Abstract
BACKGROUND:
Long noncoding RNAs (lncRNA) have been verified to be involved in hepatocellular carcinoma (HCC) progression. However, the potential biologic function of PVT1 in HCC is not still fully known.
METHODS:
PVT1 and miR-214 were detected by qRT-PCR assays in HCC tissues and adjacent normal tissues. CCK8, cell colony and transwell invasion assays were performed to evaluate cell proliferation and invasion abilities. Western-blot assay was performed to detect the protein of E-cadherin and Vimentin. QRT-PCR, RNA immunoprecipitation (RIP) and chromatin immunoprecipitation (ChIP) assays demonstrated PVT1 regulated miR-214 expression.
RESULTS:
The results showed that PVT1 was increased in HCC tissues and higher PVT1 expression was associated with tumor size, histological differentiation grade and advanced TNM stage. Furthermore, we revealed that PVT1 promoted cell proliferation and invasion in HCC. RIP and ChIP assays demonstrated that PVT1 significantly inhibited miR-214 expression by interacting with enhancer of zeste homolog 2 (EZH2).
CONCLUSIONS:
Thus, these results demonstrated that PVT1/EZH2/miR-214 regulatory pathway might serve as new target for HCC treatment.
Introduction
Hepatocellular carcinoma (HCC) is the predominant subject of liver malignancies and ranks as the fifth most common malignancy and the second leading cause of cancer death deaths globally [1]. Surgical operation, liver transplantation, and radiofrequency ablation are the effective methods for HCC treatment and the 5-year survival rates higher than 50% can be achieved [2]. However, the prognosis of advanced HCC remains poor outcome, due to the late detection of the tumors and high rate of recurrence and metastasis [3]. Therefore, further investigation of the molecular mechanisms underlying HCC progression is needed for the development of effective treatment for HCC.
Advanced studies have shown that long non-coding RNAs (lncRNAs), longer than 200 nt in length and with a lack of capability for protein coding, play a critical biological role in carcinogenesis and development [4]. Misregulated lncRNA expressions are identified as major contributors to tumorigenesis [5, 6]. Some literatures indicate lncRNAs can be used for cancer diagnosis and prognosis, and serve as potential target of treatment in tumors. In hepatocellular carcinoma, some differentially expressed lncRNAs including highly upregulated in liver cancer (HULC) [7], maternally expressed gene 3 (MEG3) [8], growth arrest-specific 5 (GAS5) [9], antisense non-coding RNA in the INK4 locus (ANRIL) [10], and so on, have been highlighted as tumor promoter or suppressor in HCC.
The plasmacytoma variant translocation 1 gene (PVT1) is 1,716 nt in length and located in the chr8q24.21 region. PVT1 has been identified as an oncogene due to its contribution to the phenotype of multiple cancers. For example, gastric cancer [11], non-small cell lung cancer [12], acute promyelocytic leukemia [13], bladder cancer, thyroid cancer [14]. PVT1 has been discovered to be associated with HCC progression and elucidated PVT1 promoted proliferation and stem cell-like property of hepatocellular carcinoma cells by stabilizing NOP2 [15]. Ding et al. pointed out that that PVT1 was associated with tumor progression and predicted recurrence in hepatocellular carcinoma patients [16]. Another study focused on that the PVT1 and uc002mbe.2 in sera provide a new supplementary method for hepatocellular carcinoma diagnosis [17]. However, the role of biology and clinical signification of PVT1 in HCC need to be well investigated.
In our study, the results showed PVT1 was notably upregulated in HCC tissues and PVT1 silencing inhibited HCC cell proliferation and invasion. Furthermore, we showed that PVT1 recruited EZH2 to miR-214 promoter and inhibited miR-214 expression. Based on the above findings, the results indicated that PVT1/EZH2/ miR-214 regulatory axis might serve as new target for HCC treatment.
