Abstract
Breast cancer resistance protein 1 (BCRP1/ABCG2) is used to identify the side population (SP) within a population of cells, which is enriched for stem and progenitor cells in different tissues. Here, we investigated the role of extracellular signal-regulated kinase (ERK) 1/2 in the signaling mechanisms underlying ischemic/hypoxic conditions in kidney SP cells. Kidney SP cells were isolated using Hoechst 33342 dye-mediated fluorescein-activated cell sorting and then incubated under hypoxia/reoxygenation (H/R) with or without verapamil, a selective BCRP1/ABCG2 inhibitor. ABCG2 expression, ERK activity, cell viability, metabolic activity, and membrane damage were tested after H/R treatment. To evaluate the role of ERK 1/2 on the expression and function of ABCG2, the expression of mitogen-activated protein kinase (MAPK)/ERK kinase (MEK), which preferentially activates ERK, was upregulated by transfection with the recombinant sense expression vector pcDNA3.1-MEK and downregulated by pretreatment with U0126, a specific MEK inhibitor. We found that hypoxia activated ERK activity in the kidney SP cells but not in non-SP cells both in vitro and in vivo. Overexpression of MEK mimicked hypoxia-induced ABCG2 expression. Contrarily, U0126 inhibited hypoxia- and MEK-upregulated ABCG2 expression. Furthermore, H/R induced significant increases in nuclear, metabolic, and membrane damage in both SP cells and non-SP cells; however, this H/R-induced cytotoxicity was much more severe in non-SP cells than in SP cells. Notably, the viability of kidney SP cells was enhanced by MEK overexpression and inhibited by U0126. Verapamil treatment reversed MEK-induced viability of kidney SP cells. When administered systemically into animals with renal ischemia/reperfusion injury, the SP cells significantly improved renal function, accelerated mitogenic response, and reduced cell apoptosis. However, this improved therapeutic potential of SP cells was significantly reduced by pretreatment with verapamil. Collectively, these findings provide evidence for a crucial role for the MEK/ERK-ABCG2 pathway in protecting kidney SP cells from ischemic/hypoxic injury.
Keywords
Introduction
Side population (SP) cells, originally identified in murine bone marrow by their ability to efficiently efflux the DNA binding dye Hoechst 33342, have been proposed to be a stem cell-rich population (26). Several groups have confirmed the existence of SP cells in the kidneys of mice (2, 4, 9, 10, 14, 19), rats (16), and humans (15). The kidney SP cells can differentiate into multiple lineages (4, 10) and accelerate the recovery of acute kidney injury (4, 9, 14), suggesting that kidney SP cells are good targets for clinical renal regenerative therapy. The ability of SP cells to efflux Hoechst 33342 dye is dependent on the expression of the ATP-binding cassette, subfamily G, member 2 transporter (ABCG2), also known as the breast cancer resistance protein (BCRP) (12). Recent reports have demonstrated that the SP cells of various cell lines and primary cells have increased antiapoptotic activity compared with non-SP cells, and this is correlated with ABCG2 expression (20, 22, 24). Furthermore, renal ABCG2 protein levels are upregulated after ischemia/reperfusion (I/R) injury (13), suggesting that the upregulation is an adaptive response to accumulation of harmful factors and that ABCG2 can protect cells from hypoxic damage.
Extracellular signal-regulated kinase (ERK) 1/2, the only known target of mitogen-activated protein kinase (MAPK)/ ERK kinase (MEK) 1/2 phosphorylation, is involved in the regulation of cell proliferation, differentiation, and survival, and it is typically activated in response to cellular stress (5). ERK 1/2 activation is induced by ischemia in vivo and has been implicated as a modulator of renal I/R injury (1). However, the role of ERK 1/2 signaling in regulating ABCG2 expression in ischemic/hypoxic kidney SP cells is far from being fully understood.
