Abstract
OBJECTIVE:
Guanylate binding protein-1 (GBP1) is highly associated with cell proliferation, and can modulate growth and invasiveness of gliomas. The relationship between GBP1 expression and the prognosis of glioma patients is further evaluated for the purpose of investigating whether GBP1 can serve as an predictor for evaluating prognosis of glioma patients.
METHODS:
GBP1 expression in 528 glioblastoma multiforme (GBM) patients of The Cancer Genome Atlas (TCGA) database were investigated, then 103 surgical specimens from glioma patients in our center were further evaluated. The effect of GBP1 on proliferation, invasion and migration of glioma cells in vitro was analyzed, and the effects of GBP1 on sensitivity of radiotherapy and chemotherapy on glioma cells in vitro were also analyzed. GBP1 associated signaling pathways were identified with Gene Set Enrichment Analysis (GSEA). Besides, the effect of GBP1 expression on proliferation of glioma cells in vivo was analyzed.
RESULTS:
In both TCGA database and our clinical data, GBM tissues exhibited increased mRNA expression of GBP1 gene, its expression level was co-related to PETN deletion and EGFR amplification, and was associated with prognosis of GBM patients. GBP1 overexpression can enhance migration and invasion ability of tumor cells in vitro, and in vivo studies showed that GBP1 can promote tumor proliferation, decrease survival in tumor-bearing mice. GSEA analysis predicted that GBP1 may play its biological roles via toll-like receptor pathway.
CONCLUSION:
This study provides new insights and evidences that high level expression of GBP1 is significantly correlated with progression and prognosis in GBMs. Furthermore, transfection of GBP1 revealed its regulation on migration and invasiveness of glioma cells, decreasing sensitivity of chemotherapeutic agent, shortening survival of tumor-bearing animals. These data demonstrate that GBP1 may serve as a novel prognostic biomarker and a potential therapeutic target for gliomas.
Introduction
Primary glioblastoma multiforme (GBM) is the worst brain malignancy, which characterized for high proliferation, broad invasiveness, complex tumor vasculature, strong resistance to current therapies [1, 2]. Although more and more advancements against GBMs have been made in surgery, radiochemotherapy, immunotherapy, and multiple molecular targeting therapy, poor prognosis still remains characteristic of GBMs and results in a median patient survival of no more than 14 month [3, 4]. Although many molecular marks have been reported to be closely related with fundamental characters of GBMs, however, much more genetic alterations need to be further identified to elucidate the key molecules and their relevant pathways, for the purpose of elucidating the key pathways controlling GBM cell invasion, affecting its growth and angiogenesis and searching therapies that improve patient survival. In the era of precision medicine, more efforts need to be made to elucidate various and sophisticated molecular characteristics in order to obtain the core genetics alterations of GBMs and the important molecular predictors relevant with both therapeutic effects and overall survival [5, 6]. In addition, various available database have collected abundant molecular information of GBMs, which offer great convenience for us to correlate levels of certain gene expression with molecular diagnosis, target therapy, and prognosis evaluation. These most commonly used public database includes the Cancer Genome Atlas (TCGA) network and the Repository of Molecular Brain Neoplasia Data (REMBRANDT) database [7, 8]. Molecular subtyping of GBM in TCGA database revealed four different molecular subtypes of GBM patients: i.e., neural, mesenchymal, classical, and proneural [9]. Thus, further searching the core biomarkers and genetic alterations responsible for treatments and prognosis in GBMs, and pursuing the correlation among their expression level, molecular structural changes and genetic subtypes of GBMs will help to screen out new therapeutic targets and improve clinical prognosis for patients suffering from malignant gliomas.
