Abstract
Background:
Long non-coding RNA (lncRNA) plays a very important role in the occurrence and development of various tumors, and is a potential biomarker for cancer diagnosis and prognosis. The purpose of this study was to investigate the relationship between the expression of lncRNA plasmacytoma variant translocation 1 (PVT1) and the prognostic significance in patients with colorectal cancer.
Methods:
The expression of PVT1 was measured by real-time quantitative reverse transcription-polymerase chain reaction (qRT-PCR) in cancerous and adjacent tissues of 210 colorectal cancer patients. The disease-free survival and overall survival of colorectal cancer patients were evaluated by Kaplan–Meier analysis, and univariate and multivariate analysis were performed by Cox proportional-hazards model.
Results:
Our results revealed that PVT1 expression in cancer tissues of colorectal cancer was significantly higher than that of adjacent tissues (P<0.001). High PVT1 expression was increased by 51.4% (108/210), which was significantly correlated with the tumor differentiation, the depth of invasion, the stage of tumor, node, metastasis (TNM), and lymphatic metastasis. The Kaplan–Meier analysis showed that high PVT1 expression resulted in a shorter disease-free survival (Log-rank test P<0.001) and overall survival (Log-rank test P<0.001) compared with the low PVT1 expression group in colorectal cancer patients, whether at TNM I/II stage or at TNM III/IV stage. A multivariate Cox regression analysis demonstrated that high PVT1 expression was an independent predictor of poor prognosis in colorectal cancer patients.
Conclusions:
Our results suggest that high PVT1 expression might be a potential biomarker for assessing tumor recurrence and prognosis in colorectal cancer patients.
Introduction
Colorectal cancer (CRC) is a common disease that seriously affects human health and quality of life worldwide. In the past two decades, the incidence and mortality of CRC have been on the rise rapidly in the Chinese population; >300,000 new cases are reported and >80,000 cases end in death each year, accounting for one-quarter of the mortality of malignant tumors.1,2 The American Joint Committee on Cancer reported that CRC patients in stage I of clinical tumor, node, metastasis (TNM) have a 5-year survival rate of 93.2%, whereas patients in stage IV have only 8.1%. 3 Thus, the TNM stage is an important index for clinicians to evaluate the prognosis of CRC patients, but it cannot distinguish the biological characteristics and has significant heterogeneity in assessing the survival rate.
Long non-coding RNA (lncRNA) is a newly discovered class of non-coding RNA. 4 With the rapid development and widespread application of gene (mRNA) sequencing technologies, human understanding of the transcriptome of organisms is more profound. lncRNA is involved in the development of human diseases by regulating gene expression, especially in tumors. 5 lncRNA constructs sophisticated and complex regulatory network systems in different tumor tissues, which regulate the tumor cell differentiation, proliferation, and apoptosis, and participates in tumor evolution. 6 Therefore, lncRNA is expected to become a potential indicator of tumor diagnosis, treatment, and prediction of prognosis.
Plasmacytoma variant translocation 1 (PVT1) transcripts are homologous to murine PVT1 genes and belong to the intergenic lncRNA. The PVT1 gene is located in the 8q24 region on the chromosome sense strand and spans >300 kb of the genome. 7 In recent years, relevant studies reported that PVT1 displays high expression in a variety of tumor tissues and cells, such as ovarian cancer, 8 lung cancer, 9 prostate cancer, 10 cervical cancer, 11 gastric cancer, 12 and liver cancer. 13 PVT1 is identified as an activator of MYC and is encoded by the PVT1 oncogene in mice. PVT1 is linked to the protein of c-Myc and c-Myc FUBP1, an upstream regulator of EZH2 and NPM1 to promote cell proliferation by inhibiting the TGF-β pathway. 14 PVT1 plays a role similar to the relevant proto-oncogene to promote the metastasis, proliferation, and invasion of malignant cells, and enhances the resistance of tumor cells to chemotherapeutic drugs.
However, the relationship between PVT1 expression and the prognostic significance of CRC patients remains unclear. Therefore, the purpose of this study was to detect PVT1 expression in the cancer tissues of CRC patients, and elucidate the significance of PVT1 in the clinicopathology and prognosis of CRC patients.
Methods
Patients and sample collection
A total of 210 cases of fresh specimens were obtained from patients with primary CRC from the General Surgery of Ningbo First Hospital and the Guangdong Provincial Hospital during September 2010 and December 2012. A total of 210 matched adjacent normal tissues served as controls. Exclusion criteria were patients who: received neoadjuvant chemotherapy or radiotherapy prior to surgery; who had an additional diagnoses of oncological diseases; or in whom CRC was not a primary tumor. All specimens were selected by a pathologist and were rapidly frozen in liquid nitrogen and immediately stored at −80°C after surgical resection. All patients’ clinical pathology and follow-up data were entered into a database that updated their status of life every month. Our study was approved by the Ethics Committee of the Ningbo First Hospital (Ningbo, China) and the Guangdong Provincial Hospital (Guangzhou, China). All patients agreed to participate in this study and signed the informed consent.
