Abstract
BACKGROUND:
It is well known that some circulating microRNAs (miRNAs) are highly stable and might serve as promising biomarkers for many types of human cancer including glioblastoma (GBM). However, the potential clinical significance of serum miR-100 in GBM remained unknown.
OBJECTIVE:
We aimed to detect the expression level of serum miR-100 in patients with GBM and assess its potential diagnostic and prognostic value.
METHODS:
Quantitative real-time PCR was performed to measure serum miR-100 levels in 95 GBM patients and 60 healthy volunteers. The association between serum miR-100 level and clinicopathological parameters as well survival of GBM patients was evaluated.
RESULTS:
Our results revealed that serum miR-100 levels were significantly decreased in GBM patients compared with the healthy controls. Additionally, miR-100 levels were significantly elevated after treatment. Low miR-100 expression was closely correlated with worse clinicopathological characteristics. Further receiver operating characteristic (ROC) curve analysis showed that serum miR-100 could effectively discriminate GBM cases from normal controls. Moreover, survival analyses revealed that patients with high serum miR-100 levels had significantly longer survival time than those with low serum miR-100 levels. Finally, multivariate analysis identified serum miR-100 as an independent prognostic indicator for GBM.
CONCLUSIONS:
Our findings suggested that serum miR-100 might serve as promising biomarker for GBM diagnosis and prognosis.
Introduction
Glioblastoma (GBM) is the most aggressive type of glioma, and the most common brain malignant tumor in both adults and young patients [1]. Despite advances in surgical excision, radiotherapy and chemotherapy, the prognosis of GBM patients remains very unfavorable, with a median overall survival of 15 months or less [2, 3, 4]. To improve the clinical outcome of GBM patients, it is urgent to identify novel biomarkers for diagnosis and prognosis of this malignancy.
MicroRNAs (miRNAs) are a class of small endogenous noncoding RNA molecules that are 18–25 nucleotides in length [5, 6]. Growing evidence have showed that miRNAs participate in a variety of biologic processes including cell growth, migration, invasion and apoptosis [7, 8]. Recently, more and more studies have demonstrated that some circulating miRNAs in serum, plasma or cerebrospinal fluid (CSF) are differentially expressed in GBM patients, and serve as biomarkers for diagnosis and prognosis in GBM. For example, loss of miR-106a [9], miR-485-3p [10], miR-328 [11], miR-137 [12] were strongly correlated with poor prognosis of GBM, while miR-196b [13] and miR-15b [14] were significantly upregulated in GBM patients.
MiR-100 has been reported to be aberrantly dysregulated in GBM. Luan et al. revealed miR-100 was significantly downregulated in both GBM cell lines and tissue. Overexpression of miR-100 resulted in decreased cell growth, migration, and increasing chemosensitivity to cisplatin in vitro and in vivo [15]. Moreover, miR-100 upregulation and subsequent elevated expression of SMRT/NCOR2 reduced cell proliferation and stimulated cell apoptosis in vitro. In vivo evidence showed re-expression of miR-100 greatly inhibited GBM tumorigenicity and improved animal survival [16]. These data suggested that miR-100 was a powerful tumor suppressor in GBM. However, the potential clinical significance of serum miR-100 was unknown. The present study aimed to detect serum miR-100 expression levels in GBM patients and analyze its association with clinical variables as well as survival.
Materials and methods
Ethics statement
This study was approved by the Ethics Committee of Xianyang Hospital of Yan’an University (Xianyang, China), and written informed consent was provided by each participant. All specimens were handled and made anonymous according to the ethical and legal standards.
Correlation of serum miR-100 expression with clinicopathological characteristics of 95 GBM cases
Correlation of serum miR-100 expression with clinicopathological characteristics of 95 GBM cases
MGMT, O6-methylguanine-DNA methyltransferase; IDH1/2, isocitrate dehydrogenase; KPS, karnofsky performance scale; NS, not significant.
