Abstract
Background:
The Glasgow prognostic score (GPS) is an established inflammatory prognostic index in cancer patients. Most studies have only measured GPS at baseline (B-GPS). Effective cancer therapy may reduce inflammation, and we investigated whether re-assessing GPS after first-line chemotherapy (E-GPS) provided more prognostic information than B-GPS in a phase III trial of advanced non-squamous non-small cell lung cancer (NSCLC).
Methods:
Glasgow prognostic score was assessed before and after carboplatin/vinorelbine chemotherapy. When assessing GPS, C-reactive protein (CRP) ⩾ 10 mg/L and albumin < 35 mg/L are defined as abnormal values. GPS 0: both values normal, GPS 1: one abnormal value, and GPS 2: both values abnormal.
Results:
Glasgow prognostic score at baseline and E-GPS were available in 138 patients. Median age was 67 years, 51% were women, and 94% had performance status 0-1. B-GPS was not a statistically significant prognostic factor (B-GPS 1 vs 0: hazard ratio [HR] = 1.32, 95% confidence interval [CI] = 0.9-2.0; B-GPS 2 vs 0: HR = 1.46, 95% CI = 0.9-2.3), while E-GPS was (E-GPS 1 vs 0: HR = 1.57, 95% CI = 1.0-2.4; E-GPS 2 vs 0: HR = 2.77, 95% CI = 1.7-4.5). E-GPS was associated with treatment response (P < .01), whereas B-GPS was not.
Conclusion:
Glasgow prognostic score at baseline after first-line chemotherapy provided more prognostic information than baseline GPS in patients with advanced non-squamous NSCLC and was associated with treatment response.
ClinicalTrials.gov Identifier:
NCT02004184.
Keywords
Background
Inflammation plays an essential role in cancer development and progression,1-3 and the development and maintenance of a systemic inflammatory response has been consistently associated with poorer outcome in both early and advanced disease. 4
Glasgow prognostic score (GPS) is an inflammatory score based on values of C-reactive protein (CRP) and albumin. 5 Mounting evidence has shown that it is an independent prognostic factor in numerous cancers, different disease stages, and treatment settings.6-13 An important aspect is that it is objectively assessed, affordable, and easy to implement in clinical practice.
Almost all previous studies have measured GPS only once and mainly before start of treatment (at baseline, “B-GPS”).5-9,11-23 However, GPS is believed to reflect inflammation as an expression of cancer activity, and hence, in patients who respond to cancer treatment, a reduction in inflammation and thereby in GPS is to be expected. 4 Thus, GPS measured after treatment (at evaluation, “E-GPS”) might capture the effect of treatment and be a more precise prognostic factor than B-GPS.
Lung cancer is marked by high inflammation and poor survival,24,25 and a high proportion of patients have elevated GPS as compared with other cancer types. 25 Therefore, in a randomized phase III trial comparing immediate maintenance pemetrexed with pemetrexed at progression in patients with advanced non-squamous non-small cell lung cancer (NSCLC), 26 we measured B-GPS and E-GPS after induction chemotherapy. The aims were to assess whether E-GPS provides better prognostic information than B-GPS and whether there were associations between response to chemotherapy and B-GPS, E-GPS, or change in GPS.
Methods
Approvals
This open randomized phase III multicenter trial was approved by the Regional Committee for Medical Research Ethics in Central Norway (ID 2013/645, approved on June 17, 2013) and The Norwegian Medicines Agency.
Patients
From May 2014 to September 2017, a total of 232 patients were enrolled at 19 hospitals in Norway. Eligible patients were treatment naïve, had stage IIIB-IV non-squamous NSCLC, no known activating EGFR-mutation or ALK-translocation, WHO performance status (PS) 0-2, and adequate bone marrow/liver/kidney function. Patient were to receive 4 courses of induction chemotherapy with carboplatin AUC 5 (Calvert’s formula) IV and vinorelbine 25 mg/m2 IV day 1 and vinorelbine 25 mg/m2 IV or 60 mg/m2 PO day 8, every 3 weeks. Patients who completed 4 courses, had PS 0-2 and non-progression were randomized to immediate maintenance pemetrexed therapy or observation. Pemetrexed was the treatment of choice at progression. Patients who were not randomized were treated according to each hospital’s routines. The study closed prematurely when immunotherapy became available in Norway and replaced pemetrexed as standard relapse treatment. In the randomized trial, there was no significant difference in overall survival (OS) (P = .10) between treatment arms. Thus, in the present study, all patients were analyzed as one cohort. 26
For our main analyses, we included patients who received 3 or 4 courses of carboplatin/vinorelbine if GPS was scored both at baseline and evaluation (main study cohort) (Figure 1). In a sensitivity analysis of B-GPS and survival, we included all patients with a B-GPS, independent of number of completed chemotherapy courses (Figure 1).