Quantitative reverse transcription-polymerase chain reaction
Total RNA was isolated from cancer tissues or adjacent normal tissues using Trizol according to the protocol of the manufacturer (Invitrogen, Waltham, MA, USA). We used Nanodrop-spectrophotometer to detect the RNA concentration and purity, and Agilent 2100 Bioanalyzer (Santa Clara, CA, USA) to determine the RNA integrity. According to the protocol of the manufacturer, we synthesized complementary DNA (cDNA) from 1 µg of total RNA using the PrimeScript-RT Kit (Madison, WI, USA), and then stored at −20°C. We performed a quantitative reverse transcription-polymerase chain reaction (qRT-PCR) using the SYBR® Premix-Ex-Taq™ (Takara, TX, USA) and the ABI 7300 system. The specific primers for PVT1 and GAPDH were as follows: PVT1 forward 5′-AGA GTA CAG ACC TAT CCA CTGC-3′ and reverse 5′-TCA CCT GCA TGA GTG CCA CA-3′; and GAPDH forward 5′-ATG CTA ACG AGC TAG CAC ACA-3′ and reverse 5′-TGT CAT CTT TAA AGC GTC CTT-3′. The total volume of the PCR system was 30 µl and each sample contained 30 ng cDNA. The amplification procedure was initial denaturation at 95°C for 10 min and followed by 45 cycles of 95°C for 10 sec, 60°C for 30 sec, 85°C for 20 sec. The RT-PCR products sizes were 130 bp. We converted all the fluorescent data into relative quantitation and normalized PVT1 expression by the 2-∆∆Ct method relative to GAPDH. We repeated all qRT-PCR reactions three times and used the median level of PVT1 expression as a cutoff to determine high or low PVT1 expression.
Measurement of endpoints
We defined disease-free survival (DFS) as the time between excision of tumor and the first finding of a CRC with local recurrence, distant metastasis, or death from non-CRC causes. The diagnosis of recurrence and distant metastasis was mainly based on computed tomography, positron emission tomography, tissue biopsy, or cytology. The censored time of patients with no tumor recurrence was death or the last follow-up. We defined overall survival (OS) as the time between tumor removal and the death of the CRC patient.
Statistical analysis
We performed all statistical analysis using SPSS 16.0 (SPSS Inc., Chicago, IL, USA). We presented categorical variables as numbers (percentages), and continuous variables as median (interquartile range). We used the independent t-test to evaluate the PVT1 expression differences between adjacent tissues and CRC tissues. We analyzed the expression level of PVT1 from different tissues by one-way analysis of variance with least significant difference post-hoc test, and used Pearson’s χ 2 tests to analyze the relationship between PVT1 expression and categorical variables. We estimated survival curves by Kaplan–Meier analysis and assessed the different significances in survival distribution by Log-rank test, and used the Cox proportional hazards model to analyze univariate and multivariate survival. We defined P<0.05 as statistically significant.
Results
The characteristics of CRC patients
According to the standard, a total of 210 CRC patients were enrolled, including 98 males (46.67%) and 112 females (53.33%). There were 145 (69.04%) cases of colon cancer and 65 (30.95%) cases of rectal cancer. Of these CRC patients, 68 (32.38%) cases had a tumor size of <3 cm and 142 (67.62%) cases ⩾3 cm. Of the 210 CRC patients, 131 (62.38%) had poorly/moderately differentiated tumors and 79(37.62%) had high differentiated tumors. A total of 72 (34.29%) cases were classified as TNM stages I/II, and 138 (65.71%) were classified as TNM stages III/IV (Table 1).
The correlation between lncRNA PVT1 expression and clinicopathological factors of colorectal cancer.
