector groups (Fig. 4A–C). The Ki-67 analysis showed that the miR-383-5p group exhibited fewer Ki-67-positive areas than the NC or vector groups (Fig. 4D and E). Size, weight, and volume of tumors were bigger in the anti-miR-383-5p group (Fig. 4F–H). The Ki-67 assay showed that there were more Ki-67-positive cells in the anti-miR-383-5p group (Fig. 4I and J). These findings confirmed that miR-383-5p exert a suppressive impact on in vivo cell growth.
miR-383-5p interact with PI3KR1 protein directly in BC cells
To make the prediction of the miR-383-5p targets and their possible binding sites, we adopted starBase v2.0 database. Our team found that PI3KR1 (also known as PI3K-p85) was among the potential targets of miR-383-5p (Fig. 5A). Significant inhibition of luciferase activity was seen in wild-type PI3KR1 after treatment with a miR-383-5p mimic (Fig. 5B), while no difference in the luciferase activity of mut-PI3KR1 was witnessed. The miR-383-5p-mimic group showed dramatic reductions in PI3KR1 expression, whereas the miR-383-5p inhibitor group showed significant increases (Fig. 5C and D). Our study demonstrated that miR-383-5p regulates PI3KR1 expression directly.
miR-383-5p controls BC cell fate through PI3K/AKT pathway
We conducted rescue experiments in order to determine whether miR-383-5p achieve its function via PI3KR1 in BC cells. Overexpression of miR-383-5p significantly inhibits UMUC3 cell proliferation and increased cell death, while the anti-proliferative effect and pro-apoptotic effect were reversed by administration of the AKT/mTOR pathway agonist SC79 (Fig. 6A–C). Knockdown of miR-383-5p dramatically promoted the proliferation of T24 cell and inhibited cellular apoptosis, and the proliferative effect and antiapoptotic effect were reversed by administration of the AKT/mTOR pathway inhibitor LY294002 (Fig. 6E and F). Targeted proteins involved in the AKT/mTOR signaling pathway were identified using Western blot in BC cells with various transfections. The variation in gene expression within the AKT/mTOR pathway caused by overexpression and knockdown of miR-383-5p was reversed by treatment with an AKT/mTOR pathway agonist or inhibitor (Fig. 6D and H). These results showed that AKT/mTOR pathway agonists or inhibitors are able to reverse miR-383-5p’s suppressive effect on BC cellular growth or promote the apoptosis of BC cells.
Discussion
Biomarkers are crucial tools for the early diagnosis of BC and could be therapeutic targets to improve the prognosis and survival of BC patients. In our research, we aimed to investigate how miR-383-5p works in regulating the PI3KR1/AKT pathway by acting as a tumor suppressor. MiRNAs can play a dual role in cancer by either promoting or suppressing tumor growth. They can influence cancer cells by regulating various cellular processes such as the cell cycle, proliferation, apoptosis, and also modulate the tumor microenvironment, which in turn can affect cancer progression and metastasis [19, 20]. MiR-383-5p is a member of a group of miRNAs that are commonly downregulated in cancer and have been shown to inhibit tumor growth and invasion [21]. Previous study showed that miR-383-5p expression is frequently decreased in various types of cancers [22]. A previous study demonstrated that miR-383-5p inhibited multiple mechanisms which promote tumor progression to suppress tumor cell growth and cancer malignant development [23]. MiR-383-5p expression loss is frequently observed in prostate cancer [21]. The abundant expression of CREPT in colorectal cancer progression is facilitated by the decreased levels of miR-383-5p [24]. MiR-383-5p has been shown to induce apoptosis and reduce cell viability in lung cancer cells [25]. Decreased miR-383-5p expression was shown in BC tissues in our research. Moreover, the proliferation of BC was inhibited, while apoptosis was promoted in vitro and in vivo by overexpression of targeted miRNA. Knockdown of miRNA expression promoted BC cellular growth and inhibited cellular apoptosis.
The PI3K signaling has been widely verified as a common pathway regulating diverse biological processes in numerous kinds of cancers, including bladder cancer, especially in regards to cell growth, proliferation, and apoptosis [26, 27]. In bladder cancer tissue, PI3K/AKT signaling is commonly overactivated. Patients with activated PI3K/AKT/mTOR signaling in BC tissue have an unfavorable prognosis [28]. The effect of the PI3K signaling pathway could be affected by many genes, miRNAs, and compounds that could modulate PI3K, AKT, mTORC1 and mTORC2 [17]. In this study, enhanced miR-383-5p levels suppressed PI3KR1 expression, thus suppressing Akt phosphorylation. miR-383-5p knockdown enhanced PI3KR1 expression and promoted Akt phosphorylation. miR-383-5p mediated the phosphorylation of AKT, suggesting that PI3K/AKT signaling was involved in miR-383-5p function in bladder cancer. AKT agonist increased bladder cancer proliferation and inhibited apoptosis in cells overexpressing miR-383-5p. However, how miR-383-5p functions have never been studied in bladder cancer, as the expression of certain microRNA is tissue-specific. Our data could be deemed as a supplement to our knowledge of miR-383-5p.
In conclusion, we study reveals that miR-383-5p expression level is decreased in BC tissues. Overexpression of miR-383-5p decreased survival and growth of BC cells not only in vitro but also in vivo and accelerated cellular apoptosis. Knockdown of miR-383-5p enhanced cell viability and its proliferative property and inhibited cellular apoptosis. The tumor-suppressing effect of miR-383-5p might be realized by suppressing PI3K/AKT signaling in bladder cancer. Research on targeted drugs that promote miR-383-5p may provide new options for clinical BC treatment.
Ethics approval and consent to participate
The Animal Management Rules of the Ministry of Health of the People’s Republic of China were followed in all animal care and experimental methods. Ethical assessments of animal experiments were authorized by the ethics committee of the First Affiliated Hospital of Chongqing Medical University.
Consent for publication
The research was done adhered to the Declaration of Helsinki-approved procedure and the requirements of the First Affiliated Hospital of Chongqing Medical University’s Ethics Review Committee.
Availability of data and materials
For reasonable requests, the corresponding author will make available the datasets used and/or analyzed during the current study.
Competing interests
All of the authors declare that there are no competing interests.
Authors’contributions
Conception: Fangchao Yuan, Yu Liu, Ke Ren.
Interpretation or analysis of data: Yu Liu; Haitao Yu; Zeng Bin.
Preparation of the manuscript: Haitao Yu.
Revision for important intellectual content: Xin Gou, Ke Ren.
Supervision: Fangchao Yuan; Ke Ren.
All authors have read and approved the final version of this manuscript.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-220379.
sj-docx-1-cbm-10.3233_CBM-220379.docx - Supplemental material
Supplemental material, sj-docx-1-cbm-10.3233_CBM-220379.docx
Footnotes
Acknowledgments
None.
