Abstract
BACKGROUND:
Breast cancer heterogeneity is well documented and to some extent is attributed to the presence of cancer stem cells (CSCs). Breast cancer stem cells are identified by the presence of cell surface molecule CD44 and absence of CD24.
METHODS:
In the present study a flowcytometric analysis was done to study the expression distribution of CSC phenotype of CD44+/CD24-/low, among different molecular subtypes of breast cancer and to find a correlation with clinicopathological features.
RESULTS:
CSCs were observed in all the molecular subtypes of breast cancer. The highest population of CSCs was noted in luminal B (3.4%), followed by TNBC (1.7%), and Her-2 subtype (1.6%). The least number of CD44+/CD24- cells were seen in Luminal A subgroup (1.3%).
CONCLUSION:
Existence of cancer stem cells in all the subtypes may suggest the possibility of failure of current therapies in treatment of patients.
Introduction
Based on immunohistochemistry (IHC) as a surrogate marker for molecular classification, breast cancer has been divided into four subtypes; luminal A, luminal B,HER2neu overexpressing and triple-negative breast cancer (TNBC). Over the last few decades, ER and HER2 expression profile of the breast cancer assumed greater prognostic and predictive importance. The TNBC subtype however exhibits pathological, molecular, and clinical heterogeneity [1]. Cumulative data indicated tumours of different histological grades show distinct molecular profiles at the genomic, transcriptomic and immunohistochemical levels [2–4]. Despite molecular classification, the tumors exhibit heterogenicity due to clonal evolution, microenvironmental differences, and the hierarchical organization. Tumor initiating stem cells also known as Cancer stem cells (CSCs) are defined as a population of cells present in tumours, which can drive the initiation, progression, metastasis, recurrence and association with poor prognosis. Identification and isolation of these CSCs using putative surface markers have been a priority of research in cancer. The existence of cancer stem cells (CSCs) in breast cancer has been demonstrated by several studies [5–7]. With advancement of the knowledge in this concept, the well-accepted cancer stem cell surface markers are CD44, CD24, CD133, CD166, EpCAM, and so forth. However, the prognostic value of these markers is still under investigation [5]. In the present study, a flowcytometric analysis was done to study the expression distribution of CSC phenotype of CD44+/CD24-/low, among different molecular subtypes of breast cancer and to find a correlation with clinicopathological features.
Methods
A prospective study was performed on 143 breast cancer patients who underwent mastectomy or lumpectomy as a therapeutic procedure for breast cancer at our centre. Treatment naïve patients, at the time of surgery, were included in the study. All the patients who have received neo-adjuvant Chemotherapy (NACT) prior to surgery (mostly with TNM stage IV) were excluded from the study. Fresh tumour tissue was collected and used for flowcytometric analysis and remaining specimen was grossed as per the standard protocol for histopathological analysis. All tumours were examined by the experienced pathologists (AB, AD) and were graded as per Elston and Ellis modified Bloom-Richardson (MBR) grading system. The tumor size, grade, histology and lymph node involvement was recorded. For Surrogate molecular subtyping immunoreactivity for Ki67, ER, PR, HER2neu status was studied on VENTANA BenchMark-XT computerized automated system. The anti-estrogen receptor (Dako, SP1 clone, 1:20 dilution), anti-progesterone receptor (Dako, IE2 clone, 1:50 dilution), and anti- HER2neu (Dako, 4B51 clone, 1:250 dilution) antibodies were used for immunohistochemistry. For secondary antibody, the ultra-View Universal 3, 3'- diaminobenzidine tetrahydrochloride (DAB) chromogen detection kit was used. The immunohistochemistry results and scores were recorded as per American Society of Clinical Oncology/College of American Pathologists Clinical Practice Guideline Focused Update.
