Abstract
BACKGROUNDS:
Upregulation of lncRNA AB073614 is found in some cancer types and involved in tumor development and progression including ovarian cancer. However, the clinical value and functional role of lncRNA AB073614 in epithelial ovarian cancer (EOC) still needed to be investigated.
METHODS:
We examined lncRNA AB073614 expression using quantitative real time polymerase chain reaction (qRT-PCR) in 75 paired of EOC tissue samples and adjacent normal tissues. Association of lncRNA AB073614 expression with overall survival (OS) was evaluated using Kaplan-Meier analysis. Univariate and multivariate analysis of factors associated with OS were assessed in EOC patients. After lncRNA AB073614 knockdown using siRNAs, the cell viability and cell colony forming assays were performed. Western blot analysis was used to assess relative protein expression.
RESULTS:
In present study, we demonstrated that lncRNA AB073614 was significantly upregulated in ovarian cancer tissues compared to adjacent normal tissues in patients. Higher lncRNA AB073614 expression significantly associated with tumor size, lymph node invasion, FIGO stage, and shorter OS rate of EOC patients. Furthermore, multivariate Cox regression analysis results showed that higher lncRNA AB0736141 was identified as an independent risk factor of OS in EOC patients. Moreover, we demonstrated that lncRNA AB0736141 knockdown suppressed EOC cell proliferation ability and cell colony formation in vitro. In vivo, we showed that AB0736141 knockdown suppressed tumor growth. We also revealed that lncRNA AB0736141 knockdown inhibited the PTEN/PI3K/AKT signaling pathway in EOC.
CONCLUSIONS:
Thus, these results indicated that LncRNA AB073614 may serve as a prognostic biomarker and potential target of treatment for EOC.
Introduction
Ovarian cancer is the most lethal gynecologic malignancy in female and the epithelial ovarian cancer is the one of major tumor types [1]. Approximately 240,000 cases occur and 150,000 cases were deaths in 2012 worldwide [2]. Due to lack of reliable biomarkers at the early stage and chemotherapy resistance for the disease, patients that are diagnosed at the advanced stages have a low 5-year survival rate with less than 30% [3]. Thus, to explore early stage biomarkers and underlying molecular mechanisms of tumorigenesis and progression of EOC is necessary.
The transcripts that lack protein coding capacity are called non-coding RNAs, and they are further classified based on their length. Long non-coding RNAs (lncRNAs) that more than 200 nucleotides in length have been functionally identified to play crucial regulators of human cancers [4]. In ovarian cancer, previous studies have suggested that lncRNAs affect tumor development and carcinogenesis. For example, Zhang et al. reported that lncRNA HOTAIR controls the expression of Rab22a by sponging miR-373 in ovarian cancer [5]. MALAT-1 is highly expressed in ovarian cancer and induces cell growth and migration [6]. UCA1 functions as a competing endogenous RNA to suppress epithelial ovarian cancer metastasis [7]. In addition, more and more lncRNAs are identified to be participated in ovarian cancer progression.
Long non-coding RNA AB073614 is reported in some cancer types, Cheng et al. revealed that lncRNA AB073614 promotes tumorigenesis and predicts poor prognosis in ovarian cancer [8, 9]. Hu et al. revealed that higher AB073614 expression predicts a poor prognosis in patients with glioma [10]. Knockdown of long non-coding RNA AB073614 inhibits glioma cell proliferation and migration via affecting epithelial-mesenchymal transition (EMT) [11]. Wang et al. showed that lncRNA AB073614 regulates proliferation and metastasis of colorectal cancer cells via the PI3K/AKT signaling pathway [12]. However, the role of lncRNA AB073614 in EOC still needs to be well investigated.
In present study, we demonstrated that lncRNA AB073614 was significantly upregulated in EOC tissues and associated with poor prognosis of EOC patients. Function assays demonstrated that lncRNA AB0736141 knockdown suppressed cell proliferation ability in EOC. Additionally, knockdown of AB0736141 significantly inhibited the PTEN/PI3K/ AKT signaling pathway of EOC cells. Thus, these results indicated that lncRNA AB073614 may act as a prognostic biomarkers and potential target of treatment in EOC.
