Abstract
Objective
To compare the serum mean platelet volume (MPV) in patients with testicular torsion and healthy control subjects.
Methods
This retrospective study compared clinical and demographic data from patients who underwent surgery for testicular torsion and healthy controls. Testicular torsion was diagnosed with a physical examination and Doppler ultrasonography. Age, leukocyte count, platelet count and MPV were recorded for all participants.
Results
A total of 50 patients with testicular torsion and 51 healthy control subjects were enrolled. There was no significant difference in age and platelet count between the two groups. Patients with testicular torsion had a significantly higher leukocyte count and MPV, compared with controls. According to receiver operating characteristic curve analysis performed for the prediction of testicular torsion, the best cut-off point for MPV was 7.7 fl (sensitivity 62%, specificity 96%), and the best cut-off point for leukocyte count was 9.5 × 109/l (sensitivity 58%, specificity 80%).
Conclusions
The MPV may be useful as an ancillary test for the diagnosis of testicular torsion. Further studies are needed in order to confirm these preliminary results.
Introduction
Testicular torsion is a clinical condition that is characterized by reduced testicular blood flow as a result of twisting of the spermatic cord around its own axis. Twisting of the spermatic cord is followed by testicular venous occlusion, congestion, and, ultimately, arterial ischaemia and testicular infarction. 1 Testicular torsion is more common in males aged <25 years; 1 affected patients typically present with testicular tenderness and pain. Testicular torsion should be differentiated from other conditions that feature acute scrotal pain including epididymo-orchitis and appendicitis. 2 Differentiation can be achieved by imaging modalities such as scrotal Doppler ultrasonography and scintigraphy. The use of laboratory parameters (e.g. leukocyte count, erythrocyte sedimentation rate, and alkaline phosphatase, creatine kinase and C-reactive protein measurement) has been investigated in the diagnosis of testicular torsion and acute scrotum. 3
Mean platelet volume (MPV) is a marker of platelet activation. 4 Although routinely analysed along with other indices of the complete blood count (CBC), the MPV is often overlooked by clinicians. Research has suggested the usefulness of MPV in the diagnostic processes of various inflammatory and/or vascular disorders.4,5 This current retrospective study aimed to investigate whether the MPV value, and the leukocyte and platelet counts, could be used for diagnosing testicular torsion.
Patients and methods
Patient population
This study retrospectively reviewed the medical records of patients with acute scrotal pain, who presented to the Department of Urology and the Department of Emergency Medicine, Medical and Research Centre, Baskent University, Konya, Turkey and the Department of Urology, Faculty of Medicine, Baskent University, Ankara, Turkey, between February 2005 and December 2013. Patients diagnosed with testicular torsion underwent orchiectomy and/or surgical detorsion and bilateral testicular fixation. Exclusion criteria included manual detorsion followed by surgery on an elective basis at a later date, myeloproliferative disorders, leukaemia, and Bernard–Soulier Syndrome. A control group included healthy male volunteers selected from individuals who attended the Department of Infectious Diseases and Clinical Microbiology, Medical and Research Centre, Baskent University, Konya, Turkey for an outpatient appointment. The control subjects had no epididymo-orchitis, testicular trauma, testicular tumour, previous scrotal surgery, or abnormal renal or hepatic function tests. Demographic features and findings of a preoperative CBC (which included leukocyte count, platelet count, and MPV) were recorded for all participants. Age, leukocyte count, platelet count, and MPV were compared between the two groups.
Consent was not required from the Ethics Committee of Baskent University, Ankara, Turkey because of the retrospective nature of this study. Written informed consent was routinely obtained from each surgical patient. Verbal informed consent was obtained from all subjects in the control group.
Laboratory measurements
Peripheral venous blood samples (3 ml) were collected into tubes containing 5.4 mg ethylenediaminetetra-acetic acid, and a rapid laboratory analysis was applied to each sample. An electronic cell counter (CELL-DYN® 3700; Abbott Laboratories, Abbott Park, IL, USA) was used to measure the CBC. At Baskent University, the expected MPV range was 7.0–12.0 fl, the expected leukocyte count was 4.5–11.0 × 109/l and the expected platelet count was 150–400 × 109/l.
