Abstract
Objective
Cervical cancer (CC) is among the most prevalent malignancies globally. Public sequencing data indicate that PAX8-AS1 is associated with gynecological cancers, including CC, but its specific function and mechanism in cervical cancer remain unclear. The present study aimed to elucidate the role of PAX8-AS1 and its target axis in the CC.
Methods
A total of 104 CC patients were included. The levels of PAX8-AS1, miR-675-3p, and decorin (DCN) were quantified by quantitative reverse transcription polymerase chain reaction. Survival curve and Cox regression predicted prognostic factors. Proliferation and invasion/migration were assayed by CCK-8 and Transwell in CC cell lines. The target relationship was verified by dual-luciferase reporter assay and co-transfection.
Results
PAX8-AS1 was declined in CC tumor tissue and cell lines. PAX8-AS1 was an independent prognostic factor. PAX8-AS1 expression was associated with the CC pathological including the international federation of gynecology and obstetrics staging system (FIGO), tumor size, and lymph node metastasis. Patients with higher PAX8-AS1 levels had better survival outcomes. Upregulation of PAX8-AS1 inhibited the CC cell invasion, migration, and proliferation. miR-675-3p was predicted and verified as sponged of PAX8-AS1. miR-675-3p was negatively related to PAX8-AS1. PAX8-AS1 impeded the CC cellular function by regulating miR-675-5p. DCN was confirmed as the target of miR-675-5p. DCN was negatively and positively related to miR-675-3p and PAX8-AS1, respectively. PAX8-AS1 suppressed the CC cell invasion, migration, and proliferation by targeting miR-675-3p/DCN axis.
Conclusion
In summary, PAX8-AS1 was related to tumor progression and inhibited CC development. As a potential biomarker, PAX8-AS1 impeded CC cell invasion, migration, and proliferation by regulating the axis of miR-675-3p/DCN.
Introduction
Cervical cancer (CC) is a prevalent malignant tumor among middle-aged and elderly women, with morbidity and mortality rates ranking high among various cancers. 1 In 2020, over 600,000 new patients of CC were diagnosed globally, with more than half of those cases resulting in death. 2 Compared to previous years, both the incidence and mortality rates of CC have risen, with an increasing trend in younger populations. 1 According to statistics, the majority of CC cases occur in developing countries. 3 In particular, China, with its large population, uneven regional development, and complex healthcare environment, faces a significant challenge in addressing CC as a serious threat to women's health. 4 Early-stage CC is primarily treated with surgical resection, while late-stage disease is managed with radiotherapy and chemotherapy. The prognosis largely depends on the clinical stage at which treatment begins. 5 The irreversible side effects of radiotherapy and chemotherapy highlight the need for novel, effective treatment strategies to improve patient prognosis. 6 However, there remains a lack of reliable biomarkers that can effectively guide therapeutic decisions and predict prognosis in CC. Therefore, identifying new biomarkers and thoroughly investigating their mechanisms is crucial for improving treatment outcomes and patient survival in CC.
Long non-coding RNAs (lncRNAs) influence disease onset and progression through various mechanisms, such as alternative splicing, transcriptional activation, protein interaction, and modulation of mRNA stability. 7 MicroRNAs (miRNAs) are pivotal regulators that control RNA levels by inducing messenger RNA (mRNA) degradation and inhibiting translation. 8 Notably, lncRNAs can interact with miRNAs and compete for binding to their target mRNAs, thereby alleviating the repressive effects of miRNAs on gene expression.7,8 This interplay affects protein levels and cellular functions, leading to alterations in various biological processes. For instance, linc00958 targets miR-185-5p to regulate the AKT1/GSK3β pathway, inhibiting oncogenesis and angiogenesis in a xenograft experiment of CC. 9 Similarly, high expression of LNC-POTEM-4 correlates with poor prognosis, and it promotes liver cancer cell activities, and epithelial-mesenchymal transition by targeting miR-149-5p. 10 In thyroid cancer, restoring ACTA2-AS1 expression inhibits cell proliferation and invasion, with ACTA2-AS1 acting as a tumor suppressor through sponged miR-4428. 11 These examples underscore the significant potential of non-coding RNAs in disease diagnosis, therapy, and prognosis.
PAX8-AS1, located on chromosome 2q14.1, has attracted attention as a candidate gene for CC following recent GWAS meta-analyses, 12 prompting further investigation into its role. Additionally, PAX8-AS1 has been identified as a key lncRNA in endometriosis, with its expression found to be downregulated in this condition. 13 Literature suggests a close association between PAX8-AS1 and the malignant progression of ovarian cancer, and it has been proposed as a prognostic factor in multi-genomic analyses of adrenocortical carcinoma. 14 However, the specific function and mechanism of PAX8-AS1 in CC remain poorly understood. Some studies have indicated that miR-675-3p may be involved in neoplastic growth and resistance to therapy.15–17 For instance, immune infiltration analysis has suggested that miR-675-3p is a critical factor in ovarian cancer, 16 while bioinformatics analysis of public datasets has identified it as a potential biomarker for melanoma. 17 During our database search, we discovered target binding sites for PAX8-AS1 and miR-675-3p, raising the possibility that PAX8-AS1 may regulate CC progression via miR-675-3p. However, this hypothesis requires further validation.
