Abstract
BACKGROUND:
Breast cancer is one of the most commonly diagnosed cancers, and the fourth leading cause of cancer deaths in females worldwide. Activated platelets play a key role in tumor growth and tumor metastasis. Platelet distribution width (PDW) is a platelet index, and is altered in patients with malignancies. The aim of this study was to explore whether PDW can effectively predict death outcome of breast cancer patients.
STUDY DESIGN:
The clinical data of 271 breast cancer patients in our hospital between January 2009 and December 2009 were retrospectively analyzed. Survival analysis was performed using the log-rank test and Cox proportional hazards regression analysis.
RESULT:
There were significant correlations between increased PDW and tumor size, molecular subtype, differentiation grade, and cancer stages (T, N, or TNM). Moreover, survival analysis revealed that the overall survival of patients with PDW
CONCLUSIONS:
Elevated PDW may serve as a marker of adverse prognosis in breast cancer. However, these data are preliminary and should be interpreted with caution pending validation by additional clinical and molecular/genomics studies in various populations.
Introduction
Breast cancer (BC) is one of the most commonly diagnosed cancers, and the fourth leading cause of cancer deaths in females worldwide [1]. Although many advancements have been made in diagnosis and treatment, more than 50% of invasive BC patients have developed distant metastases within ten years of diagnosis [2]. Accurate assessment of individual patient prognosis is an extremely important aspect of clinical decision-making in care and treatment of cancer patients. Diagnostic tools that are can be easily conducted and at low cost would be beneficial in the practice [3].
Platelets have been involved in the mechanisms leading to carcinogenesis, tumor growth, tumor angiogenesis, and tumor metastasis, as well as in the outcome of therapy [4]. Elevated platelets are associated with poor prognosis in a wide spectrum of malignancies, such as pancreatic cancer, gastric cancer, colorectal cancer, endometrial cancer, and ovarian cancer [5, 6, 7, 8, 9]. Platelet number is determined by the balance between the rates of production versus consumption of platelets. A normal platelet count could not be interpreted as the presence of hypercoagulative and pro-inflammatory state in the presence of efficient compensatory mechanisms [10].
Platelet distribution width (PDW), a platelet index, indicates variation in platelet size [11]. Altered PDW levels have been reported in gastric cancer, lung cancer, thyroid cancer, and breast cancer [12, 13, 14, 15]. In addition, increased PDW levels are associated with poor prognosis in melanoma, laryngeal cancer, gastric cancer, and non-small cell lung cancer [16, 17, 18, 19]. However, the clinical implications of PDW in breast cancer have not been well defined. The aim of the present study was to determine whether preoperative PDW could predict survival of BC patients.
Patients and methods
Study population
We retrospectively analyzed 271 consecutive BC patients who underwent complete surgical resection at Harbin Medical University Cancer Hospital from January 1, 2009 to December 31, 2009. All of these patients were histologically confirmed as having breast cancer by two experienced pathologists. None of the patients received any adjuvant therapy before resection. Exclusion criteria included: hematological disorders, hypertension, diabetes mellitus, and medical treatment with anticoagulants, statins, and acetylic salicylic acid. Overall survival (OS) was calculated from the date of surgery to the date of either death or last follow-up. Final follow-up was completed on December 31, 2014. The median follow-up duration was 60 months.
The Institutional Ethics Review Board of Harbin Medical University Cancer Hospital approved this study prior to commencement of data collection, and waived the informed consent requirement because it was a retrospective study.
Biochemical measurements
Venous blood samples taken after a 10-hour over-night fasting were collected from the individuals within 1 week prior to surgery. White blood cell (WBC), hemoglobin, and platelet indices were measured by an autoanalyzer (Sysmex XE-2100, Kobe, Japan). The whole-blood samples were collected in EDTA-containing tubes, and all samples were processed within 30 minutes of blood collection.
Immunohistochemistry and FISH analysis
Human epidermal growth factor receptor 2 (HER2) was evaluated by immunohistochemistry (IHC) with the polyclonal antibody EP3 (Maixin Laboratories, China). HER2-positive was defined as a score of 3+ on IHC or score 2+ and confirmed HER2 amplification by FISH (Jinpujia Laboratories, China). The immunohistochemical assessment of estrogen receptor (ER) and progesterone receptor (PR) was made using the SP1 (Maixin Laboratories, China) and 1D5 (Maixin Laboratories, China) antibodies, respectively. ER and PR expression were considered positive if at least 1% immunostained tumor nuclei were detected in the sample [20].
