Abstract
Long non-coding RNAs play important roles in the regulation of cellular processes including cell proliferation, differentiation, and metastasis. The dysregulation of long non-coding RNAs, such as the SPRY4-IT1 (SPRY4 intronic transcript 1), has been associated with various types of malignancies. However, the functional roles and regulatory mechanism of SPRY4-IT1 in ovarian cancer remain to be elucidated. Here, we quantified the expression level of SPRY4-IT1 in ovarian cancer patients and found its downregulation in ovarian cancer tissues compared to the adjacent normal tissues. Patients with lower SPRY4-IT1 expression were associated with a relatively poor prognosis. In consistency, the expression of SPRY4-IT1 was found to be reduced in four human ovarian cancer cell lines compared to normal ovarian epithelial cells. Next, two ovarian cancer cell lines SKOV3 and HO8910 were employed in vitro assays to investigate biological functions of SPRY4-IT1 in ovarian cancer. The cell proliferation was reduced following SPRY4-IT1 overexpression in SKOV3/HO8910 cells based on 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide and colony formation assays. The SPRY4-IT1 overexpression also dramatically arrested cell cycle and promoted cell apoptosis. Both wound-healing and transwell-based assays demonstrated that cell migration and invasion were inhibited following SPRY4-IT1 overexpression. Meanwhile, overexpression of SPRY4-IT1 increased E-cadherin and decreased N-cadherin and vimentin protein levels, indicating that SPRY4-IT1 may regulate ovarian cancer cell metastasis through the inhibition of epithelial–mesenchymal transition. Taken together, our findings suggest that SPRY4-IT1 regulates various cellular processes of ovarian cancer cells and its downregulation may contribute to ovarian cancer progression and metastasis partly via affecting the epithelial–mesenchymal transition.
Introduction
Ovarian cancer is the seventh most commonly diagnosed cancer in females worldwide, with an estimated occurrence of 238,700 new cases and 151,900 deaths in 2012. 1 In 2017, the American Cancer Society estimates an occurrence of 22,400 new cases of ovarian cancer and 14,080 deaths in the United States. 2 Lack of efficient screening and early diagnosis leads to the diagnosis of most ovarian cancer patients at advanced stages. 3 Routine treatments for ovarian cancer include surgery and chemotherapy, which are ineffective for advanced-stage patients. 4 Recent studies have suggested that the epithelial–mesenchymal transition (EMT) may play an important role in epithelial cancer metastasis and chemoresistance. 5 Cancer cells may acquire and maintain mesenchymal phenotypes through the EMT, such as invasiveness and resistance to apoptosis. 6 EMT signaling pathways have also been associated with various epithelial cancers including ovarian cancer. 7 Reversal of EMT in ovarian cancer may represent a novel strategy for the treatment of late-stage patients. Therefore, a better understanding of molecules involved in ovarian cancer progression and mechanisms underlying its EMT process are critical for development of new methods to improve therapeutic interventions of ovarian cancer.
Long non-coding RNAs (lncRNAs) belong to a new class of non-coding RNAs with more than 200 nucleotides in length.8,9 They have been recently demonstrated to play important roles in the regulation of chromatin remodeling, gene transcription, and pre-messenger RNA (mRNA) processing.10,11 Dysregulation of lncRNAs is associated with various types of malignancies.12–14 LncRNAs can regulate both oncogenic and tumor-suppressive pathways. 15 A number of lncRNAs have been described to be involved in ovarian cancer. 16 For example, the human ovarian cancer specific transcript 2 (HOST2) was reported to be overexpressed in ovarian cancer and promotes proliferation and migration of ovarian cancer cells as well as tumor growth in xenografted mice. 17
Recently, increasing evidences have shown that the SPRY4 intronic transcript 1 (SPRY4-IT1), which belongs to a group of intron-retained lncRNAs, is dysregulated in various cancers. For example, SPRY4-IT1 was found with enhanced expression in human melanoma, renal cancer, and gastric cancer tissues.18–20 The in vitro assays using the respective cell lines of these cancers demonstrated that knockdown of SPRY4-IT1 inhibited cell proliferation, migration, and invasion and induced cell apoptosis. The results suggested that in these types of cancers, SPRY4-IT1 may play an important role in the promotion of cancer progression and metastasis. In some other cases, the SPRY4-IT1 expression was shown to be downregulated in gastric cancer and non–small cell lung cancer tissues.21,22 Overexpression of SPRY4-IT1 suppressed trophoblast cell migration and invasion, whereas reduced expression of SPRY4-IT1 prevented the EMT process. 23 To date, the functional roles and regulatory mechanism of SPRY4-IT1 in ovarian cancer have remained poorly understood.