Consort flow diagram.
Glasgow prognostic score
According to the GPS, an elevated CRP of ⩾10 mg/L and hypoalbuminemia of <35 mg/L are considered abnormal values. If both values are normal, GPS is 0. If one value is abnormal, GPS is 1, and when both are abnormal, GPS is 2. A higher score is associated with shorter survival. 5
Blood samples for assessing GPS were collected within 2 weeks before chemotherapy commenced (B-GPS) and within 3 weeks after the last chemotherapy course was administered (E-GPS).
Endpoints
Overall survival was defined as time from inclusion until death of any cause in the analyses with B-GPS and as time from evaluation after first-line chemotherapy until death of any cause in analyses with E-GPS. Response to treatment was assessed according to the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. 27
Statistical considerations
Survival was estimated using the Kaplan-Meier method and compared using the Cox proportional hazard model. To assess the prognostic value of B-GPS and E-GPS in our main study cohort, multivariable Cox proportional hazards models for survival were adjusted for sex, age (continuous variable), and stage of disease (III vs IV). In addition, we adjusted for PS scored at baseline when examining B-GPS, and PS at evaluation in models with E-GPS. Performance status at evaluation was missing for 6 patients who were excluded from multivariable analyses.
As E-GPS was not measured in all patients with a B-GPS, we performed sensitivity survival analyses including all patients with a B-GPS (n = 208) to account for a potential selection bias.
Finally, we performed sensitivity survival analyses in the main study cohort (n = 138), adjusting for randomization (no/observation-arm/maintenance-arm), and whether patients received immunotherapy after chemotherapy, as this has been shown to significantly improve survival in some patients with advanced NSCLC.28,29
Associations between B-/E-GPS and response to chemotherapy were compared using Pearson’s chi-square test or Fisher’s exact test. A 2-sided P < .05 was considered statistically significant. SPSS Version 27.0 (Armonk, NY: IBM Corp) was used for all statistical analyses.
Results
Patients
For 138 (59%) of the 232 patients enrolled in the randomized controlled trial (RCT), both B-GPS and E-GPS were available. These patients were included in the present study as our main study cohort. B-GPS was measured in an additional 70 patients, who were also included in sensitivity analyses, whereas 24 patients had no GPS measures and were excluded altogether (Figure 1).