The results of this study confirmed that ABCG2 expression in the kidney SP cells contributes to protection against hypoxia/reoxygenation injury by acting through the MEK/ERK pathway.
Materials and Methods
Isolation of Kidney SP Cells
C57BL/6 mice (8 weeks of age) were provided by the Experimental Animal Center of the Fourth Military Medical University (Xi'an, China). All the procedures were approved by the animal ethics committee of the university. The protocol was based on a report by Challen et al. (4). Mice were anesthetized and perfused via the abdominal aorta with normal saline. The kidneys were minced and digested enzymatically in Hank's balanced salt solution (HBSS) (Invitrogen, Stockholm, Sweden) containing 7.5 mg/ml collagenase B (Roche Diagnostics, Milan, Italy), 1.2 U/ml dispase II (Roche Diagnostics), and 0.01% DNAse type I (Sigma, St. Louis, MO, USA) for 20 min at 37°C. The cell suspensions were filtered through 40-μm strainers (BD Falcon 2350; BD Pharmingen, San Diego, CA, USA) to remove debris and washed in HBSS containing 2% fetal bovine serum (FBS) (Gibco, Grand Island, NY, USA) and 10 mmol/L HEPES (Sigma). To isolate SP cells, the cells were resuspended in Dulbecco's modified Eagle's medium (DMEM) (Hyclone, Logan, UT, USA) containing 10 mM HEPES, 2% FBS and 5 μg/ ml Hoechst 33342 (Sigma), with or without 50 μM verapamil (Sigma), at 1 × 106 cells/ml and incubated at 37°C for 90 min. After staining, the cells were washed with cold phosphate-buffered saline (PBS) and 2% FBS and stained with antibodies for 20 min on ice. The following antibodies were used: cluster of differentiation 45-fluorescein isothiocyanate (CD45-FITC), v-kit Hardy-Zuckerman 4 feline sarcoma viral oncogene homolog (c-kit)-FITC, and isotype controls (BD Pharmingen). Propidium iodide (PI) (2 μg/ml) (BD Pharmingen) was added prior to fluorescence-activated cell sorting (FACS) analysis to exclude dead cells and to ensure that all quantified and studied SP cells were viable. The results are given as the average of six experiments.
FACS Analysis
Cell analysis and sorting were performed on a duallaser FACS Vantage (BD Biosciences, Franklin Lakes, NJ, USA). Hoechst 33342 was excited at 355 nm UV light, fluorescence emission was collected with a 450/20 bandpass filter (Hoechst blue) and a 675-nm long-pass filter (Hoechst red), and a610-nm dichroic mirror short pass was used to separate the emission wavelengths. Phycoerythrin (PE), FITC, and PI fluorescence were detected using a 488-nm argon laser, and the live cell gate was defined as that which excluded the PI-positive cells.
Cell Culture
Cells were plated in DMEM containing 10% FBS, 5 μg/ ml transferrin, 5 μg/ml insulin, 5 pM thyronine, 50 nM prostaglandin, 50 nM selenium, and 50 nM hydrocortisone (all Sigma) at 37°C in a 95% air/5% CO2 incubator for 48 h. For normoxia exposure, cells were maintained at 37°C in a 95% air/5% CO2 incubator for 16 h. For hypoxia exposure, cells were maintained with or without verapamil under hypoxic conditions in an airtight chamber (Billups-Rothenberg, Del Mar, CA, USA) gassed with a mixture of 95% N2/5% CO2 at 37°C for 4 h. For reoxygenation, plates were removed from the hypoxic chamber and were incubated under standard conditions at 37°C (95% air/5% CO2) for 12 h.