Guanylate-binding protein 1 (GBP1) is a guanosine-5’-triphosphate (GTP)-binding protein with a strong GTPase activity that hydrolyzes GTP specifically, mainly producing GMP [10, 11]. GBP1 can be induced by interferon-gamma (IFN-
In the current study, we investigated GBP1 expression levels in human surgical glioma specimens using a tissue microarray and evaluated its prognostic value, then validated the differential expression and prognostic significance of GBP1 in two other independent datasets: namely, TCGA cohort and the REMBRANDT cohort. For TCGA cohort, we additionally analyzed the expression profile of GBP1 according to subtypes and genetic alterations of GBM, and performed different assays to further explore the biological functions of GBP1 in GBM cells.
Materials and methods
Cell culture
Human GBM cell line U251 (ATCC), and SHG44 [21] were cultured in Dulbecco’s modified Eagle’s medium (DMEM; Life Technologies) supplemented with 10% fetal bovine serum (FBS; Life Technologies) and penicillin (100 units/mL)/streptomycin (100
Patients and tissue samples
For our single center glioma patients in this study, 103 tumor specimens were obtained at the time of surgery at the Department of Neurosurgery, the Second Affiliated Hospital of Soochow University between January 2009 and December 2016, and brain tissues of 10 non-tumor samples were-taken from surgical decompression with informed consent. Clinicopathological data (age, gender, clinical manifestations, and extent of resection) were obtained from patients’ medical records. Tumor histology was confirmed independently by two neuropathologists. The study protocol and acquisition of tissue specimens were approved by the Specialty Committee on Ethics of Biomedical Research of the Second Affiliated Hospital of Soochow University (Suzhou, China).
Stable transfection of GBP1
Transfections were performed using LipofectAmine Plus reagents (Life Technologies). Approximately 5
Western blot
Cells were lysed in the radioimmunoprecipitation assay (RIPA) buffer containing 1
Real-time polymerase chain reaction (PCR)
Snap frozen tissues from ten human high grade (grade III–IV), ten low grade (grade I–II) glioma patients and ten non-tumor samples were selected randomly for total RNA extraction with Trizol reagent (Invitrogen) and reverse transcribed (SuperScript II First Strand kit; Invitrogen) according to the manufacturer’s instructions. Real-time PCR of GBP1 was performed using the Bio-Rad CFX96 Touch Real-Time PCR Detection system with human GBP1 forward primer: 5
Immunohistochemistry
Briefly, after verification under microscopic view with hematoxylin and eosin staining of each specimen, a 1.5 mm core punch sample was taken from each specimen, cut as 4
Cell proliferation assay
Colorimetric cell counting (CCK)-8 kits (Dojindo Laboratories, Japan) were applied to assess cell proliferation. Briefly, cells were seeded at a density of 1000 cells/100
Cell invasion and migration
For in vitro transwell invasion assays, 2.5
Transwell migration assay was used to assess chem-otactic and migration capacities of tumor cells. Cells were seeded in upper part of transwell (8
Cell cycle