In brief, the study explored the potential role of PAX8-AS1 and its regulatory axis in CC by examining the levels of PAX8-AS1/miR-6675-3p and conducting cell function assays. The findings will promote the comprehension of the pathogenesis of CC and targeted therapeutic strategies.
Material and methods
Volunteer recruitment
From June 2017 to June 2019, 104 CC volunteers were recruited from the Department of Obstetrics and Gynecology at Northwest University First Hospital. The diagnosis of CC was confirmed based on pathological examination. All participants gave their signed written consent. The study was approved by the hospital’s Ethics Committee (Approval no. 2019036). Follow-up data were collected over a period of 5 years.
Collection of samples
The inclusion criteria for patients were as follows: (a) diagnosis of CC based on pathological examination; (b)o prior conization of the cervix, cryosurgery, or preoperative chemoradiotherapy; (c) absence of other malignant tumors or severe underlying diseases. Sample were collected from the tumor and adjacent tissue. Tumor and adjacent tissue samples were obtained using separate instruments. The specimens were washed with phosphate-buffered saline (PBS) (Solarbio, Beijing, China) and immediately immersed in TRIzol reagent. Following thorough homogenization at low temperatures, total RNA was extracted and stored at −80°C.
Cell cultured
Five CC cell lines (CaSki, C33A, C4-1, HeLa, ME180) were purchased from ATCC (Manassas, VA, USA), and the human cervical epithelial cell line was obtained from ScienCell (Carlsbad, CA, USA). The HeLa cell line was cultured in high-glucose Dulbecco’s modified eagle medium (DMEM) (Hyclone, Logan, UT, USA) supplemented with 10% fetal bovine serum (FBS) (Hyclone), while the other cell lines were cultured in RPMI-1640 (Hyclone) medium with 10% FBS. All cells were cultured at 37°C with 5% CO2. Mycoplasma contamination (Servicebio, Wuhan, China) was tested every 3 months.
Cell transfection
Plasmids used for cell transfection were purchased from Genepharma (Shanghai, China). The overexpression plasmid for PAX8-AS1 was constructed using the pcDNA3.1 vector containing the target lncRNA sequences. A short hairpin RNA (shRNA) targeting PAX8-AS1 was constructed using the pGCsiLence 4.1-CMV vector for lncRNA knockdown. Additionally, miRNA inhibitors, mimics, and corresponding negative controls were provided by Genepharma. For transfection, 1 × 105 cells/well were seeded in 24-well plates. When cells reached approximately 70% confluence, transfection was carried out using Lipofectamine 3000 (Invitrogen, Carlsbad, CA, USA) according to the manufacturer's instructions.
Quantitative reverse transcription polymerase chain reaction
Total RNA was extracted using TRIzol (Thermo, Waltham, MA, USA). RNA concentration was determined using NanoDrop 2000 spectrophotometer (Thermo). Reverse transcription and polymerase chain reaction (PCR) mixture were prepared with PrimeScript RT reagent kit and TB Green Premix Ex Taq II kit (Takara, Dalian, China). Quantitative reverse transcription PCR was undertaken using the ABI 7500 Real-Time PCR System (Applied Biosystems, Waltham, MA, USA). Relative abundance of molecular was calculated using the 2−ΔΔCt method, with GAPDH or U6 as the internal reference.
Dual-luciferase reporter assay
The ENCORI database was used to predict the putative binding sites. The PAX8-AS1 or decorin (DCN) sequence containing miR-675-3p binding sites, along with their mutated versions, were cloned into the pmirGLO vector to establish constructs (WT/MT-PAX8-AS1 and WT/MT-DCN) (GenePharm, Pallini, Greece). Co-transfection of the constructs and miR-mimic/NC was performed by Lipofectamine 2000 (Invitrogen). Relative luciferase activity was measured using the Dual-Luciferase Reporter Assay System (Promega, WI, USA).
CCK-8 assays
The transfected cells with 2 × 103 cells/well were seeded into the 96-well plates. The different time points (0, 24, 48, 72 h) of proliferation were measured. 100μL medium was supplemented with 10μL CCK-8 (Beyotime, Shanghai, China) and incubated in the cell incubator. After 2 h, the optical density value at 450 nm was recorded using microplate absorbance reader (BioTek, Winooski, VT, USA).
Transwell assays
Cell migration and invasion assays were performed using 24-well Transwell chambers with 8-µm pore size polycarbonate membranes (Corning, NY, USA). For the invasion assay, the upper chambers were pre-coated with Matrigel (BD Bioscience, San Jose, CA, USA). Cells (4 × 104 for migration assays or 1 × 105 for invasion assays) were seeded into the upper chamber in serum-free medium. The lower chamber was filled with medium containing 20% FBS as a chemoattractant. The non-migrated or non-invaded cells on the upper side of the membrane were gently removed with a cotton swab. Cells that had migrated or invaded the lower surface of the membrane were fixed with 4% paraformaldehyde for 20 min, stained with 0.1% crystal violet (Solarbio) for 10 min, and washed with PBS. The stained cells were then counted under a microscope.
Statistical analysis
Data were expressed as means ± standard deviation for continuous variables and as frequencies for categorical variables. Statistical analysis was performed using GraphPad Prism 9.0 and R version 4.0.0. The t-test and chi-square test were used to compare the two groups. One-way of variance with Bonferroni's post hoc test was applied for comparisons among multiple groups. Survival analysis was performed using Kaplan–Meier curves and the Log-rank test. Cox univariate analysis was used to evaluate hazard ratios. P < 0.05 was considered statistically significant.