Materials and methods
Patient tissue samples
The 92 cases HCC tissues and non-tumorous normal tissues specimens were obtained from patients who had underwent radical resection at between Feb 2010 and March 2014 at West China School of Preclinical and Forensic Medicine, Sichuan University, All of the patients were diagnosed as HCC. The tumor stage was staged according to the seventh edition of the AJCC Cancer Staging Manual by histopathological evaluation. No radiotherapy or chemotherapy treatment was performed for patients before the operation. All tissues specimens were immediately frozen in liquid nitrogen and stored at
Cell culture
Two Human HCC cells (SMCC-7721, Huh-7 and SK-hep-1) and a normal liver cell LO2 were purchased from the Institute of Biochemistry and Cell Biology of the Chinese Academy of Sciences (Shanghai, China). Cells were maintained in Dulbecco’s modified Eagle’s medium (DMEM) with 10% fetal bovine serum (Thermo Scientific HyClone, Beijing, China), 100 U/ml penicillin and 100 mg/ml streptomycin in humidified air at 37
Cell transfection
Cells (1
Cell proliferation assay
1
Cell invasion assay
Cell invasion were assessed by transwell chambers precoated with Matrigel (BD, Franklin Lakes, NJ, USA; without Matrigel for cell migration assay). After cells incubation for 48 hours, cells on the upper surface of inserts were removed, and cells on the lower surface were fixed and then stained with 0.5% crystal violet solution. Cells were counted in five random fields in each well.