The relation between clinico-pathological parameters and the PDW levels of BC patients before treatment
The relation between clinico-pathological parameters and the PDW levels of BC patients before treatment
PDW, platelet distribution width; TNBC, triple-negative breast cancer; ER, estrogen receptor; PR, progesterone receptor; HER2, human epidermal growth factor receptor 2. p value was obtained using Chi-square test.
The descriptive statistics are presented as means
Results
The characteristics of BC patients are summarized in Table 1. Overall, there were 109 (40.2%) pre-menopausal and 162 (59.8%) post-menopausal patients, and the mean age was 50.7
Optimized cut-off value was determined for PDW using standard ROC curve analysis.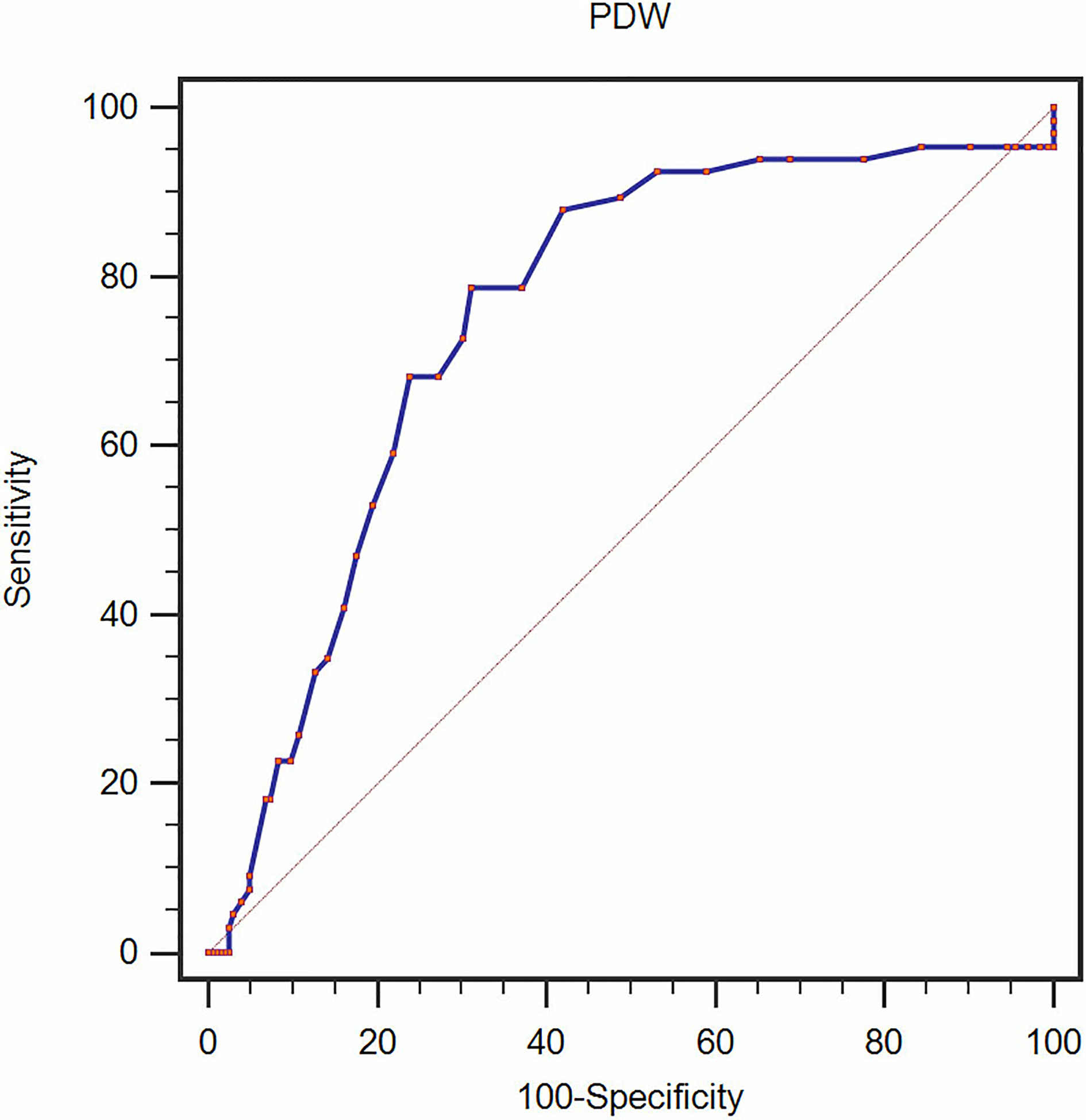
According to the ROC curve analysis, we identified that the optimal cut-off value of 16.8 for PDW had a 78.8% sensitivity and 68.8% specificity for survival rate (AUC
The relation between clinical parameters and the pretreatment PDW levels of BC patients
P value was obtained using Student’s
