Abstract
OBJECTIVE:
This study aimed to explore the correlation of circular RNA ABCB10 (circ-ABCB10) expression with clinicopathological features and survival, as well as its impact on regulating cell proliferation and apoptosis in epithelial ovarian cancer (EOC).
METHODS:
A total of 103 EOC patients were consecutively recruited, then their tumor tissues were obtained for circ-ABCB10 detection using qRT-PCR. Additionally, 53 EOC adjacent tissues were collected as control. Patients’ clinicopathological and survival data were recorded. In vitro, circ-ABCB10 expression was detected in OVCAR3, UWB1.289, SKOV3, CAOV3 and IOSE80 cell lines by RT-qPCR, and the effect of circ-ABCB10 on cell proliferation and apoptosis was detected through circ-ABCB10 overexpression and silencing by plasmids transfection into SKOV3 cells.
RESULTS:
Circ-ABCB10 was upregulated in tumor tissues compared with adjacent tissues, and presented with good value in distinguishing tumor tissues from adjacent tissues (AUC
CONCLUSIONS:
Circ-ABCB10 correlates with advanced clinicopathological features and unfavorable survival, and promotes proliferation, reduces apoptosis and negatively regulated miR-1271, miR-1252 and miR-203 in EOC.
Introduction
Epithelial ovarian cancer (EOC), accounting for 90% of all ovarian cancer cases, is the second most common gynecological cancers and the leading cause of gynecological cancer-related deaths [1]. EOC patients are commonly treated by standardized therapy including surgical resection and chemotherapy, and are always assisted by targeted therapy, endocrine therapy and radiotherapy [2]. Although therapeutic and diagnostic approaches for EOC have progressed and improved the clinical outcomes for EOC patients during the past decades, it remains a troublesome clinical challenge that EOC is commonly diagnosed at late-stage and is under high risk of recurrence and metastasis, resulting in an unfavorable prognosis in EOC patients [3]. In order to conquer these problems, accurate and reliable biomarkers are critical for prediction of disease risk and prognosis in EOC patients.
Of note, increasing evidences have revealed the close association of non-coding RNAs with the pathogenesis of human diseases [4]. one sort of the abundant and evolutionary conserved non-coding RNAs, circular RNAs (CircRNAs) are characterized by covalent loops formed by back-splicing without 3’ and 5’ ends [5]. They have been discovered to play key roles in various human diseases including cancers, cardiovascular diseases and autoimmune diseases through acting as microRNA (miRNA) sponges, protein sponges, protein scaffolds etc., and particularly in cancer, an increasing number of dysregulated circRNAs and their putative functions have been disclosed [5, 6, 7, 8, 9, 10]. CircRNA ABCB10 (circ-ABCB10) is a novel circRNA initially found to promote breast cancer proliferation and progression by sponging miR-1271, a miRNA that has been reported to participate in the development of various cancers including EOC, breast cancer, pancreatic cancer and so on [10, 11, 12, 13, 14]. And it is also observed to promote non-small-cell lung cancer cell proliferation and migration via regulating miR-1252/Forkhead box 2 (FOXR2) axis, and enhances osteosarcoma progression through sponging miR-203 [15, 16]. These studies indicate the oncogenic role of circ-ABCB10 in cancers, while the expression pattern and regulatory role of circ-ABCB10 in EOC is still unknown. Therefore, this study aimed to explore the correlation of circ-ABCB10 expression with clinicopathological features and survival, as well as its function on regulating cell proliferation and apoptosis in EOC.
Methods
Patients
Between Jan 2014 and Dec 2018, 103 EOC patients who underwent resection at Lishui People’s Hospital were reviewed in this retrospective study. The inclusion criteria were: (1) confirmed EOC by histopathological findings; (2) received resection, and the fresh-frozen tumor tissue was available; (3) with complete clinical and follow-up data; (4) no neoadjuvant therapy. The patients were excluded if they had relapsed or secondary EOC. Informed consents (paper version or electronic edition) were obtained from all patients or their guardians after the study was approval by the Ethics Committee of the Hospital.
Clinical data collection
Patients’ clinical data, which included age, histological subtype, pathological differentiation, tumor size, the volume of ascites, peritoneal cytology, FIGO stage and CA125 level were collected from electronic medical records. All patients were followed up to 2018/12/31, and the survival data were obtained from follow-up records, which was used to calculate overall survival (OS). The OS was defined as the time interval from resection to death.