RNA extraction and real-time quantitative PCR analysis (qRT-PCR)
Total RNA was extracted from cultured cells or tissues using TRIzol Reagent (Takara, Dalian, China). The reaction mixture (20
PVT1-forward: 5
Western blot analysis
Protein samples were harvested from cells and were lysed in RIPA buffer and concentration was detected using BCA kit (Thermo Fisher Scientific). Equal amounts (45
RNA immunoprecipitation (RIP)
RIP was performed in SMCC-7721 cells as previously described method [18]. RNA immunoprecipitation (RIP) assays were performed using the Magna RIP RNA-Binding Protein Immunoprecipitation Kit (Millipore) following manufacturer’s instructions. Total cell lysate was used for immunoprecipitation with EZH2 antibodies (same as for Western blotting). Rabbit IgG antibody was as a control.
Chromatin immunoprecipitation (ChIP)
ChIP was performed in SMCC-7721 cells as previously described method [19]. The supernatants were subjected to immunoprecipitation overnight with antibodies against EZH2 (CST, USA) or with isotype rabbit IgG (CST, USA) at 4
Statistical analysis
The SPSS18.0 software was used for statistical analysis. The significance of differences between groups was performed using Student’s t-test, oneway analysis of variance (ANOVA),
Correlation of PVT1 expression with clinicopathological parameters
Correlation of PVT1 expression with clinicopathological parameters