Cells were detached and washed with ice-cold phosphate buffered saline (PBS) once and fixed in 70% ethanol, then were washed in PBS and incubated with 1 mg/mL of RNase A and 0.1 mg/mL of propidium iodide (PI, Sigma, College Park, MD, USA) for 30 min at 37
Cell viability assay
The cytotoxic effect of temozolomide (TMZ, Sigma) was determined using CCK-8, and TMZ was dissolved in dimethyl sulfoxide. Cells were seeded at a density of 5
X-ray irradiation
Briefly, cell suspensions of the different cell lines were adjusted to appropriate concentrations and inoculated into a 6-well plate (
Xenograft model
All animal experiments conformed to ethical principles and guidelines and were approved by the Soochow University Institutional Animal Care and Use Committee. For the survival study, U251-pIRES, SHG44-pIRES, U251-GBP1, and SHG44-GBP1 cells in 10
GBP1 expression level increased in TCGA GBMs cohort and associated with several molecular features of GBM. A, GBP1 mRNA expression level was detected in 528 clinical GBM specimens and 10 cases of normal brain tissue obtained by TCGA. The value represents log 2 of the gene expression level of each GBM sample to the average mRNA of 10 normal samples. Red samples (
SPSS version 20.0 (SPSS Inc., Armonk, NY, USA) and GraphPad Prism version 6.01 (GraphPad Software Inc., La Jolla, CA, USA) were performed to analyze the data. Expression differences were evaluated by paired t-test. Clinicopathological characteristics were compared by chi-squared test. Kaplan-Meier survival curves were computed with log-rank test and Cox regression analyses were used to assess overall survival (OS) based on different GBP1 expressions and other clinical features. Two-tailed P-values of
Results
GBP1 expression is correlated with several molecular features of GBM in TCGA cohort
When the expression profile of the GBP1 gene in TCGA cohort was examined, the mRNA expression of GBP1 was discovered to demonstrate more than 2-fold upregulation in 94.5% (499/528 for the Affymetrix platform) of GBMs compared to normal controls (Fig. 1A). To better understand the significance of GBP1 expression in GBMs, a correlation analysis was additionally conducted to analyze the relationship between GBP1 expression and molecular features of GBMs. TCGA network has identified a robust molecular classification of GBMs based on gene expression, which classified GBMs into four different subtypes: namely, classical, mesenchymal, neural, and proneural. Therefore, we screened GBP1 expression according to distinct molecular subtypes of GBM and observed relatively lower GBP1 expression in the proneural subtype compared with the other three subtypes, although GBP1 expression of each subtype remained noticeably higher than that of normal controls (Fig. 1B).
Univariate and multivariate Cox regression of GBP1 immunoreactivity for overall survival in our single-center glioma cohort
Univariate and multivariate Cox regression of GBP1 immunoreactivity for overall survival in our single-center glioma cohort
95% CI, 95% confidence interval; GBP1, guanylate-binding protein 1; HR, hazard ratio.
High expression of GBP1 predicts poor clinical outcomes in human gliomas. A–C, Kaplan-Meier plots were estimated according to different GBP1 gene expressions for overall survival. D–F, progression-free survival of all GBM patients, considering the mutation status of PTEN (B and E) or EGFR (C and F) simultaneously.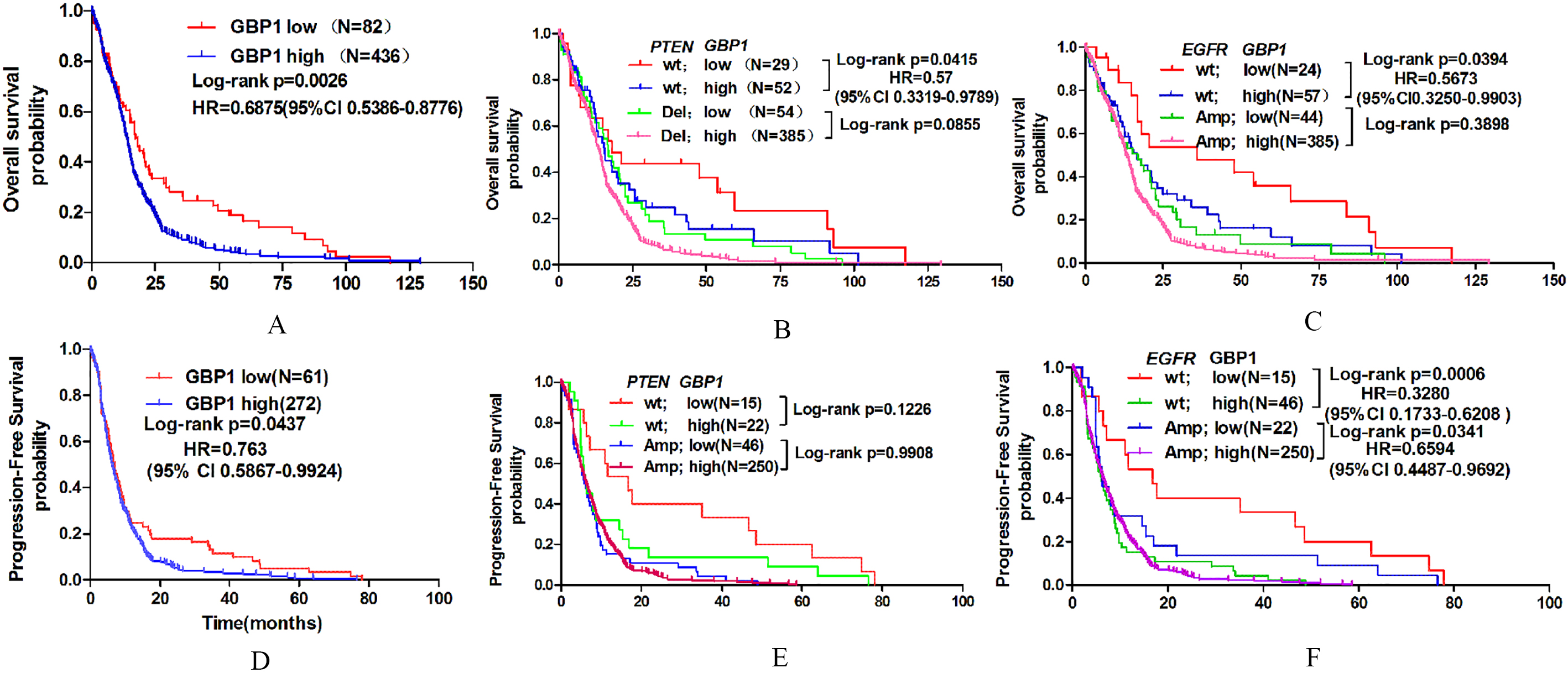