Detection of circ-ABCB10 relative expression in EOC tumor and adjacent tissues
Circ-ABCB10 relative expression in fresh-frozen tumor tissues from 103 EOC patients was detected by Quantitative Real-time Reverse Transcription (qRT-PCR), meanwhile, 52 EOC adjacent tissues were collected as controls, and the circ-ABCB10 relative expression in the adjacent tissues was detected using qRT-PCR as well. All tumor tissues and adjacent tissues were collected from storage compartment after approval by the Pathology Department of the Hospital.
Cell source and culture
Human EOC cell lines OVCAR3, UWB1.289, SKOV3 and CAOV3 were purchased from American Type Culture Collection (ATCC) (Manassas, USA), while human normal ovarian epithelial cell line IOSE80 was purchased from BioVector NTCC (Beijing, China). OVCAR3, UWB1.289, SKOV3 and IOSE80 cell lines were cultured in 90% RPMI-1640 medium (Gibco, USA) and 10% fetal bovine serum (FBS) (Gibco, USA) under 95% air and 5% CO
Detection of circ-ABCB10 relative expression in human EOC cell lines and normal ovarian epithelial cell line
Circ-ABCB10 relative expression was detected in OVCAR3, UWB1.289, SKOV3, CAOV3 and IOSE80 cell lines by RT-qPCR.
Cell transfection
The control overexpression, circ-ABCB10 overexpression, control shRNA and circ-ABCB10 shRNA plasmids were constructed by Shanghai GenePharma Company (Shanghai, China) using pEX-2 and pGPU6. Then these plasmids were transfected into SKOV3 cells and UWB1.289 cells using HilyMax (Dojindo, Japan) and were divided into NC (
Cell proliferation and apoptosis
SKOV3 cell and UWB1.289 cell proliferation was detected by OD value at 0 h, 24 h, 48 h and 72 h post transfection using Cell Counting Kit-8 (Sigma, USA) according to the instructions of manufacturer. SKOV3 cell and UWB1.289 cell apoptosis rate was detected at 24 h post transfection using Annexin V-FITC Apoptosis Detection Kit (Sigma, USA) according to the instructions of manufacturer. Besides, in order to further confirm the apoptosis, cleaved caspase 3 (C-Caspase 3) and B-cell lymphoma-2 (Bcl-2) protein expressions were detected at 24 h post transfection using Western blot as well.
qRT-PCR process
The qRT-PCR was performed as follows: first, TRIzol reagent (Invitrogen, USA) was used to extract total RNA according to the manufacturer’s instructions; then the linear RNA was digested using the RNase R enzyme (Epicentre, USA); subsequently, cDNA was synthesized from the RNA using the reverse transcription kit (Qiagen, German); finally, ABI 7900HT Real-time PCR system (Applied Biosystems, USA) was used for PCR reaction by use of QuantiNova SYBR Green PCR Kit (Qiagen, German). GAPDH or U6 was used as normalized internal reference and the relative expression of circ-ABCB10 or miRNAs was calculated using the 2
Western blot process
Proteins were extracted from tissue using RIPA Lysis and Extraction Buffer (Thermo, USA) and were then quantified using Pierce
Antibodies applied in Western Blot
Antibodies applied in Western Blot
Circ-ABCB10 expression in distinguishing tumor tissue and adjacent tissue in EOC patients. Circ-ABCB10 expression was higher in tumor tissue compared with adjacent tissue (A), and it presented good value in distinguishing EOC tumor tissue from adjacent tissue (B). Comparison of circ-ABCB10 expression between tumor tissue and adjacent tissue was performed using Wilcoxon rank-sum test. ROC curve and AUC were used to assess the value of circ-ABCB10 relative expression in distinguishing tumor tissue from adjacent tissue. 
All data analyses were performed using SPSS 22.0 software (SPSS Inc., USA), and all figures were made with the use of GraphPad Prism 7.00 software (GraphPad Software, USA). Data were presented as mean value
Results
Patients’ baseline characteristics
The mean age of 103 EOC patients was 56.6
Baseline characteristics of EOC patients
Baseline characteristics of EOC patients
Data were presented as mean value
Circ-ABCB10 expression in tumor tissue was 2.017 (1.028–3.377), which was higher compared to adjacent tissue (0.883 (0.249–1.575)) (
Correlation of circ-ABCB10 expression with patients’ characteristics
EOC patients were divided into circ-ABCB10 high expression group (
Correlation of circ-ABCB10 expression with clinicopathological characteristics
Correlation of circ-ABCB10 expression with clinicopathological characteristics
Data were presented as count (percentage). Comparisons were determined by Chi-square test.