The relationships between PDW and clinical characteristics are summarized in Tables 1 and 2. Our study revealed that PDW was associated with tumor size, T stage, N stage, molecular subtype, differentiation grade, and TNM stage (
With a median of up to 60 months follow-up, 66 (24.4%) patients had died. Of the deceased patients, those with PDW
Kaplan–Meier analysis of overall survival in breast cancer patients.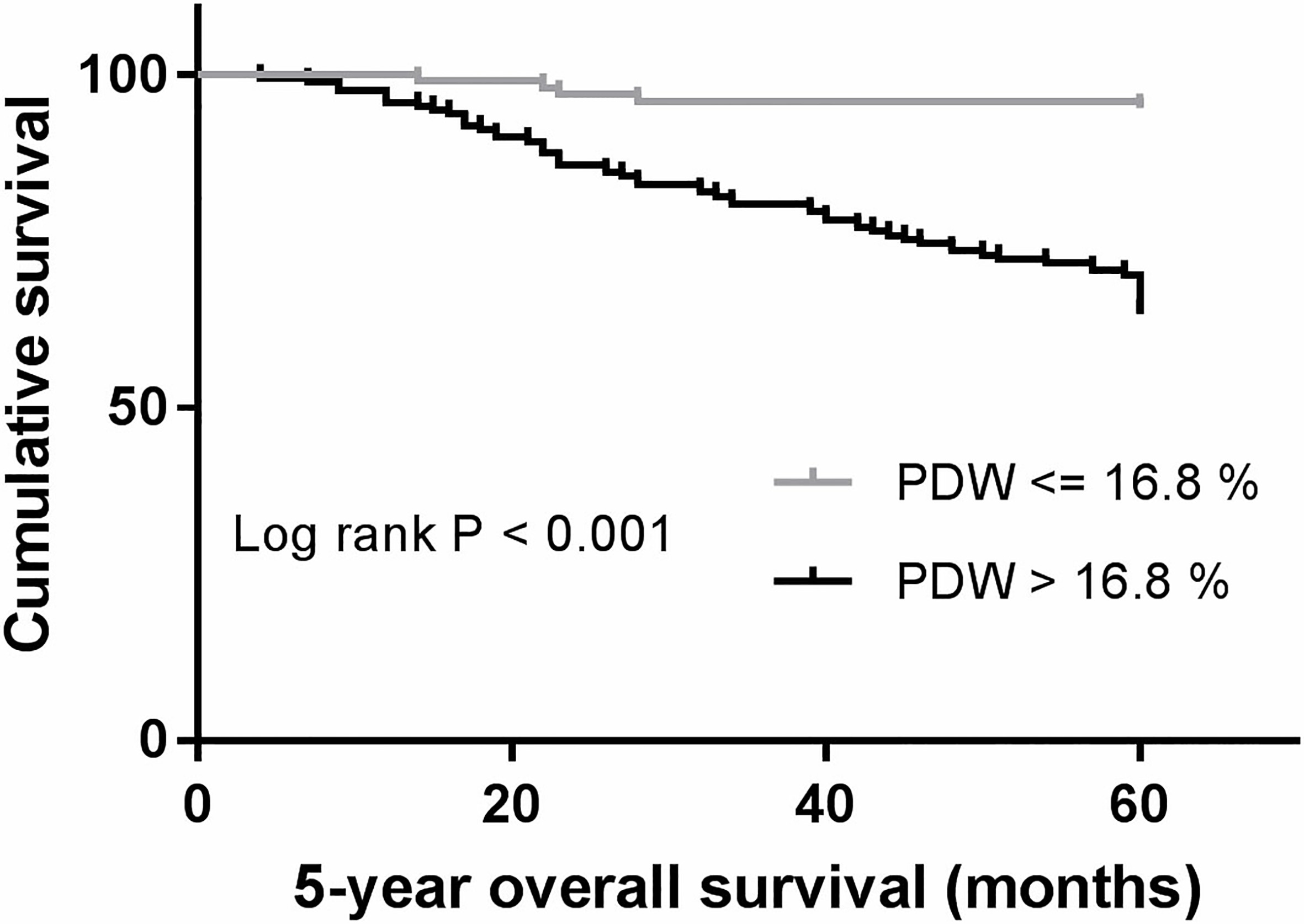
Cox univariate and multivariate regression analyses were performed for the factors that could impact survival. Cancer stage (T, N, TNM), tumor size, molecular subtype, differentiation, PR status, Ki-67, MPV, MPV to platelet count ratio, PDW to platelet count ratio, and PDW were determined, which were significant in univariate analyses (Table 3). Next, all the factors in univariate analysis with a p-value less than 0.10 were included in multivariate analysis (Table 4). Multivariate analysis showed that PDW was an independent variable associated with survival. Patients with PDW
The univariate analysis of overall survival in BC patients
MPV/P, mean platelet volume to platelet count; PDW/P, platelet distribution width to platelet count.