In our main study cohort, median age was 67 years (range, 47-83), 70 (51%) were women, 130 (94%) had stage IV disease, and 50 (36%), 80 (58%), and 8 (6%) had PS 0, 1, and 2, respectively (Table 1). After completing induction chemotherapy, 80 (58%) of the patients were randomized to immediate maintenance pemetrexed therapy (n = 42) or observation (n = 38). Thirty-six (26%) of the patients received immunotherapy after the study therapy (Table 1). Mean follow-up time was 14.1 months (95% confidence interval [CI] = 12.3-15.9). Eighteen of 138 patients were alive when follow-up was completed.
Patient characteristics of all patients in the main study cohort.
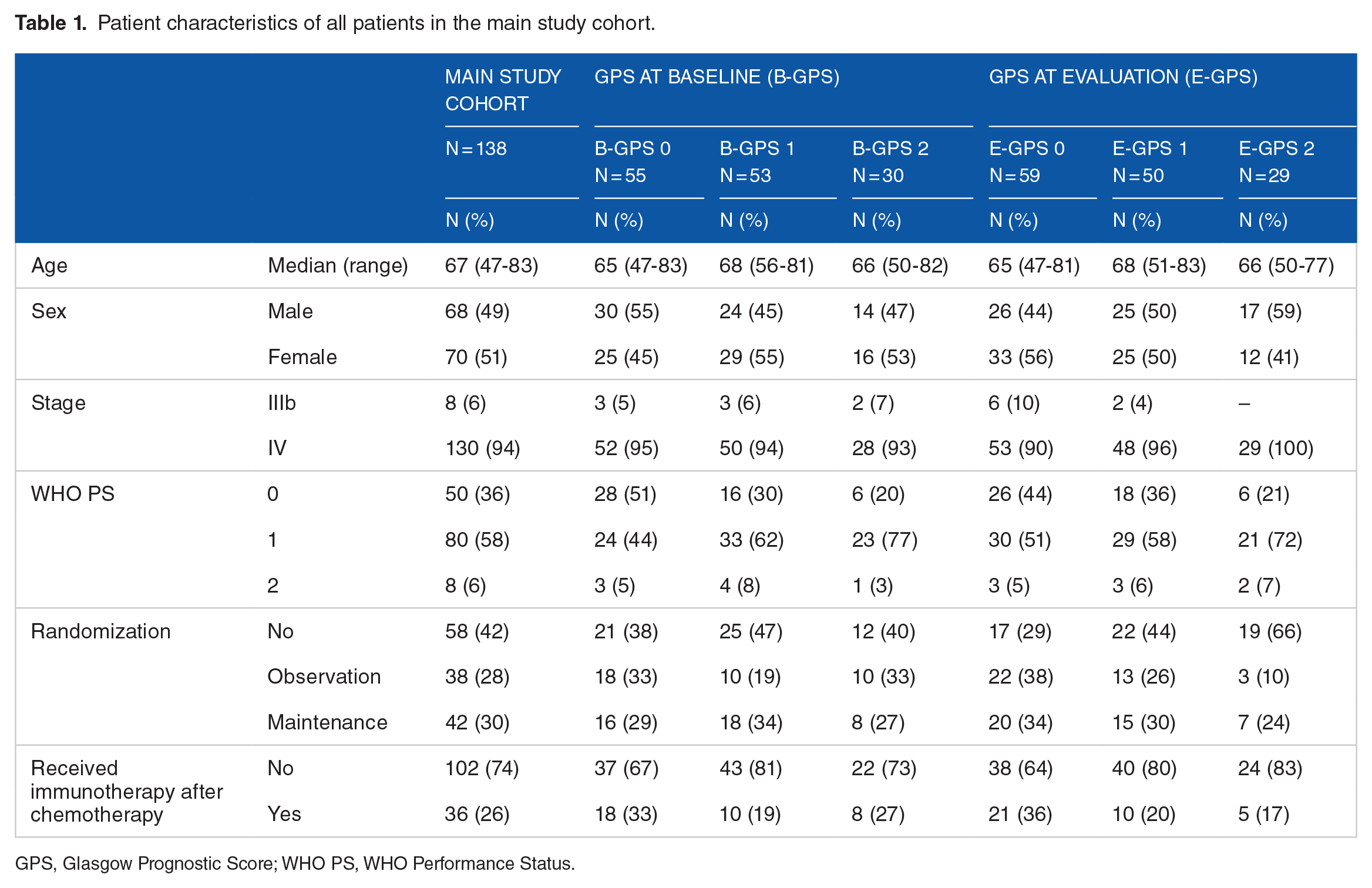
GPS, Glasgow Prognostic Score; WHO PS, WHO Performance Status.
In our main study cohort, 55 (40%) patients had B-GPS 0, 53 (38%) B-GPS 1, and 30 (22%) B-GPS 2. At evaluation after induction chemotherapy, 59 (43%) patients had E-GPS 0, 50 (36%) E-GPS 1, and 29 (21%) E-GPS 2 (Table 1). Patients with B-GPS 0 were more likely to have PS 0 than patients with B-GPS 1-2. Otherwise, baseline and treatment characteristics were balanced between patients with B-GPS 0, 1, and 2.
Seventy-three patients (53%) had no change in GPS. Thirty-three patients (24%) improved their GPS; 19 (14%) from 1 to 0, 8 (6%) from 2 to 1, and 6 (4%) from 2 to 0. Glasgow prognostic score deteriorated in 32 (23%) patients; 19 (14%) from 0 to 1, 2 (1%) from 0 to 2, and 11 (8%) from 1 to 2.
Baseline characteristics of the 70 patients included in the sensitivity analysis and the 24 excluded patients (Figure 1) were comparable to the characteristics of the main study cohort (Supplementary Table).
Overall survival
B-GPS and survival
Overall, median OS was 10.6 months (95% CI: 9.2-11.9) in the main study cohort (n = 138). Patients with B-GPS 0, 1, and 2 had median OS of 13.5 (95% CI: 9.9-17.1) months, 9.8 (95% CI: 8.0-11.6) months, and 8.7 (95% CI: 5.8-11.6) months, respectively (Figure 2A). There were no statistically significant differences in OS according to B-GPS in univariable or multivariable analyses (Figure 2A and Table 2), nor in a post hoc multivariable analysis in which B-GPS 1 and 2 were pooled and compared with B-GPS 0 (hazard ratio [HR] = 1.27, 95% CI: 0.87-1.88, P = .22 in multivariable analysis).