**
PVT1 expression was increased in HCC tissues and cell lines. (A) QRT-PCR was used to evaluate the expression of PVT1 in HCC tissues compared with adjacent normal HCC tissues. GAPDH was used as a reference control. (B) PVT1 was upregulated in three HCC cell lines (SMCC-7721, Huh-7 and Sk-hep-1) compared with normal LO2 cell line. GAPDH was used as a reference control. (C) The expression of PVT1 was detected after PVT1 silencing by two si-PVT1 oligos in SMCC-7721 cells. GAPDH was used as a reference control. (D)The expression of PVT1 was detected after PVT1 overexpression by pcDNA3.1-PVT1 plasmid in Sk-hep-1 cells. GAPDH was used as a reference control. The data derived from the results of 3 independent experiments, **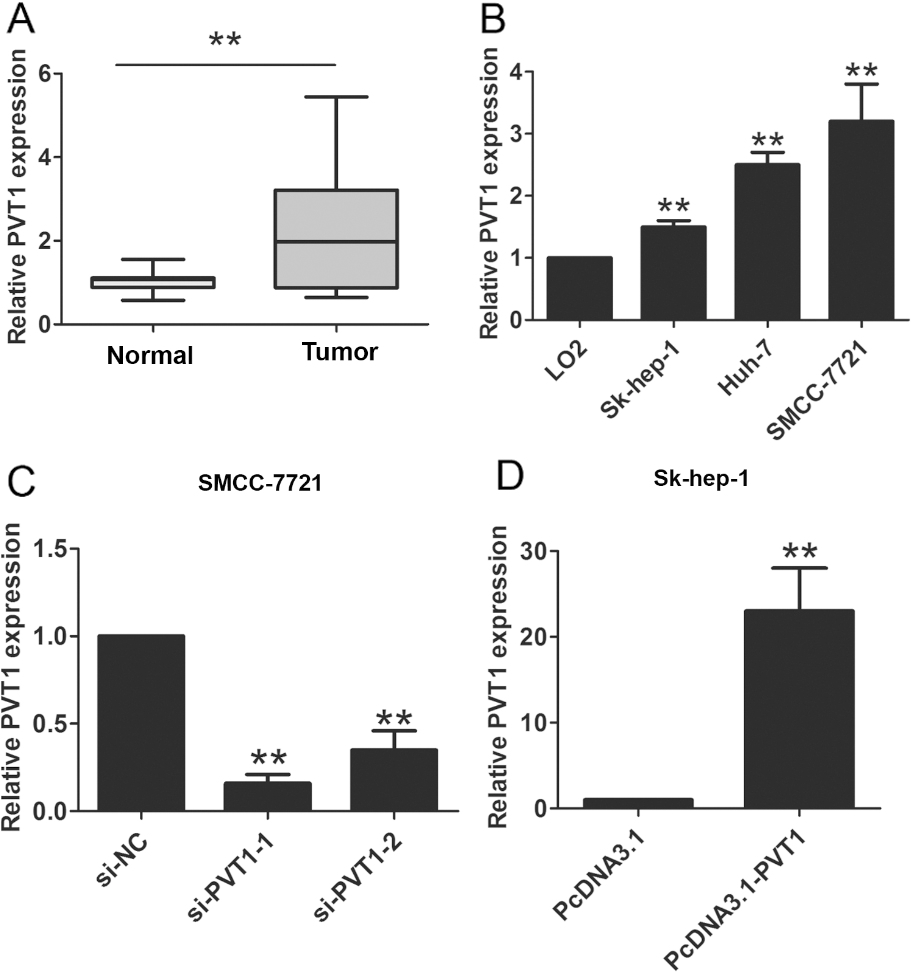
PVT1 promoted HCC cells proliferation and invasion. (A) CCK8 cell proliferation assays were performed after introducing si-PVT1 or si-NC into SMCC-7721 cells. (B) CCK8 cell proliferation assays were performed after introducing pcDNA-PVT1 or pcDNA vector plasmid into Sk-hep-1 cells. (C) Cell colony formation assay was performed after introducing si-PVT1 or si-NC into SMCC-7721 cells. (D) Cell colony formation assay was performed after introducing pcDNA-PVT1 or pcDNA vector plasmid into Sk-hep-1 cells. The data derived from the results of 3 independent experiments, **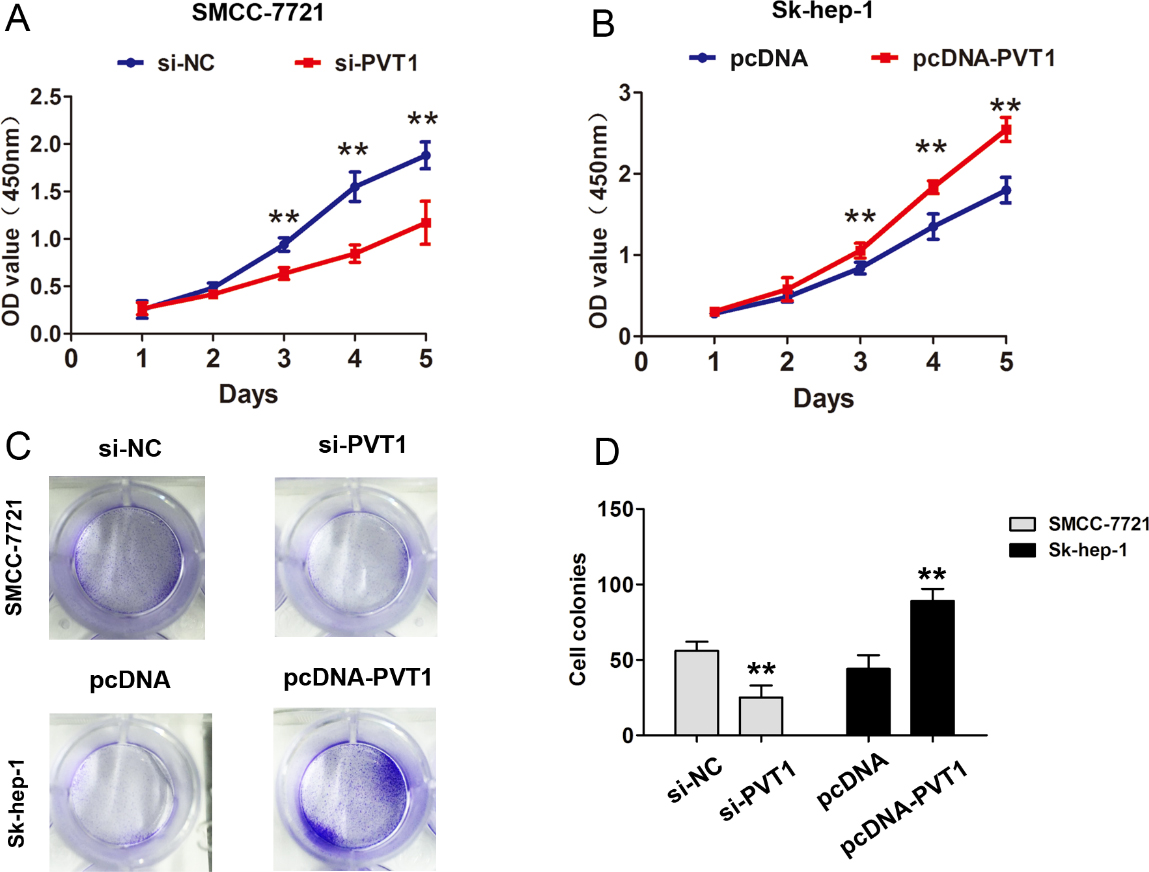
PVT1 promoted HCC invasion by regulating EMT process. (A) Transwell cell invasion assays were performed after introducing si-PVT1 or si-NC into SMCC-7721 cells or were performed after introducing pcDNA-PVT1 or pcDNA vector plasmid into Sk-hep-1 