Methods
Patients and tissue samples
A total of 75 paired of EOC tissue samples (the control groups were adjacent normal tissues including fallopian tube/non-tumor section of ovary, etc.) were obtained from patients who had received surgery at the Department of Gynecology, Cangzhou Central Hospital, Hebei Medical University. All of cases were diagnosed as EOC by two professional pathologists. All tissues samples were immediately frozen in liquid nitrogen and stored at
Association of AB073614 expression with clinicopathological variables in 75 ovarian cancer patients
Association of AB073614 expression with clinicopathological variables in 75 ovarian cancer patients
Three EOC cell lines (SKOV3 and HO-8910, OVCAR3) and a normal ovarian cell line HOEpiC were purchased from American Type Culture Collection (ATCC, Manassas, VA, USA). Cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM) supplemented with 10% FBS (Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA), 100 U/ml penicillin and 100
RNA isolation and quantitative RT-PCR (qRT-PCR)
The qRT-PCR assay was performed as previous describe [11]. Briefly, total RNA from tissues and cell lines was extracted using Trizol reagent (Invitrogen; Carlsbad, CA, USA) according to the manufacturer’s instructions. RNA was reversed using First Strand cDNA System (Invitrogen; Carlsbad, CA, USA) The SYBR
Cell transfection
Two siRNAs oligos targeting human AB073614 and a negative control were designed and synthesized by GenePharma (Shanghai, China). Cells were transfected with siRNA-AB073614-1, siRNA-AB073614-2 and si-negative control (NC) using Lipofectamine 2000 (Invitrogen; Carlsbad, CA, USA) according to the manufacturer’s instruction.
Cell viability assay
Cells proliferation was assessed using CCK8 Kit (Beyotime, Shanghai, China). SKOV3 and HO-8910 were seeded in 96-well plates. Following cells were cultured for 24 h overnight, 2000 cells were transfected with si-NC and si-AB073614. After cell transfection at 24, 48, 72, and 96 h, the cell viability was detected, Absorbance values at 450 nm and were analyzed under a microplate reader (BioTek, VT, USA).
Cell colony forming assay
Cells were plated in 6-well plate at a concentration of 500 cells/well. Cells were transfected with si-NC and si-AB073614. Two weeks later, cell colonies were fixed with methanol, stained using 0.1% crystal violet and were counted.
Western blot analysis
After cell transfection at 48 h, SKOV3 and HO-8910 cells were harvested and lysed using radioimmunoprecipitation assay (RIPA) buffer (Beyotime, Shanghai, China). Equal protein was separated on 10% SDS denaturing polyacrylamide gel (SDS-PAGE) and transferred onto PVDF membrane (Bio-Rad, Hercules, CA, USA). The 5% nonfat milk was used to block the membranes for 1 h and incubated with primary antibodies with PTEN (sc-133242, 1:1000, Santa Cruz, Biotechnology, Inc., Dallas, TX, USA), AKT (sc-5298, 1:500, Santa Cruz, Biotechnology, Inc., Dallas, TX, USA), BCL-2 (sc-509, 1:300, Santa Cruz, Biotechnology, Inc., Dallas, TX, USA), CCND1(sc-786, 1:400, Santa Cruz, Biotechnology, Inc., Dallas, TX, USA), and CyclinD1 (sc-8396, 1:300, Santa Cruz, Biotechnology, Inc., Dallas, TX, USA), p-AKT (sc-271966, 1:500, Santa Cruz, Biotechnology, Inc., Dallas, TX, USA). The protein blots were visualized by enhanced chemiluminescence (ECL) according to the manufacturer’s instruction. GAPDH was normalized to the other protein in the study.