TCGA analyses of GBM have identified several critical genetic aberrations, including mutations in TP53, PTEN, NF1, EGFR, RB1, PIK3R1, IDH1, PIK3CA, SPTA1, ATRX, KEL, GABRA6, LZTR1, CTNND2, and BRAF; amplifications of EGFR, CDK4, PDGFRA, MDM2, MET, MDM4, CDK6, MYCN, CCND2, PIK3CA, and AKT3; and deletions of CDKN2A, CDKN2B, PTEN, CDKN2C, RB1, PARK2, and NF1. To further explore the expression profile of the GBP1 gene, we examined the association between its expression and these common genetic alterations in GBM. Consequently, we found that GBP1 expression was significantly associated with PTEN deletion and EGFR amplification (Fig. 1C and D).
Univariate Cox regression of GBP1 expression for OS and PFS in GBM patients of TCGA cohort
95% CI, 95% confidence interval; GBM, glioblastoma; GBP1, guanylate-binding protein 1; HR, hazard ratio; OS, overall survival; PFS, progression-free survival; TCGA, The Cancer Genome Atlas.
Expression and prognostic value of GBP1 in single-center glioma cohort. A, GBP1 protein expression was analyzed by immunohistochemistry staining in human glioma specimens and normal brain samples, and representative images showed high expression of GBP1 in GBMs. Magnification: 
To investigate the further relationship between GBP1 expression and clinical prognosis, the prognostic significance of GBP1 expression was also analyzed in our single-center glioma cohort. Kaplan-Meier analysis showed that in all glioma patients, patients with high GBP1 expression had obviously shorter OS than those with low GBP1 expression. Moreover, subsequent univariate and multivariate Cox regression analyses were additionally conducted to determine the independence of the prognostic value of GBP1. After correction for clinical characteristics that were suggested to be significant prognostic factors in the univariate Cox regression, high GBP1 expression was an independent risk predictor of OS (
Meanwhile, the correlation between GBP1 expression and GBM patients’ clinical outcomes in TCGA cohort was also analyzed. High GBP1 expression was confirmed to be an unfavorable predictor for patients’ OS and progression-free survival (PFS) (Fig. 2A and D). Results of Kaplan-Meier survival analyses stratified by the status of common genetic aberrations of GBM showed that the effect of GBP1 on patients’ prognosis was correlated with several molecular features including PTEN deletion and EGFR amplification (Fig. 2B–F). It was rather remarkable that the prognostic significance of GBP1 was highly pronounced in individuals with wild type PTEN (OS, log-rank
Cox regression further confirmed the prognostic value of GBP1 as an independent predictor for GBM patients’ OS (Affymetrix platform,
GBP1 could not promote glioma cell proliferation in vitro.A, Overexpression of GBP1 in U251 and SHG44 human glioma cell lines was validated by Western blot analysis. B, The cell growth curve of GBP1 overexpression cells was determined by CCK-8 assay. C, The indefinite proliferation ability of GBP1-overexpressed cells was examined using clonogenic cell survival assay. Bars represent the number of colonies (lower panels). D, The cell cycle of SHG44 and U251 glioma cells after upregulating GBP1 expression. 
