Abstract
Breast cancer as the most common female cancer is a malignancy with heterogeneous course. Dysregulation of several genes has been associated with development of this malignancy. Among these genes are the stem cell markers CD61 and breast cancer resistance protein (BCRP or ATP-binding cassette super-family G member 2 (ABCG2)). ABCG2 is one of the major efflux transporters implicated in multidrug resistance in cancer cells. In the present study, we compared expression of CD61 and ABCG2 transcripts between 30 breast cancer tissues and matched adjacent non-cancerous tissues (ANCTs) using real time qPCR technique. There was no significant difference in expression of CD61 or ABCG2 between tumoral tissues and ANCTs (Expression ratios = 1.21 and 0.98, P values = 0.55 and 0.96, respectively). There was a trend toward association between relative expression of CD61 (tumoral tissues versus ANCTs) and patients’ age (P = 0.05) in a way that older patients tended to over-express this marker in their tumoral tissues compared with the matched ANCTs. Moreover, there was a significant association between expression of this gene and tumor size (P = 0.04) in a way that all tumors with sizes less than 2 cm showed down-regulation of CD61 (as compared with the matched ANCTs). Expression of CD61 was significantly higher in tumor tissues with extracapsular nodal extension compared with confined lesions (P = 0.007). Moreover, expression of ABCG2 was significantly higher in tumor tissues of patients aged less than 55 years compared with older patients (P = 0.04). There was no significant correlation between expression of CD61 and ABCG2 either in tumoral tissues or in ANCTs. The current investigation shows association or trends toward association between expression of two cancer stem cell markers and some clinical data of breast cancer patients such as extracapsular nodal extension, age and tumor size which might imply their importance in the pathogenesis of breast cancer.
Keywords
Introduction
Nucleotide sequences of primers and the corresponding amplicon sizes
Nucleotide sequences of primers and the corresponding amplicon sizes
Breast cancer is the most frequent malignancy among females and the second principal reason of cancer-related death according to the International Agency for Research on Cancer [1]. In Iran, breast cancer is the 5th principal cause of cancer death among women [2]. Most cases of breast cancer have been recognized in omen between 40 to 50 years [2]. This kind of cancer is among human malignancies in which the role of cancer stem cells (CSCs) in their evolution has been acknowledged [3]. Previous studies have identified a number of surface markers for CSCs. For instance, over-expression of the integrin CD61 (β3) has been reported as a feature of a mammary progenitor cell population. This surface marker is merely expressed in normal epithelial cells of this tissue [4]. Tumorigenic ability of CD61 over-expressing cells has been also verified in mouse models of mammary tumorigenesis [5]. Further investigations have shown that the CD61 signaling pathway has a crucial role in maintenance of active TGF-β signaling and self-renewal of tumor initiating cells [6]. Another important factor in sustaining stem cell properties in breast cancer cells is the ATP-binding cassette (ABC) super-family G member 2 (ABCG2) [7]. This member of ABC transporter superfamily also participates in multidrug resistance (MDR) in cancer chemotherapy [8]. Burger et al. have reported association between expression level of ABCG2 in breast cancer and response to anthracycline-based chemotherapy [9]. Faneyte et al. demonstrated extensive variation in expression of this gene in clinical samples and no association between its levels and response of patients to therapeutic options. While they detected ABCG2 expression in vessels and normal breast epithelium, they could not detect its expression in tumor cells [10]. On the contrary, Yuan et al. appreciated the role of ABCG2 in determination of response of breast cancer cells to 5-fluorouracil [11].
Based on the putative importance of ABCG2 in response of patients to chemotherapeutic options and lack of consistent data in this regard, we selected this gene to compare its expression levels between breast cancer tissues and adjacent non-cancerous tissues (ANCTs). Moreover, we compared expression of the CSC marker CD61 in these two sets of tissues to assess its relative expression and its association with clinicopathological data of breast cancer patients.