Xenograft tumor formation assay
2
Statistical analysis
All data was analyzed SPSS version 18.0 (SPSS Inc., IL, USA). Data were shown as the mean
Expression of lncRNA AB073614 is upregulated in human EOC tissues and cells. (A) The expression of lncRNA AB073614 in 75 EOC tumor tissues and paired adjacent normal tissues were detected by qRT-PCR. GAPDH was used as internal control. (B) The expression of AB073614 in three EOC cell lines (SKOV3 and HO-8910, OVCAR3) and a normal ovarian cell line HOEpiC were detected by qRT-PCR. GAPDH was used as internal control. (C) Association of AB073614 expression with overall survival rate in EOC patients was shown using Kaplan-Meier analysis and log rank test. (D) and (E) The expression of AB073614 was detected by qRT-PCR when cells were transfected with si-NC or si-AB073614-1 and si-AB073614-2 in OVCAR3 and HO-8910 cells. All data are representative of three independent experiments, 
The expression of lncRNA AB073614 is upregulated in human EOC tissues and cells
The relative lncRNA AB073614 expression was detected using qRT-PCR methods in 75 EOC tissue specimens and adjacent normal tissue specimens. The results indicated that AB073614 expression significantly higher in tumor tissues than that in normal ovarian tissues (2.876
The median lncRNA AB073614 expression (2.98 fold) in patients was used as a cut off value, we divided EOC patients into higher expression group (lncRNA AB073614 expression
Univariate and multivariate analysis of factors associated with overall survival
Univariate and multivariate analysis of factors associated with overall survival
AB073614 downregulation suppresses EOC cells proliferation and increases cisplatin sensitivity. (A) and (B) Representative CCK8 assays were performed when cells were transfected with si-NC or si-AB073614 in OVCAR3 and HO-8910 cells. (C) and (D) Representative cell colony forming assays were performed when cells were transfected with si-NC or si-AB073614 in OVCAR3 and HO-8910 cells. All data are representative of three independent experiments, 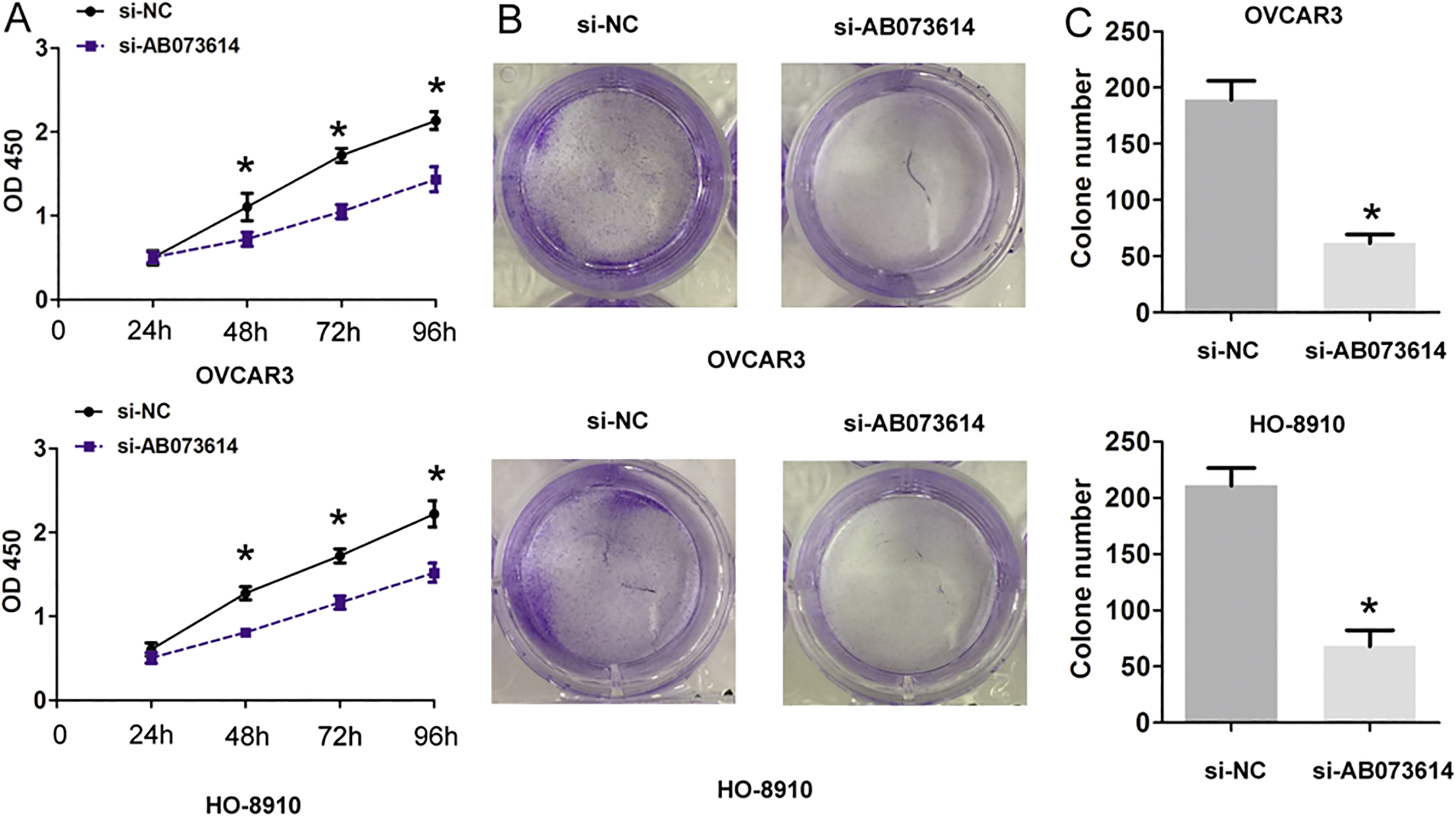