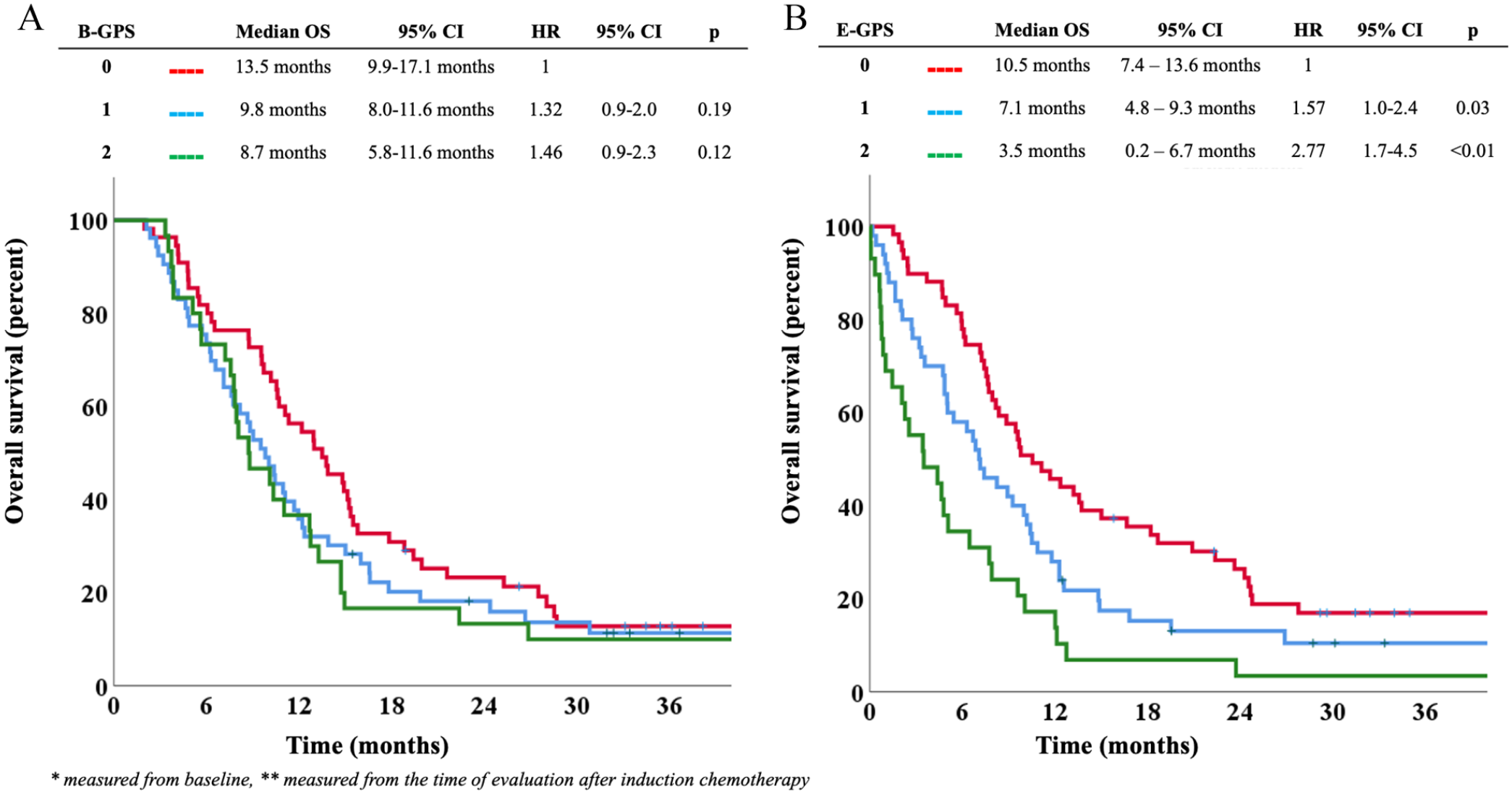
(A) B-GPS and overall survival in the main study cohort.* (B) E-GPS and overall survival in the main study cohort.** B-GPS indicates Glasgow Prognostic Score at baseline; CI, confidence interval; E-GPS, Glasgow Prognostic Score at evaluation; HR, hazard ratio; OS, overall survival. *Measured from baseline. **Measured from the time of evaluation after induction chemotherapy.