Results
PAX8-AS1 was decreased in CC
The abundance of PAX8-AS1 was significantly decreased approximately 25% in adjacent tissues compared to the tumor tissue (Figure 1(a), P < 0.05). PAX8-AS1 was an independent prognostic factor, alongside the international federation of gynecology and obstetrics staging system (FIGO) stage, lymph node metastasis, and recurrence (Figure 1(b), P < 0.05). Patients with higher PAX8-AS1 expression had better survival probabilities versus those with lower expression (Figure 1(c), P < 0.05). The levels of PAX8-AS1 were reduced across all CC cell lines, with the most pronounced decreases observed in C33A and C4-1 cells (Figure 1(d), P < 0.05), which were used for the following study. Based on the median abundance of PAX8-AS1, the samples were divided into high and low-level groups. PAX8-AS1 was associated with features including FIGO (P = 0.018), tumor size (P = 0.030), and lymph node metastasis (P = 0.019). There was no significant correlation with characteristics including age, differentiation, and recurrence (Table S1, P > 0.05).
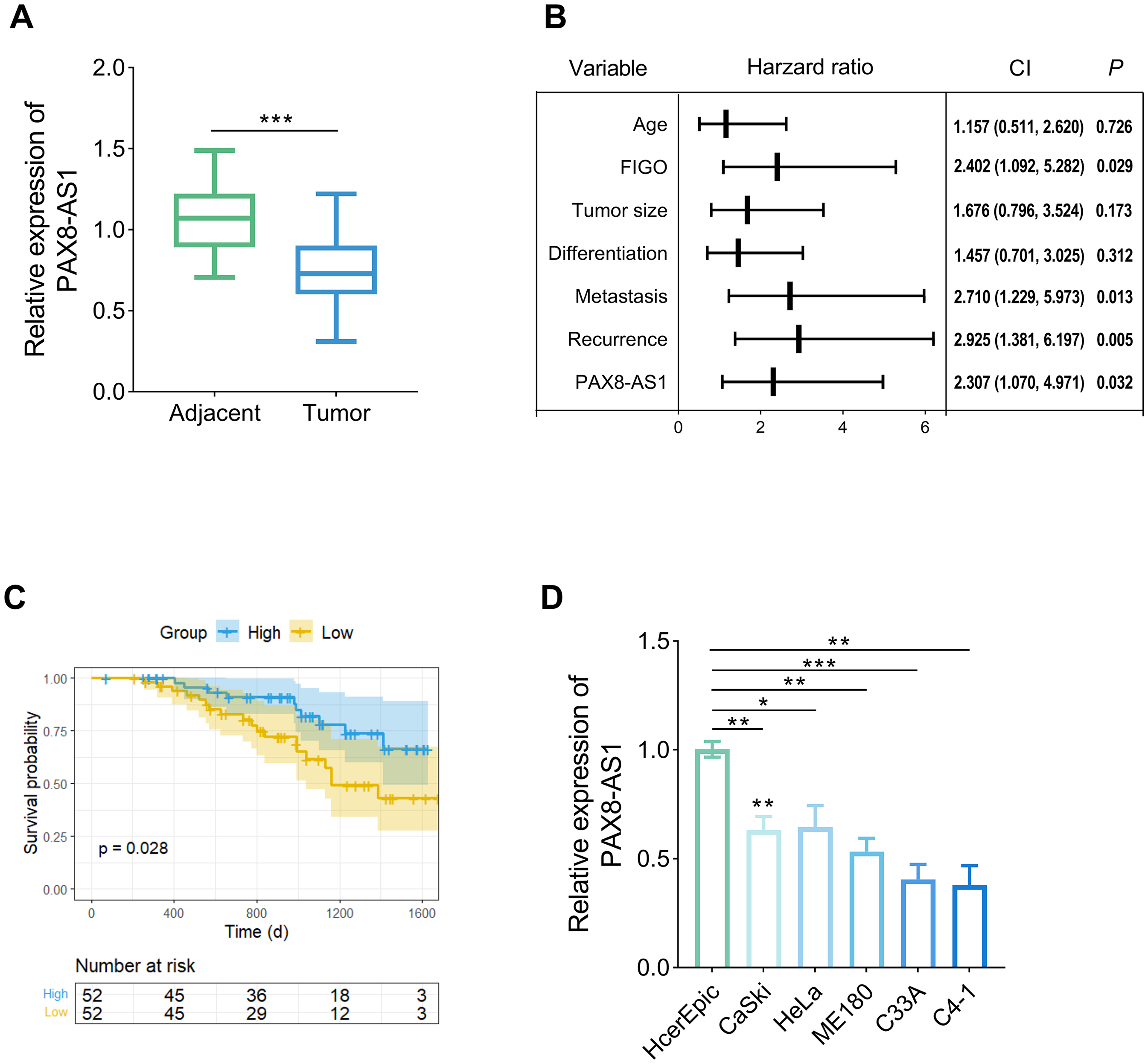
PAX8-AS1 was significantly decreased in the CC. (a) The abundance of PAX8-AS1 was reduced in the tumor tissue of the CC patients (n = 104). (b) PAX8-AS1 was an independent prognostic factor based on the Cox regression analysis. (c) Survival analysis showed that patients with higher PAX8-AS1 expression had better survival probabilities. (d) The expression of PAX8-AS1 declined in the CC cell lines (n = 3). Data are presented as the mean ± SD. *P < 0.05, **P < 0.01, ***P < 0.001 vs. adjacent/HcerEpic cell.