Blood samples were obtained from 95 patients with diagnosed GBM before surgery, 2 weeks after surgery and 30 days after radiotherapy combined with chemotherapy, respectively. None of the patients had received any treatment prior to blood sampling. All the GBM patients were confirmed and classified based on 2016 WHO classification of tumours of the central nervous system. Detailed characteristics of all patients were listed in Table 1. The average age of GBM patients was 58.6 y (36.2 y
RNA extraction and real-time PCR (qRT-PCR)
Total RNA was isolated from 250
Primer sequences used in real-time PCR analysis of miR-100
Primer sequences used in real-time PCR analysis of miR-100
(a) Serum miR-100 levels were significantly lower in GBM patients in high combined risk factors (CRF) group. (b) A significant increase in serum miR-100 levels was noted in the blood samples collected after treatment.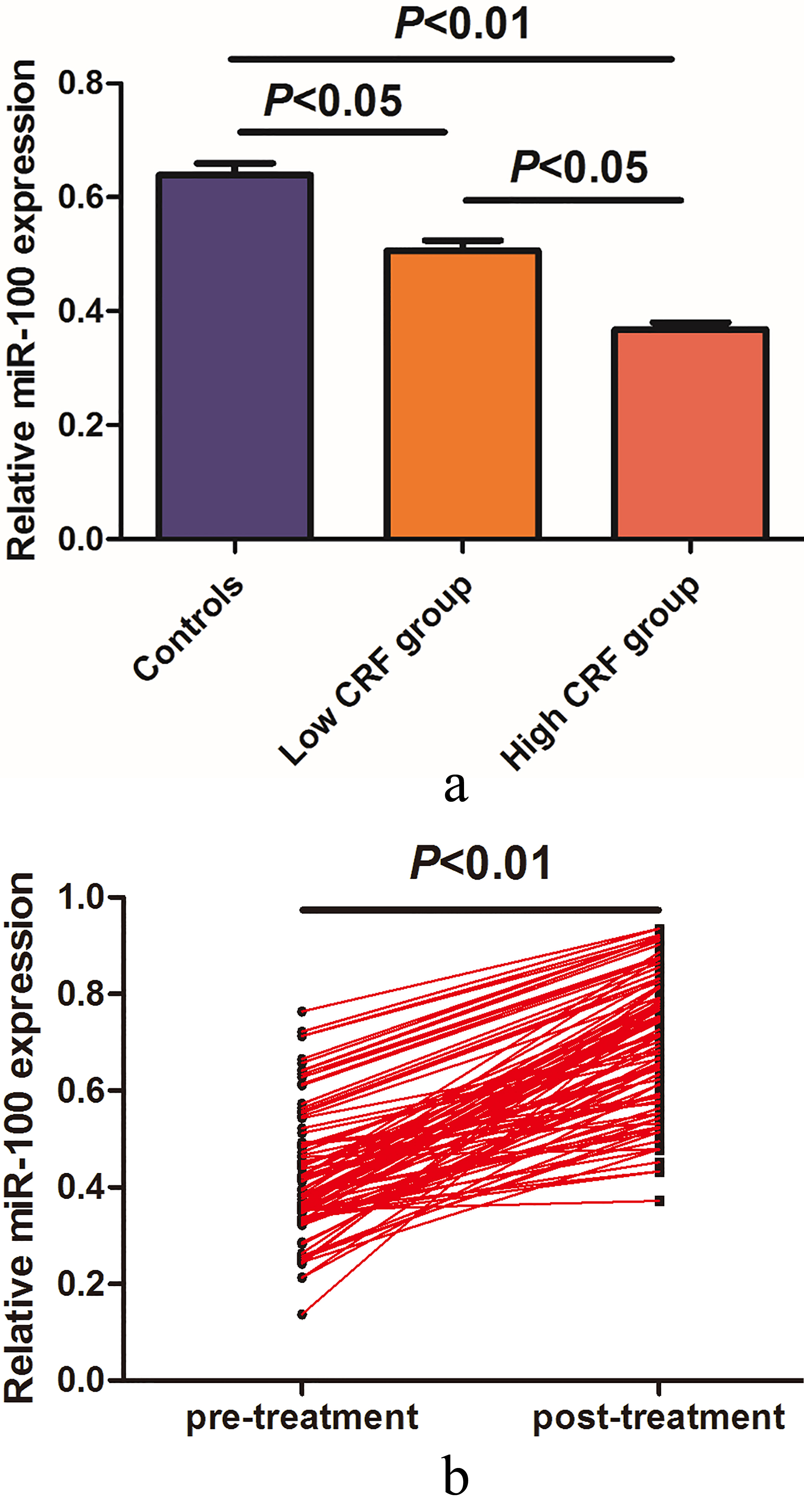
Statistical analyses were conducted using GraphPad Prism version 4 (GraphPad Software, San Diego, CA, USA). Statistical significance was defined as
Results
Down-regulation of miR-100 in GBM patients and the alterations of miR-100 levels
The serum miR-100 levels in all participants were detected by qRT-PCR. As shown in Fig. 1a, the relative serum miR-100 levels were significantly lower in GBM patients from the high CRF group compared with those in the low CRF group (
Diagnostic accuracy of miR-100 in GBM patients
ROC curve analysis demonstrated that serum miR-100 expression was a promising indicator for identifying GBM cases from normal controls, with the area under the ROC curve (AUC) of 0.839. In addition, the specificity and sensitivity were 83.33% and 77.89%, respectively (Fig. 2).