The multivariate analysis of overall survival in BC patients
Variables that showed a
This study showed that increased PDW was significantly correlated with tumor size, molecular subtype, differentiation grade, cancer stages. In addition, PDW was an independent risk factor that influences BC prognosis.
Despite best current medical and surgical treatment, the overall prognosis of BC patients remains poor. It is therefore of great importance to identify novel prognostic biomarkers. There is a complex interplay between platelet-induced tumor growth and tumor cell-induced platelet activation [21]. In BC, platelet-derived growth factor (PDGF) expression is associated with biological aggressiveness, and it functions through NFkappaB signaling pathway [22, 23]. Moreover, platelet-derived lysophosphatidic acid has been shown to accelerate osteolytic bone metastases [24]. These discoveries make the PDGFR/PDGF system an attractive therapeutic target [25]. In deed, a recent study demonstrated that combinations of anti-platelet drugs inhibit BC cell-induced platelet aggregation [26].
In agreement with the studies cited above, our study indirectly confirmed these findings using a simple platelet indicator. Our results are also consistent with a recent report published by Takeuchi et al. [27]. In this interesting paper, an elevated PDW/PLT was found to reduce disease-free survival in small set of 275 Japanese BC patients with a follow-up of 120 months. In addition, this report showed that PDW correlated with tumor size, estrogen receptor status and progesterone status [27]. Our study found that there were significant correlations between increased PDW and tumor size and molecular subtypes. Furthermore, our results provided further evidence for the role of PDW in decreasing OS in Chinese BC patients with a follow-up of 60 months. These studies underscore the importance of PDW in prognosis prediction of BC patients, which warrants further mechanistic investigations, and may ultimately lead to new personalized therapeutic strategies.
Currently, the underling mechanisms that link PDW and survival are unknown. PDW is a measure of platelet heterogeneity caused by heterogeneous demarcation of megakarocytes [28]. Altered PDW may be attributed to dysfunction of bone marrow cells (including megakaryocytes). Recent studies showed several cytokines, such as interleukin-6 (IL-6), granulocytes colony stimulating factor (G-CSF) and macrophage colony stimulating factor (M-CSF), regulate megakaryocytic maturation, platelet production and platelet size [29]. IL-6 is also known to promote tumor angiogenesis, metastasis and metabolism [30]. In addition, G-CSF and M-CSF secreted by tumor cells could stimulate megakaryopoiesis and subsequent thrombopoiesis in cancer [31]. In addition, local inflammation play a key role in tumor microenvironment. During tumor progression, some inflammatory cytokines like IL-1, IL-6, IL-8, IL-12, interferon-
Our study has some limitations. First, this was a single-center retrospective study, and additional large-scale, multicenter prospective trials are needed to confirm our results. Second, further mechanistic studies are needed to determine why PDW is associated with survival rate of BC patients. Third, in agreement with the report of Takeuchi et al. in Japanese BC patients, our study has confirmed that PDW is associated with survival in Asian BC patients [27]. The study is needed to be repeated in other ethnic groups in order to determine whether our conclusion is genetic ethnic-specific.
In conclusion, our study showed that elevated PDW may serve as a marker of adverse prognosis in breast cancer. However, these data are preliminary and should be interpreted with caution pending validation by additional clinical and molecular/genomics studies in various populations.
Footnotes
Acknowledgments
This work was supported by Science Foundation of Heilongjiang Academy of Medical Science (Grant No. 201714).
Conflict of interest
All authors declare no conflict of interest.