Overexpression of PAX8-AS1 impedes the invasion, migration, and proliferation
Following treatment with OE-PAX8-AS1, there was a significant elevation in the levels of PAX8-AS1, exceeding double the original levels, which confirmed the success of the transfection (Figure 2(a), P < 0.05). The overexpression of PAX8-AS1 markedly reduced the invasive and migratory capabilities of both the C33A and C4-1 cell lines (Figure 2(b) and (c), P < 0.05). Concurrently, the cell proliferation exhibited a decline in the overexpression group over time (Figure 2(d), P < 0.05).
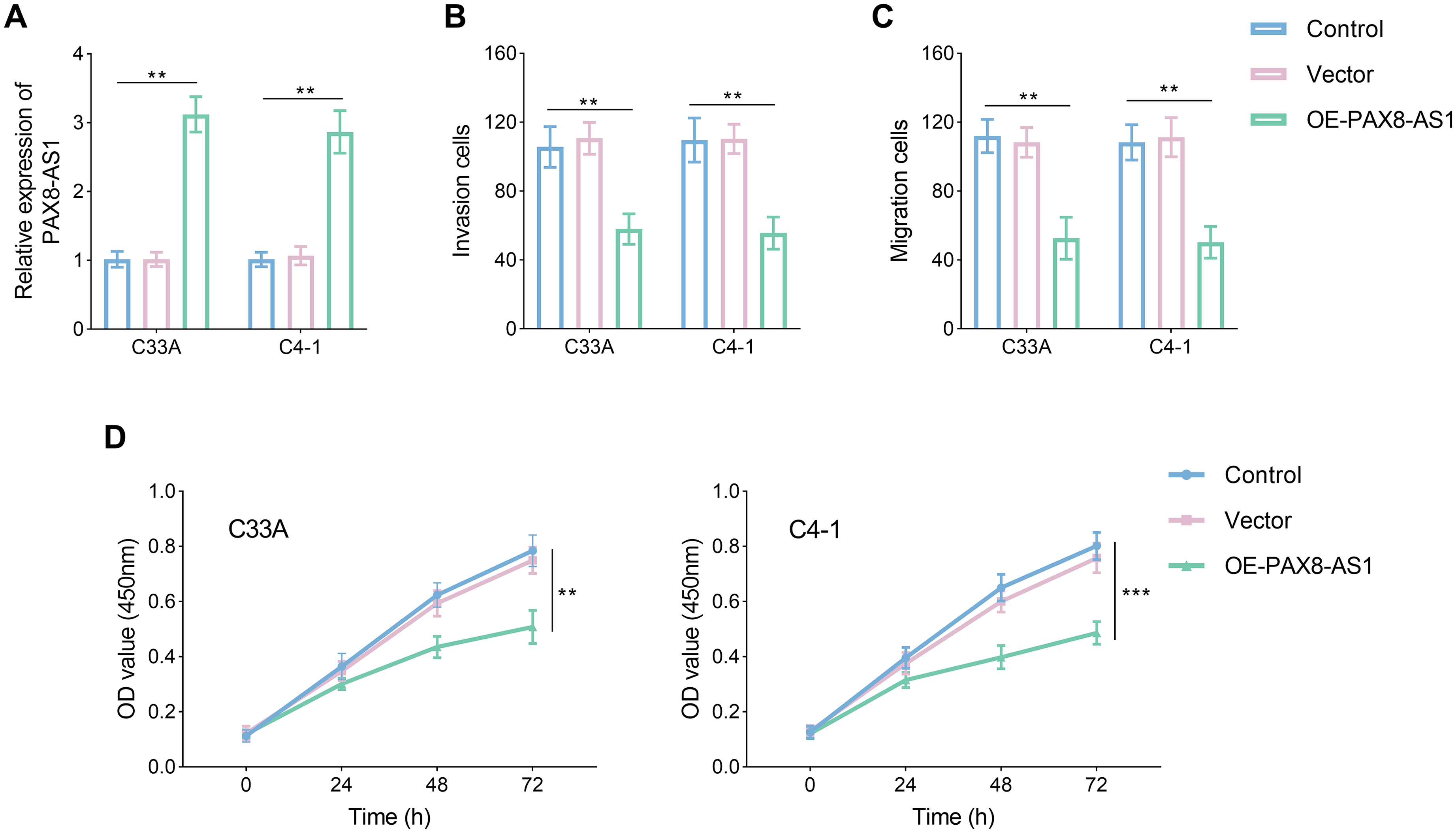
PAX8-AS1 inhibited the invasion, migration, and proliferation in the CC cell lines. (a) PAX8-AS was increased after treatment OE-PAX8-AS1. (b) to (d) Upregulation of PAX8-AS impeded the invasion (b), migration (c), and proliferation (d) of CC cell lines. Data are presented as the mean ± SD (n = 3). **P < 0.01, ***P < 0.001 vs. control.