Multivariate analysis of prognostic factors for OS and PFS in GBM patients
Multivariate analysis of prognostic factors for OS and PFS in GBM patients
CI, confidence interval; RR, risk ratio.
The diagnostic value of serum miR-100.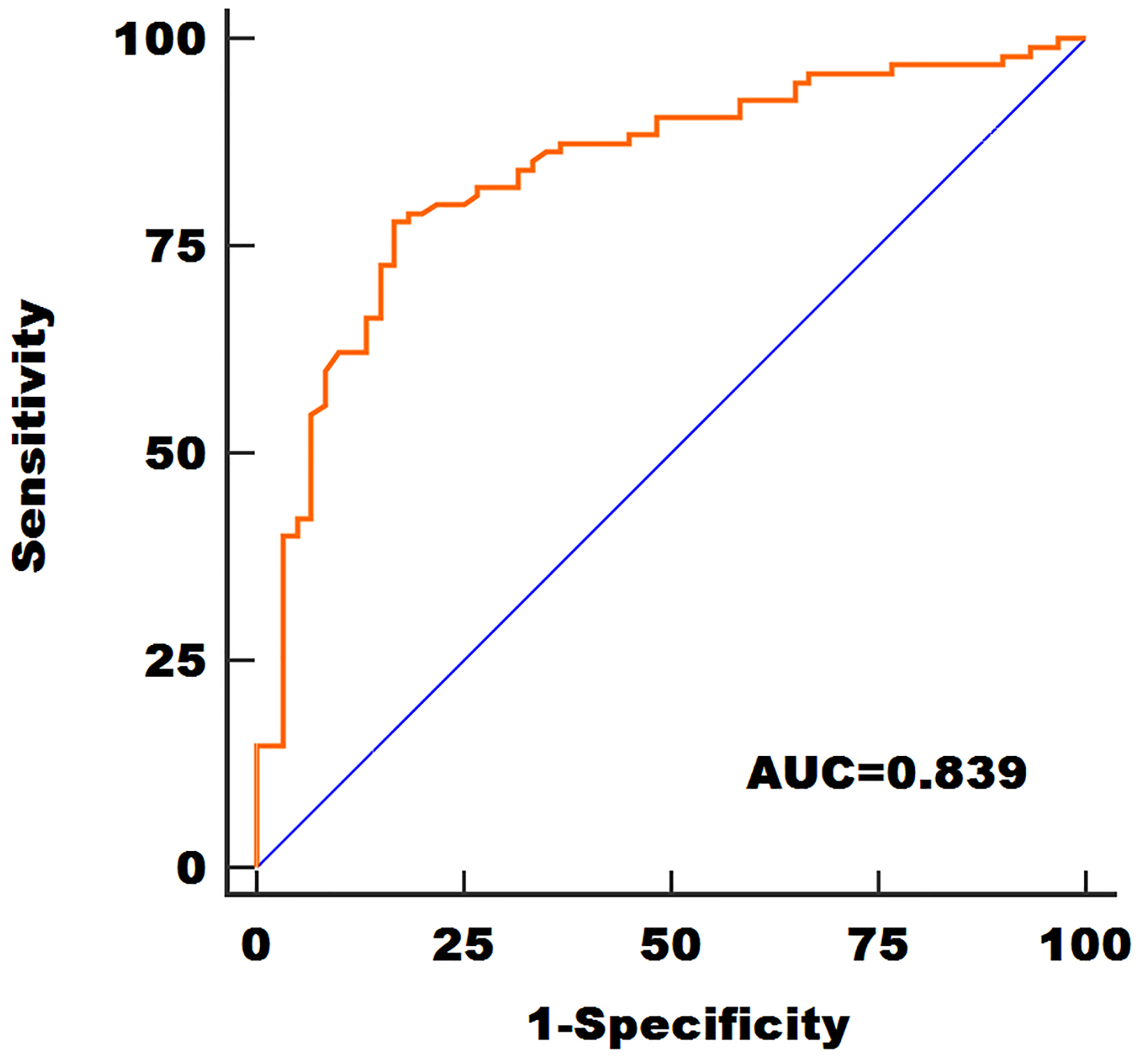
(a) Kaplan-Meier plot of overall survival for patients with GBM. (b) Kaplan-Meier plot of progression-free survival for patients with GBM.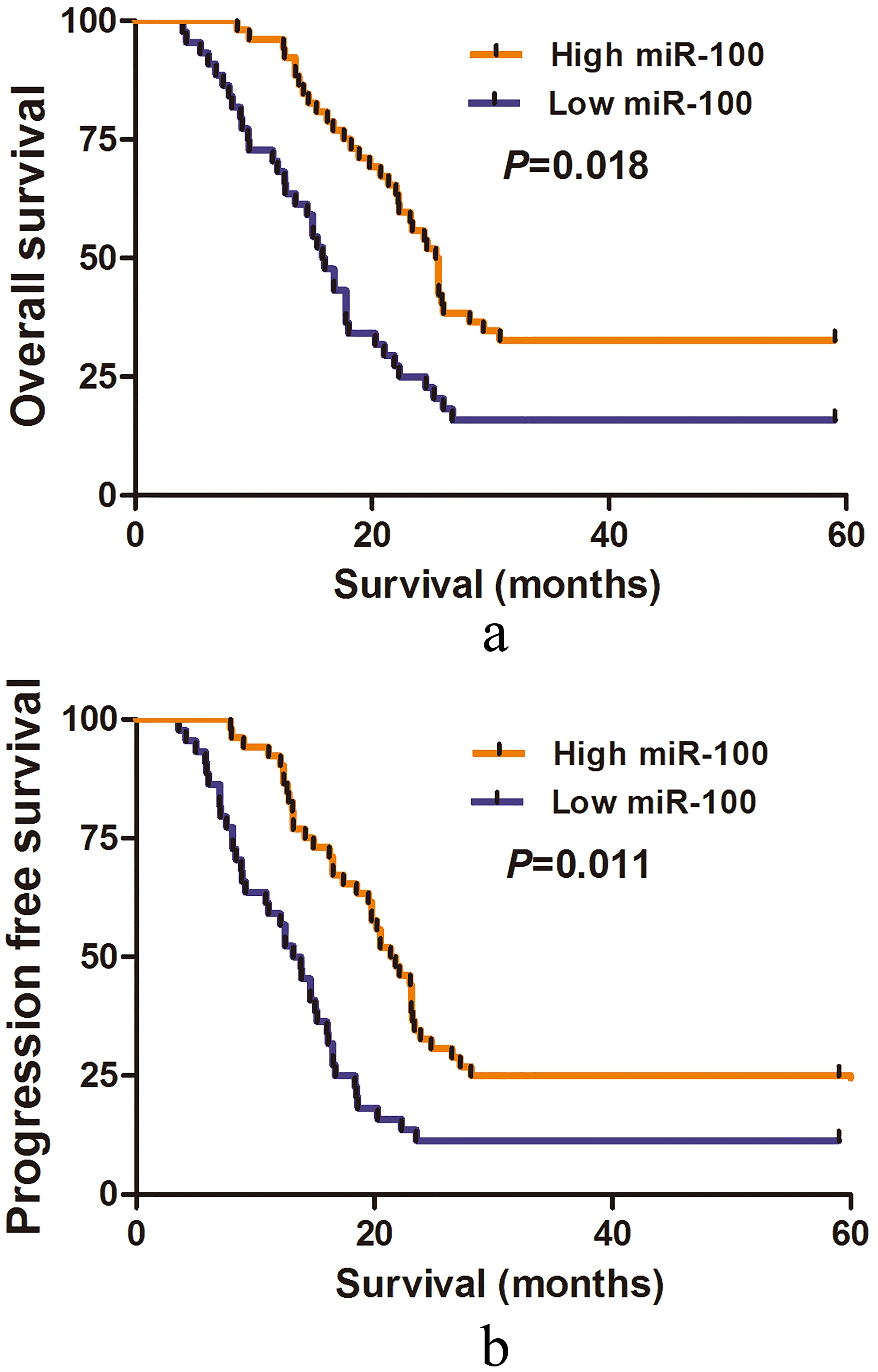
Next, whether serum miR-100 expression correlated with clinical characteristics of GBM patients was investigated. All 95 GBM subjects were divided into two groups based on median expression levels of miR-100. As presented in Table 1, low serum miR-100 expression was significantly correlated with Karnofsky Performance Scale (KPS) (
Association between miR-100 expression and prognosis in GBM patients
The association between serum miR-100 expression and OS/PFS in patients with GBM was evaluated by Kaplan-Meier method. GBM patients with high serum miR-100 had significantly longer OS (