In this study, we wanted to investigate the expression correlation of SPRY4-IT1 with ovarian cancer and to further explore the biological functions and regulatory mechanism of SPRY4-IT1 in this type of cancer. We first examined the expression level of SPRY4-IT1 in ovarian cancer tissues versus the adjacent normal ovarian tissues using the method of quantitative real-time polymerase chain reaction (qRT-PCR). We observed reduced SPRY4-IT1 expression in ovarian cancer tissues as well as in several ovarian cancer cell lines. Next, we employed ovarian cancer cells SKOV3 and HO8910 in vitro assays to investigate biological functions and regulatory mechanism of SPRY4-IT1 in ovarian cancer. Cell proliferation, apoptosis, migration, invasion, and EMT following SPRY4-IT1 overexpression were analyzed, which suggested that SPRY4-IT1 regulated these cellular processes and contributed to ovarian cancer cells metastasis, at least partly, via regulating the EMT process. Our findings established the correlation between reduced SPRY4-IT1 expression and ovarian cancer and revealed the mechanism of how downregulation of SPRY4-IT1 may contribute to the development of ovarian cancer.
Materials and methods
Human tissue sample collection
Human tissue samples were collected from 15 ovarian cancer patients. Informed consent was obtained in all cases, and protocols were approved by medical ethics committee of The Second Affiliated Hospital of Harbin Medical University (ethics number: K1 /2017-148). Then, the cancer and normal tissues were collected for paired analysis in this study. The excised tissue specimens were snap-frozen in liquid nitrogen and stored until the RNA extraction.
Cell culture
In total, four human ovarian cancer cell lines including SKOV3, HO8910, ES-2, and CAOV3 and normal human ovarian epithelial cells IOSE80 were purchased from the Institute of Biochemistry and Cell Biology, Chinese Academy of Sciences (Shanghai, China). Cells were maintained in Dulbecco’s modified Eagle’s medium (DMEM) or RPMI-1640 media plus 10% fetal bovine serum (FBS) according to the manufacturer’s instructions and cultured in a CO2 incubator.
RNA extraction and qRT-PCR
Total RNA was extracted from human tissues or cells using TRIzol reagent (Invitrogen, USA). The complementary DNA (cDNA) was reverse transcribed from total RNA using the Prime-Script™ RT-PCR Kit (TaKaRa, Dalian, China). The qRT-PCR reactions were performed using the SYBR Green PCR Master Mix (TaKaRa) on an ABI7900 instrument (Applied Biosystems, USA). The expression of target genes was normalized to that of a house-keeping gene glyceraldehyde 3-phosphate dehydrogenase (GAPDH). The sequences of the qRT-PCR primers are listed in Table 1.
q-PCR primer sequences.

q-PCR: quantitative polymerase chain reaction; SPRY4-IT1: SPRY4 intronic transcript 1; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
SPRY4-IT1 plasmid preparation and transfection
The sequence of SPRY4-IT1 was synthesized and subcloned into the pCMV vector. The SPRY4-IT1 plasmid was transfected into ovarian cancer cells using Lipofectamine 2000 reagent (Thermo Fisher Scientific, USA) for overexpression. The empty pCMV vector was employed as the negative control in all SPRY4-IT1 overexpression experiments.
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assays
After transfection with pCMV-SPRY4-IT1 or empty vector for 48 h, the ovarian cancer cells were trypsinized and seeded onto 96-well plates at 1 × 104 cells per well. 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) solution (100 µg) was added to each well containing 150 µL medium at 24, 48, 72, and 96 h later, respectively. After incubation for 3 h at 37°C, the medium was removed and 150 µL of dimethyl sulfoxide (DMSO) solution was added to lyse the cells for 15 min. The absorbance at 490 nm was detected using a microplate reader.
