Abstract
BACKGROUND:
The aim in this study was to explore the role of long non-coding RNA GHET1 in development of non small cell lung cancer (NSCLC).
METHODS:
LncRNA GHET1 expression levels were analyzed by qRT-PCR in tumor tissues and adjacent normal tissues in NSCLC. Measuring the cell proliferation and invasion abilities by CCK8, cell colony formation and transwell invasion assays. Relative protein expression was analyzed by western blot assays.
RESULTS:
Expression of lncRNA GHET1 was notably higher in NSCLC tissues compared with adjacent normal tissues by using qRT-PCR analyses. Higher lncRNA GHET1 expression associated with lymph node metastasis, TNM stage and showed poor outcome in NSCLC patients. Knockdown of lncRNA GHET1 suppressed cell proliferation and invasion capacity and Epithelial-Mesenchymal Transition (EMT) phenomenon of NSCLC cells. Moreover, we demonstrated that knockdown of lncRNA GHET1 suppresses LATS1/YAP pathway signaling pathway by downregulating YAP1 expression in NSCLC cells.
CONCLUSIONS:
GHET1 predicted a poor outcome and acted as a tumor-promoting gene in NSCLC. Thus, inhibition of GHET1 may be a potential target of NSCLC treatment.
Introduction
Lung cancer is the leading cause of cancer- associated mortality worldwide. Lung cancers are divided into two types including non small cell lung cancers (NSCLC) and small cell lung cancers [1]. Non-small cell lung cancer accounts for more than 80% of lung cancer new cases worldwide. In spite of many therapeutic strategies have exhibited larger advances, the 5-year survival rate for NSCLC remains poor [2]. Therefore, to explore potential targets for NSCLC treatment will be urgent.
Recently, increasing evidence has verified that many long non-coding RNAs are involved in the development and biological functions in NSCLC. For example, silencing long non-coding RNA ROR improves sensitivity of non small cell lung cancer to cisplatin resistance by inhibiting PI3K/Akt/mTOR signaling pathway [3]. Downregulation of long non-coding RNA TRPM2-AS promotes cell apoptosis in non-small cell lung cancer [4]. Highly expressed long non-coding RNA FOXD2-AS1 promotes non-small cell lung cancer progression via Wnt/
GHET1, a long non-coding RNA, was found to act as oncogenes in some types of tumors. Long non-coding RNA GHET1 promotes gastric carcinoma cell proliferation by increasing c-Myc mRNA stability [6]. Knockdown of long non-coding RNA GHET1 inhibits cell proliferation and invasion of colorectal cancer [7]. Overexpression of long non-coding RNA GHET1 promotes the development of multidrug resistance in gastric cancer cells [8]. However, the understanding the role of GHET1 in NSCLC is still limited.
In the study, lncRNA GHET1 expression was found to be upregulated in NSCLC tissues compared with adjacent normal tissues. Knockdown of lncRNA GHET1 suppresses cell proliferation and invasion. Moreover, we demonstrated that knockdown of lncRNA GHET1 suppresses LATS1/YAP signaling pathway by downregulating YAP1 expression in NSCLC cells. Thus, these results indicated that inhibition of GHET1 may be a potential target of NSCLC treatment.
Materials and methods
Patient tissue samples
Fifty-two NSCLC tissue and adjacent normal tissue samples were collected between June 2011 and April 2015 during radical surgery at the PLA, Second Military Medical University. Tissue samples were immediately snap-frozen in liquid nitrogen and stored at
Cell line culture
Human NSCLC cell lines A549 (KRAS mut), H1299 (EGFRwtKRASwtALKwt), and (SK-MES-1) (EGFRwtKRASwtALKwt) and a human bronchial epithelial cell line (BEAS-2B) were obtained from American type culture collection (ATCC) (Manassas, VA, USA). The cell lines were cultured in RPMI 1640 (Gibco, Grand Island, NY, USA) with 10% fetal bovine serum (FBS) (HyClone, Camarillo, CA, USA). Cells were cultured in 5% CO
Cell transfection
The siRNA against GHET1 and negative control oligonucleotides (NC group) were synthesized by Shanghai GenePharma Co. Ltd (Shanghai, China). Cells transfection were performed using Lipofecta- mineTM2000 (Invitrogen, Carlsbad, CA, USA) transfection reagent according to the manufacturer’s instructions. The siRNA sequences were si-GHET1-1: 5’-CGGCAGGCATTAGAGATGAACAGCA-3’.