The accumulating OS in circ-ABCB10 high expression group (mean: 42.6 months, 95% CI: 36.2–49.0 months) was reduced compared with circ-ABCB10 low expression group (mean: 52.5 months, 95% CI: 48.8–56.1 months) (
Comparison of OS between circ-ABCB10 high expression and low expression group in EOC patients. OS was lower in circ-ABCB10 high expression group compared with circ-ABCB10 low expression group. The difference of OS between two groups was determined by Kaplan-Meier method and Log-rank test. 
Univariate Cox’s regression model analysis displayed that high circ-ABCB10 expression (HR
Cox’s regression model analysis of factors affecting OS
Cox’s regression model analysis of factors affecting OS
Factors affecting OS were determined by univariate and multivariate Cox’s proportional hazards regression model analyses.
In vitro experiments were further carried out to investigate the function of circ-ABCB10 in EOC. It was observed that circ-ABCB10 expression was higher in OVCAR3 (
Circ-ABCB10 relative expression in human EOC cell lines and normal ovarian epithelial cells. Compared with normal ovarian epithelial cells IOSE80, the expression of circ-ABCB10 was elevated in OVCAR3, SKOV3 and CAOV3 cell lines but not in UWB1.289 cells. Comparison of circ-ABCB10 expression between two cell lines were performed by 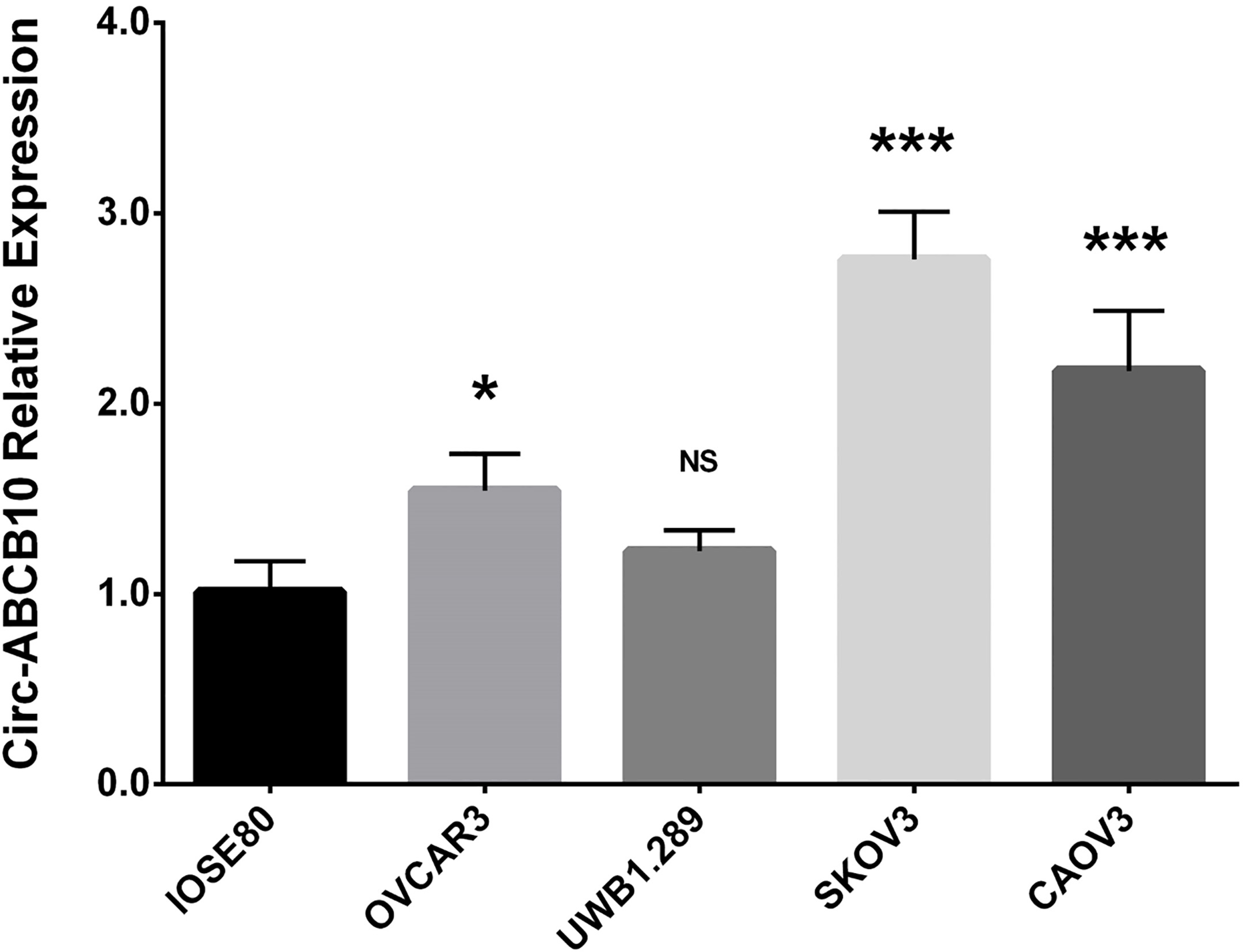
In SKOV3 cells, circ-ABCB10 expression was increased in ABCB10 (
Circ-ABCB10 expression, cell proliferation and apoptosis after transfection in SKOV3 cells. Circ-ABCB10 relative expression was elevated by circ-ABCB10 overexpression transfection and lowered by circ-ABCB10 silencing transfection (A). Cell proliferation was promoted by circ-ABCB10 overexpression transfection and reduced by circ-ABCB10 silencing transfection (B). Cell apoptosis was reduced by circ-ABCB10 overexpression transfection and increased by circ-ABCB10 silencing transfection (C and E). C-caspase 3 expression was lowered by circ-ABCB10 overexpression transfection and raised by circ-ABCB10 silencing transfection; Bcl-2 expression was increased by circ-ABCB10 overexpression transfection and decreased by circ-ABCB10 silencing transfection. Comparison of circ-ABCB10 expression, cell proliferation and apoptosis between two groups were performed by 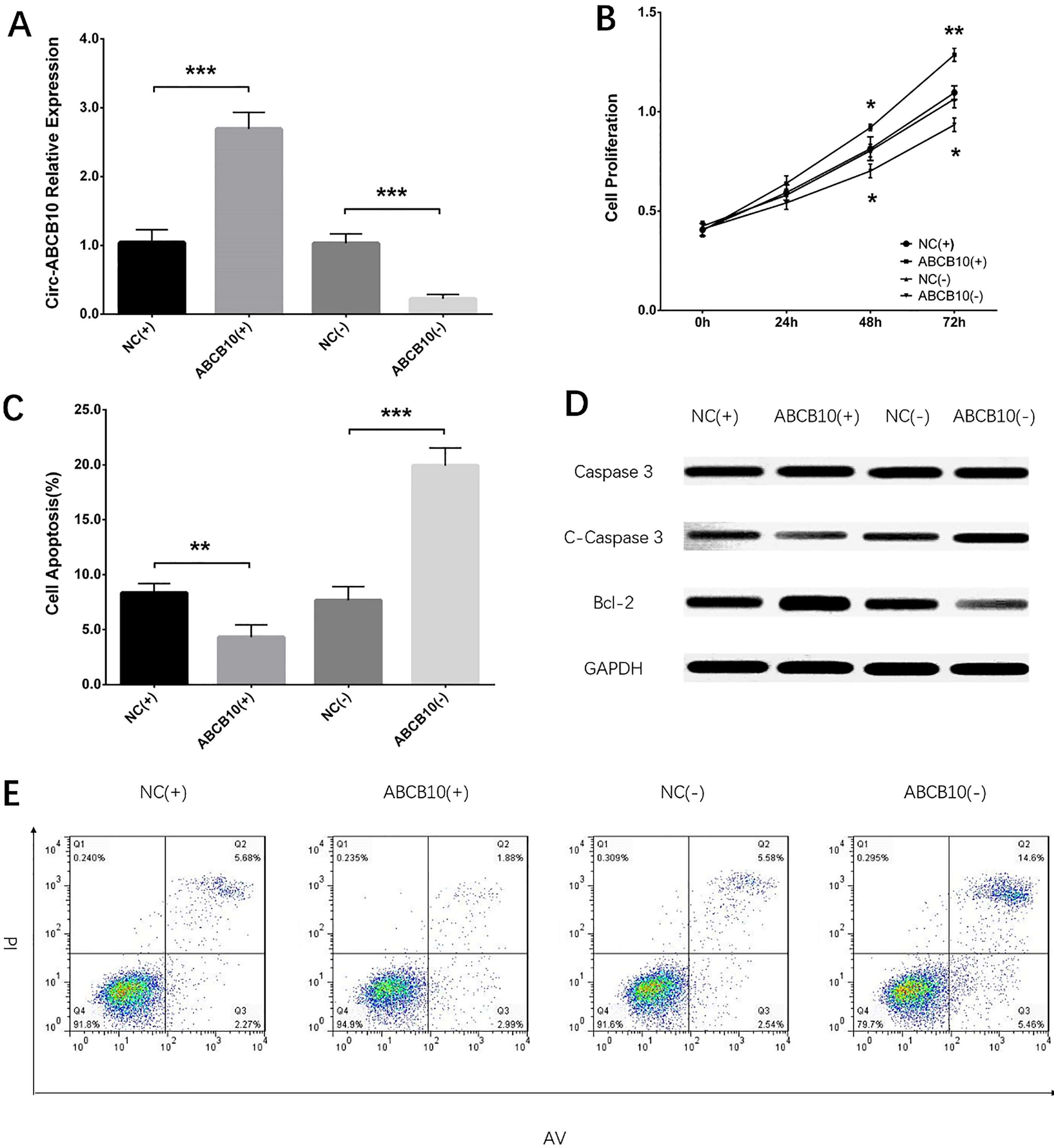