Statistical analyses
All statistical analyses were performed using the SPSS® statistical package, version 19.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Data were expressed as mean ± SD. Student’s t-test was used for continuous variables. The leukocyte and platelet counts and the MPV were not normally distributed and thus were compared with the nonparametric Mann–Whitney U-test across the two study groups. The correlation between the MPV and the other variables was analysed using Spearman’s rank correlation coefficient. A P-value <0.05 was considered statistically significant. Receiver operating characteristic (ROC) curve analysis was used to determine the relationship between testicular torsion and MPV and leukocyte count.
Results
Clinical and demographic characteristics of patients with testicular torsion (n = 50) compared with a group of healthy control subjects (n = 51).

Data presented as mean ± SD.
Student’s t-test was used for continuous variables. Leukocyte and platelet counts and mean platelet volume were not normally distributed and thus were compared with the nonparametric Mann–Whitney U-test across the study groups.
NS, no significant between-group differences (P ≥ 0.05).
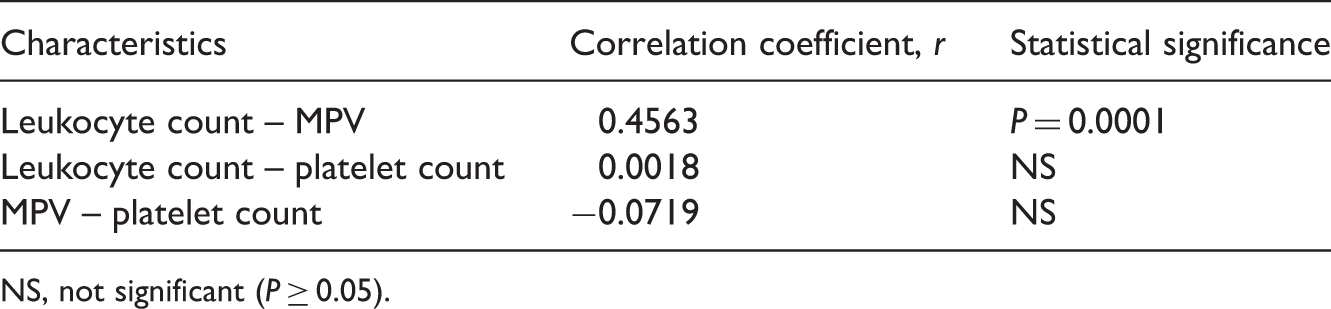
According to the ROC curve analysis performed for the prediction of testicular torsion, the best cut-off point for the leukocyte count was 9.5 × 109/l (sensitivity 58%, specificity 80%, area under curve [AUC]: 0.7) (Figure 1). The same analysis showed that the best cut-off point for MPV was 7.7 fl (sensitivity 62 Receiver operating characteristic curve of mean platelet volume level (MPV) and leukocyte count. Spearman’s rank correlation coefficient analysis of the correlation between mean platelet volume (MPV), leukocyte count and platelet count in patients with testicular torsion (n = 50). NS, not significant (P ≥ 0.05).
Discussion
Measuring the CBC is a frequently used, rapid and inexpensive way of providing valuable information with regard to the diagnosis and follow-up of various disorders. Urologists frequently use CBC for determining inflammatory pathologies and for undertaking routine preoperative assessments of patients scheduled for surgery. Along with providing leukocyte, neutrophil and platelet counts, haemoglobin concentration and haematocrit levels, the CBC also includes some parameters that are often overlooked, such as MPV and red cell distribution width. In common urological practice, leukocyte count is frequently used for assessing complicated urinary tract infections.6,7 Leukocytes are immune-system cells that are involved in protecting the body against infectious disease and foreign invaders. 8 The leukocyte count has also been reported to be increased in testicular torsion. 9 An increased leucocyte count as a result of torsion in the cremasteric muscle tissue surrounding testicular tissue has been reported in Wistar rats; this increase worsened hypoxia, reduced microperfusion and lowered the number of functional capillary vessels. 10 The current study similarly observed that the leukocyte count was significantly higher in patients with testicular torsion when compared with the control group.