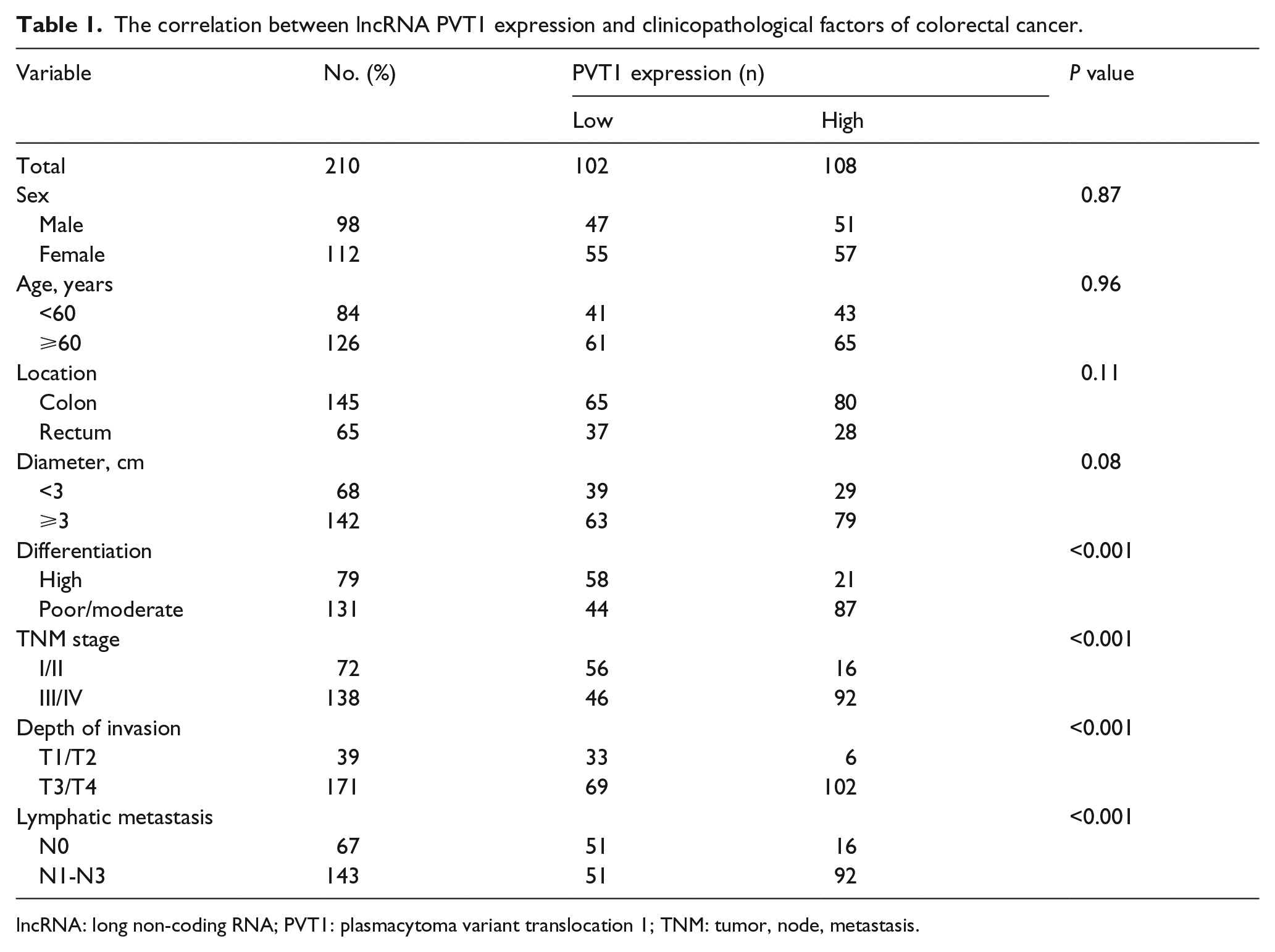
lncRNA: long non-coding RNA; PVT1: plasmacytoma variant translocation 1; TNM: tumor, node, metastasis.
The relationship between PVT1 expression and clinical features in CRC patients
To determine the clinical value of PVT1 expression in CRC patients, we used qRT-PCR to detect PVT1 expression in cancerous and adjacent tissues of 210 CRC patients. Following normalized GAPDH expression, the PVT1 expression in cancer tissues was significantly higher than matched adjacent tissues (5.01 (3.71–7.36) vs. 2.36 (1.72–3.18), P<0.001, Figure 1(a)). The 210 CRC patients were divided into two groups based on their PVT1 expression compared to the median PVT1 expression (6.75): high PVT1 expression group (>6.75, n=108), and low PVT1 expression group (<6.75, n=102). As show in Table 1, PVT1 expression was not correlated with sex, age, and tumor location (P>0.05). However, the expression of PVT1 was significantly related to the degree of tumor differentiation, depth of invasion, the stage of TNM, and lymphatic metastasis (P<0.001). Moreover, PVT1 expression was significantly higher in CRC tissues with a tumor size ⩾3 cm than in CRC tissues with a tumor size <3 cm (6.02 (4.76–8.21) vs. 4.58 (2.43–6.72), P<0.001; Figure 1(b)), and PVT1 expression in cancer tissues of CRC patients with stage III/IV was significantly higher than those CRC patients with stage I/II (7.63 (5.02–9.86) vs. 5.31 (3.43–7.65), P<0.001; Figure 1(c)).