Circ-ABCB10 expression, cell proliferation and apoptosis after transfection in UWB1.289 cells. Circ-ABCB10 expression was elevated by circ-ABCB10 overexpression transfection and suppressed by circ-ABCB10 silencing transfection (A). Cell proliferation was increased by circ-ABCB10 overexpression transfection and decreased by circ-ABCB10 silencing transfection (B). Cell apoptosis was reduced by circ-ABCB10 overexpression transfection and promoted by circ-ABCB10 silencing transfection (C and D). Comparison of circ-ABCB10 expression, cell proliferation and apoptosis between two groups were performed by t test. 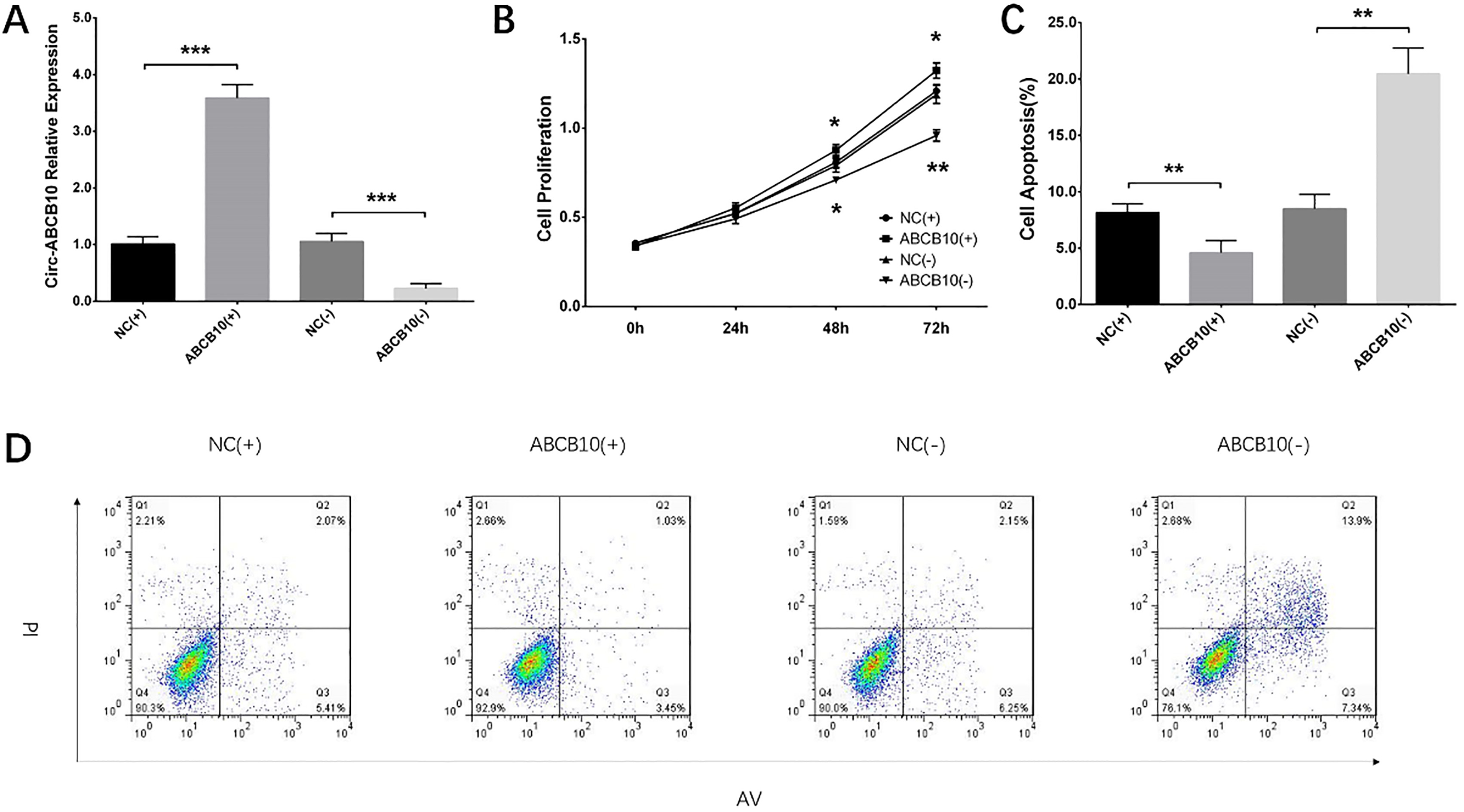
MiR-1271, miR-1252 and miR-203 expression after transfection in SKOV3 cells. MiR-1271 (A), miR-1252 (B) and miR-203 (C) expressions were suppressed by circ-ABCB10 overexpression transfection but promoted by circ-ABCB10 silencing transfection. Comparison of miRNA expression between two groups was performed by 
MiR-1271, miR-1252 and miR-203 expression after transfection in UWB1.289 cells. MiR-1271 (A), miR-1252 (B) and miR-203 (C) expressions were decreased by circ-ABCB10 overexpression transfection but increased by circ-ABCB10 silencing transfection. Comparison of miRNA expression between two groups was performed by 
MiR-1271, miR-1252 and miR-203 that were previously shown as target miRNAs of circ-ABCB10 were selected, and the regulatory effect of circ-ABCB10 on these miRNAs in SKOV3 cells and UWB1.289 cells was investigated. In SKOV3 cells, miR-1271 (Fig. 6A), miR-1252 (Fig. 6B) and miR-203 (Fig. 6C) expressions were downregulated in ABCB10 (
Discussion
Our study disclosed that in EOC patients, (1) circ-ABCB10 was upregulated in tumor tissues and correlated with advanced clinicopathological features; (2) circ-ABCB10 was correlated with unfavorable OS but not an independent predictive factor; (3) Circ-ABCB10 was upregulated, and it promoted proliferation but reduced cell apoptosis as well as negatively regulated miR-1271, miR-1252 and miR-203 in EOC cells.
