Abstract
BACKGROUND:
Esophageal cancer (EC) is the sixth most common cause of death from cancer. Altered mean platelet volume (MPV) levels were found in patients with malignancies.
OBJECTIVE:
The present study investigated whether MPV can predict the survival in EC patients.
MEHTODS:
The clinical data of 236 consecutive EC patients between January 2009 and December 2009 in our center were retrospectively analyzed. The overall survival rate was estimated using Kaplan-Meier method. Cox proportional hazards models were fitted to model the relationships between patient characteristics and prognosis.
RESULTS:
Decreased MPV was significantly correlated with tumor location and tumor differentiation (
CONCLUSIONS:
Reduced MPV is associated with worse survival outcome in EC.
Introduction
Esophageal cancer (EC) is the sixth most common cause of death from cancer and the eighth most common cancer worldwide [1]. Prognosis of EC is poor with a 5-year survival rate of less than 20% even with multiple treatment modalities [2, 3]. Therefore, novel prognostic biomarkers could improve prognosis for EC patients.
Emerging evidence demonstrated that platelets contribute to circulating tumor cell growth, survival and invasiveness, and lead to tumor metastasis [4]. Thrombocytosis correlates with poor prognosis in patients with gastric, pancreatic, colorectal, ovarian, endometrial cancer [5, 6, 7, 8, 9]. Since platelet count is determined by the balance between the rate of production and consumption of platelets, in the presence of efficient compensatory mechanisms, a normal platelet count could conceal the presence of highly hypercoagulative and pro-inflammatory cancer phenotypes [10].
A most commonly used measurement of platelet size is mean platelet volume (MPV) in clinical practice. MPV is an indicator of platelet activation [11]. Alteration of MPV levels in breast, lung, gastric, colon, and ovarian, cancer has been reported [12, 13, 14, 15]. However, the significance of the change in these cancers remain elusive. The aim of this study was to assess the relationship between preoperative MPV and the long-term survival of EC patients.
Patients and methods
Study population
The study was comprised of 236 consecutive EC patients who underwent surgery between January 2011 and December 2011 at Harbin Medical University Cancer Hospital (Harbin, Heilongjiang, China). Clinical, demographic and pathological data were obtained from the patients’ medical records. Histopathological evaluation was carried out independently by two pathologists. None of the patients received preoperative chemotherapy or radiation therapy. Exclusion criteria included: hematological disorders, hypertension, diabetes mellitus, and medical treatment with anticoagulant, statins, and acetylic salicylic acid. Disease-free survival (DFS) time was calculated from the day of surgery to the day of definitive diagnosis of recurrent tumor. Overall survival (OS) time was measured from the date of surgery to the date of death or the date of the last clinical follow-up. The last clinical follow-up was completed on December 31, 2016. The median follow-up duration was 60 months.
The Institutional Ethics Review Board of Harbin Medical University Cancer Hospital approved this study prior to commencement of data collection and waived the informed consent requirement because it was a retrospective study. All methods were conducted according to guidelines (Declaration of Helsinki) for biomedical research.
Biochemical measurements
Fasting venous blood samples were collected in the morning after a 10-h fast within 1 week prior to surgery. White blood cell (WBC), haemoglobin, and platelet indices were measured by an autoanalyzer (Sysmex XE-2100, Kobe, Japan). The whole blood samples were collected in EDTA-containing tubes, and all samples were processed within 30 minutes after blood collection.
Statistical analysis
The descriptive statistics were presented as means
Results
Of 236 enrolled primary EC patients, 94.5% (
The relation between clinico-pathological parameters and the pretreatment MPV levels in EC patients
The relation between clinico-pathological parameters and the pretreatment MPV levels in EC patients
Optimized cut-off value was determined for MPV using standard ROC curve analysis.
The relation between clinical parameters and the pretreatment MPV levels in EC patients
According to the ROC curve analysis, we identified that the optimal cut-off value of MPV was 7.4 fL, with a 28.2% sensitivity and 85.2% specificity for survival rate (AUC
The relationship between MPV and clinical characteristics was shown in Tables 1 and 2. MPV was associated with differentiation tumor location. However, there were no significant differences between two groups in terms of age, tumor size, tumor invasion depth, lymph node metastasis, and clinical stage.
With a median follow up of 60 months, 135 (57.2%) patients had death events. The association between MPV and survival of EC patients was investigated by Kaplan-Meier analysis and log-rank test. We found that both the DFS and OS in the normal MPV group are significantly longer than those in the low MPV group (Fig. 2, 5-year DFS rates, 47.0% vs 11.3%,
Kaplan-Meier analysis of disease-free survival in EC patients.