Colony formation assay
After transfection with pCMV-SPRY4-IT1 or empty vector for 48 h, the ovarian cancer cells were trypsinized and seeded onto six-well plates at 1 × 103 cells per well. The cells were grown for 2 weeks with medium renewal for every 3 days. Colonies were fixed with methanol and stained with 0.1% crystal violet in phosphate-buffered saline (PBS). Photographs were taken and the number of colonies at each well was counted.
Western blot analysis
Cells were lysed with the radioimmunoprecipitation assay (RIPA) buffer supplemented with protease inhibitors for 15 min and then centrifuged at 14,000g for 10 min at 4°C. The total cell extracts were run on a sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and transferred onto polyvinylidene difluoride (PVDF) membranes. Western blotting was performed by blocking the membranes with 5% milk for 1 h and then incubating with primary antibodies at 4°C overnight. The next day, after washing thrice with 1× Tris-buffered saline (TBS) buffer supplemented with 0.1% Tween (TBST), the membranes were incubated with the horseradish peroxidase (HRP)-conjugated secondary antibodies at room temperature for 1 h. After washing thrice with the TBST buffer, the protein bands were visualized by the enhanced chemiluminescence (ECL) kit (Thermo Fisher Scientific).
Cell cycle analysis
After transfection with pCMV-SPRY4-IT1 or empty vector for 48 h, the ovarian cancer cells were harvested, washed once with cold PBS, and fixed with 70% ice-cold ethanol. Cell pellets were washed once with PBS and stained with propidium iodide (PI; BD Biosciences, USA). The cell cycle profile was analyzed by flow cytometry (Beckton Dickinson, USA), and the percentages of cells in G0–G1, S, and G2–M phases were quantified.
Cell apoptosis analysis
After transfection with pCMV-SPRY4-IT1 or empty vector for 42 h, the ovarian cancer cells were harvested by trypsinization. Cell apoptosis was analyzed by the fluorescein isothiocyanate (FITC)-Annexin V/PI double staining followed by flow cytometry analysis (BD Biosciences).
Transwell assays
After transfection with pCMV-SPRY4-IT1 or empty vector for 48 h, around 1 × 105 cells were placed into the upper chamber of inserts in serum-free medium (Millipore, USA). The bottom chamber contained medium with 10% FBS as a chemotactic factor. After incubation for 24 h, cells on the lower surface of the chamber were stained with 0.1% crystal violet and counted using a digital microscope. For the invasion assay, transfected cells were plated into the top chamber with a Matrigel-coated membrane.
Wound-healing (scratch) assay
After transfection with pCMV-SPRY4-IT1 or empty vector for 48 h, around 5 × 105 cells were seeded in six-well plates and cultured overnight. The next day, once the cells reached around 90% confluency, the cell layer was scratched with a sterile plastic tip. After removing the floated cells by washing thrice with PBS, the cells were added to the culture media and incubated in a CO2 incubator at 37°C. Images were acquired after 0 and 24 h using a microscope.
Statistical analysis
All statistical analyses were performed using the GraphPad Prism 5 software (GraphPad Software, USA). Comparison of the SPRY4-IT1 expression level in ovarian tissues and adjacent normal tissues was performed using the paired t test. All in vitro cell assays were conducted in triplicate and the data were presented as mean ± standard deviation (SD). The significance of difference between two groups was analyzed by Student’s t test.
Results
Downregulation of SPRY4-IT1 in ovarian cancer tissues and its correlation with patients’ survival rate
To evaluate the association of SPRY4-IT1 with ovarian cancer, we examined the expression level of SPRY4-IT1 in tissue samples from 15 patients using the qRT-PCR approach. By the paired comparison of ovarian cancer tissues versus adjacent ovarian normal tissues for each patient, we found that the expression level of SPRY4-IT1 in ovarian cancer tissues was significantly reduced compared to that in the adjacent normal tissues (Figure 1(a)). Furthermore, based on the analysis of Kaplan–Meier overall survival and disease-free survival curves, the expression level of SPRY4-IT1 showed a correlation with the survival rate in these ovarian cancer patients (Figure 1(b) and (c)). Both overall and disease-free survival rates were higher for patients with high SPRY4-IT1 expression and lower for those with less SPRY4-IT1 (Figure 1(b) and (c)). These results suggest that the SPRY4-IT1 expression level is correlated with ovarian cancer, and downregulation of SPRY4-IT1 may contribute to ovarian cancer progression.