Different from linear RNAs, circRNAs are considered to function independent of their host genes via multiple means, such as acting as miRNA sponges, protein sponges or decoys etc, and they have been implicated in several hallmarks in cancers due to these unique properties [1]. For instance, circ-SMARCA5 is downregulated in tumor tissues and is negatively correlated with aggressive clinical characteristics in hepatocellular carcinoma (HCC) patients [17]. Conversely, circ-BPTF expression is increased in bladder cancer tissues and it predicts higher tumor grades in bladder cancer patients [18]. In addition, the expression of circ_0067934 is remarkably higher in tumor tissues compared with paired adjacent normal tissues, and is associated with poor disease stage in esophageal squamous cell carcinoma patients [19]. These previous studies have implied the aberrant expression and the close correlation of several circRNAs with human cancers. As for circ-ABCB10, it has been observed to be significantly upregulated in breast cancer tissues and plays oncogenic role in NSCLC as well as osteosarcoma, whereas its role in EOC is still obscure [10, 15, 16]. Therefore, in this study, we investigated the function of circ-ABCB10 in EOC and discovered that it was upregulated in tumor tissue and was correlated with poor pathological differentiation, large tumor size and advanced FIGO stage in EOC patients. This might result from that (1) circ-ABCB might sponge miRNAs such as miR-221-3p and miR-1276 (retrieved from Tissue-Specific CircRNA Database,
Although the aberrant expression of a number of circRNAs and their correlations with clinicopathological status in several human cancers have been revealed, the prognostic roles of circRNAs, especially their predictive value on survival in EOC patients are sparsely reported. One previous study illustrates that circ-ITCH independently predicts favorable OS in EOC patients [20]. Another study performs circRNA profiling and screens out that circ-EXOC6B and circ-N4BP2L2 are correlated with longer OS and progression free survival in EOC patients [21]. As for circ-ABCB10, its prognostic role has not been reported in any cancer researches except for osteosarcoma, in which high circ-ABCB is associated with shortened survival [16]. In our study, we first evaluated the predictive role of circ-ABCB10 on survival in EOC patients and disclosed that circ-ABCB10 was associated with unfavorable OS in EOC patients, but was not an independent predictive factor for OS. This might be attributed to that: (1) circ-ABCB10 might facilitate cancer cell proliferation and inhibit apoptosis, promoted cancer progression and higher disease stage, which brought in poor OS in EOC patients. The effect of circ-ABCB10 on cancer cell proliferation and apoptosis was validated in the following in vitro experiments. (2) Circ-ABCB10 might alter the sensitivity to drugs in EOC cells, reducing the treatment responses and worsening OS in EOC patients. (3) Circ-ABCB10 might affect survival via interacting or correlating with other tumor features such as tumor size, differentiation and FIGO stage, thus, it was not an independent predictive factor for OS. Other than that, patients’ characteristics including poor differentiation, large tumor size, high volume of ascites, peritoneal cytology test positive and advanced FIGO stage were independent predictive factors for shorter OS in EOC patients. The possible reason might be that advanced disease stage might reduce patients’ compliance to treatment, attenuated the treatment efficacy and increase the risk of recurrence, hence worsened the survival in EOC patients.
It is considered that circRNAs take part in tumorigenesis mainly via altering cancer cell activities by absorbing the functional miRNAs in the cytoplasm [6]. In line with that, circ-ABCB10 promotes NSCLC cell proliferation and migration by sponging miR-1252 and increasing FOXR2 levels [15]. Besides, circ-ABCB10 sponges miR-203 in osteosarcoma, and knockdown of circ-ABCB10 suppresses cell proliferation, migration and invasion but promotes cell apoptosis [16]. n addition, circ-ABCB10 is also discovered to increase cell proliferation and suppress cell apoptosis in breast cancer via directly binding miR-1271 [10]. These data suggest that circ-ABCB10 participate in carcinogenesis of NSCLC, osteosarcoma and breast cancer via regulating cell activities, whereas the cellular influence of circ-ABCB10 in EOC is still obscure. Known from our findings that circ-ABCB10 is correlated with increased disease risk, poor clinicopathological features and unfavorable survival in EOC patients, in vitro experiments were conducted to further validate the function of circ-ABCB10 in EOC. We observed that circ-ABCB10 was upregulated in EOC cell lines, and it promoted cell proliferation but inhibited cell apoptosis. The possible reason was that circ-ABCB10 might serve as sponge to tumor suppressive miRNAs, such as miR-1252, miR-1271, and miR-203, as previously reported, in the cytoplasm and influence the downstream protein expression, which subsequently promoted cell proliferation and inhibited cell apoptosis in EOC. This explanation was further validated by our following experiments, which disclosed that circ-ABCB10 negatively regulated miR-1252, miR-1271, and miR-203 levels in both SKOV3 cells and UWB1.289 cells.
In conclusion, circ-ABCB10 correlates with advanced clinicopathological features and unfavorable survival in EOC patients, and it promotes proliferation, reduces apoptosis and negatively regulates miR-1271, miR-1252 and miR-203 in EOC.
Footnotes
Conflict of interest
No potential conflict of interest was reported by the authors.