We further explored whether lncRNA AB073614 expression affected EOC cell biological functions. We knocked down lncRNA AB073614 by two siRNAs oligos and found that the si-AB073614-1 showed a higher knockdown efficiency in EOC cells (Fig. 1D and E). At 48 h and 72 h after cell transfection, CCK8 assay showed that cell viability in both OVCAR3 and HO-8910 cells were significantly inhibited when lncRNA AB073614 was downregulated (Fig. 2A). Cell colony forming assays analysis results indicated that cell colonies number in both OVCAR3 and HO-8910 cells were significantly reduced when AB073614 was downregulated (Fig. 2B and C). Thus, these results indicated that lncRNA AB073614 knockdown suppressed EOC cells proliferation ability in EOC.
Knockdown of AB073614 suppresses tumor growth in vivo
To explore the effects of AB073614 expression in vivo, we constructed the nude xenograft model using BALB/c nude mouse inoculated subcutaneously into the flanks in OVCAR3 cells that stably transfected with sh-negative control and sh-AB073614 plasmids. After 4 weeks, the mice were killed. Our results indicated that tumor volume was smaller and tumor growth was slower in nude mice injected with sh-AB073614 plasmid compared the empty vector group (Fig. 3A and B). These results indicated that knockdown of AB073614 suppressed tumor growth in vivo.
Knockdown of AB073614 suppresses tumor growth in vivo. (A) Tumor formation was performed in nude mice and the tumor volume was shown. (B) Tumor growth was slower from nude mice in sh-AB073614 group each week after injection compared with the empty control. 
AB073614 affects PTEN/PI3K/AKT signaling pathway in EOC cells. (A) The relative protein of PTEN, p-AKT, AKT, CCND1, CyclinD1, and BCL-2 were detected using western blot analysis when cells were transfected with si-NC or si-AB073614 in OVCAR3. (B) The relative protein of PTEN, p-AKT, AKT, CCND1, CyclinD1, and BCL-2 were detected using western blot analysis when cells were transfected with si-NC or si-AB073614 in HO-8910 cells. All data are representative of three independent experiments.