cells. (B) Transwell cell invasion analysis were calculated after introducing si-PVT1 or si-NC into SMCC-7721 cells or after introducing pcDNA-PVT1 or pcDNA vector plasmid into Sk-hep-1 cells. (C) The expression of EMT marker E-cadherin or Vimentin were performed after introducing si-PVT1 or si-NC into SMCC-7721 cells. (D) The expression of EMT marker E-cadherin or Vimentin were performed after introducing pcDNA-PVT1 or pcDNA vector plasmid into Huh-7 cells. The data derived from the results of 3 independent experiments, **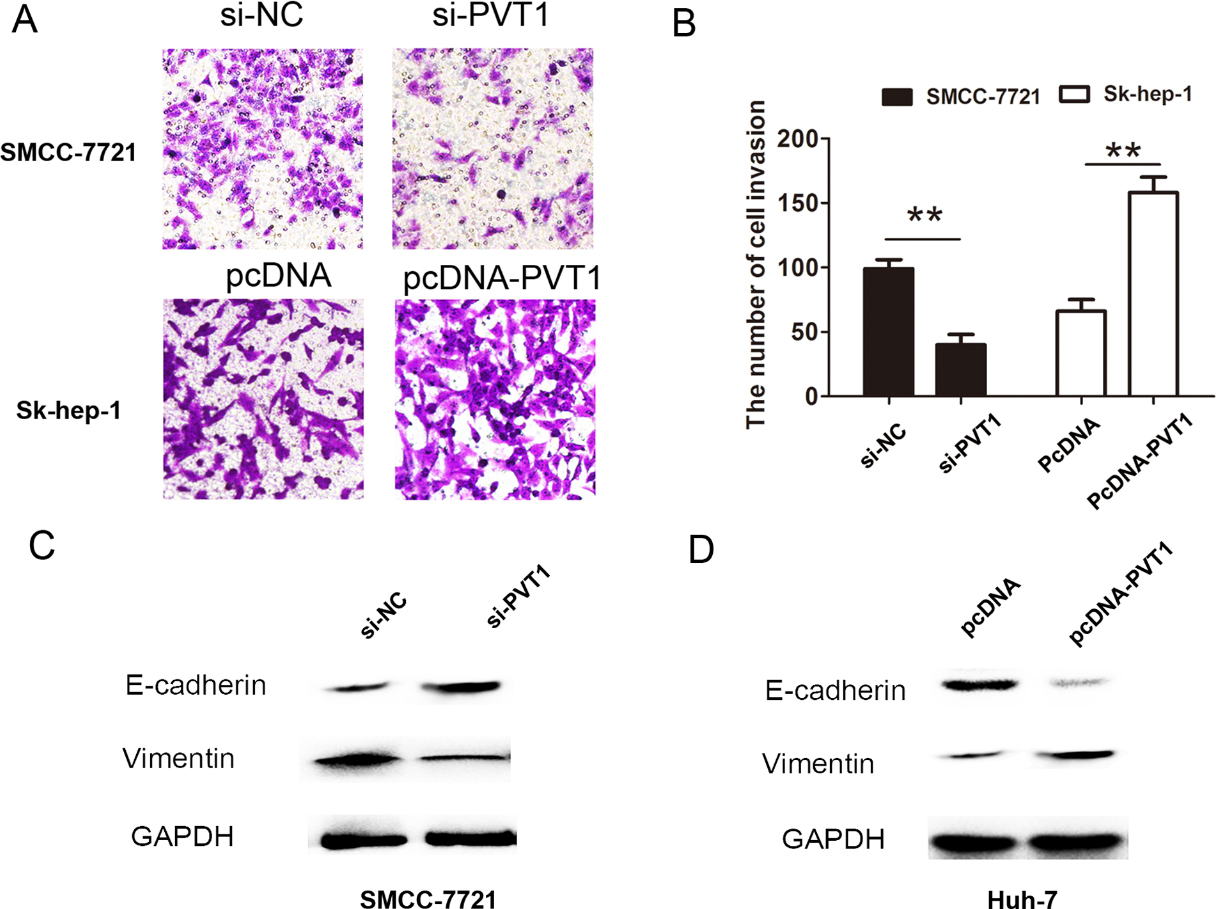
PVT1 inhibited the miR-214 expression via EZH2 in HCC cells. (A) RNA immunoprecipitation was performed using anti-EZH2 or anti-IgG antibody, and RNA was extracted from the pellets. The expression of PVT1 was analyzed by qRT-PCR.(B) The expression of miR-214 were analysis in HCC tissues compared with adjacent normal HCC tissues. U6 was used as a reference control. (C) The expression of miR-214 was analysis after introducing si-PVT1 or si-NC into SMCC-7721 cells. (D) The expression of miR-214 was analysis after introducing si-EZH2 or si-NC into SMCC-7721 cells. (E) Chromatin immunoprecipitation was performed using anti-EZH2 antibody after knockdown of PVT1 in SMCC-7721 cells. The promoter region of miR-214 was detected by qRT-PCR. The data derived from the results of 3 independent experiments, **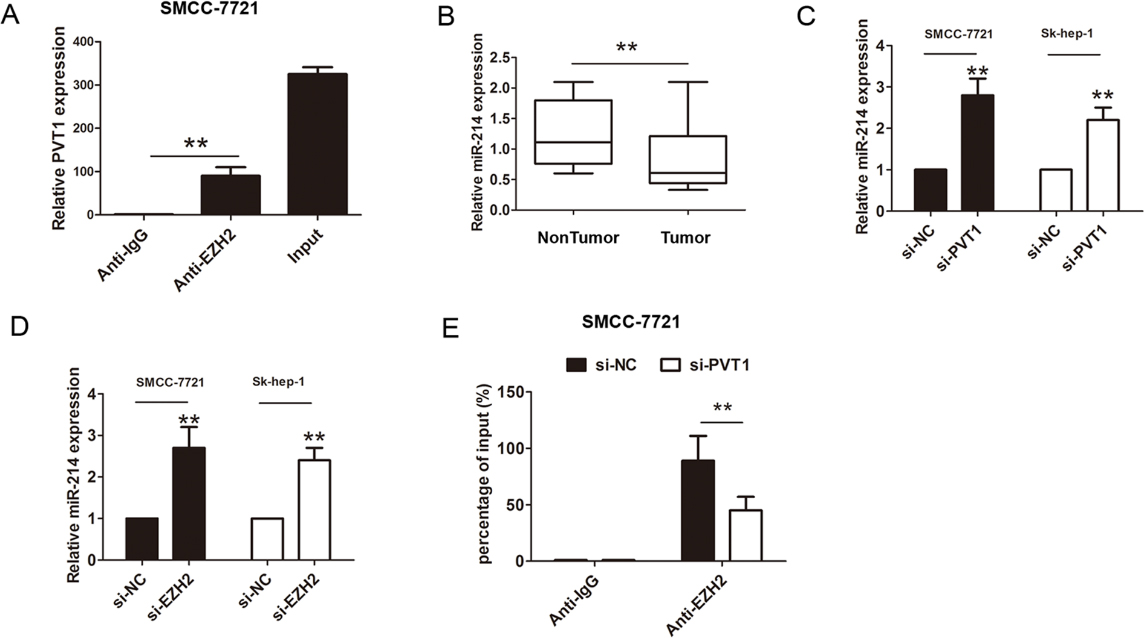