Univariable and multivariable analyses of B-GPS and overall survival in the main study cohort. a
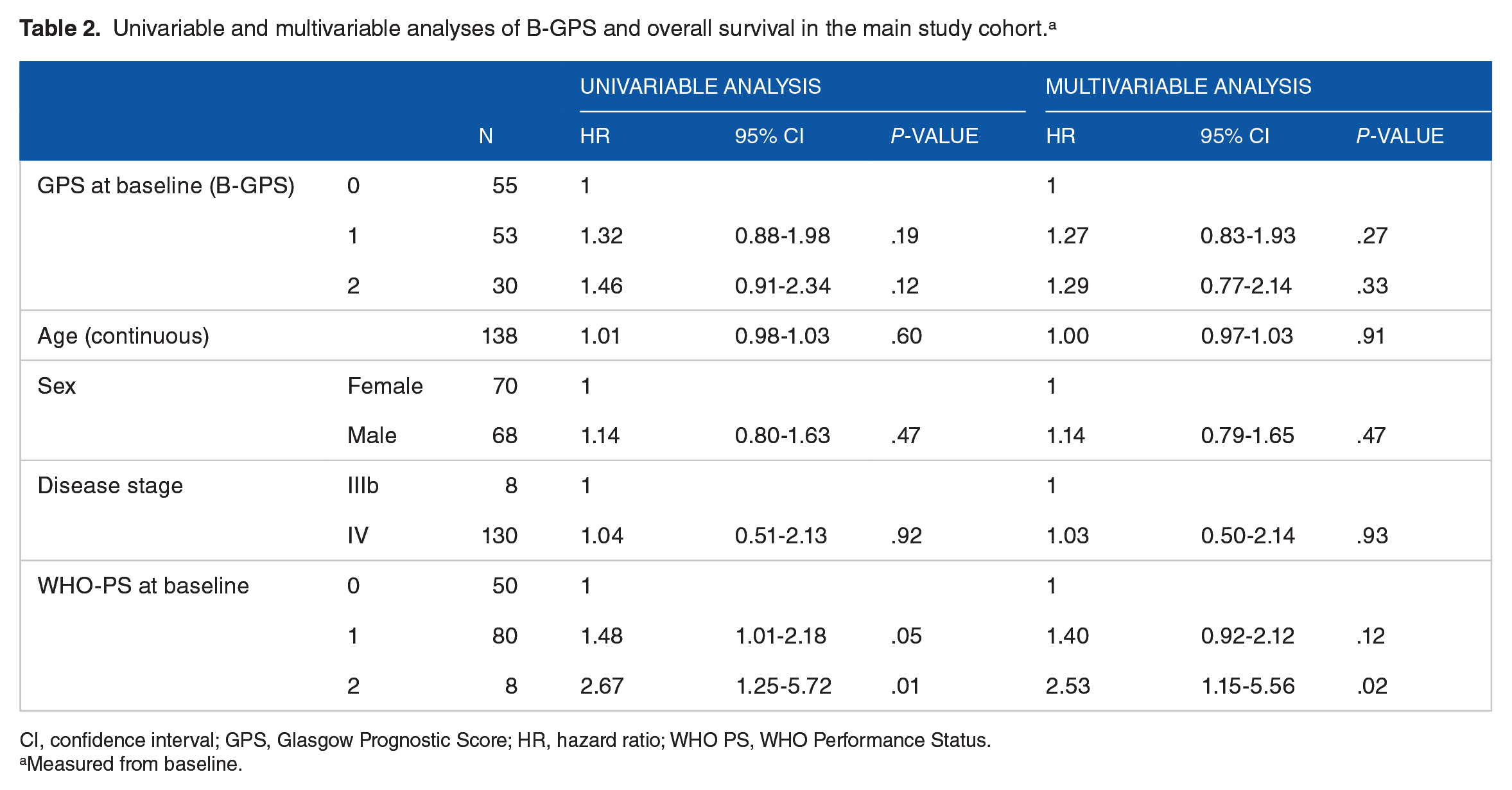
CI, confidence interval; GPS, Glasgow Prognostic Score; HR, hazard ratio; WHO PS, WHO Performance Status.
Measured from baseline.
Sensitivity survival analysis of all patients with B-GPS measured (n = 208) showed that patients with B-GPS 0, 1, and 2 had median OS of 13.8 (95% CI: 11.1-16.5) months, 7.1 (95% CI: 4.8-9.4) months, and 8.2 (95% CI: 7.1-9.3) months, respectively. For this group, the lower survival in patients with B-GPS 1 compared with B-GPS 0 reached statistical significance (HR = 1.51, 95% CI: 1.1-2.1, P = .01) (Supplementary Figure).
In the final sensitivity multivariable survival analysis of the main study cohort (n = 138) adjusting for randomization and whether patients later received immunotherapy, B-GPS was still not a significant prognostic factor (data not shown).