Downregulation of SPRY4-IT1 in ovarian cancer tissues and its correlation with patient survival rate. (a) Reduced expression of SPRY4-IT1 in ovarian cancer tissues (OEC) compared to adjacent normal ovarian tissues based on the qRT-PCR analysis (n = 15, p < 0.05 as analyzed by the paired t test indicates significant difference between the two groups). (b) Kaplan–Meier overall survival curves showing the correlation between decreased SPRY4-IT1 expression and lower survival rate in patients. (c) Disease-free survival curves showing the correlation between lower SPRY4-IT1 expression and less survival of patients.
Overexpression of SPRY4-IT1 inhibited cell proliferation in ovarian cancer cell lines
To investigate the functional role of SPRY4-IT1 in ovarian cancer, we employed ovarian cancer cell lines for the in vitro assay of cell proliferation. First, we analyzed the expression level of SPRY4-IT1 in four ovarian cancer cell lines including SKOV3, HO8910, ES-2, and CAOV3 relative to the normal human ovarian epithelial cells IOSE80. As shown in Figure 2(a), SPRY4-IT1 exhibited a lower expression level in each of the ovarian cancer cell lines compared to IOSE80. This result was in agreement with the reduced expression of SPRY4-IT1 in ovarian cancer tissues (Figure 1). The SKOV3 and HO8910 cells showed the lowest expression of SPRY4-IT1 and were therefore employed for cell proliferation assays. Next, the SKOV3 and HO8910 cells were transiently transfected with the pCMV-SPRY4-IT1 construct or the empty vector (mock; Figure 2(b)). The overexpression of SPRY4-IT1 was confirmed by qRT-PCR, which was three- to four-fold higher than that of mock-transfected control cells (Figure 2(c)). Cell proliferation was evaluated by the MTT and colony formation assays. Based on the MTT assay, cell viability was significantly decreased by SPRY4-IT1 overexpression both in SKOV3 and HO8910 cells (Figure 2(d)). Fewer colonies were formed in cells transfected with SPRY4-IT1 compared to mock-transfected cells, with a significant reduction of around 50% (Figure 2(e)). Collectively, these results support that SPRY4-IT1 plays an inhibitory role in the proliferation of ovarian cancer cells, and its downregulation may result in enhanced cell proliferation, thus contributing to ovarian cancer progression.

Overexpression of SPRY4-IT1 inhibited cell proliferation in ovarian cancer cell lines. (a) Lower expression level of SPRY4-IT1 in the four ovarian cancer cell lines SKOV3, HO8910, ES-2, and CAOV3 in comparison to that of IOSE80 by qRT-PCR analysis (n = 3, *p < 0.05 and **p < 0.01 indicate significant difference from the IOSE80 cells). (b) HO8910 and SKOV3 cells observed under fluorescence microscopy (100×) 48 h after transfection. (c) Transient transfection of pCMV-SPRY4-IT1 resulted in a remarkable increase of SPRY4-IT1 expression in HO8910 and SKOV3 cells compared to the mock-transfected control cells (n = 3, **p < 0.01 indicates significant difference between the two groups by Student’s t test). (d) MTT assay showing that the overexpression of SPRY4-IT1 reduced cell viability (n = 3, *p < 0.05 and **p < 0.01 indicate significant difference between the two groups by Student’s t test). (e) Colony formation assay showing fewer colonies in cells transfected with SPRY4-IT1 compared to control (n = 3, **p < 0.01 indicates significant difference between the two groups by Student’s t test).
Overexpression of SPRY4-IT1 arrested cell cycle of ovarian cancer cells
To further explore the mechanism underlying the inhibitory activity of SPRY4-IT1 on cell proliferation, we analyzed the cell cycle profile of cells transfected with pCMV-SPRY4-IT1 by flow cytometry and compared with that of mock-transfected cells. As shown in Figure 3, HO8910 cells transfected with pCMV-SPRY4-IT1 were mostly arrested in the S phase. The percentage of cells in the S phase increased from around 28% to 39% (Figure 3(a)). In SKOV3 cells, overexpression of SPRY4-IT1 led to an increase of cells in the G2–M phase, from about 17% to 40% (Figure 3(b)). These results suggest that SPRY4-IT1 could arrest cell cycles of ovarian cancer cells, which may consequently inhibit the cell proliferation.