Quantitative reverse transcription-polymerase chain reaction (QRT-PCR)
Total RNA from NSCLC tissues and cells was iso- lated by using TRIzol
Cell proliferation assay
Cell proliferation rate was evaluated by CCK8 cell proliferation kit (KeyGEN biotech, Nanjing, China). Briefly, 2000 cells/well were seeded in 96-well plates and cultured overnight. Cells were transfected with si-GHET1 or si-NC and cells were detected at 1, 2, 3 and 4 days. Added 10
Colony formation assay
For colony formation assay, transfected 200 cells/ well were seeded in 6-well plates and were cultured for 14 days in RPMI-1640 medium containing 10% FBS. Cells were fixed with 4% paraformaldehyde and stained with 1% crystal violet for 20 min, and colonies were measured.
Cell invasion assay
Transwell invasion assay was performed by a 24-well chamber by a polycarbonate membrane (8
Expression of GHET1 is upregulated in NSCLC tissues and cells. (A) Expression of GHET1 was analyzed by qRT-PCR in 52 NSCLC tissues and adjacent normal tissues. (B) Expression of GHET1 was analyzed by qRT-PCR in three human NSCLC cell lines (A549, H1299, and SK-MES-1) and a human bronchial epithelial cell line (BEAS-2B). (C) Survival time was showed in higher GHET1 expression group when compared with lower GHET1 expression group in NSCLC patients. 