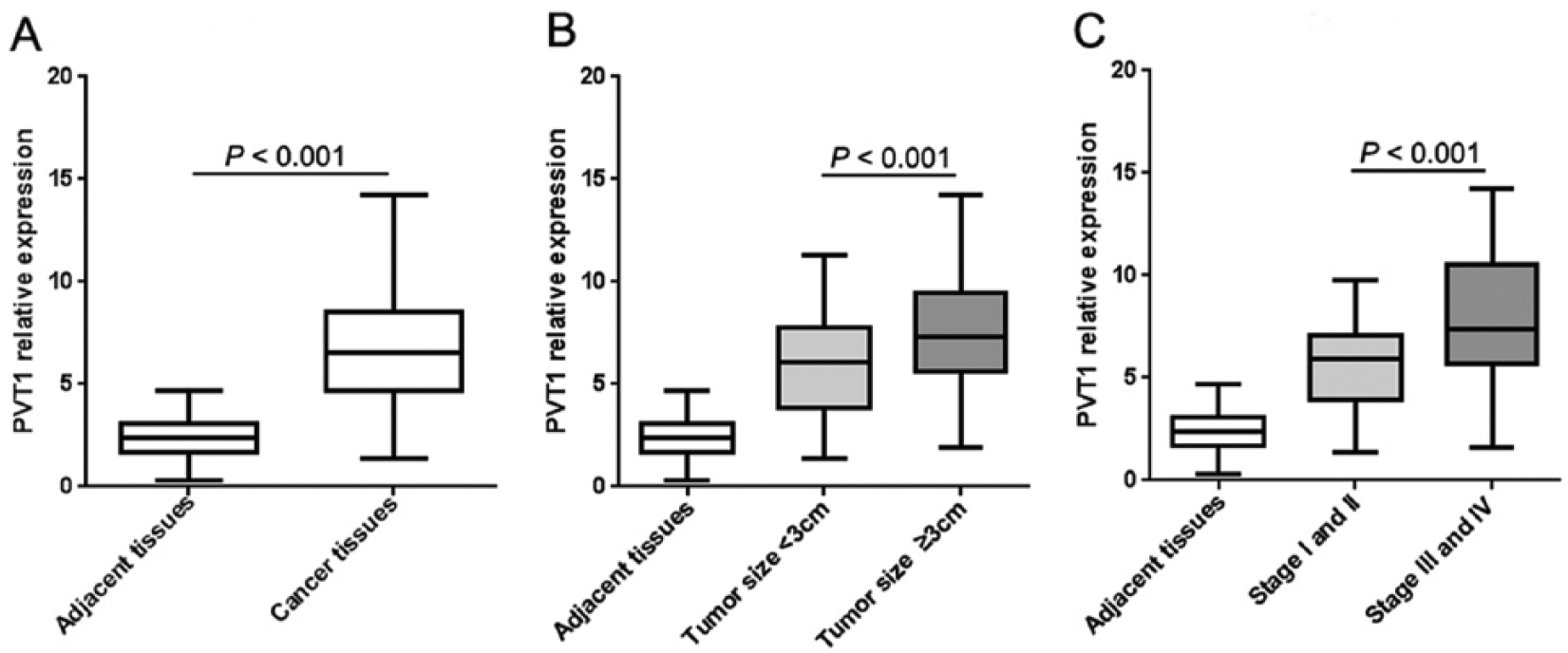
lncRNA PVT1 expression in CRC tissues. (a) The significant differences of PVT1 expression between adjacent tissues and cancer tissues were evaluated using an independent t-test. (b) PVT1 expression in adjacent tissues, with tumor size ⩾3 cm and tumor size <3cm, and the levels of PVT1 in different tissues were analyzed using a one-way analysis of variance. (c) PVT1 expression in adjacent tissues, TNM I/II stage group and III/IV stage group, and PVT1 expression levels in different tissues were analyzed using a one-way analysis of variance.