Patients
The current study was performed on 30 breast cancer samples and 30 matched ANCTs. All tissues were acquired from Tumor Bank of Cancer Research Institute, Imam Khomeini Hospital, Tehran, Iran. Comprehensive histopathological features were also gathered. The study protocol was approved by the institutional ethic committee. Patients signed the informed consent forms.
RNA extraction, cDNA synthesis and expression study
Total RNA was isolated from both tumor tissues and ANCTs using TRIzolTM Reagent (Invitrogen, Paisley, UK). Then, 500 ng−1 μg of the extracted RNA was used for cDNA synthesis using the RevertAid First Strand cDNA Synthesis Kit (TaKaRa, Tokyo, Japan). Expression of CD61 and ABCG2 were measured in tumoral tissues and ANCTs using RealQ Plus 2x PCR Master Mix Green Without ROXTM PCR Master Mix (Ampliqon, Odense, Denmark). Expression levels were normalized to the expression amounts of the housekeeping gene glyceraldehyde 3-phosphate dehydrogenase (GAPDH). The nucleotide sequences of primers and the corresponding amplicon sizes are summarized in Table 1.
Statistical methods
SPSS v.18.0 (IBM Corp., Armonk, NY, USA) was used for statistical analyses. Efficiencies of each primer set were used for calculation of relative expression levels. Associations between clinical data and relative expression of CD61 and ABCG2 were evaluated using Chi-square test. Mean values of expression of genes in tumoral tissues were compared between subgroups using independent sample T test. Correlation between relative expressions of CD61 and ABCG2 was assessed using the regression model. P values less than 0.05 was considered as significant. P values were calculated using Chi-square test for categorical variables [12] and using T test when comparing the means of two groups, which were related in certain features [13].
Results
General features of breast cancer patients
Patients enrolled in the current study had the mean age of 51.82 years. Tumors were originated from right and left sides in 53.3% and 46.7% of patients, respectively. Cancer stage, grade, tumor size and immunohistochemical parameters were obtained from medical records. Table 2 summarizes the clinical data of breast cancer patients.
Clinical data of enrolled individuals in the current study
Clinical data of enrolled individuals in the current study
There was no significant difference in expression of CD61 or ABCG2 between tumoral tissues and ANCTs (Expression ratios = 1.21 and 0.98, P values = 0.55 and 0.96, respectively). Figure 1 shows relative expression levels of these genes in each group of specimens.
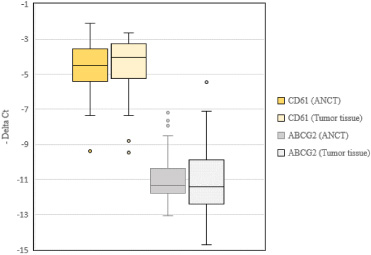
Relative expression levels of CD61 and ABCG2 genes in tumoral tissues and ANCTs.
There was a trend toward association between relative expression of CD61 (tumoral tissues versus ANCTs) and patients’ age (P = 0.05) in a way that older patients tended to over-express this marker in their tumoral tissues compared with the matched ANCTs. Moreover, there was a significant association between expression of this gene and tumor size (P = 0.04) in a way that all tumors with sizes less than 2 cm showed down-regulation of CD61 (as compared with the matched ANCTs). Table 3 shows the results of association analysis between expression of genes and clinical data.
The results of association analysis between expression of genes and clinical data (For some parameters, there was a number of missing or inconclusive data)
Expression of CD61 was significantly higher in tumor tissues with extracapsular nodal extension compared with confined lesions (P = 0.007). Moreover, expression of ABCG2 was significantly higher in tumor tissues of patients aged less than 55 years compared with older patients (P = 0.04). Table 4 shows the results of association analysis between expression of CD61 and ABCG2 genes in tumoral tissues and clinical data.
There was no significant correlation between expressions of CD61 and ABCG2 in ANCTs or tumoral tissues (Figs 2A and 2B, respectively).