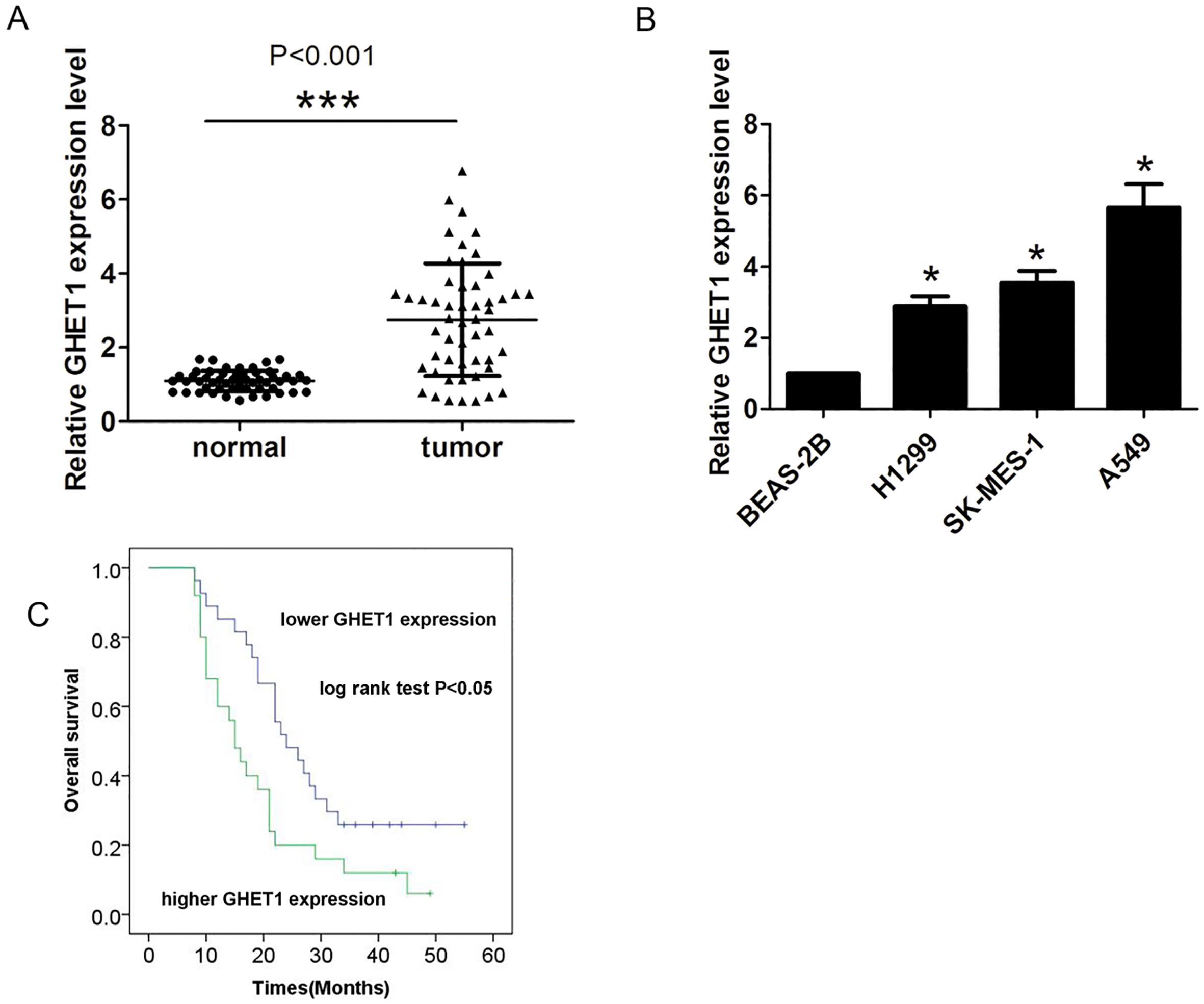
Total cell were lysed with radioimmunoprecipitation buffer (Beyotime Institute of Biotechnology, Nantong, China). The protein concentrations were detected using bicinchoninic acid assay (BCA, Beyotime Institute of Biotechnology, Nantong, China). The proteins were separated by SDS-PAGE and transferred onto nitrocellulose membranes. Then, the membranes were incubation with antibodies specific for Twist1 (Santa Cruz Biotechnology, CA, USA), E-cadherin (Cell Signaling Technology, Boston, MA, USA), N-cadherin (Cell Signaling Technology, Boston, MA, USA), LATS1 (Santa Cruz Biotechnology, CA, USA), YAP1 (Cell Signaling Technology, Boston, MA, USA), and GAPDH (Santa Cruz Biotechnology, CA, USA). The membranes were incubated with corresponding horseradish peroxidase-conjugated secondary antibody (Santa Cruz Biotechnology, CA, USA) and visualized by enhanced chemiluminescence (Pierce; Thermo Fisher Scientific, Inc. Carlsbad, CA, USA) on a FluorChem imaging system.