High PVT1 expression related to poor prognosis of CRC patients
To investigate the effect of PVT1 expression on the prognosis of CRC patients, we evaluated the association of increased PVT1 expression with DFS and OS. Of 210 CRC patients followed up, 91 (43.33%) died. A Kaplan–Meier analysis showed that high PVT1 expression in CRC patients had significantly shorter DFS (Figure 2(a) and (c); Log-rank test P<0.001) and OS (Figure 2(b) and (d); Log-rank test P<0.001) than the low PVT1 expression group, whether TNM I/II stage or TNM III/IV stage, suggesting that high PVT1 expression had an adverse effect on the prognosis of CRC patients. Moreover, we used Cox proportional hazards models to perform univariate and multivariate analyses for DFS and OS. Our univariate analysis results revealed that high PVT1 expression, the tumor differentiation, depth of invasion, the stage of TNM III/IV, and lymphatic metastasis were significantly related to shortened DFS (Table 2) and OS (Table 3) in CRC patients. We used multivariate analysis to explore the underlying prognostic predictors. High PVT1 expression in cancer tissues was an independent predictor of poor prognosis for DFS (Table 2, hazard ratio (HR) 2.15; 95% confidence interval (CI) 1.39, 2.76, P<0.001) and OS (Table 3; HR 1.76; 95% CI 1.16, 2.37; P<0.001). Furthermore, on the basis of controlling other clinicopathological factors, the tumor differentiation, depth of invasion, the stage of TNM III/IV, and lymphatic metastasis were also significantly related to shortened DFS and OS.
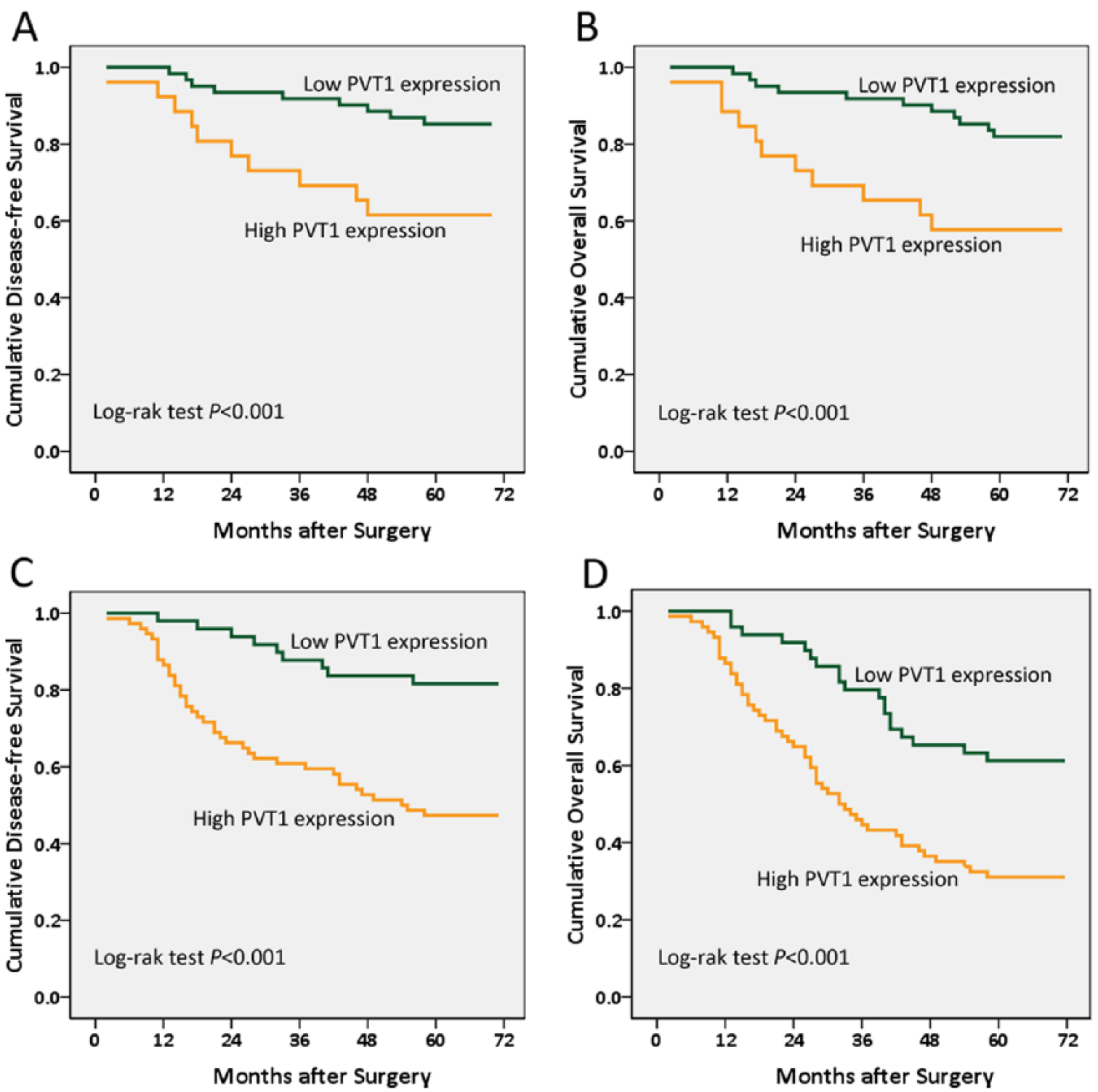
Kaplan–Meier survival curve in CRC patients based on lncRNA PVT1 expression. (a) The disease-free survival curve. (b) The overall survival curves were analyzed based on the relative PVT1 expression in CRC patients with TNM I/II stage. (c) The disease-free survival curve. (d) The overall survival curves were analyzed in CRC patients with TNM III/IV stage.