PAX8-AS1 acts as a sponge for mir-675-3p
The potential binding sites between PAX8-AS1 and miR-675-3p were identified, as depicted in Figure 3(a). This putative interaction was subsequently validated. The dual-luciferase assay revealed an increase in relative luciferase activity following the introduction of miR-mimics in both the C33A and C4-1 cell lines, with this change observed exclusively in the WT-PAX8-AS1 group (Figure 3(b) and (c), P < 0.05). In contrast to the expression patterns of lncRNA in tissue samples, miR-675-5p expression was elevated by approximately 30% in tumor tissues (Figure 3(d), P < 0.05). The negative correlation was observed between PAX8-AS1 and miR-675-3p (Figure 3(e), P < 0.05). Under transfection conditions, miR-675-5p was reduced by more than one-half (Figure 3(f), P < 0.05).
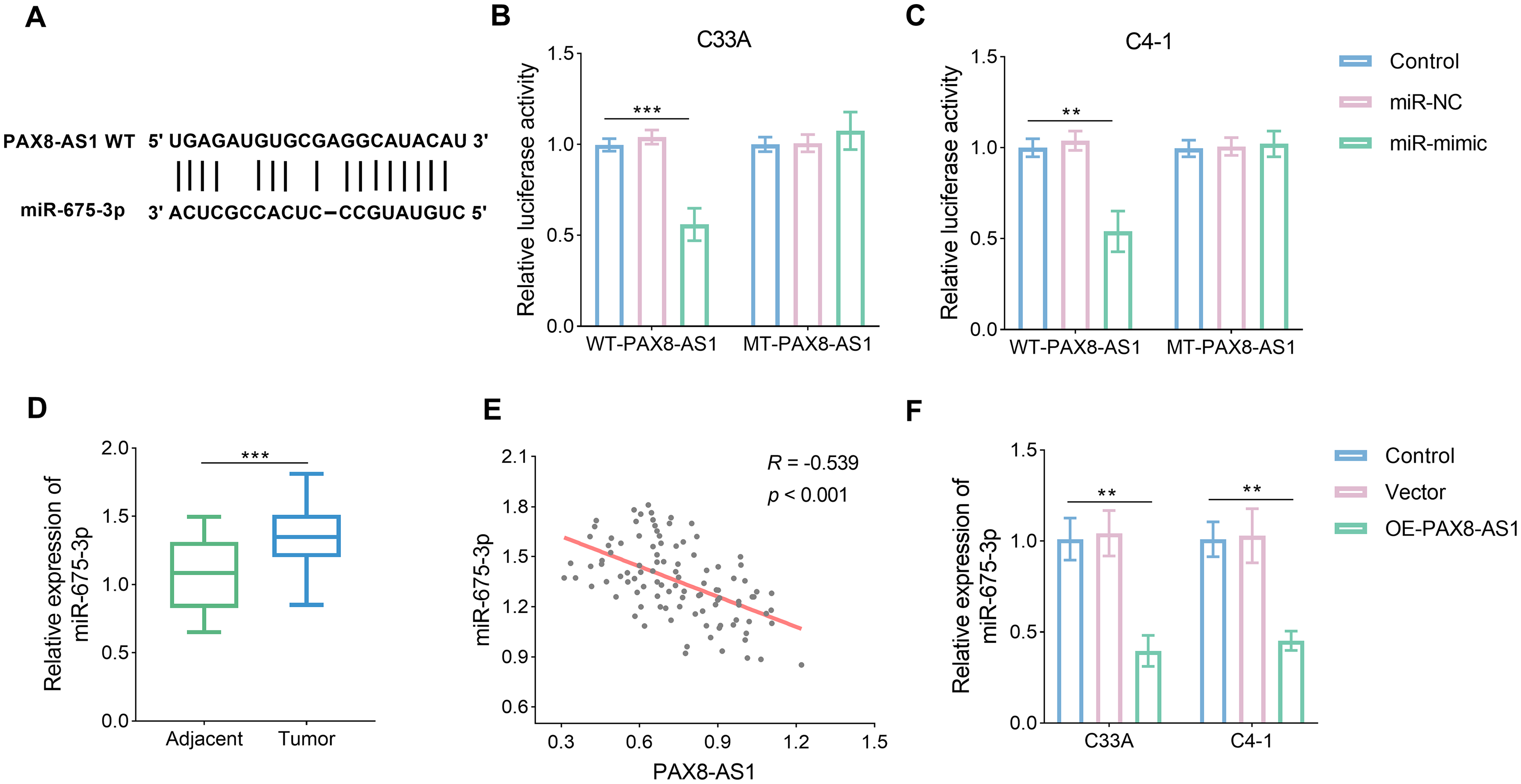
PAX8-AS1 targeted the miR-675-3p. (a) The potential binding sites were predicted according to ENCORI database. (b) and (c) The relationship between PAX8-AS1 and miR-675-3p was verified by dual-luciferase (n = 3). (d) miR-675-3p was increased in the CC tumor tissue. (e) miR-675-3p was negatively related to PAX-AS1 based on Pearson correlation (n = 104). (f) miR-675-3p was decreased in the CC cell lines after treatment with OE-PAX8-AS1 (n = 3). Data are presented as the mean ± SD. **P < 0.01, ***P < 0.001 vs. control.