Cytotoxicity Assays
Cells in the dish (105 cells/well) were treated with or without verapamil (50 μM) under hypoxia/reoxygenation (H/R) or normoxic conditions. The cytotoxicity studies of the cells were performed as described (25). The nuclear viability of the cells was measured using an automated cell counter (NucleoCounter, New Brunswick Scientific, Edison, NJ, USA), and the metabolic activity was measured using the 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) assay (Sigma). The cell counter technique uses PI, which binds to cellular nuclei. Depending on sample preparation, the counts provide the total number of cells or the fraction of viable cells. To better compare cell metabolic activity in different groups, the MTT results in other groups were expressed as a percentage compared to SP cells in normoxia group. Cytotoxicity was also determined by measuring the amount of lactate dehydrogenase (LDH) released in the cell culture medium using a Sigma assay kit by measuring the absorption at 340 nm.
Western Blot Analysis
For the detection of protein expression, the harvested cells were lysed on ice in a radio-immunoprecipitation assay (RIPA) buffer that included protease inhibitors (Sigma-Aldrich). The resulting supernatants were resolved by 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and blotted onto a polyvinylidene difluoride membrane (all Invitrogen). The membranes were treated with 1 × Tris-buffered saline-Tween 20 (TBST, Cell Signaling Technology Inc., Beverly, MA, USA) containing 5% nonfat milk for 2 h at room temperature and incubated with antibodies against p-ERK 1/2, total ERK 1/2 (Cell Signaling Technology Inc.), ABCG2 (BXP-53, Santa Cruz Biotech, Santa Cruz, CA, USA), and β-actin (Cell Signaling Technology Inc.). The membranes were washed and incubated with horseradish peroxidase-conjugated secondary antibodies (Santa Cruz Biotech). Enhanced chemiluminescence (Amersham, Freiburg, Germany) was used for detection.
RT-PCR Analysis
Total RNA was extracted from kidney SP cells after 24 h of exposure to hypoxia or MEK overexpression and after pretreatment with or without the MEK inhibitor U0126 (New England Biolabs Inc., Beverly, MA, USA). RNA was extracted using RNAzol (Molecular Research Center, Cincinnati, OH, USA) according to the manufacturer's instructions. First-strand cDNA was synthesized using oligo dT primers with reverse transcriptase (Promega Corp., Fitchurg, WI, USA). The PCR primers used were as follows: 5′-CCA TAG CCA CAG GCC AAA GT-3′ (forward) and 5′-GGG CCA CAT GAT TCT TCC AC-3′ (reverse) for ABCG2 product size 235 bp; 5′-TCG TGC GTG ACA TCA AAG AGA-3′ (forward) and 5′-GAA CCG CTC GTT GCC AAT AGT-3′ (reverse) for β-actin. The RT-PCR conditions for ABCG2 were as follows: 94°C for 5 min followed by 35 cycles of 94°C for 45 s, 58°C for 45 s, and 72°C for 1 min. After 35 cycles of amplification, additional extensions were carried out at 72°C for 10 min. The RT-PCR products were separated by 2% agarose gels and stained with ethidium bromide (both Invitrogen). Quantification of differences in mRNA levels was performed using the Kodak Digital Science Electrophoresis Documentation and Analysis System 290 (Eastman Kodak Co., Rochester, NY, USA).
Plasmid Constructs and Transfection
To upregulate MEK expression, the recombinant sense expression vector plasmid Cytomegalovirus promoter DNA 3.1 for MEK (pcDNA3.1-MEK) (Invitrogen) was constructed by subcloning the complementary deoxyribonucleic acid (cDNA) fragment of MEK that contained the complete coding sequence between KpnI and BamHI. All cells were transfected using Lipofectamine 2000 (Invitrogen) according to the manufacturer's instructions.