Multivariate analysis was used to explore the impact of clinical parameters on OS and PFS. In GBM patients, low serum miR-100 expression was strongly correlated with worse OS (RR
Discussion
In the current study, we have demonstrated that serum miR-100 expression levels were dramatically downregulated in GBM compared to the healthy controls. Furthermore, serum miR-100 levels were significantly increased following treatment, indicating that serum miR-100 levels might be sensitive to treatment response. ROC analysis revealed serum miR-100 had a good performance to differentiate GBM patients from healthy controls. In addition, low serum miR-100 was closely correlated with shorter survival, and serum miR-100 was identified as an independent prognostic biomarker for GBM patients. In consistent with previous reports, miR-100 exhibited tumor suppressive properties in GBM [15, 16]. To the best of our knowledge, our study demonstrated for the first time that serum miR-100 might serve as a diagnostic and prognostic prediction biomarker for GBM. There are two possible mechanisms accounting for the deceased miR-100 in the serum. First, miR-100 might be highly methylated in the GBM, thus the level of miR-100 is significantly downregulated in GBM cells, resulting less miR-100 is secreted into the circulation system. In addition, exosome is very important for the substance transmission between cancer cells and cancer cells, or cancer cells and other cell types in the tumor microenvironment. It is possible that less miR-100 is packaged into the exosomes, leading to the downregulation of miR-100 in the serum. MiR-100 functions as a tumor suppressor in GBM and serum miR-100 might be a good indicator of the miR-100 levels in the cancer cells. Therefore, it is reasonable to observe that decreased serum miR-100 level is associated with unfavorable clinical outcome in GBM.