Univariate and multivariate analyses of disease-free survival in CRC patients (Cox proportional-hazards model).
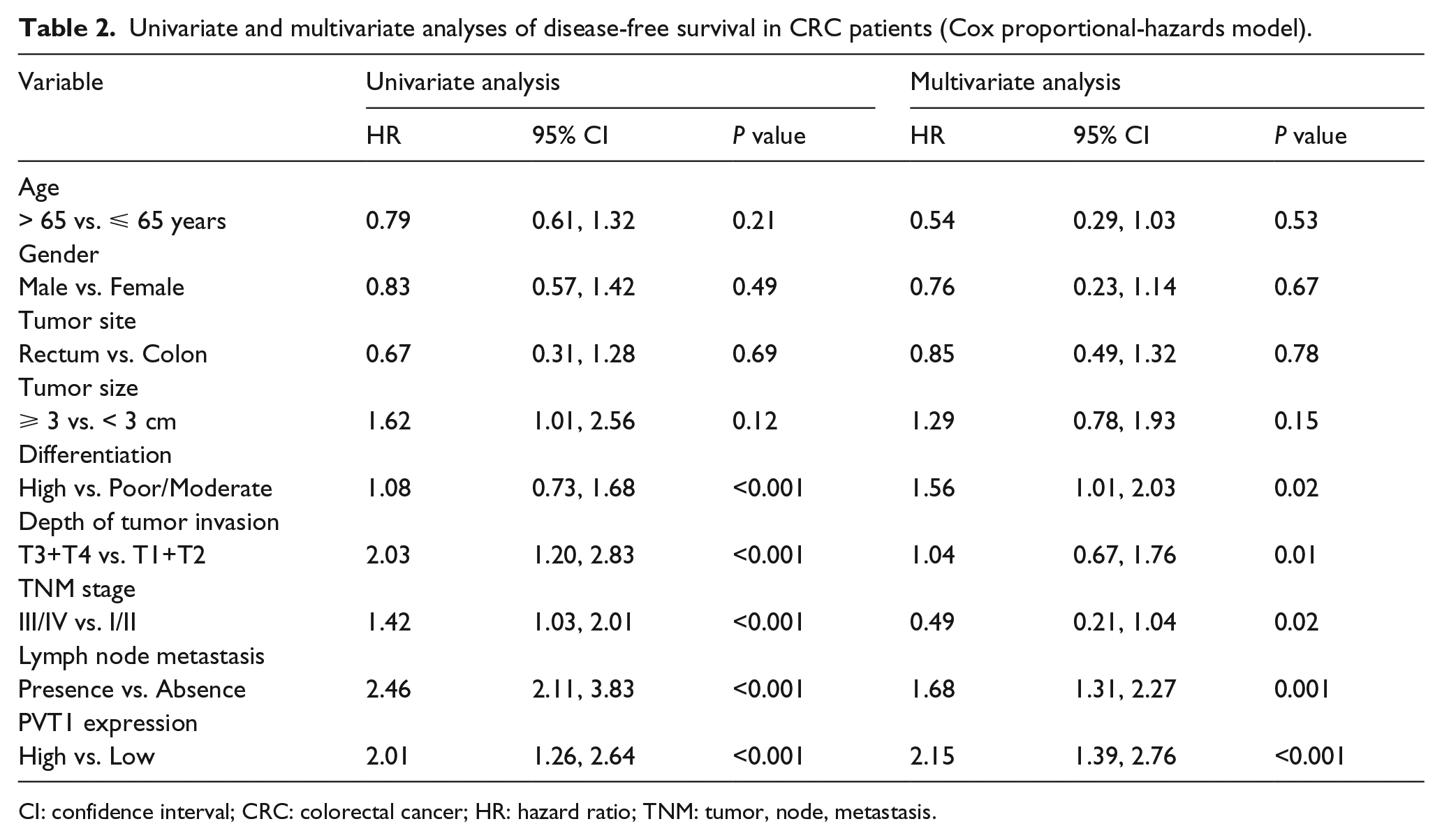
CI: confidence interval; CRC: colorectal cancer; HR: hazard ratio; TNM: tumor, node, metastasis.