Renal I/R Injury
Models of renal I/R injury were performed in female, 6- to 8-week-old C57BL/6 mice by clamping the unilateral (left) or bilateral renal pedicle for 30 min followed by clamp release to allow reperfusion, as described previously (18). On the third day after unilateral renal I/R injury, kidney SP and non-SP cells were isolated from the ischemic (left) and nonischemic (right) kidneys, respectively. The aim was to compare the effect of in vivo hypoxia on the activation of ERK1/2 in kidney SP and non-SP cells. Since only bilateral renal I/R injury could cause severe deterioration of renal function, especially elevation in blood urea nitrogen (BUN) and serum creatinine (Scr) levels, to determine the potential for kidney SP cells to act as functional stem cells, cell transplantation was performed in animals 1 day after bilateral renal I/R injury. To ensure complete intravenous administration of the SP and non-SP cells, a 27-gauge cannula connected to a short polyethylene catheter (B Braun Melsungen AG, Melsungen, Germany) was used. At 24 h after surgery, the cells [1 × 106/0.5 ml low glucose (L)-DMEM; Invitrogen] were infused via a cannulated tube that was then flushed with 0.3 ml L-DMEM to infuse the cells remaining in the tubing. The animals were randomly assigned to one of five experimental groups as follows: group 1, serum-free L-DMEM; group 2, non-SP cells; group 3, SP cells; group 4, non-SP cells pretreated with verapamil; and group 5, SP cells pretreated with verapamil. Cells were pretreated with verapamil (50 μM/106 cells) for 90-min incubation at 37°C. Renal function was monitored by measurement of serum creatinine (scr) using the Crea Plus kit (Roche, Mannheim, Germany) and blood urea nitrogen (BUN) using a urea estimation kit (Abcam, Cambridge, UK) according to the manufacturers' procedure.
Histological Score for the Kidney (HSK)
The excised kidneys were fixed in phosphate-buffered 10% formalin (Sigma), sectioned, and subsequently stained with hematoxylin and eosin (Sigma). The determination of a histological score for the kidney (HSK) was performed in a blind manner by a pathologist. HSK was graded on a 4-point scale (7): 0 = normal histology, 1 = mild damage [less than one third of nuclear loss (necrosis) per tubular cross section], 2 = moderate damage [greater than one third and less than two thirds of tubular cross section showing nuclear loss (necrosis)], and 3 = severe damage [greater than two thirds of tubular cross section shows nuclear loss (necrosis)]. The total score per kidney section was calculated by the addition of all 10 scores, with a maximum possible injury score of 30.
Immunohistochemical Staining
Tissue sections were subjected to immunohistochemical staining for proliferating cell nuclear antigen (PCNA) 2 days after cell transplantation. For immunohistochemical staining, the rabbit-specific horseradish peroxidasediaminobenzidine (HRP-DAB) detection immunohistochemical kit (ab64261, Abcam) was used. Briefly, after deparaffination, 4-μm sections of kidneys were hydrated using decreasing concentrations of ethanol and incubated with a peroxidase-blocking reagent for 30 min. Sections were incubated overnight at 4°C with a rabbit polyclonal PCNA (FL-261) antibody (1:200, sc-7907, Santa Cruz Biotech.). Control experiments included omission of either the primary or secondary antibody. The reaction sections were incubated with biotinylated goat anti-rabbit IgG (H + L) (Abcam) as a secondary antibody for 10 min. Visualization of specific binding at the site of primary antibody binding was performed by an enzymatic conversion of DAB into a brown precipitate by streptavidin peroxidase. After counterstaining with hematoxylin, the sections were mounted and cleared, and subsequently the number of positive nuclei in 10 randomly chosen sections of kidney cortex and outer medulla was counted; this number was converted to the mean number of positive cells in highpower fields (HPFs, 20× magnification). Apoptotic scores were assessed using a terminal transferase-mediated dUTP nick-end labeling (TUNEL) assay with an in situ cell death detection kit (Roche). TUNEL-stained sections were screened for positive nuclei under a fluorescence microscope, and 10 random fields in the corticomedullary area were counted for every kidney at 40× magnification.