Some signaling pathway including PI3k/Akt, Ras/ MAPK, and JAK/STAT pathways, could increase cell proliferation, invasion, and chemotherapy resistance in tumor cells [13, 14]. Furthermore, we tested whether lncRNA AB073614 expression affected the PTEN/ PI3K/AKT signaling pathway. After OVCAR3 and HO-8910 cells was transfected with si-AB073614 at 48 h, the results demonstrated that the expression of AKT had no significant change, but the expression of p-AKT, CCND1, CyclinD1, BCL-2 was significantly downregulated in VCAR3 and HO-8910 cells compared with NC group. Moreover, the PTEN was increased when lncRNA AB073614 was knocked down in VCAR3 and HO-8910 cells compared with NC group (Fig. 4A and B). These results indicated that lncNRA AB073614 knockdown affected PTEN/PI3K/ AKT signaling pathway in EOC cells.
Discussion
Dysregulation of long noncoding RNAs frequently is found in many types of tumors including ovarian cancer [15]. The aberrant expression of lncRNAs in ovarian cancer could mark the spectrum of disease progression and these lncRNAs may serve as independent biomarkers for diagnosis and prognosis. Such as, Li et al. showed that long non-coding RNA C17orf91 is a potential prognostic marker and functions as an oncogene in ovarian cancer [16]. Downregulation of lncRNA TUBA4B is associated with poor prognosis for epithelial ovarian cancer [17]. Upregulation of the long non-coding RNA SPRY4-IT1 indicates a poor prognosis and promotes tumorigenesis in ovarian cancer [18]. These previous findings indicated that the clinical significance and functional effects of lncRNAs in ovarian cancer.
LncRNA AB073614 is firstly identified as a Homo sapiens primary hepatoblastoma cDNA (clone: HMFN1050) and is reported to be higher expression in ovarian cancer [8]. In the study, by qRT-PCR analysis, our results also showed that lncRNA AB073614 expression was significantly upregulated in EOC tissues and cells. Association of lncRNA AB073614 with clinical data showed that higher lncRNA AB073614 expression associated with tumor size, lymph node invasion, FIGO stage, and shorter overall survival rate of EOC patients. By multivariate Cox regression analysis, we showed that lncRNA AB073614 expresssion was an independent risk factor of overall survival in EOC patients. These above finding indicated that lncRNA AB073614 expression may serve as prognostic biomarker for EOC.
In previous study, some lncRNAs was found to affect PI3K/AKT signaling pathway in epithelial ovarian cancer progression. For instance, lncRNA MALAT1 promotes proliferation and metastasis in epithelial ovarian cancer via the PI3K-AKT pathway [19]. In colorectal cancer, lncRNA AB073614 regulates tumor proliferation and metastasis of colorectal cancer cells via the PI3K/AKT signaling pathway [12]. In the study, functional assays demonstrated that lncRNA AB0736141 expression knockdown suppressed cell proliferation ability in EOC. We further explored if lncNRA AB073614 knockdown suppressed cell proliferation ability associated with PTEN/PI3K/AKT signaling pathway or not. The results showed that the results demonstrated that the expression of AKT had no significant change, but the expression of p-AKT, CCND1, and CyclinD1 was significantly downregulated compared with NC group in EOC cells. Meanwhile, the PTEN was increased when lncRNA AB073614 was knocked down in EOC cells compared with NC group, which indicated that lncRNA AB0736141 could associate with PTEN/PI3K/AKT signaling pathway in EOC. In the previous study, Cheng et al. showed that lncRNA AB073614 was upregulated in ovarian cancer and promoted cell proliferation, invasion by targeting ERK1/2 and AKT-mediated signaling pathway [9]. Our results also showed that lncRNA AB073614 knockdown reduced cell proliferation by regulating the PTEN/PI3K/AKT signaling pathway. In the further, the underlying regulator mechanism between lncRNA AB073614 expression and PTEN/PI3K/AKT signaling pathway need to be discovered.
In conclusion, we demonstrated that lncRNA AB073614 was higher in EOC tissues and closely correlated with poor prognosis of patients. Moreover, lncRNA AB0736141 knockdown suppressed cell proliferation ability and PTEN/PI3K/AKT signaling pathway. These results indicated that AB0736141 may serve as prognostic maker and potential therapeutic target for EOC.
Footnotes
Conflict of interest
The authors declare no conflict of interest.