In total, 103 cases of human glioma patients were enrolled in our single-center glioma cohort, and 72 (70%) were male patients. Details of clinical characteristics were presented in Table 3. The median OS was 21 months for all glioma patients. Expression levels of GBP1 protein were investigated by immunohistochemical staining. GBP1 was mainly expressed in the cytoplasm of tumor cells. Compared to 10 cases of non-tumor brain tissues, the immunoreactivity of GBP1 was unequivocally elevated in human gliomas and increased according to World Health Organization (WHO) grades, while the highest expression of GBP1 was observed in GBM samples (Fig. 3). Besides, we investigated whether the GBP1 gene was augmented at the transcriptional level. Total RNA was extracted from a subset of high grade, low grade glioma specimens and non-tumor brain tissues that were randomly selected from this cohort, then subjected to real-time quantitative reverse transcription (RT)-PCR assay. The mRNA expression of GBP1 also elevated significantly in human GBM and increased in parallel according to WHO grades (Fig. 3). Taken together, these results suggest that GBP1 elevated in human gliomas at both transcriptional and protein levels.
Clinicopathologic characteristics of glioma patients in our single-center glioma cohort (
103)
Clinicopathologic characteristics of glioma patients in our single-center glioma cohort (
GBP1, guanylate-binding protein 1; WHO, World Health Organization.
GBP1 expression-modulated cell migration and invasiveness in both SHG44 and U251 glioma cells. A–B, Cell invasion and migration assay showed that GBP1 overexpressed glioma cells could promote cell migration (A) and invasiveness (B). C, Western blot showed that cell migration and invasiveness related proteins were upregulated in GBP1 overexpressed cells.
GSEA of genes ranked by positive correlation with GBP1 expression in glioma. A, GSEA analysis showed that MT1H might has a regulative role in TOLL-like receptor signaling in glioma cells. B, GBP1 was related with TLR1, IRAK4 and IRF1. C, TLR1, IRAK4 and IRF1 were upregulated in GBP1 overexpressed cells.
To explore the biological significance of GBP1 in glioma, we investigated whether GBP1 could affect cell proliferation. First, GBP1 stably overexpressed U251 and SHG44 cells were established in transfected tumor cells respectively. The empty vector (pIRES) transfected tumor cells served as control groups. The efficiency of GBP1 overexpression was validated with Western blot (Fig. 4A) assay. The impact of GBP1 on glioma cell proliferation in vitro was then examined. Cell growth was determined by CCK-8 assay within a 4-day monitoring period. Results showed that enforced expression of GBP1 could not promote the proliferation of U251 and SHG44 cells compared with control groups (Fig. 4B), which were accordance with the previous study [20]. The effect of ectopic GBP1 expression on long-term cell growth was examined using a clonogenic assay in U251 cells. The colony-forming ability of U251 cells with GBP1 overexpression was unenhanced (Fig. 4C). These results suggest that GBP1 could not promote the proliferation of glioma cells in vitro. Cell cycle analysis was also applied to investigate whether GBP1 could affect cell proliferation. Compared with control pIRES-infected cells, glioma cells with GBP1 overexpression showed no difference in percentages of G0/G1, S, and G2/M phases (Fig. 4D).