Univariate and multivariate analyses for overall survival in CRC patients (Cox proportional-hazards model).
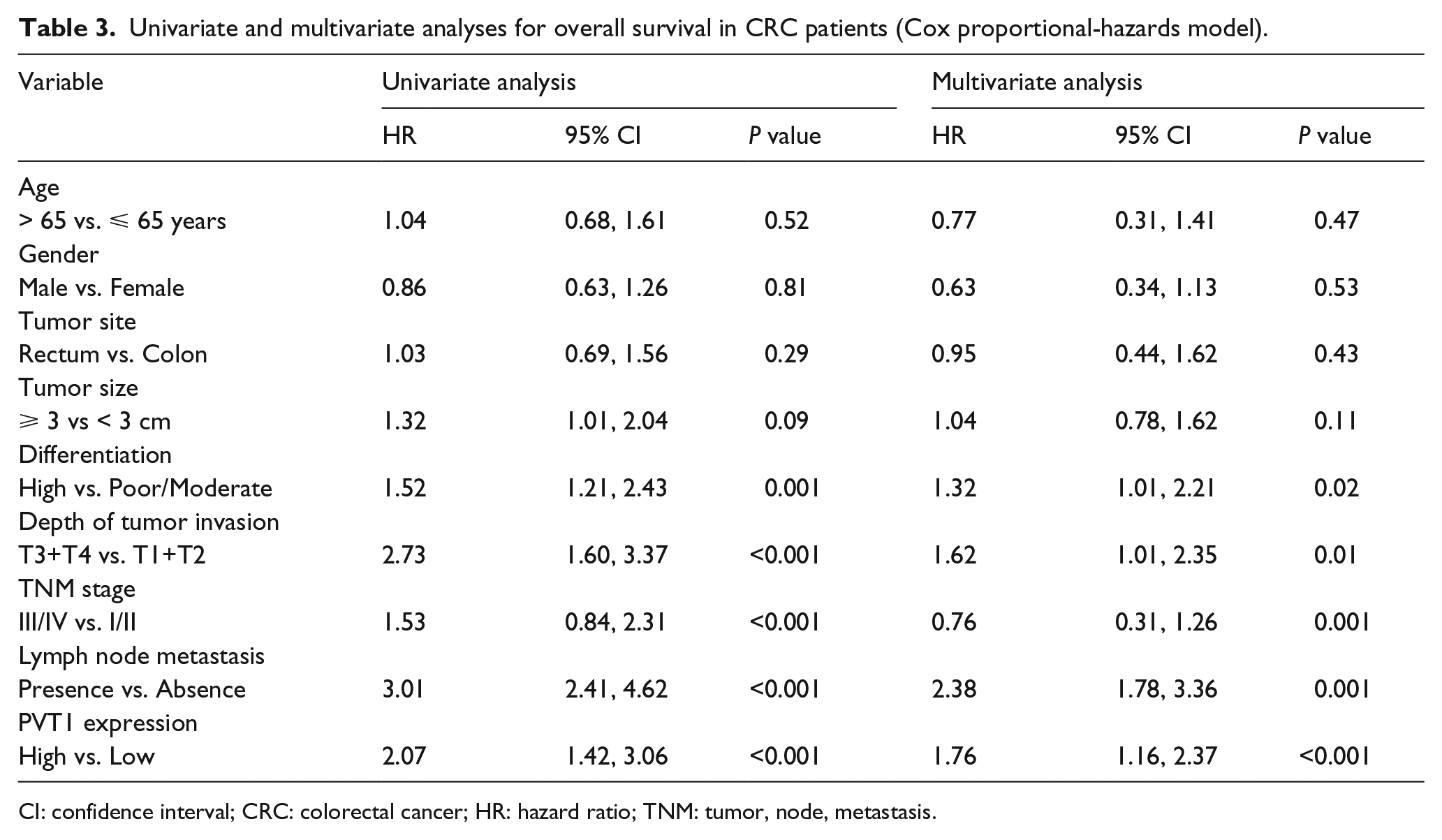
CI: confidence interval; CRC: colorectal cancer; HR: hazard ratio; TNM: tumor, node, metastasis.