For Flowcytometric analysis, fresh tissue was digested with trypsin (0.5%Gibco) for three hours at 37 °C. Cells were separated from undigested tissue and washed with 1X PBS. Cell pellet was re-suspended and incubated with antibodies: APC conjugated CD44, PE conjugated CD24, FITC conjugated lineage positive population (BD Biosciences, USA), for 30 minutes at 37 °C. Excess antibody was washed with 1X PBS and cell pellet was re-suspended in 1X PBS. Immediately after tagging with antibodies, the cells were analysed using FACS Aria (Becton Dickinson, USA) flow cytometer.
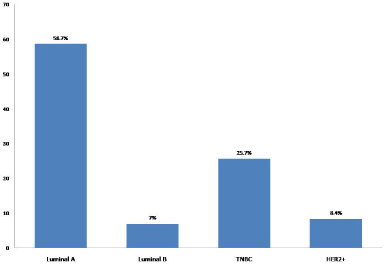
Patient categorization based on surrogate molecular subtyping of breast tumours.
The histologically proven breast cancer patients enrolled in this study were in age range of 20–80 years (mean age of 50.5 ± 13.17 years). Majority of cases were in age group of 41–60 years (n = 78), followed by 20–40 years (n = 35 cases), and 61–80 years (n = 30). Tumor size varied between 0.3 cm–7.0 cm in the largest dimensions. Majority (69.9%) had tumour size in range of 2.0–5.0 cm (pT2), 30.6% had tumour <2.0 cm (pT1) and 1.4% had tumours greater than 5.0 cm (pT3) in largest dimensions. Histologically, most frequent subtype was infiltrating ductal carcinoma-No special type (IDC-NST, 89.5%). Rest of the cases were of invasive cribriform carcinoma (0.7%), apocrine carcinoma (0.7%), solid papillary carcinoma (1.4%), mucinous carcinoma (0.7%) and metaplastic carcinoma (1.4%). The majority tumours were grade III (48.2%) followed by grade II category (44.0%), and grade I (7.0%) tumours. In 40.4% case axillary lymph node involvement was observed. Less than 4 lymph nodes were involved in 23.7% and more than 4 lymph nodes were involved in 16.7%. None of the cases showed distant metastasis at the time of diagnosis.
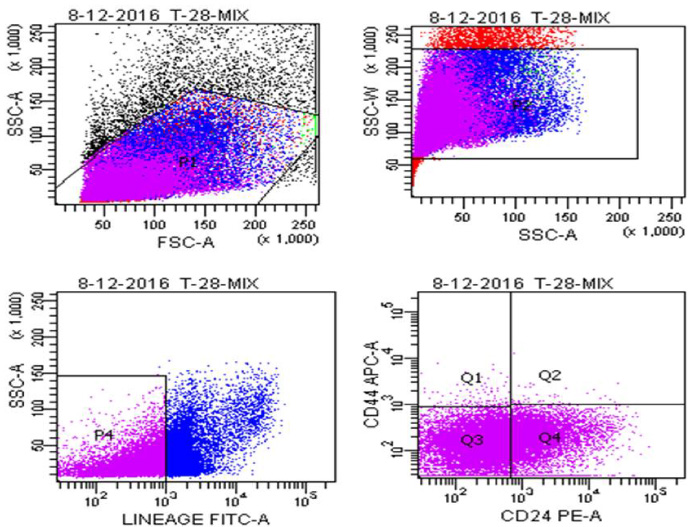
Gating strategy used for enumerating cancer initiating cells by flowcytometery.