PVT1 inhibited cell proliferation by regulating miR-214 expression in HCC cells. (A) CCK8 cell proliferation assays were performed after introducing si-NC, si-PVT1 or si-PVT1 and miR-214 inhibitor into SMCC-7721 cells. (B)–(C) Cell colony formation assays were performed after introducing si-NC, si-PVT1 or si-PVT1 and miR-214 inhibitor into SMCC-7721 cells. (D) Cell invasion assays were performed after introducing si-NC, si-PVT1 or si-PVT1 and miR-214 inhibitor into SMCC-7721 cells. The data derived from the results of 3 independent experiments, **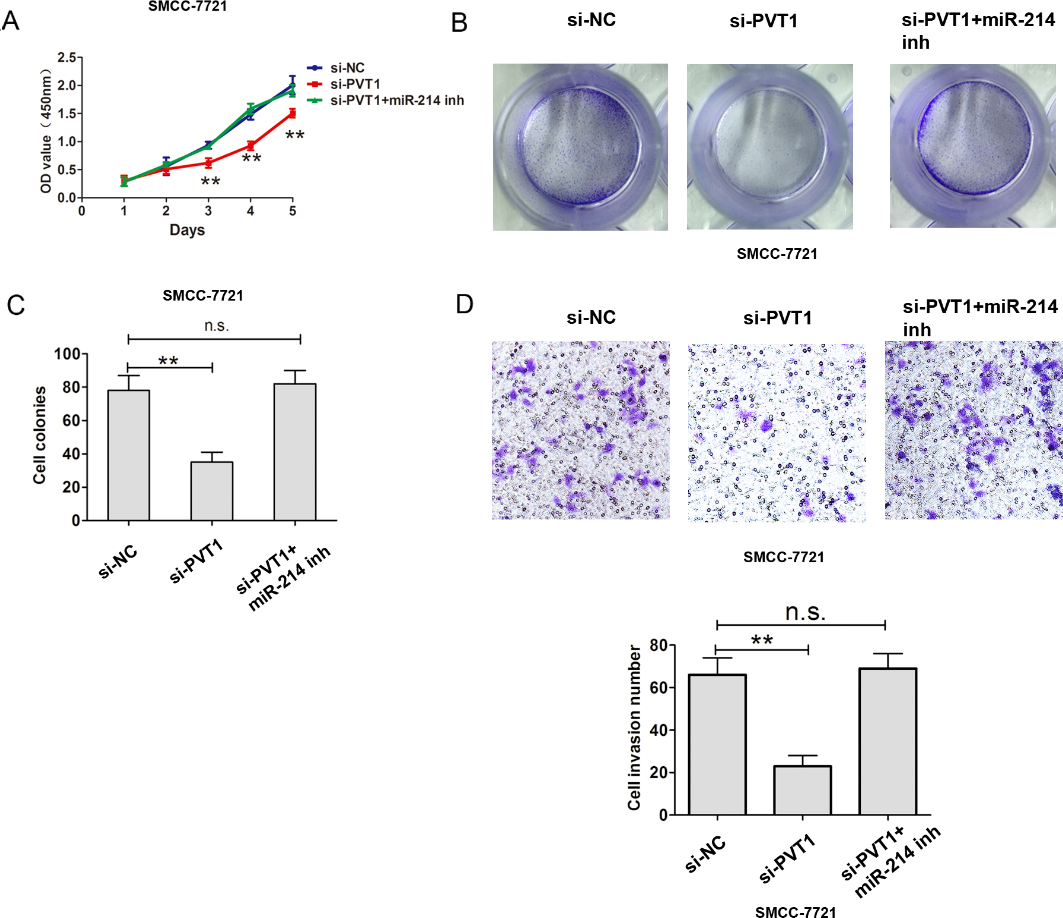
PVT1 interacted with EZH2 repressed miR-214 expression to promote cell proliferation and invasion of HCC.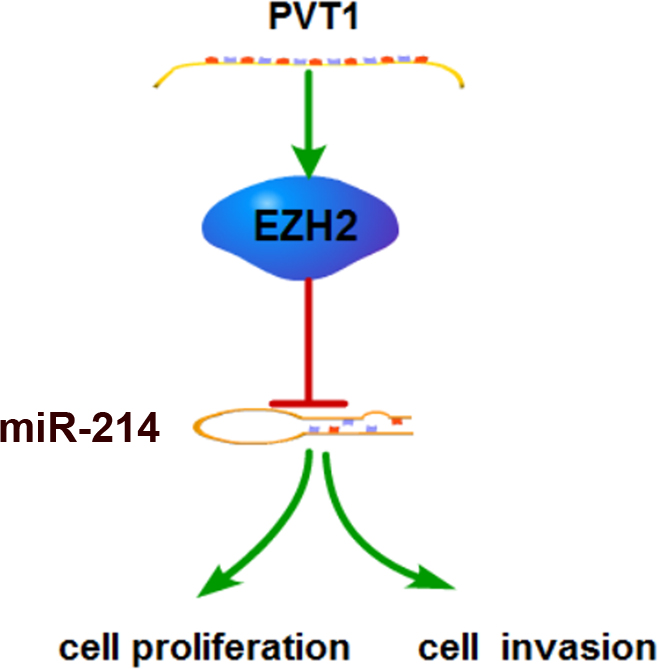
PVT1 is overexpressed in HCC tissues and cells
To evaluate the clinical role of PVT1 expression levels in HCC, PVT1 were detected by qRT-PCR assays in 92 cases HCC tissues and adjacent normal tissues. The results showed that expression levels of PVT1 were significantly higher in tumors tissues compared to adjacent normal tissue in patients (Fig. 1A,
Upregulation of PVT1 promotes the HCC cell proliferation and invasion in vitro
To further demonstrate the biological function of PVT1 in HCC, we evaluated the cell proliferation and invasion capacity in HCC cells after down-regulation or upregulation of PVT1. The results showed that PVT1 silencing inhibited SMCC-7721 cell proliferation using CCK8 assays, but upregulation of PVT1 expression promoted the cell proliferation in Sk-hep-1 cells (Fig. 2A and B). Cell colonies were smaller after PVT1 silencing than that in