Overexpression of SPRY4-IT1 arrested cell cycles of ovarian cancer cells. (a) HO8910 cells that overexpressed SPRY4-IT1 were arrested in the S phase compared to the mock-transfected cells by flow cytometry analysis. (b) SKOV3 cells that overexpressed SPRY4-IT1 were arrested in the G2–M phase compared to the mock-transfected cells by flow cytometry analysis (n = 3, *p < 0.05 indicates significant difference between the two groups by Student’s t test.
Overexpression of SPRY4-IT1 induced cell apoptosis in ovarian cancer cells
To assess the effect of SPRY4-IT1 on cell apoptosis, HO8910 and SKOV3 cells were overexpressed with SPRY4-IT1 and analyzed for cell apoptosis by the Annexin V–FITC/PI double staining assay. Cells were collected 48 h after transfection, and the Annexin V–FITC/PI staining was detected by flow cytometry. The percentage of apoptotic cells increased from around 9% to 16% and 4% to 25% by SPRY4-IT1 overexpression in HO8910 and SKOV3 cells, respectively (Figure 4(a)). In addition, western blotting was performed to examine the protein levels of apoptotic markers caspase-3 and caspase-9 as well as the anti-apoptotic protein Bcl-2. The expression of cleaved caspase-3 and cleaved caspase-9 significantly increased by SPRY4-IT1 overexpression, while the Bcl-2 protein level decreased in pCMV-SPRY4-IT1-transfected cells compared to control cells (Figure 4(b) and (c)). These results demonstrated that overexpression of SPRY4-IT1 induced apoptosis in ovarian cancer cells.

Overexpression of SPRY4-IT1 induced cell apoptosis in ovarian cancer cells. (a) Significant increase of the portion of apoptotic cells in pCMV-SPRY4-IT1-transfected cells compared to mock-transfected cells by the Annexin V–FITC/PI staining assay (n = 3, **p < 0.01 indicates significant difference between the two groups by Student’s t test). (b) Representative western blot results showing the decrease of Bcl-2 and the increase of cleaved caspase-3 and cleaved caspase-9 proteins following SPRY4-IT1 overexpression in HO8910 and SKOV3 cells. (c) Quantitative analysis of relative band intensity demonstrated the significant increase of cleaved caspase-3 and cleaved caspase-9 and the decrease of Bcl-2 protein levels upon SPRY4-IT1 overexpression (n = 3, *p < 0.05 indicates significant difference between the two groups by Student’s t test).
Overexpression of SPRY4-IT1 inhibited migration and invasion of ovarian cancer cells
To investigate the role of SPRY4- IT1 in ovarian cancer metastasis, we analyzed changes in cell motility and invasiveness upon SPRY4-IT1 overexpression in SKOV3 and HO8910 cells. The wound-healing (scratch) assay revealed slower wound closure of cells with the SPRY4-IT1 overexpression in comparison to control (Figure 5(a)). Consistently, cell invasion was considerably decreased in pCMV-SPRY4-IT1-transfected cells compared to mock-transfected cells based on the transwell experiments (Figure 5(b)). These results revealed that SPRY4-IT1 could inhibit ovarian cancer cell migration and invasion.