Upregulation of GBP1 was related to TMZ resistance but not RT resistance. A–B, Survival rates of tumor-bearing mice of GBP1-overexpressed cells were significantly higher than those of pIRES transfected cells. C–D, Survival rates of these cells showed no difference under different radiation doses. 
Inhibition of cell-cell and/or cell-matrix adhesive functions correlated with tumor migration and invasion. However, it was unclear whether GBP1 could affect the migration and invasive ability of glioma cells to influence patient prognosis. As shown in Fig. 5A, our results showed that cell migration enhanced significantly as a result of GBP1 overexpression in both SHG44 and U251 cells. Invasiveness evaluation by transwell invasion assay showed same significant difference in both SHG44 and U251 cells between GBP1 and empty vector transfection (Fig. 5B). Furthermore, Western blot was applied to measure protein levels of migration/invasiveness-related factors such as MMP-9, Twist, and Snail (Fig. 5C), which showed the protein levels of these migration/invasiveness related molecules increased obviously in GBP1 overexpression cells. The relevant data revealed that GBP1 promoted the migration and invasion capabilities of both U251 and SHG44 glioma cells.
Identification of GBP1-associated biological pathways by Gene Set Enrichment Analysis (GSEA)
To assess GBP1-related pathways on an unbiased basis, we performed GSEA using data from TCGA cohort. GBP1 gene expression level was used as the phenotype label. Toll-like pathways were found to be significantly associated with GBP1 expression in TCGA cohort (Fig. 6).
To validate the GSEA analysis of GBP1, we analyzed the protein levels of pathway-related factors in GBP1-transfected SHG44 and U251 cells. As shown in Fig. 6C, the protein levels of Toll-like-related factors TLR1, IRAK4, and IRF1 altered significantly in GBP1-overexpressed glioma cells (Fig. 6C).
Survival analysis of tumor-bearing mice with intracranial inoculation of GBP1 overexpressed SHG44 cells. A, H&E staining of brain sections 1 month after intracranial inoculation of SHG44-pIRES (left) or SHG44-GBP1 (right). B, Immunohistochemical staining of MMP9, twist in the transplanted tumor sections of in vivo xenografts showed high expression level of invasion associated molecules in the in vivo tumor models after GBP1 transfection (right). C, Kaplan-Meier survival curve of mice after intracranial implantation of GBP1 or blank vector transfected SHG44 cells. Statistical comparisons were performed using a log-rank test. 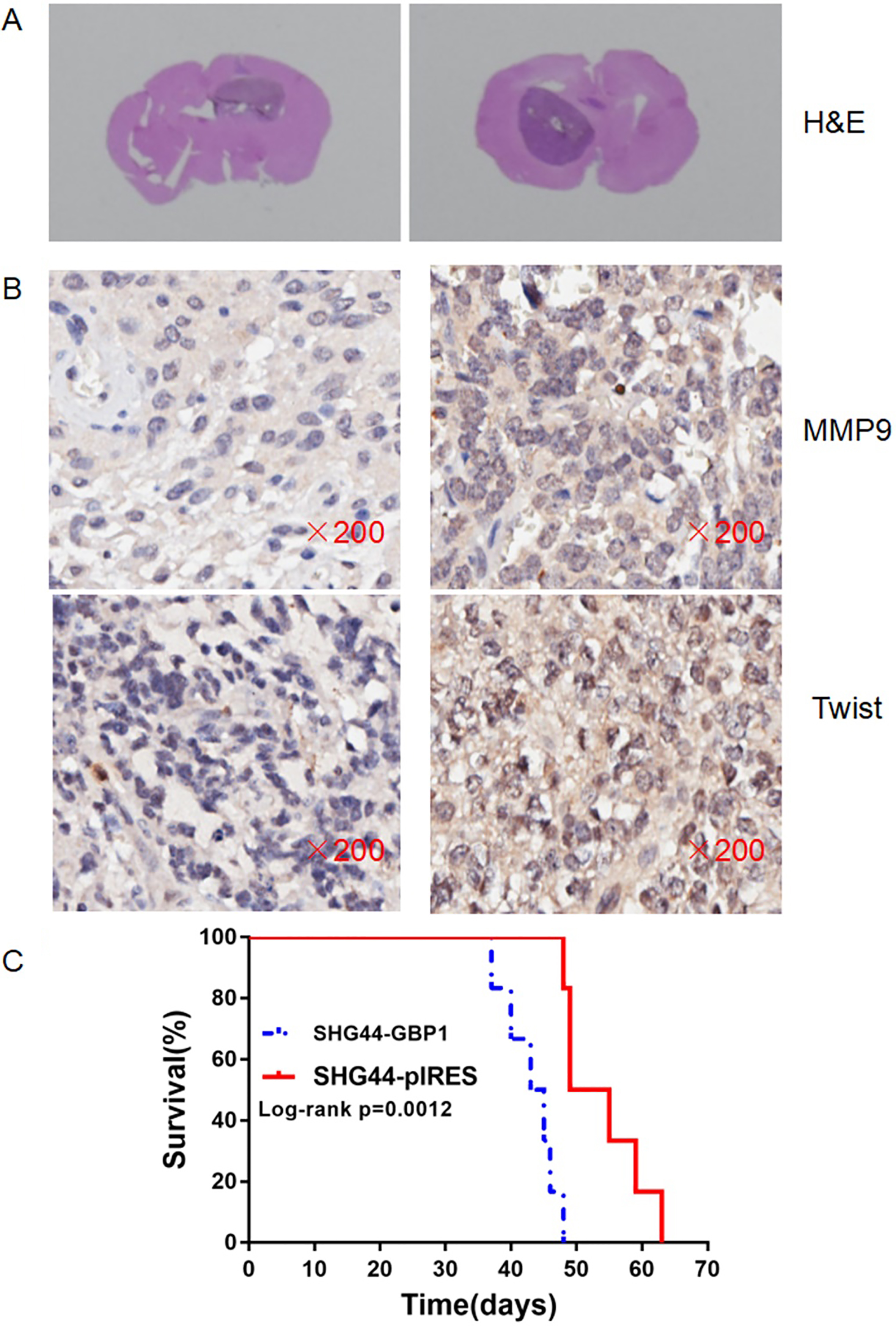