Kaplan-Meier analysis of overall survival in EC patients.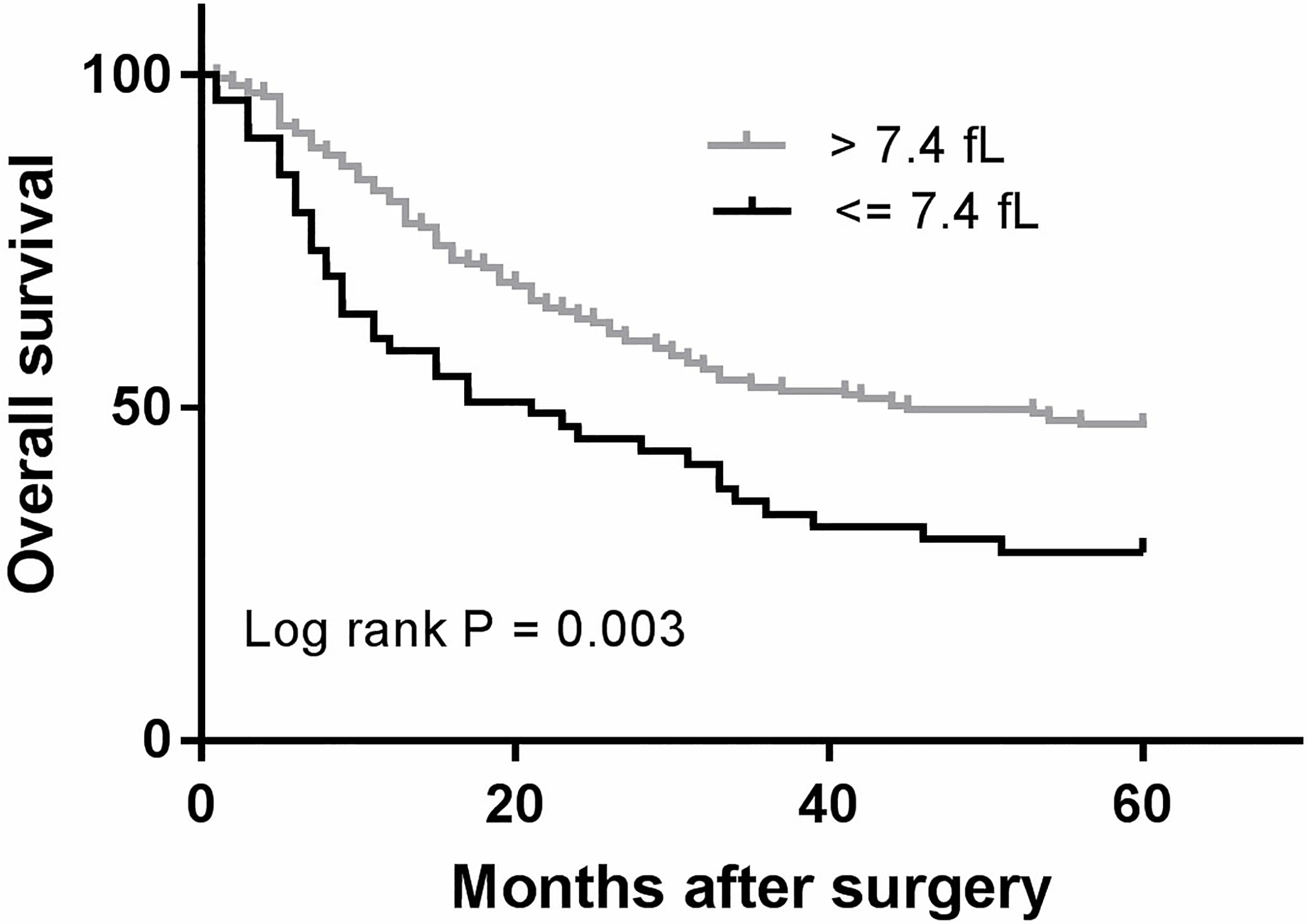
We preformed Cox univariate and multivariate regression analyses for factors that could impact survival including tumor invasion depth, lymph node metastasis, TNM stage, and MPV. We found that MPV is significantly associated with survival in univariate analyses (Table 3). Next, we performed a multivariate analysis including all factors with a
The univariate analysis of overall survival in EC patients
The multivariate analysis of overall survival in EC patients
Variables that showed a p-value
The main finding of this study is that reduced MPV is significantly associated with shorter survival in EC patients.
It was well recognized that platelets act as a key modulator in tumor cell growth, angiogenesis, and metastasis. In EC, platelets were associated with lymphangiogenesis and lymphovascular invasion [16]. High expression of platelet-derived growth factor-BB (PDGF-BB) predicts lymph node metastasis independently from platelet count [17]. Cyclooxygenase-2 expression in esophageal squamous cell carcinoma cells was significantly increased following treatment with platelet activating factor [18]. However, measurement of most parameters reflecting activated platelets was expensive and not commonly used in clinical practice. Whereas MPV is available in routine praxis and can be easily obtained prior to treatment.
The reasons for reduced MPV in EC are unclear. Chronic inflammation has been implicated in the development and progression of cancer including EC [19]. MPV is an early indicator of activated platelets. Reduced MPV could be regarded as an enhanced consumption of large platelets in inflammatory states [11]. Recent studies showed that MPV were reduced in high-grade inflammatory diseases, which could be reversed in the course of anti-inflammatory therapy [11].
In accord with our results, Hyder et al. showed that changes in neutrophil-to-lymphocyte and platelet-to-lymphocyte ratios during chemoradiation predict survival in trimodality EC patients [20]. McLaren et al. found that neutrophil-to-lymphocyte and platelet-to-lymphocyte ratios predict treatment response to neoadjuvant therapy in EC [21]. These data are also consistent with the current knowledge that anti-platelet is considered to be a part of cancer adjuvant therapy [22]. However, the association between MPV and the prognosis varies in different cancer types. Low MPV level in non-small-cell lung cancer (NSCLC) was associated with unfavorable survival [14, 23], whereas high MPV in blood tumor, renal cancer, hepatocellular carcinoma, and lung cancer is associated with advanced stage cancers (or unfavorable disease-like thrombotic state) [24, 25, 26, 27].
The present study is not without limitations. First, our study was single-center retrospective in nature. Larger prospective randomized studies are needed to validate and extend our findings. Second, the patients were composed of Chinese. The application to other ethnic groups still needs further investigation.
Taken together, MPV may serve as a promising biomarker for predicting prognosis of EC patients.
Footnotes
Acknowledgments
This work was supported financially by grants from the Harbin Medical University Cancer Hospital (JJZD 2017-05).
Conflict of interest
All authors declare no conflict of interest.