E-GPS and survival
Overall, median OS from evaluation after first-line chemotherapy was 7.7 months (95% CI: 6.3-9.2). Patients with E-GPS 0, 1, and 2 had median OS of 10.5 (7.4-13.6) months, 7.1 (4.8-9.3) months, and 3.5 (0.2-6.7) months, respectively. Higher E-GPS was significantly associated with shorter survival time; HR = 1.57 (95% CI: 1.04-2.37, P = .03) for E-GPS 1 as compared with E-GPS 0, and HR = 2.77 (95% CI: 1.73-4.45, P < .01) for E-GPS 2 as compared with E-GPS 0 (Figure 2B). In the multivariable analysis, the survival difference between E-GPS 2 vs 0 remained statistically significant (P < .01), while there was a trend toward a significant difference between E-GPS 1 and 0 patients (P = .08) (Table 3).
Univariable and multivariable analyses of E-GPS and overall survival in the main study cohort. a
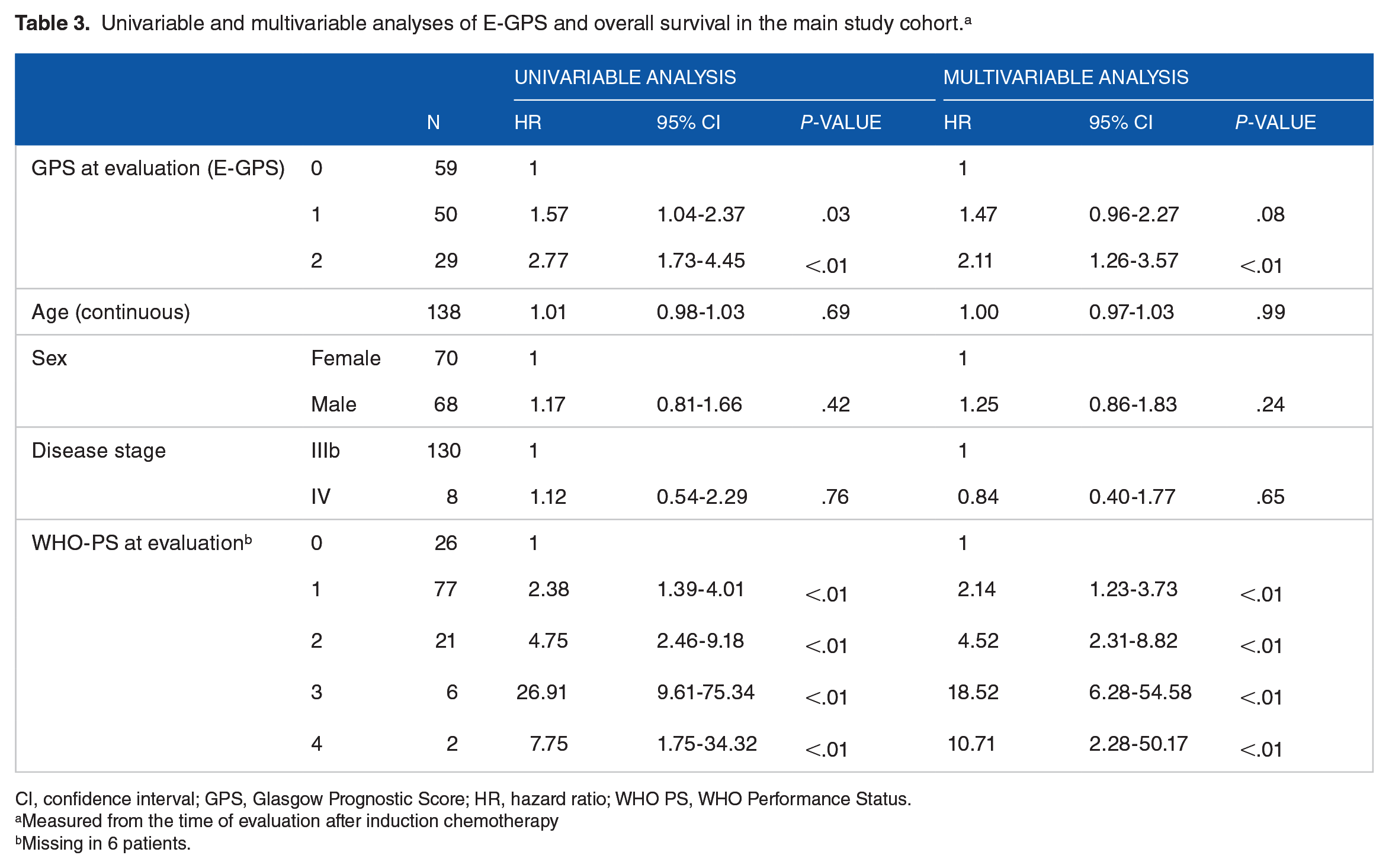
CI, confidence interval; GPS, Glasgow Prognostic Score; HR, hazard ratio; WHO PS, WHO Performance Status.