Correlation of GHET1 expression with clinicopathological factors
Correlation of GHET1 expression with clinicopathological factors
All experiments were independently performed at least three times and all statistical analysis was analyzed using the SPSS 20.0 (SPSS, Inc., Chicago, IL, USA). Results were expressed using means
Multivariate Cox proportional hazards analysis of GHET1 expression and overall survival for NSCLC patients
Multivariate Cox proportional hazards analysis of GHET1 expression and overall survival for NSCLC patients
Expression of GHET1 in NSCLC tissues and cells
Increased GHET1 expression promotes cell proliferation and invasion. (A)–(B) Expression of GHET1 was analyzed by qRT-PCR after introducing si-GHET1 or si-NC into A549 and SK-MES-1 cells. (B)–(C) Cell colony number was evaluated by introducing si-GHET1 or si-NC into A549 and SK-MES-1 cells. (D)–(E) Cell proliferation was evaluated by CCK8 assays after introducing si-GHET1 or si-NC into A549 and SK-MES-1 cells. 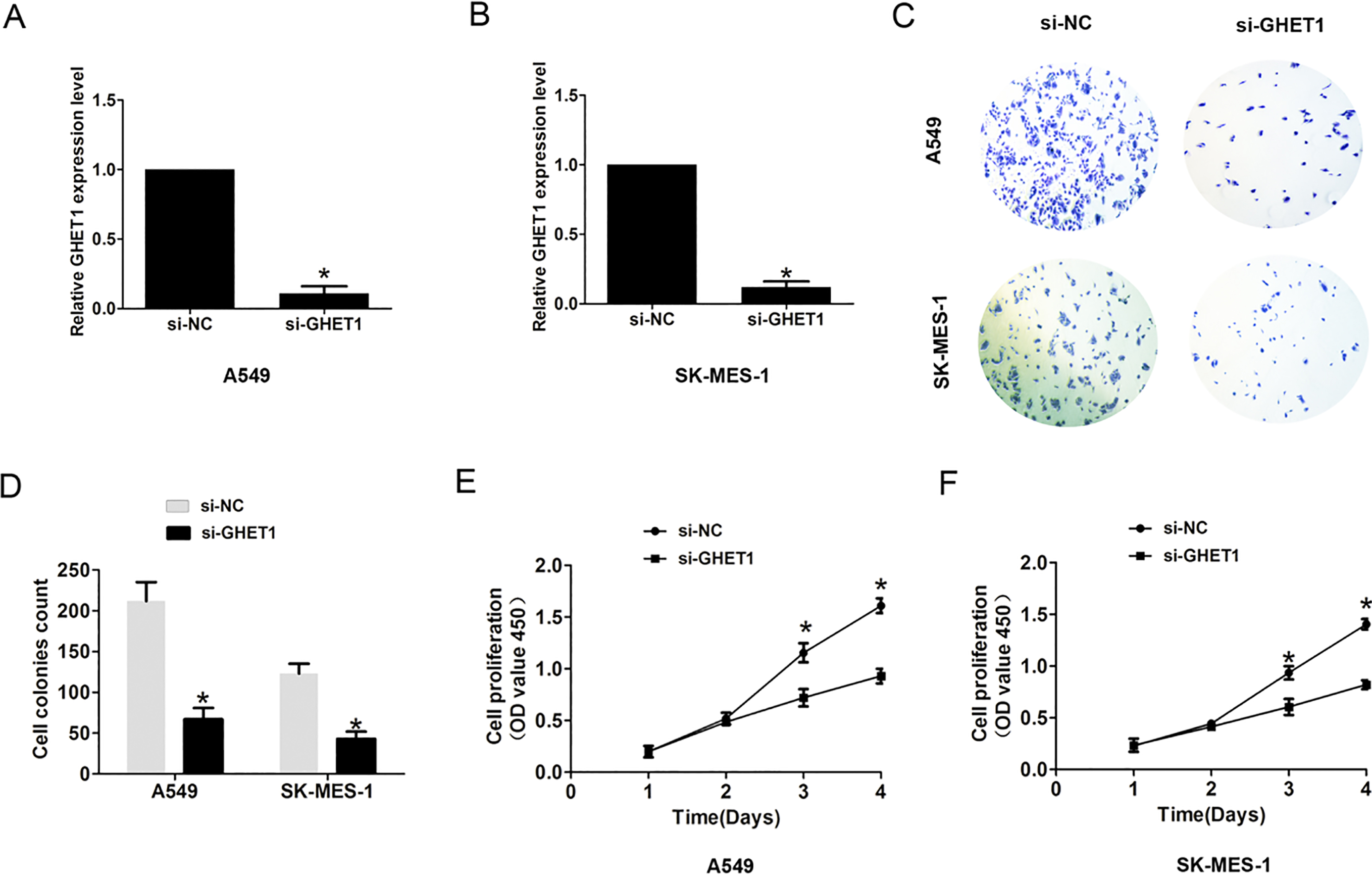