si-NC group in SMCC-7721 cells, but were larger after upregulation of PVT1 in Sk-hep-1 cells (Fig. 2C and D). Furthermore, cell invasion analysis showed that cell invasive number was decreased in SMCC-7721 cells after PVT1 was downregulated using transwell invasive assays. However, cell invasive number was increased after PVT1 was overexpressed in Sk-hep-1 cells (Fig. 3A and B). These results showed that PVT1 promoted the cell proliferation and invasion in HCC. In addition, we detected EMT relative markers and found that knockdown of PVT1 inhibited the Vimentin expression and upregulated the E-cadherin expression in SMCC-7721 cells, due to be difficult to detect E-cadherin expression in Sk-hep-1 cells, we selected Huh-7 cells to detect the EMT makers after upregulation and the result showed that overexpression of PVT1 enhanced the Vimentin expression and downregulated the E-cadherin expression in Huh-7 cell (Fig. 3C and D). Thus, these results showed that PVT1 promoted the HCC cell proliferation and invasion.
PVT1 inhibits the miR-214 expression by interacting with EZH2 in HCC cells
To further detect whether PVT1 affects gene expression by recruiting EZH2 to its target genes, we performed RNA immunoprecipitation (RIP) using an antibody against EZH2 in SMCC-7721 cells. The results showed that PVT1 was enriched in the anti-EZH2 RIP fraction compared to the anti-IgG fraction in SMCC-7721 cells (Fig. 4A). Therefore, these results demonstrated that PVT1 was significantly associated with EZH2.