PAX8-AS1 inhibits the invasion, migration, and proliferation of choriocarcinoma by targeting mir-675-3p
After transfection with sh-PAX8-AS1, knockdown efficiency was assessed. The results revealed an inverse relationship between PAX8-AS1 and miR-675-3p expression. PAX8-AS1 decreased by approximately 50%, while miR-675-3p levels more than doubled (Figure 4(a) and (b), P < 0.05). In terms of cellular activity, inhibition of miR-675-3p expression led to decreased migration, invasion, and proliferation (Figure 4(c) and (d), P < 0.05). However, the addition of additional sh-PAX8-AS1 material reversed these effects, restoring cell activity. Notably, sh-PAX8-AS1 significantly increased cell migration, invasion, and proliferation (Figure 4(e), P < 0.05).
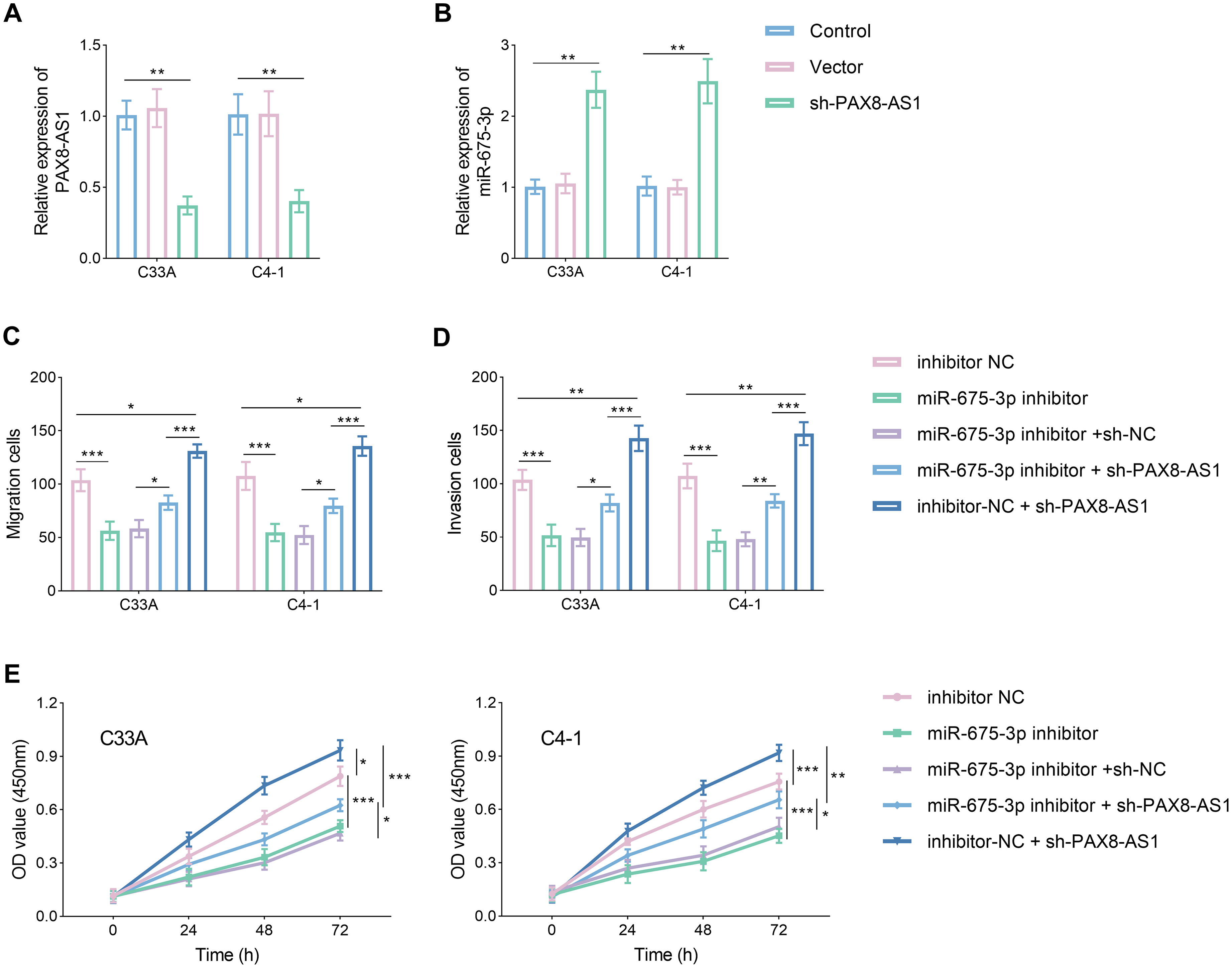
PAX8-AS1 impeded the CC cell invasion, migration, and proliferation by regulating miR-675-3p. (a) and (b) PAX8-AS was decreased (a) and miR-675-3p was increased (b) after silencing PAX8-AS1. (c) to (e) PAX8-AS1 inhibits the invasion (c), migration (d), and proliferation (e) of CC cell lines by targeting miR-675-3p. Data are presented as the mean ± SD (n = 3). *P < 0.05, **P < 0.01, ***P < 0.001 vs. control.