Reduced GHET1 expression suppressed cell EMT process. (A)–(B) Cell invasion number was evaluated by transwell assays after introducing si-GHET1 or si-NC into A549 and SK-MES-1 cells. (C)–(D) The protein expression of Twist1, E-cadherin and N-cadherin was evaluated by western blot assays after introducing si-GHET1 or si-NC into A549 and SK-MES-1 cells. 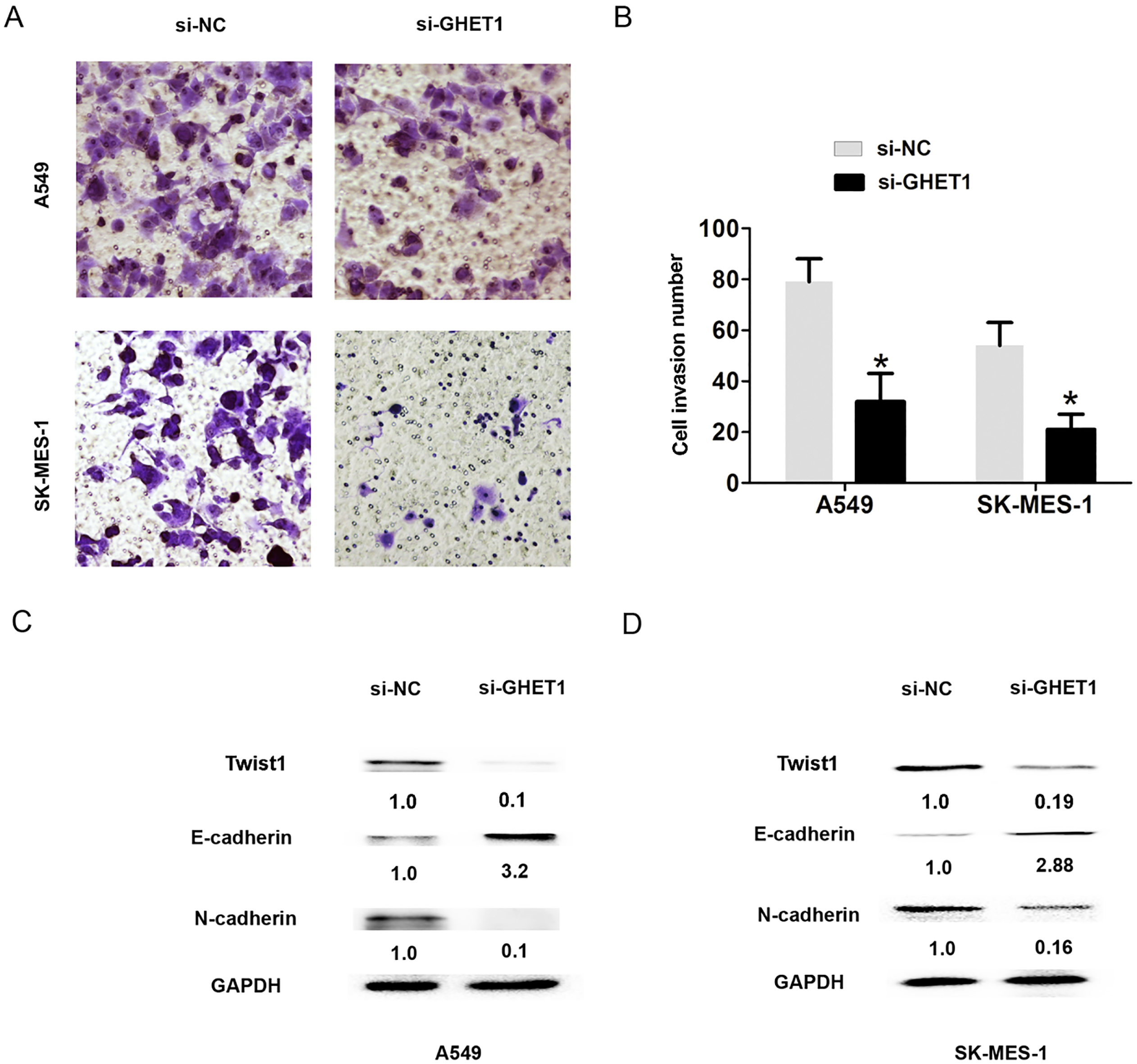
Reduced GHET1 expression suppressed LATS1/YAP signaling pathway. (A) The protein expression of LATS1 and YAP1 was evaluated by western blot assays after introducing si-GHET1 or si-NC into A549 cells. (B) The protein expression of LATS1 and YAP1 was evaluated by western blot assays after introducing si-GHET1 or si-NC into SK-MES-1 cells. 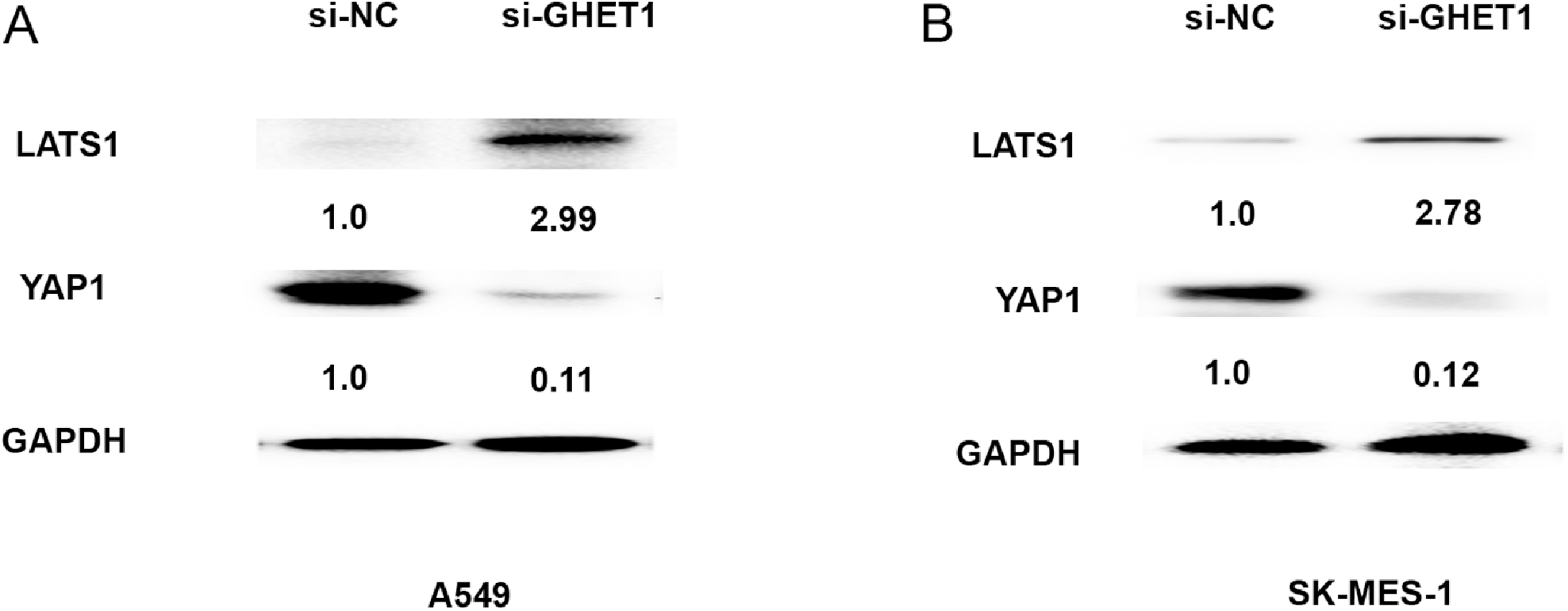
To explore the role of GHET1 in NSCLC development and progression, we first detected the expression of GHET1 in NSCLC tissues compared with adjacent normal tissues. The results showed that GHET1 expression was notably increased in NSCLC tissues compared with adjacent normal tissues (Fig. 1A,
To identify whether GHET1 affected NSCLC cell biology behaviors, CCK8 assay, cell colony and cell invasion assay were performed. We used a siRNA targeting GHET1 to knockdown it in A549 and SK-MES-1 cells and observed that the GHET1 expression was effectively reduced in the si-GHET1 group when compared to the si-NC group (Fig. 2A and B,
Effects of GHET1 expression on cell invasion and EMT process in NSCLC
Moreover, cell invasion assay was evaluated by transwell invasion. We found that cell invasion number was reduced in the si-GHET1 group when compared to the si-NC groups (Fig. 3A and B,
Effects of GHET1 expression on LATS1/YAP signaling pathway in NSCLC
Hippo signaling pathway was found to be involved in NSCLC progression [9]. We measured the effects of GHET1 expression on Hippo cores protein LATS1 and YAP1 by western blot analyses. The results demonstrated that the protein expression levels of YAP1 were downregulated, however, LATS1 expression was upregulated After A549 and SK-MES-1 transfected with si-GHET1 at 48h (Fig. 4A and B). Thus, these results showed that GHET1 expression in NSCLC cells was related to LATS1/YAP signaling pathway.