Discussion
CRC is one of the main causes of death worldwide in patients with digestive cancer, and the TNM stage of cancer is an important indicator of the prognosis of CRC patients. 1 Due to the limitations of the treatment and the lack of simple and effective biomarkers in advanced CRC patients, the diagnosis of CRC is very difficult in its early stage. Therefore, identifying the function of potential biomarkers may facilitate the early diagnosis of CRC.
lncRNA is a subset class of non-coding RNAs with >200 nucleotides. 4 PVT1 belongs to the lncRNA class and is encoded by the PVT1 oncogene. 5 In human genes, PVT1 is located on chromosome 8q24.21, which is a downstream gene of the c-Myc gene. 7 The Wnt signaling pathway is an important pathway for CRC carcinogenesis and progression, and PVT1 plays a carcinogenic role by regulating Wnt downstream targets. However, PVT1 is different from most non-coding genes upstream of MYC (such as CCAT1 and CCAT2). PVT1 mainly controls the MYC protein levels by protecting MYC proteins against degradation. 14
The dysregulation of PVT1 plays an important role in human diseases, especially in the development of malignancies. Alvarez and DiStefano 15 revealed that PVT1 is lowly expressed in the normal tissues of patients with diabetic nephropathy, but is highly expressed in extracellular matrix-transformed cells. However, PVT1 is highly expressed in a variety of malignancies, such as cervical cancer, 11 gastric cancer, 12 thyroid cancer, 16 lung cancer, 17 and pleural mesothelioma. 18 Increased expression of PVT1 promotes the proliferation, migration, and invasion of these tumor cells. 19 The high expression of PVT1 enhances the malignancy of breast cancer and ovarian cancer, and induces the drug resistance of chemotherapy, 8 which is significantly related to the poor prognosis of cervical cancer. 11
Recent studies have indicated that there are many kinds of gene expression abnormalities in the occurrence and development of gastrointestinal tumors.20-22 PVT1 mediated epigenetics is the focus field of researchers worldwide. Guo et al. 23 found that PVT1 is highly expressed in CRC tissues and cells, and its level is closely related to vascular invasion and lymph node metastasis; the down-regulation of PVT1 induces apoptosis and inhibits proliferation in CRC cells. Takahashi et al. 24 showed that silence of the PVT1 gene significantly attenuates the ability of CRC cells to proliferate and invade, and their multivariate analysis results demonstrated that PVT1 expression is negatively associated with the prognosis of CRC patients, which is an important prognostic indicator of CRC.
In the present study, we detected the expression of PVT1 in cancer tissues and matched adjacent normal tissues of 210 CRC patients, and aimed to investigate the relationship between PVT1 expression and prognosis of CRC patients. Our results revealed that PVT1 expression in cancer tissues of CRC is significantly higher than that in matched adjacent normal tissues, and PVT1 expression in CRC patients with tumor size ⩾3 cm and TNM stage III/IV is significantly higher than those of tumor size <3 cm and TNM stage I/II. These data suggest that PVT1 expression is correlated with the progression of CRC. Moreover, our study also found that high PVT1 expression is related to poor tumor differentiation, depth of invasion, the stage of TNM, and lymphatic metastasis, but is not associated with age, sex, cancer size, and location. These finding suggest that high PVT1 expression promotes CRC metastasis.
In our study, a Kaplan–Meier analysis demonstrated that high PVT1 expression in CRC patients has significantly shorter DFS and OS than the low PVT1 expression group regardless of TNM stage. Univariate and multivariate analyses indicated that high PVT1 expression significantly affects the prognosis of CRC patients. The multivariate analysis suggested that high PVT1 expression indicates an increased risk of cancer recurrence and death. These results suggested that high PVT1 expression promotes the development of CRC, and is a potential target for progression in CRC patients. Moreover, after adjusting for other clinicopathological factors, high PVT1 expression is still significantly associated with shortened DFS or OS. These data indicated that PVT1 is an independent risk factor for the prognosis of CRC patients, and it can be used for predicting recurrence and death, and early aggressive prevention therapy after CRC surgery.
To date, little is known about the biomarkers used to predict the recurrence of CRC patients. Biomarkers that are used to predict DFS also mean that they can be used to predict the metastasis or the recurrence of the tumor. Our results suggested that high PVT1 expression is helpful to predict the early metastasis or recurrence of CRC after radical resection. Therefore, the level of PVT1 in tumor tissue will provide a choice of individualized treatment for CRC patients, thus prolonging the survival time of the patients.
In conclusion, our study showed that lncRNA PVT1 is highly expressed in the tumor tissues of CRC patients. High PVT1 expression is not only a prospective biomarker for the recurrence of CRC metastasis, but also an independent risk factor for assessing the prognosis of CRC. Our findings also provide a clinical basis for exploring the potential of PVT1 as a new therapeutic target, encouraging further study of the biological functions of PVT1.
Footnotes
Author contributions
Heng Fan and Jian-hua Zhu contributed equally to this work.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Zhejiang Provincial Medical and Health Science Fund (2018KY667, 2017KY134, 2018KY669, 2018KY685), the Ningbo Natural Science Fund (2017A610188) and the Zhejiang Natural Science Fund (LQ18H150001).