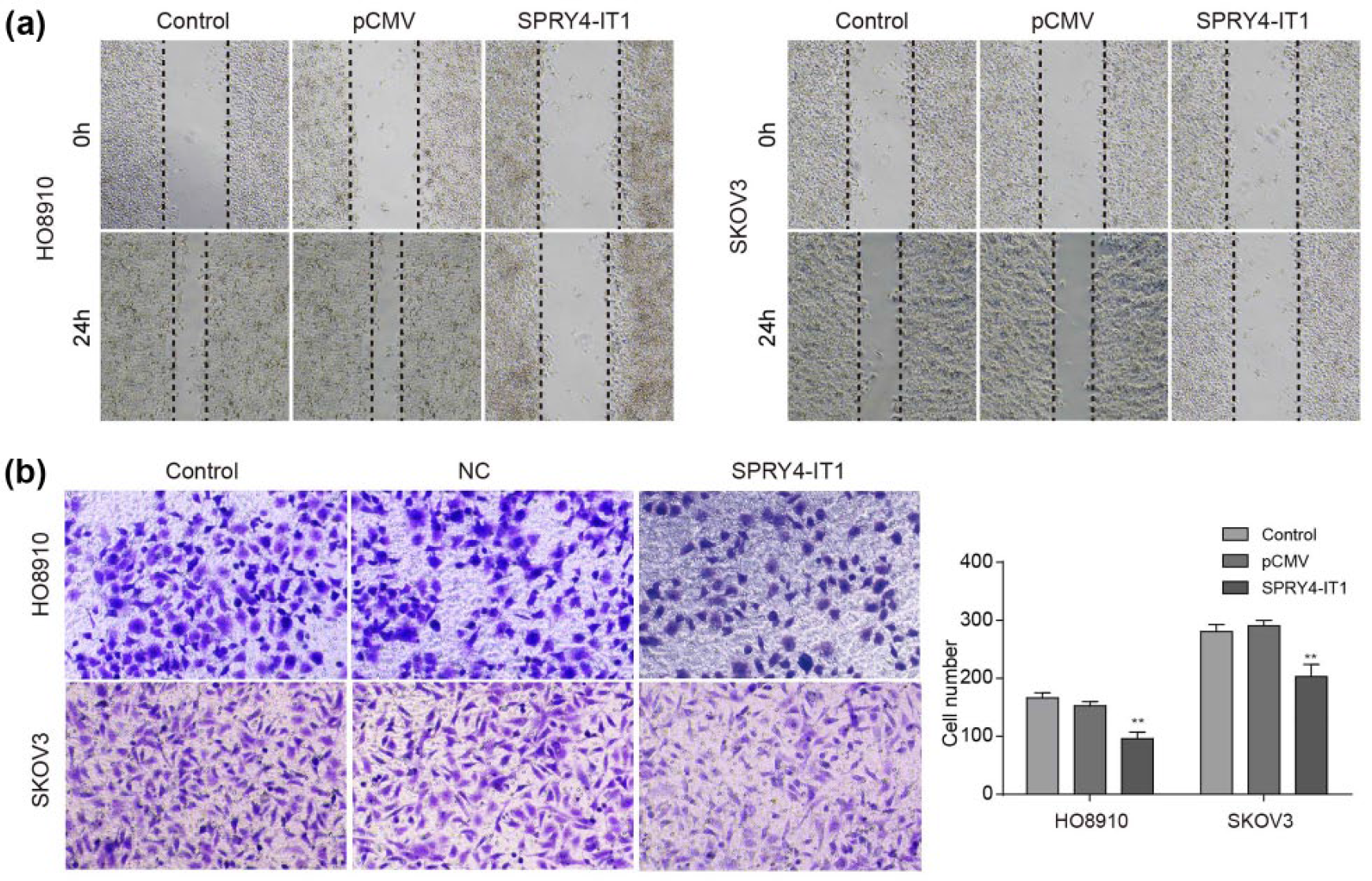
Overexpression of SPRY4-IT1 inhibited migration and invasion of ovarian cancer cells. (a) SPRY4-IT1 reduced the wound-healing (scratch) ability of HO8910 and SKOV3 cells. Images were obtained at 0 and 24 h. (b) Transwell-based cell invasion assay demonstrated the inhibition of invasiveness of HO8910 and SKOV3 cells by SPRY4-IT1 overexpression.
SPRY4-IT1 influenced ovarian cancer cell EMT
EMT plays a key role in cancer cell invasion and metastasis.24,25 Therefore, we examined changes in the expression of EMT-induced markers E-cadherin, vimentin, and N-cadherin following the SPRY4-IT1 overexpression. The qRT-PCR analysis was performed to examine the mRNA level of EMT-related makers in HO8910 and SKOV3 cells. As expected, SPRY4-IT1 remarkably increased the expression of E-cadherin and meanwhile decreased the expression of N-cadherin and vimentin in both the cells (Figure 6(a)). In consistency, the western blot analysis demonstrated that the SPRY4-IT1 overexpression induced an increase of E-cadherin and decrease of N-cadherin and vimentin proteins, indicating that SPRY4-IT1 blocked EMT in ovarian cancer cells (Figure 6(b) and (c)). These findings suggest that SPRY4-IT1 may contribute to ovarian cancer cell metastasis partly via regulating the EMT process.