In an effort to further understand whether GBP1 expression could affect the sensitivity of TMZ and radiotherapy treatment, GBP1 overexpressed U251, SHG44 glioma cells and blank vector transfected U251, SHG44 cells were used to examine the relative cell viability as well as colony formation following either TMZ treatment or irradiation. When compared to blank vector transfected SHG44 and U251 cells, the GBP1-overexpressed cells significantly enhanced the viability of TMZ concentrations up to 200
Survival analysis of tumor-bearing mice with intracranial inoculation of GBP1 overexpressed U251 cells. A, H&E staining of brain sections 1 month after intracranial inoculation of U251-pIRES (left) or U251-GBP1 (right). B, Immunohistochemical staining of MMP9, twist in the transplanted tumor sections of in vivo xenografts showed high expression level of invasion associated molecules in the in vivo tumor models after GBP1 transfection (right). C, Kaplan-Meier survival curve of mice after intracranial implantation of GBP1 or blank vector transfected U251 cells. Statistical comparisons were performed using a log-rank test. 
Although GBP1 overexpression could not promote GBM cell growth in vitro, its effect on cell growth in vivo was investigated. 1
Kaplan-Meier survival studies were performed on intracranial tumor bearing mice. Average survival time was significantly reduced in the SHG44-GBP1 group compared to the control group of blank vector transfected SHG44 cells(median survival 44 vs. 52 days,
Discussion
Although various clinical trials have been performed to treat gliomas over the past several decades, patients’ median survival rate has not significantly improved [26]. In recent years, greater attention has been paid to molecular target therapy; however, no mature clinical application has been achieved. There is still a great need to develop new molecules related to prognosis and target therapy to improve the treatment of gliomas.