We then sought to detect potential downstream targets of PVT1. We found miR-214, which is a target of EZH2, was decreased in HCC tissues compared to the adjacent normal tissues (Fig. 4B). After PVT1 was knocked down, PVT1 was upregulated in SMCC-7721 and Sk-hep-1 cells (Fig. 4C). In addition, we transfected si-EZH2 into SMCC-7721 or SK-hep-1 cells and found that miR-214 was also significantly up-regulated (Fig. 4D). To evaluate whether PVT1 repressed miR-214 mRNA expression by recruiting EZH2 to the promoter of miR-214, we carried out Chromatin immunoprecipitation (ChIP) method in SMCC-7721 cells. The results showed that PVT1 silencing decreased the binding ability between EZH2 and miR-214 promoter compared to IgG group (Fig. 4E).
Moreover, CCK8 and colony formation assays de- monstrated that si-PVT1 inhibited the cell proliferation and miR-214 inhibitor could counteract the si-PVT1 inhibited effect in SMCC-7721 cells (Fig. 5A–C). transwell assay demonstrated that si-PVT1 inhibited the cell invasion and miR-214 inhibitor could counteract the si-PVT1 inhibited effect in SMCC-7721 cells (Fig. 5D). Thus, these results suggest that PVT1 inhibited the miR-214 expression by interacting with EZH2 in HCC.
Discussion
Many tumor relative lncRNAs have been found and characterized in recent year. PVT1 was negatively correlated with miR-146a level in prostate cancer and mediated miR-146a expression by inducing the methylation of CpG Island in its promoter [20]. High levels of PVT1 were positively associated with the status of clinical stage, lymph node metastasis, and distant metastasis in in small cell lung cancer patients and had a poor prognostic biomarker and regulated NSCLC cell migration and invasion [21]. Our results showed PVT1 was upregulated in HCC tissues and patients with higher PVT1 expression was associated with tumor size, differentiation grade and advanced TNM stage. Furthermore, functional analysis revealed that knockdown of PVT1 inhibited cell proliferation and invasion in HCC.
We also sought to determine the underlying molecular mechanisms by which PVT1 regulates downstream effectors in HCC. We focused on the PRC2 subunit EZH2 because other lncRNAs regulate downstream effectors via EZH2-driven H3K27 methylation [22]. Study found LINC01133 functioned to repress KLF2, P21 and E-cadherin transcription through binding with EZH2, LSD1 in non small cell lung cancer (NSCLC) cells [23]. Long intergenic non-coding RNA 00152 promoted gastric cancer cell cycle progression by repressing p15 and p21 via binding to EZH2 [24]. In thyroid cancer, PVT1 could be enriched by EZH2, and silencing PVT1 resulted in the decreased recruitment of EZH2 and regulated thyroid-stimulating hormone receptor (TSHR) [14]. In non-small cell lung cancer cell, PVT1 recruited EZH2 to the large tumor suppressor kinase 2 (LATS2) promoter and repressed LATS2 transcription [13]. In the study, we demonstrated that PVT1 recruited EZH2 by RIP assay. Furthermore, ChIP assay showed that PVT1 inhibited the miR-214 expression by via binding to EZH2 to recruit EZH2 the tumor suppressor miR-214 promoter in HCC cell.
MiR-214 acted as a tumor suppressor in some cancer including HCC, Yang et al. reported that microRNA-214 suppressed the proliferation of human hepatocellular carcinoma cells by targeting E2F3 [25]. In the study, we found miR-214 inhibitor promoted the cell proliferation in HCC. The knockdown of PVT1 inhibited the cell proliferation and miR-214 inhibitor could counteract the si-PVT1 inhibited effect in HCC cells.
To sum up, our results revealed that PVT1 has a higher expression levels in HCC tissues than that in the adjacent normal tissues, and functions as an oncogene by promoting HCC cell proliferation, invasion. Furthermore, we found that PVT1 interacted with EZH2 repressed miR-214 expression in HCC cells (Fig. 6). Thus, our findings indicated that PVT1/EZH2/miR-214 interactions might serve as new target for HCC patients.
Conflict of interest
The authors declare that they have no competing interests.
Footnotes
Acknowledgments
We thanks for Key Laboratory of Chronobiology, West China School of Preclinical and Forensic Medi- cine, Sichuan University, for their help.