Measured from the time of evaluation after induction chemotherapy
Missing in 6 patients.
In the sensitivity multivariable survival analysis adjusting for randomization and whether patients received subsequent immunotherapy, E-GPS but not B-GPS remained a significant prognostic factor (data not shown).
GPS and response to induction chemotherapy
At evaluation after induction chemotherapy, 38 patients (28%) had partial response (PR), 48 (35%) had stable disease (SD), 48 (35%) had progressive disease (PD), and 4 (3%) were not evaluable (Table 4).
B-GPS, E-GPS, and change in GPS and response to first-line chemotherapy in the main study cohort.

B-GPS was not significantly associated with treatment response (P = .54), whereas E-GPS was (P < .01). Forty-one percent of patients with E-GPS 0 had achieved a PR, while corresponding numbers among patients with E-GPS 1 and E-GPS 2 were 21% and 14%, respectively. Furthermore, change in GPS was associated with treatment response (P = .01). Among patients with improved GPS, 45% had achieved a PR, among those with stable GPS, 26% had a PR, while 13% of those with worse GPS had a PR.
Discussion
In this study of patients with advanced non-squamous NSCLC, we found that GPS assessed at evaluation after 3 or 4 courses of first-line platinum-doublet chemotherapy (E-GPS) was prognostic for survival, whereas GPS at baseline (B-GPS) was not. Furthermore, patients with a low E-GPS and those with an improved GPS from baseline until evaluation after chemotherapy had higher response rates to chemotherapy than other patients. There was no significant association between B-GPS and response to chemotherapy.
Studies of the prognostic role of GPS in cancer comprise more than 70 000 patients,7-9 but only a few have looked at the impact of GPS measured during or after treatment: Three small studies (n = 24-64) reported that elevated GPS measured 3 to 6 weeks after initiation of immune checkpoint inhibitor therapy was associated with poor survival in advanced NSCLC30,31 and renal cell carcinoma. 32 Others have found that elevated GPS after initiation of palliative chemotherapy for colorectal cancer 33 and after surgery for localized NSCLC 34 and gastric cancer 15 was associated with poor prognosis. A study of patients with advanced head and neck cancer found that GPS after concurrent chemoradiotherapy was associated with recurrence free and overall survival, whereas pretreatment GPS was not. 35 Moreover, elevated modified GPS (mGPS) after neo-adjuvant chemotherapy before surgery for adenocarcinoma of the esophagogastric junction was associated with reduced survival, whereas pre-neo-adjuvant mGPS was not. 16 Overall, these studies corroborate the results of our study. In contrast, Forrest and colleagues studied patients with inoperable NSCLC treated with chemotherapy 11 and found that B-GPS was associated with survival, whereas GPS measured 3 to 6 months after inclusion was not. However, only a minority (42%) of patients received active cancer treatment and a minority (38%) had GPS measured during follow-up.
Most previous studies of NSCLC have found B-GPS to be a prognostic factor,5,6,10-13,22,23 but many studies have pooled B-GPS categories when running the analyses,6,8-10,30,36,37 which limits the evidence for the prognostic value of each of the 3 different GPS values. Furthermore, most included relatively unselected populations, only subsets of patients received cancer treatment,6,11 and many included patients with PS of 3 or 4.6,11,22,23 Our main analyses only included patients who completed 3 or 4 courses of chemotherapy, and the majority (94%) had a PS of 0 to 1. Thus, it is possible that B-GPS provides less prognostic information in patients who are considered fit for systemic cancer treatment than in less selected cohorts including cancer patients unfit for palliative chemotherapy, because there might be less variation in prognostic/predictive factors including B-GPS. The potential impact of patient selection might explain why there was a statistically significant survival difference between B-GPS 0 and 1 in our expanded cohort (all 208 with a B-GPS), while this was not the case in the main study cohort.
To the best of our knowledge, no studies have assessed E-GPS and response, but neutrophil-to-lymphocyte ratio (NLR) and CRP after targeted therapy or immune checkpoint blockade were associated with overall response rate in advanced renal cell carcinoma.32,38,39 Although treatments, settings, and design are different, these studies support the hypothesis that E-GPS holds more prognostic information than B-GPS because it incorporates the treatment effect. A possible explanation, as hypothesized, is that effective systemic therapy reduces the cancer-induced inflammation. On the contrary, systemic inflammation might reduce the effect of chemotherapy, possibly due to influence on tumor microenvironment. 40 However, our study was not designed to investigate underlying mechanisms.