DCN is a target of mir-675-3p
To investigate the comprehensive regulatory axis of PAX8-AS1/miR-675-3p, the target of miR-675-3p was predicted using the ENCORI database. The prediction identified potential interaction sites between miR-675-3p and DCN (Figure 5(a), P < 0.05). This putative target relationship was subsequently validated. The assay revealed a significant decrease in relative luciferase activity only in the WT-DCN group following treatment with miR-mimic (Figure 5(b) and (c), P < 0.05). Furthermore, the levels of DCN were found to be reduced in tumor tissues, mirroring the pattern observed for lncRNA PAX8-AS1 (Figure 5(d), P < 0.05). The expression of DCN exhibited a negative correlation with miR-675-3p and a positive correlation with PAX8-AS1 (Figure 5(e) and (f), P < 0.05).
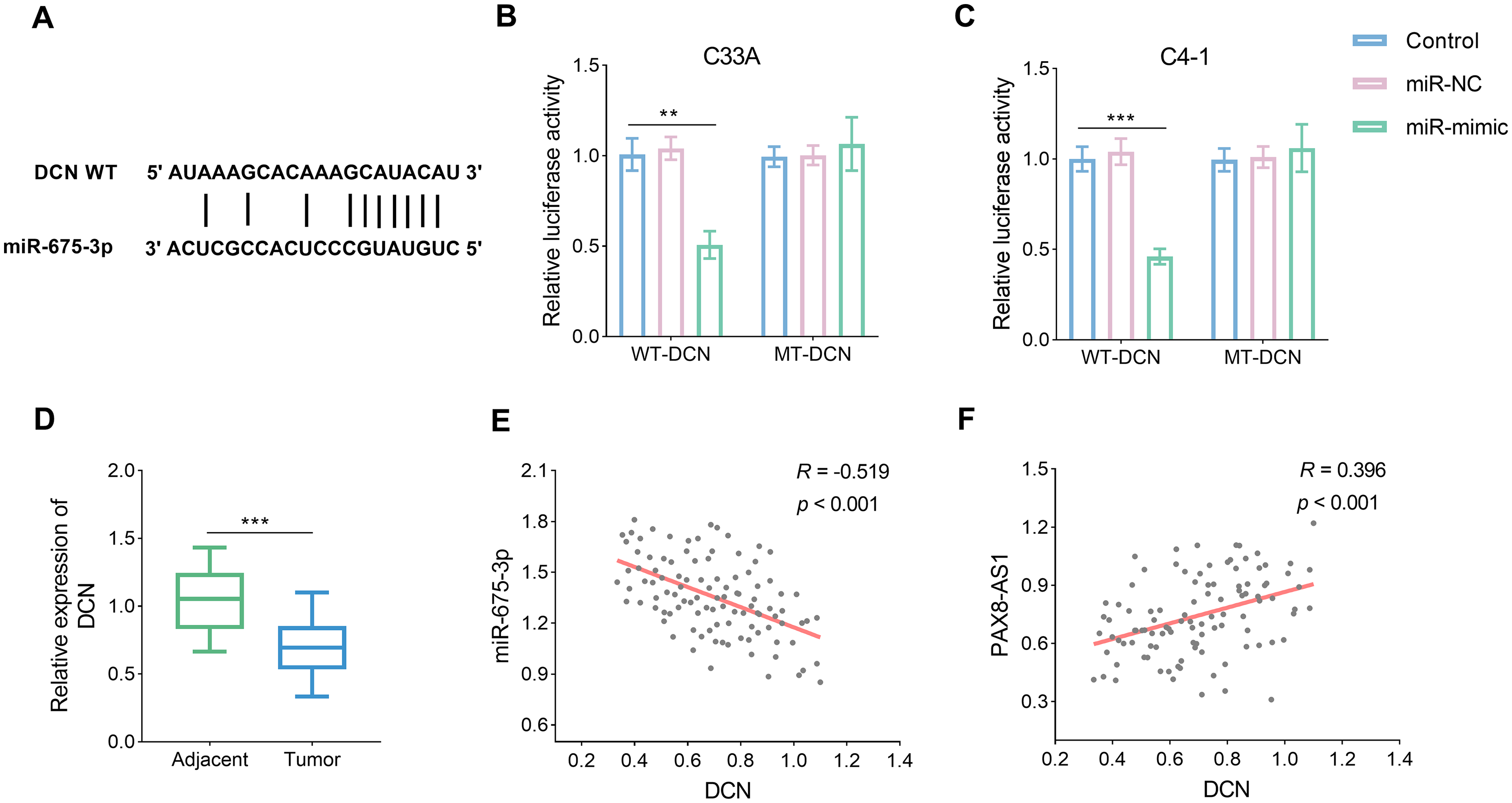
miR-675-3p targeted the DCN. (a) The potential binding sites between miR-675-3p and DCN were predicted. (b) and (c) The targeted link between miR-675-3p and DCN was confirmed by dual-luciferase (n = 3). (d) DCN was reduced in the CC tumor tissue (n = 104). (e) to (f) DCN was negatively and positively associated with miR-675-3p (e) and PAX8-AS1 (f), respectively. Data are presented as the mean ± SD. **P < 0.01, ***P < 0.001 vs. control.