It seems that downregulation of miR-100 expression was also a frequent event in multiple cancer types. In bladder cancer, the expression level of miR-100 was remarkably lower in cancerous tissues compared with normal adjacent tissues, and miR-100 downregulation was strongly associated with tumor stage, tumor grade, regional lymph node involvement and shorter survival [17, 18]. Moreover, Xu and colleagues reported miR-100 was frequently underexpressed in bladder cancer tissues. Upregulation of miR-100 significantly decreased cell proliferation, motility and stimulated cell-cycle arrest in vitro, as well as suppressed the tumor growth in vivo by targeting mTOR [19]. Chen et al. found miR-100 expression was decreased in BC tissues compared with normal specimens. MiR-182/miR-100 ratio showed good performance for discriminating BC patients from controls and was confirmed to be a prognostic factor [20]. In hepatocellular carcinoma (HCC), an under-expression of miR-100 was found in cancerous tissues and ectopic expression of miR-100 greatly restrained cell growth in HCC mouse xenograft models. In addition, both mTOR and IGF-1R were demonstrated to be its downstream targets [21]. Zhou et al. found that loss of miR-100 in HCC tissues was closely correlated with aggressive clinical variables. MiR-100 upregulation significantly inhibited carcinogenesis in vitro and suppressed HCC cell metastasis in vivo [22]. Also, Liu and colleagues revealed that miR-100 levels were decreased in non-small cell lung cancer tissues, and low miR-100 levels predicted shorter survival of patients. MiR-100 mimics significantly reduced cancer cell growth, stimulated cell apoptosis and G2/M cell cycle arrest via regulating polo-like kinase 1 [23]. In oral squamous cell carcinoma, decreased miR-100 expression was observed both in tumor and cell lines, miR-100 downregulation resulted in a decreased cell proliferation [24]. In epithelial ovarian cancer, Azizmohammadi and colleagues reported the level of miR-100 was relatively lower in cancerous tissues compared with non-cancerous specimens, and reduction of miR-100 predicted worse prognosis [25]. Moreover, Sun et al. showed miR-100 expression was significantly lower in nasopharyngeal carcinoma tissues than that in healthy tissues, and miR-100 upregulation markedly decreased the oncogenic activities of cancer cell lines by regulating IGF1R [26]. Likewise, a significant reduction in miR-100 expression was observed in tissues of gastric cancer (GC), and the deregulated miR-100 was correlated with shorter survival and aggressive clinical parameters. Furthermore, in vitro analysis revealed enforced miR-100 expression or suppression of CXCR7 dramatically attenuated GC cell proliferation [27].
In addition, the oncogenic property of miR-100 in acute myeloid leukemia (AML) was reported by some studies. MiR-100 exhibited elevated levels in AML, and in vitro and in vivo analysis revealed high miR-100 expression contributed to invasive nature of AML cells. RBSP3 was confirmed to be the target gene [28]. Likewise, Bai et al. found miR-100 expression was highly expressed in pediatric AML patients than in controls. Its expression was closely associated with poor phenotypes and worse survival of patients [29]. More importantly, the role of miR-100 in cancer development and progression remained controversial in some cancer types. In osteosarcoma, significantly lower miR-100 levels were found in cancerous specimens, and loss of miR-100 promoted U2OS cell growth and increased cell survival against Doxorubicin by inversely regulating ZNRF2 [30]. However, Kubota et al. demonstrated elevated miR-100 expression in MNNG/HOS, 143B, MG63 cell lines promoted tumorigenicity and resistance to chemotherapeutic drugs [31]. Similarly, in breast cancer (BC), ectopic expression of miR-100 suppressed the oncogenic activities of BC cells and enhanced the sensitivity of cancer cells to cisplatin treatment [32, 33, 34]. Conversely, miR-100 levels were specifically increased in SK-BR-3 BC cell lines. In addition, miR-100 silencing induced apoptosis in SK-BR-3 cells and inhibited tumorigenicity in vitro and in vivo through the MTMR3-p27 pathway [35]. These findings indicated that the role of miR-100 in tumorigenesis might be associated with tumor type and tumor microenvironment.
Currently, deregulated serum miR-100 has been associated with esophageal squamous cell carcinoma, chronic obstructive pulmonary disease and hepatocellular carcinoma [36, 37, 38]. Single biomarker is not enough to accurately detect and predict the prognosis of GBM. Combination of several serum biomarkers and molecular events (IDH1/2 mutation status, MGMT methylation etc.) might contribute to avoid the false positive and negative findings.
Conclusions
Taken together, our data provided compelling evidence that serum miR-100 might serve as a promising biomarker for the diagnosis and prognosis prediction of GBM. Further studies with larger cohort are warranted to confirm our findings.
Footnotes
Acknowledgments
This study was supported by the Grants from Bureau of Science and Technology of Xianyang City (No: 2017k02-91) and Yan’an University (No: YDY2017-12).
Conflict of interest
We deny any conflict of interest.