The main limitation of our study is the sample size. Furthermore, we cannot rule out that our results might have been influenced by a selection bias. There were only lab values for assessing E-GPS in 138 of the 232 patients included in the trial, and most common reasons for not measuring E-GPS were death, progression, or poor PS. The time frames for measuring CRP and albumin were generous. There was only a trend toward a statistically significant difference between E-GPS 0 and 1 patients in the multivariable survival analysis (P = .08). In sensitivity analyses including all patients with B-GPS measured (n = 208), there was a statistically significant difference in survival between patients with B-GPS 0 and 1, possibly indicating that B-GPS have less prognostic value among cancer patients who tolerate palliative chemotherapy than in less selected populations including patients unfit for such therapy. Finally, GPS might also be influenced by malnutrition and side effects from chemotherapy such as nausea and anorexia, and one study shows that patients with a poor B-GPS experience more toxicity from cancer therapy. 13 Unfortunately, our study was not designed to investigate such complex interactions.
Another limitation of our study is that subsequent treatment differed largely between the participants, especially as immunotherapy was introduced during the study period. However, this is not likely to affect our results, as it would rather be a mediator than a confounder of the association between GPS and OS. And in sensitivity analysis adjusting for group in our original RCT (randomized to pemetrexed maintenance therapy, randomized to observation, or did not meet criteria for randomization) and whether patients received immunotherapy or not, the prognostic value of E-GPS remained stronger than for B-GPS (data not shown). Platinum-doublet chemotherapy alone is no longer standard primary treatment for advanced NSCLC, but our and previous studies have demonstrated associations between E-GPS and response to treatment and survival in patients with several cancers receiving different therapies, indicating that E-GPS reflects treatment effect independently of treatment modality. Finally, CRP and albumin, and thereby GPS, could have been influenced by other factors, ie, infection, inflammation, comorbidity, nutrition, and medication (eg, corticosteroids), but our study was not designed to collect such data. On the contrary, this also applies to most previous studies of GPS.6,11,23,31- 37
The main strength of our study is that we have investigated a relatively uniform patient population. Furthermore, the differences in survival between the 3 E-GPS categories are relatively large and clinically meaningful and might guide clinicians when planning follow-up intervals of patients, when considering maintenance therapy, or switching ongoing treatment. However, the clinical value of E-GPS and how it should be used needs to be further evaluated, ideally in prospective trials.
Conclusion
To conclude, we found that E-GPS was a stronger prognostic factor than B-GPS and that E-GPS, but not B-GPS, was significantly associated with response to chemotherapy in patients with advanced non-squamous NSCLC.
Supplemental Material
sj-pptx-1-onc-10.1177_11795549221086578 – Supplemental material for Prognostic Value of Post First-Line Chemotherapy Glasgow Prognostic Score in Advanced Non-Small Cell Lung Cancer
Supplemental material, sj-pptx-1-onc-10.1177_11795549221086578 for Prognostic Value of Post First-Line Chemotherapy Glasgow Prognostic Score in Advanced Non-Small Cell Lung Cancer by Kristin Stokke, Marie Søfteland Sandvei, Bjørn Henning Grønberg, Marit Slaaen, Kristin T Killingberg and Tarje O Halvorsen in Clinical Medicine Insights: Oncology
Supplemental Material
sj-tiff-1-onc-10.1177_11795549221086578 – Supplemental material for Prognostic Value of Post First-Line Chemotherapy Glasgow Prognostic Score in Advanced Non-Small Cell Lung Cancer
Supplemental material, sj-tiff-1-onc-10.1177_11795549221086578 for Prognostic Value of Post First-Line Chemotherapy Glasgow Prognostic Score in Advanced Non-Small Cell Lung Cancer by Kristin Stokke, Marie Søfteland Sandvei, Bjørn Henning Grønberg, Marit Slaaen, Kristin T Killingberg and Tarje O Halvorsen in Clinical Medicine Insights: Oncology
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported byt the Liaison Committee between the Central Norway Regional Health Authority (RHA) and the Norwegian University of Science and Technology, and the Research Council of Norway.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
BHG and TOH conceived and designed the work and acquired data and handled funding and supervision. KS performed the statistical analyses and drafted the manuscript. All authors played an important role in interpreting the results. All authors revised the manuscript for important intellectual content, approved the final version, and agree to be accountable for all aspects of the work.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