Mean platelet volume is a parameter that provides information about the functional status of platelets. Assessing platelet volume and structure is helpful in diagnosing various blood disorders such as idiopathic thrombocytopenic purpura and myeloproliferative diseases.11,12 Larger platelets are both metabolically and enzymatically more active than other platelets and have a greater thrombotic potential due to their contents (i.e. thromboxane A2, P-selectin, and glycoprotein IIb/IIIa). 13 Elevated MPV values often indicate accelerated and increased production of young platelets (i.e. megakaryocytes), with a resulting disproportionate increase in the number of large hypercoagulable platelets. 14 Activated platelets also release antibacterial peptides. 15 According to some evidence, however, certain pathogens may have become able to exploit activated platelets and thus to establish or propagate infection by binding to platelet surfaces. 16 In addition, studies have reported some changes in MPV values in certain noninfectious inflammatory processes, possibly suggesting a role for MPV in reflecting the activity of certain inflammatory disorders.17–19
There is only one study in the published literature that specifically sought to investigate the relationship between testicular torsion and platelet count, clotting factors, and platelet activating factors. 20 This experimental study reported that platelet activation had a role in testicular ischaemia because ischaemia-related damage was reduced by the administration of antiplatelet activating factor. 20 To our knowledge, no previous study has examined the MPV in testicular torsion. However, the MPV has been reported to increase in urological vascular pathologies such as varicocele and erectile dysfunction.21,22 Furthermore, an increase in the MPV has also been observed in conditions like patent foramen ovale, myocardial infarction, and cerebral infarction.23–25 Therefore, increased MPV values have been considered a vascular risk factor. This current study found no significant difference between the two study groups with respect to platelet count and there was no correlation between MPV and platelet count. In contrast, the MPV was significantly higher in patients with testicular torsion compared with the control group.
Testicular torsion is characterized by the twisting of the spermatic cord around itself, leading to intratesticular venous congestion and interstitial oedema. This situation causes compression in the testicular microcirculatory system (venous–arteriolar) and alteration of the vascular diameter.26,27 Studies have shown that testicular venous congestion causes formation of vascular microthrombi that induce the intrinsic coagulation pathway.27,28 Furthermore, hypoxia has been shown to increase the formation of microthrombi and production of endothelin-1.29,30 Endothelin-1 is a vasoconstricting factor that acts via endothelin-A receptors. 30 The use of a selective endothelin-A antagonist during the reperfusion period improved the microcirculatory inflammatory reactions and caused an earlier return of pulsatile blood flow in rats. 30 These published findings suggest that the MPV is elevated in testicular torsion because this condition causes vascular pathology. 26–28
Erythrocyte sedimentation rate and C-reactive protein, which are acute-phase markers, have been shown to be elevated during the clinical examination leading to a diagnosis of testicular torsion.3,31 The MPV was shown to be significantly higher in patients with acute pancreatitis compared with control subjects, 32 but a significantly lower MPV was detected in paediatric patients with acute appendicitis. 33 This current study found a positive correlation between the MPV and leukocyte count, and the MPV had a greater specificity than the leukocyte count for diagnosing testicular torsion. These current findings suggest that the MPV may have been elevated as an acute-phase reactant. The sensitivity of the MPV to diagnose testicular torsion was low (62%) in the present study. To our knowledge, no previous study has examined MPV values in testicular torsion, so it is not possible to compare the present findings with those of other studies.
This current study had a number of limitations. First, it had a retrospective design. Secondly, it only had a small sample size. Thirdly, the control group consisted of healthy men without epididymo-orchitis, but the patient population included a low number of patients of similar age with epididymo-orchitis who underwent a complete blood count and scrotal Doppler ultrasonography. For these reasons, it is difficult to conclude how beneficial it might be to use the MPV as a parameter when excluding inflammatory and noninflammatory causes of testicular torsion. Further research in larger patients groups is required.
In conclusion, although it had a low sensitivity, the MPV may be a useful ancillary marker for diagnosing testicular torsion. Without further published reports on the role of testicular MPV values in relation to specific testicular inflammatory pathologies, the MPV might be helpful but not critical in the diagnosis of testicular torsion. The causes of the MPV elevation in testicular torsion should be more fully investigated in future studies.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