The GBP1 protein was identified to interact with a variety of diseases involved in viral infections and tumors [27, 28, 29, 30, 31, 32]. In addition, it plays varying roles in different tumors. Hence, our study aimed to unveil the indispensable roles of GBP1 in malignant progression of gliomas. We found evidence of upregulation of the GBP1 protein in human GBM tissues in TCGA cohorts; these results have been confirmed in our single centre clinical specimens. In addition, expression of GBP1 was significantly higher in high-compared to low-grade gliomas, and may serve as an independent prognostic factor for gliomas. These data suggest that GBP1 is a significant prognostic factor and might play a significant role in glioma progression. Since GBM has recently been classified into four subtypes based on gene expression profiles, in TCGA data analyses, we found a significantly reduced expression of GBP1 in the proneural subtype compared to other three subtypes, and in GBM patients, proneural subtype had relative better prognosis than the other three subtypes, suggesting GBP1 expression level had close relationship with GBM patients prognosis. Enhanced GBP1 expression has also been found to be linked closely with PTEN deletion or EGFR amplification. The impact of GBP1 expression on patients’ survival stratified by these molecular features showed that GBP1 expression could delineate GBM patients together with same other specific genetic alterations. Indeed, these patients could be classified into two subsets with completely distinct clinical outcomes, which were more pronounced than in all patients. GBP1 could facilitate predicting prognosis for patients with deleted PTEN or amplified EGFR, suggesting that the prognostic value of GBP1 was dependent on PTEN status and EGFR alteration. The current understanding of GBP1 is that its roles in tumor growth are controversial, and results of our single-center glioma cohort were consistent with TCGA results. On one hand, it facilitated tumor proliferation, tumor metastasis invasiveness, and chemotherapy resistance [33]. On the other hand, GBP1 was recently indicated as a potential tumor suppressor in colon cancer [14]. In gliomas, found that GBP1 could promote U87 cells proliferation in vivo but not in vitro [20]; our results further validate this finding in another two GBM cell lines, suggesting the possible mechanisms of GBP1 pro-proliferation on glioma cells may rely on tumor-associated microenvironment. Besides, GBP1 was able to promote invasion; further, MMP9 and Snail were upregulated in SHG44-GBP1 and U251-GBP1. In terms of the relationship between GBP1 expression and sensitivity of radiotherapy or chemotherapy, our results showed that GBP1 could contribute to TMZ resistance, but that radiotherapy sensitivity was unaffected. In the GSEA analysis, results indicated that Toll-like pathways may be involved in the role of GBP1 in GBM and that they could promote tumor invasion and immune escape. TLR1, IRAK4, and IRF1 were the key factors in Toll-like pathways, and were upregulated in GBP1 transfected cells, which had already been found in ovarian cancer that GBP1 could affect the prognosis of ovarian cancer patients and that IRF1 and GBP1 were involved in this process [27].
In summary, we conclude that GBP1 significantly elevated in human GBMs, and predicts an shorter overall survival and PFS for patients with GBM as a novel and independent prognostic factor, as found in TCGA cohort and our single-center cohort analysis. Furthermore, the expression profile and clinical significance of GBP1 are associated with several molecular features of GBM. Finally, GBP1 plays an important role in glioma cell growth in vivo, highlighting its potential application as a therapeutic target for glioma.
Footnotes
Acknowledgments
This study was supported by grants from the National Natural Science Foundation of China (NO. 81472739, 81702457, 81602183), the Natural Science Foundation of Jiangsu China (BK20151214), Science and Technology Project of Suzhou (SYS201627).
Conflict of interest
The authors have no conflicts of interest to declare.