Discussion
Recent evidence has suggested that long non-coding RNA functions as regulators of tumor suppressive and oncogenic functions [10]. In NSCLC development and progression, some lncRNAs were found to play crucial role to affect cell proliferation, cell cycle, cell apoptosis and cell invasion [11, 12]. For example, higher CRNDE expression strongly promoted NSCLC cell proliferation and growth through activating PI3K/AKT signaling [13]. Long intergenic noncoding RNA 00673 promotes non-small-cell lung cancer metastasis by binding with EZH2 and causing epigenetic silencing of HOXA5 [14]. Long non-coding RNA TCF7 promotes invasiveness and self-renewal of human non-small cell lung cancer cells [15]. In the study, Increased GHET1 expression was observed in NSCLC tissues when compared with adjacent normal tissues and associated with TNM stage, lymph node metastasis and poor outcome in NSCCL patients. GHET1 was identified as independent prognostic maker of NSCLC. Moreover, we demonstrated that inhibition of GHET1 could reduce the cell proliferation rate and cell invasion ability.
EMT process plays a critical step in tumor metastasis. Established hallmark of EMT process causes the loss of epithelial maker E-cadherin expression, and enhances expression of mesenchymal makers, such as N-cadherin and vimentin [16]. LncRNAs were demonstrated to be involved in NSCLC cell EMT process. Downregulation of BRAF activated non-coding RNA is associated with poor prognosis for non-small cell lung cancer and promotes metastasis by affecting epithelial-mesenchymal transition [17]. Downregulation of long noncoding RNA FOXF1-AS1 regulates epithelial-mesenchymal transition, stemness and metastasis of non-small cell lung cancer cells [18]. In the study, we found that reduced GHET1 expression affects cell EMT process through upregulating E-cadherin expression and downregulated the Twist1 and N-cadherin expression in NSCLC cells.
Aberrant activation of Hippo pathway was reported to be involved in NSCLC cell invasion and EMT process. RASSF1A suppresses cell invasion and meta static potential of by inhibiting YAP activation through the GEF-H1/RhoB pathway in NSCLC [9]. NCTD-mediated inhibition of YAP suppressed epithelial-to-mesenchymal transition (EMT) and downregulated the motile and invasive cellular phenotype in vitro via enhancing E-cadherin and decreasing fibronectin/ vimentin [19]. Our results showed that reduced GHET1 expression was related to inhibition of LATS1/YAP pathway by upregulated the LAST1 and downregulated the YAP1 expression.
In conclusion, our data showed that GHET1 were significantly upregulated in NSCLC tissues and cells. Furthermore, inhibition of GHET suppressed cell proliferation, invasion and EMT process. Moreover, we showed that LATS1/YAP pathway was inhibited by downregulating GHET1 in NSCLC cells. Thus, these results indicated that GHET1 may serve as a potential target of NSCLC treatment. In the further, we hope to investigate the underlying mechanism between GHET1 and LATS1/YAP pathway in NSCLC.
Footnotes
Conflict of interest
The authors declare that they have no competing interests.