Majority cases (58.7%) were Luminal A type (ER+/PR+/Her-2-ve), followed by TNBC (25.7%), Her-2 over-expressing (8.4%) and least (7.0%) were of Luminal B type (Fig. 1). Tumours with ER+/PR+/Her2+ expression were labelled as Luminal B subtype irrespective of Ki67 index. Ki67 expression was evaluated in all the subtypes of breast cancer. Expression levels <14% were taken as low Ki and >14% were taken as high Ki. The mean Ki67 expression across different subtypes were 16.4% ± 11.2 in Luminal A, 24.5% ± 9.6 in Luminal B, 21.2 ± 7.2 in TNBC, and 29.5 ± 9.2 in Her2 over-expressing subtype. The cancer initiating cells were identified as LinNegCD44+ & CD24−∕low population. The double discriminating gating was done using forward and side scatter (Fig. 2). Lineage negative population was separated and CD44+/CD24- cells were selected. The CD44+/CD24- cells were present in all the cases analyzed in the study. On comparing the mean CD44+/CD24- cell population across different molecular subtypes, the highest population was noted in luminal B (3.4%), followed by TNBC (1.7%), and Her-2 subtype (1.6%). The least number of CD44+/CD24- cells were seen in Luminal A subgroup (1.3%). However, no statistically significant correlation of number of cancer initiating cells was noted with the molecular subtypes (P > 0.080). There was a trend of high number of CD44+/CD24- cells in higher grade tumours, however statistical significance was not achieved. Proportion of CD44+/CD24- cells was higher in cases with lymph node metastasis (44% cases) however, no statistically significance was noted with lymph node status and tumour size. Correlation of BCSC with clinico-pathological parameters is detailed in Table 1.
Correlation of CD44+/CD24- cell population with different prognostic parameters
Breast cancer stem cells (BCSCs) represent a minor subset of cells in a breast tumor that is deemed as the driver of tumor initiation, displaying resistance to drugs and developing metastatic disease [8]. CD44 and CD24 are two important cell surface markers of cancer stem cells. Elevated expression of these markers has been observed in ovarian, breast, prostate, bladder, renal, non-small-cell lung carcinomas, and other human cancers [9,10]. Al-Hajj et al. observed that in the breast cancer cell lines the BCSCs can be identified as CD44+/CD24-/low cells [5]. In the present study we determined the expression of CD44+/CD24- marker in order to identify the population of CSC among different subtypes of human breast cancer samples. Also levels of expression were correlated with clinico-pathological features. BCSCs were observed in all the subtypes of breast cancer. These observations are in contrast to previous study which reported that CD44+/CD24- phenotype may be associated with only basal intrinsic subtype breast cancers [6]. To best of our knowledge, this is the first study reporting the presence of CD44+/CD24- stem cells in all the molecular subtypes of breast cancer. The population of CD44+/CD24- cells was higher in cases with lymph node involvement and these observations suggest that breast cancer stem cells present in the primary tumour may play an important role in metastasis as suggested by other studies [6,10]. High number of CD44+/CD24- cells were also found in higher grade tumours, however no statistical correlation between tumour size, lymph node status and presence of CD44+/CD 24-cells was observed.
In conclusion, we studied the distribution of CSC phenotype of CD44+/CD24- breast cancer stem cells in various breast cancer subtypes (luminal A, luminal B, Her2 overexpressing and TNBC) in Indian population. Breast cancer stem cells are known to resist the available clinical treatment thus resulting in relapse. Existence of cancer stem cells in all the subtypes, as observed in the present study, may possibly affect the efficacy of current therapies in treatment of patients. Hence there is a need to develop new treatment methods that not only affect differentiated tumour cells but also target stem cell population in the tumour.
Footnotes
Acknowledgements
None
Compliance with ethical standards
Yes
Conflicts of interest/disclosures
The authors declare that they have no financial or other conflicts of interest in relation to this research and its publication.
Sources of funding support
ICMR, India provided the funds for this study.
Contributions
Dr. Parul Gupta – Did flowcytometery, Analysed the results and prepared the manuscript
Dr. Vikram Singh – Pathologist, prepared the manuscript
Mr. Sandeep Kumar – PhD Scholar, did flowcytometery, analysed the results, prepared the manuscript
Dr. Ashim Das – Pathologist, Conceptualised and prepared the manuscript
Dr. Gurpreet Singh – Provided tissue samples and prepared the manuscript
Dr. Amanjit Bal – Pathologist, Conceptualised and prepared the manuscript