Discussion
CC is among the most prevalent and lethal malignancies affecting women, posing a significant threat to their health. 2 Despite advances in diagnosis and treatment, an ideal tumor marker for CC remains unidentified. Therefore, identifying molecules whose expression changes during CC aggravation and exploring potential biomarkers is critical for improving early diagnosis and discovering therapeutic targets. The balanced expression of lncRNAs is essential for maintaining cellular homeostasis and function. 18 Disruption of lncRNA expression is closely linked to various diseases, including cancer. For instance, lncRNA SFTA1P is upregulated in CC and promotes its progression, with high expression correlating with poor prognosis. 19 Upregulation of lncRNA ABHD11-AS1 accelerates CC tumor growth through activation of the EGFR. 20 This study confirmed that lncRNA PAX8-AS1 was significantly downregulated in CC tissues, a finding that was further confirmed in CC cell lines. A study has shown that PAX8-AS1 is down-regulated in ovarian neoplastic, where it regulates cancer development through the Hippo signaling pathway. 21 Upregulation of PAX8-AS1 suppresses cell proliferation and promotes apoptosis in papillary thyroid carcinoma. 22 A recent study has identified PAX8-AS1 as a prognostic factor in thyroid cancer. 23 Although some studies have indicated a potential association between PAX8-AS1 and CC,12,24 no research has explored its prognostic value or its role in the progression of CC. In this study, patients with high expression of PAX8-AS1 had better survival outcomes compared to those with low expression. Cox regression analysis further indicated that PAX8-AS1 serves as an independent prognostic regulator. Additionally, PAX8-AS1 expression was correlated with various pathological characteristics, including FIGO, tumor size, and lymph node metastasis. These clinical indicators, which reflect the extent of cancer progression, suggest that PAX8-AS1 expression is closely associated with the malignancy of the tumor. These findings suggested that PAX8-AS1 may serve as a promising biomarker and could be involved in CC progression.
Malignant cell migration and invasion are key characteristics of tumor biology and are central to metastasis, the leading cause of cancer-related mortality. 25 Carcinoma is a disease characterized by abnormal and uncontrolled cell proliferation, which increases the burden on the body and contributes to detrimental effects on the host. 26 The regulatory mechanisms of lncRNAs play a critical role in modulating cellular activities. For example, lncRNA HOXC-AS3 promotes CC cell proliferation and tumor growth in vivo, 27 while downregulation of HOTAIR reduces cell growth, migration, and invasion in CC cell lines. 28 The role of PAX8-AS1 in CC cell activity was investigated. Restoring PAX8-AS1 expression inhibited migration, invasion, and proliferation of CC cells, demonstrating that PAX8-AS1 functions to impede the progression of CC cell activities.
The regulatory mechanisms involving lncRNAs are complex. lncRNAs can act as “sponges,” competing binding to miRNAs, thereby reducing miRNA-mediated regulation of their target mRNAs. 8 It is reported the MALAT1/miR-124/SIRT1 axis has been shown to inhibit CC development by mediating pyroptosis. 29 STARD7-AS1 suppresses CC cell proliferation and promotes autophagy through the miR-31-5p/TXNIP axis. 30 The bioinformatics tool was used to identify potential targets of PAX8-AS1 and found that miR-675-3p is a likely candidate. The interaction between PAX8-AS1 and miR-675-3p was confirmed. Additionally, a negative correlation between their expression was observed. Upregulation of miR-675-3p promotes cell proliferation in colorectal cancer 31 and enhances migration in esophageal carcinoma. 32 In the cell functional assays of this study, down-regulated of miR-675-3p hindered CC cell proliferation, migration, and invasion. However, co-transfection with PAX8-AS1 reversed these effects. These results indicated that miR-675-3p is a direct target of PAX8-AS1 and that PAX8-AS1 suppresses CC cell proliferation, invasion, and migration by inhibiting miR-675-3p expression. Furthermore, the target of miR-675-3p was also predicted, and identified DCN, a member of the small leucine-rich proteoglycan family, as a potential target. DCN is primarily located in stromal and epithelial cells.33,34 A study has shown that DCN expression is higher in normal liver tissue compared to tumor tissue. 35 In breast cancer, downregulation of DCN activates stromal fibroblasts, promoting angiogenesis and in situ tumor growth. 36 The downregulated of DCN expression was verified in CC, and dual-luciferase assays confirmed the interaction between miR-675-3p and DCN. These results suggested that the tumor-suppressive effects of PAX8-AS1 in CC may be mediated through the miR-675-3p/DCN axis. However, this study has some limitations. It was conducted exclusively in vitro using cell models, and in vivo validation using animal experiments is needed to further confirm these findings. Future studies should also explore the signaling pathways involved in PAX8-AS1 regulation to better understand its role in CC progression.
Conclusion
In conclusion, PAX8-AS1 was associated with malignancy of the tumor. This study demonstrated that PAX8-AS1 inhibits CC cell proliferation, migration, and invasion. PAX8-AS1 suppressed CC progression by regulating the miR-675-3p/DCN axis. These findings suggested that PAX8-AS1 may serve as a promising biomarker for CC and provide valuable insights for developing new therapeutic strategies to combat the disease.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155251386471 - Supplemental material for lncRNA PAX8-AS1 suppresses cervical cancer development by regulating miR-675-3p/DCN axis
Supplemental material, sj-docx-1-jbm-10.1177_03936155251386471 for lncRNA PAX8-AS1 suppresses cervical cancer development by regulating miR-675-3p/DCN axis by Xiayang Lu, Peng Song and Qingfen Chen in The International Journal of Biological Markers
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