Expressional changes of EMT markers following SPRY4-IT1 overexpression in ovarian cancer cells. (a) The qRT-PCR analysis of E-cadherin, N-cadherin, and vimentin mRNA expression following SPRY4-IT1 overexpression in HO8910 and SKOV3 cells. (b) Western blot assay of E-cadherin, N-cadherin, and vimentin protein expression after overexpression of SPRY4-IT1 in HO8910 and SKOV3 cells. The β-actin protein was used as an internal control. (c) The relative band intensity of E-cadherin, N-cadherin, and vimentin with reference to β-actin in the western blot assay. Values were presented as mean ± SD (n = 3, *p < 0.05 and **p < 0.01 by Student’s t test).
Discussion
Ovarian cancer has a high occurrence rate as a common malignancy while the early diagnosis for ovarian cancer patients remains ineffective, which makes ovarian cancer a serious public health issue. Recently, accumulating evidences have indicated that certain lncRNAs are aberrantly expressed in human cancers. In ovarian cancer, several lncRNAs including HOST2, 17 LSINCT5, 26 FALEC, 27 PVT1, 28 XIST, 29 and HOTAIR30–32 have been involved in its progression and metastasis. SPRY4-IT1 is transcribed from the intronic region of the SPRY4 gene. Recently, it has been reported that SPRY4-IT1 is upregulated in melanoma cells. The siRNA-mediated knockdown of SPRY4-IT1 has caused cell growth arrest and cell apoptosis. 20 However, the functional roles and regulatory mechanism of SPRY4-IT1 in ovarian cancer remain poorly understood.
In this study, we first analyzed the expression level of SPRY4-IT1 in ovarian cancer patients’ tissue specimens and found that SPRY4-IT1 was significantly downregulated in ovarian cancer tissues compared to the adjacent normal tissues. In consistency, the SPRY4-IT1 expression was lower in the four tested ovarian cancer cell lines relative to the normal ovarian epithelial cells, supporting the reduction of SPRY4-IT1 expression in ovarian cancer. Moreover, based on the in vitro assays using ovarian cancer cells HO8910 and SKOV3, we found that the overexpression of SPRY4-IT1 significantly inhibited the proliferation and colony formation of ovarian cancer cells, changed their cell cycle profiles, and also promoted cell apoptosis. These findings suggest that SPRY4-IT1 plays functional roles in regulating proliferation and apoptosis of ovarian cancer cells, and its downregulation may contribute to ovarian cancer progression through affecting these cellular processes. In previous studies, SPRY4-IT1 has been shown to be downregulated in some cancers including gastric cancer and non–small cell lung cancer21,22 but has also been found with enhanced expression in some other cancers like melanoma, renal cancer, and gastric cancer.18–20 This may indicate that different mechanisms and cellular pathways are underlying the functional roles of SPRY4-IT1 in different cancer types.
Next, we determined the effects of SPRY4-IT1 on ovarian cancer metastasis. The transwell and scratch assays showed that the overexpression of SPRY4-IT1 reduced the migration and invasion of ovarian cancer cells, which was accompanied with morphological changes indicating the inhibition of EMT. In accordance with this phenomenon, overexpression of SPRY4-IT1 enhanced the expression level of E-cadherin and decreased vimentin and N-cadherin protein levels, suggesting that SPRY4-IT1 could affect ovarian cancer cell metastasis partially by regulating the EMT process, an essential process for tumor invasion and metastasis. 25
In summary, our findings have demonstrated that SPRY4-IT1 is downregulated in ovarian cancer and could act as a functional tumor suppressor gene in ovarian cancer cells. Our findings also suggest that SPRY4-IT1 may contribute to ovarian cancer cell metastasis partly via regulating the EMT process. Moreover, SPRY4-IT1 may be considered as a prognostic marker and a novel therapeutic target for ovarian cancer. However, the underlying molecular mechanism through which SPRY4-IT1 is involved in EMT requires further investigations.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