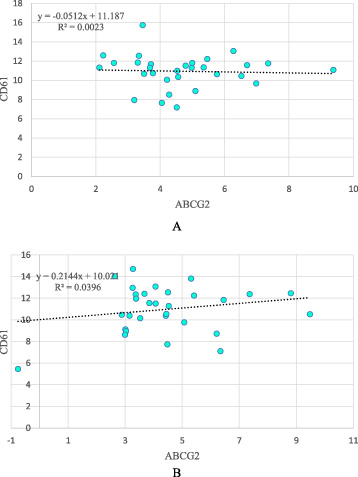
Correlations between expression levels of CD61 and ABCG2 in ANCTs (A) and tumoral tissues (B).
In the current study, we compared expression of two CSC-related markers i.e. CD61 and ABCG2 between breast cancer tissues and matched ANCTs in a cohort of Iranian patients. Based on previous studies, Iranian patients with breast cancer have typically no history of abortion, smoking, or diabetes. The majority of these patients have been estrogen and progesterone receptor positive, HER2 negative, and P53 negative [2]. Although we reported similar expression levels of these transcripts between tumoral tissues and ANCTs, expressions of these transcripts were associated with some of clinical determinants of patients’ outcome. First, there was a trend toward association between relative expression of CD61 and patients’ age in a way that older patients tended to over-express this marker in their tumoral tissues compared with the matched ANCTs. Considering the role of CD61 in the maintenance of CSCs [6], this finding is in line with the previously reported poor survival of breast cancer in elderly [14]. Consistent with the role of CD61 in breast tumorigenesis, all tumors with sizes less than 2 cm showed down-regulation of CD61 when compared with the matched ANCTs. Moreover, expression of CD61 was significantly higher in tumor tissues with extracapsular nodal extension compared with confined lesions.
Association between expression of CD61 and ABCG2 genes in tumoral tissues (designated by Ct housekeeping gene- Ct target gene) and clinical data (P values were calculated by independent samples T test)
However, expression of ABCG2 was significantly higher in tumor tissues of patients aged less than 55 years compared with older patients. This finding is in contrast with the previously reported role of estrogen in down-regulation of ABCG2 [15]. Considering the putative role of ABCG2 in determination of response to certain types of chemotherapeutic agents such as 5-fluorouracil [11], quantification of its expression levels in tumoral tissues might be a practical approach for selection of anti-cancer treatments.
Previous studies have mostly assessed expression of ABCG2 at protein level. Yoh et al. has shown association between expression of this protein in non-small cell lung cancer samples and response to platinum-based chemotherapy [16]. Omran has shown ABCG2 protein expression in the majority of patients with invasive ductal carcinoma. He also showed correlation between ABCG2 expression and tumor grade, clinical stage, lymph node metastasis, and HER2 status. However, ABCG2 expression was not correlated with PR or ER status [17]. Similar to this study, we demonstrated no association between ABCG2 expression and PR or ER status. But our results regarding lack of association with HER2 status differ from this study. Yuan et al. have examined expression of ABCG2 in 140 breast cancer samples and reported expression of this gene 47 samples at both protein and mRNA levels [18]. Our study differs with these studies as we assessed relative expression of ABCG2 in tumoral tissues compared with ANCTs to find whether its expression is influenced by the presence of malignancy. The similar levels of expression of this gene in the mentioned sets of samples might indicate the effect of tumor microenvironment on modulation of its expression. However, assessment of its expression levels in samples from cancer-free patients is needed to appraise this hypothesis.
There was no significant correlation between expression of CD61 and ABCG2 either in tumoral tissues or in ANCTs which might indicate the presence of independent mechanisms for their regulation.
Taken together, our study shows association or trends toward association between expression of two CSC marker and some clinical data of breast cancer patients such as extracapsular nodal extension, age and tumor size which might imply their importance in the pathogenesis of breast cancer. Our study had a limitation regarding the sample size.
Footnotes
Acknowledgements
This study was supported financially by Shahid Beheshti University of Medical Sciences.
Conflict of interest
The authors declare they have no conflict of interest.