Statistical Analysis
Data are presented as the mean ± SD. Differences between the data means were analyzed using Student's t test and one-way analysis of variance followed by Dunnett's multiple comparison tests using the SPSS statistical software package (SPSS, Inc., Chicago, IL, USA), and p < 0.05 was considered to be statistically significant.
Results
ABCG2 Protects Kidney SP Cells From Hypoxia/Reoxygenation-Induced Cytotoxicity In Vitro
Whole kidney cells were isolated from C57BL/6 mice and stained with Hoechst 33342 dye (Fig. 1A). The kidney SP cell fraction represented, on average, 1.36% of the total viable cell population (1.13–1.59%) and contained very low levels of cells that expressed the markers CD45 (2.8% ± 0.7%) and c-kit (0.7% ± 0.4%), which are predominant in bone marrow SP cells. When analyzed using RT-PCR, the kidney SP cells, but not the non-SP cells, were ABCG2 (+) (Fig. 1B).

Effect of ABCG2 expression on H/R-induced cytotoxicity in kidney SP cells. (A) Fluorescence-activated cell sorting isolation of side population (SP) and non-SP cells from C57BL/6 adult mouse kidneys. (B) RT-PCR analysis of the kidney SP and non-SP cells for ATP-binding cassette, subfamily G, member 2 (ABCG2; 235bp). (C–E) Hypoxia/reoxygenation (H/R)-induced cytotoxicity in kidney SP and non-SP cells. Verapamil (50 μM/L) was used to inhibit ABCG2 expression in the SP cells. Cell viability, metabolic activity, and membrane damage were determined by propidium iodide (PI) staining (C), 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) assay (D), and lactate dehydrogenase (LDH) assay (E), respectively. Vera, verapamil. *p < 0.05 versus SP.
To determine whether the essential expression of ABCG2 in kidney SP cells was functional, the cells were treated with the selective BCRP1/ABCG2 inhibitor verapamil in hypoxic culture for 4 h, followed by 12 h of reoxygenation. At the end of the treatment period, the kidney SP and non-SP cells were examined for possible cytotoxicity induced by H/R. The standard cytotoxicity tests, including PI-binding assay for nuclear viability, MTT assay for metabolic activity, and LDH release assay for membrane damage, showed that H/R induced a significant decrease in the viability and metabolic activity and an increase in membrane damage in both SP cells and non-SP cells; however, this H/R-induced cytotoxicity was much more severe in non-SP cells than in SP cells (Fig. 1C–E). Compared with control samples, verapamil treatment alone showed no effect on the standard cytotoxicity tests, suggesting that 50 μM of verapamil is not toxic to kidney SP cells. Importantly, the SP cells pretreated with verapamil showed a significant increase in H/R-induced toxicity compared with the non-verapamil control (Fig. 1C–E), suggesting a role for ABCG2 in protecting SP cells against H/R-induced cytotoxicity.
ABCG2 Confers the Potential for Kidney SP Cells to Participate in Renal Regeneration In Vivo
To investigate the potential for kidney SP cells to act as functional kidney stem cells, the cells were injected into animals 24 h after I/R via the tail vein. At 24 h after surgery (i.e., immediately after cell transplantation), renal function was identically aggravated in animals designated to receive kidney SP cells, non-SP cells, or vehicle treatment, as assessed by BUN and Scr levels (data not shown). Infusion of SP cells significantly improved renal function in animals at 2 days after transplantation, whereas infusion of non-SP cells or L-DMEM vehicle had no effect (Fig. 2A, B). To further substantiate the result described above, histological sections were prepared for HSK, PCNA, and TUNEL staining immediately or 2 days after transplantation. Compared with control kidneys from both vehicle- or non-SP-treated mice, kidneys from SP-treated mice had significantly reduced HSK, increased numbers of PCNA-positive cells, and decreased numbers of apoptotic cells by TUNEL assay (Fig. 2C–E). These results showed that administration of SP cells was associated with increased proliferation and reduced apoptosis of surviving renal tubules.
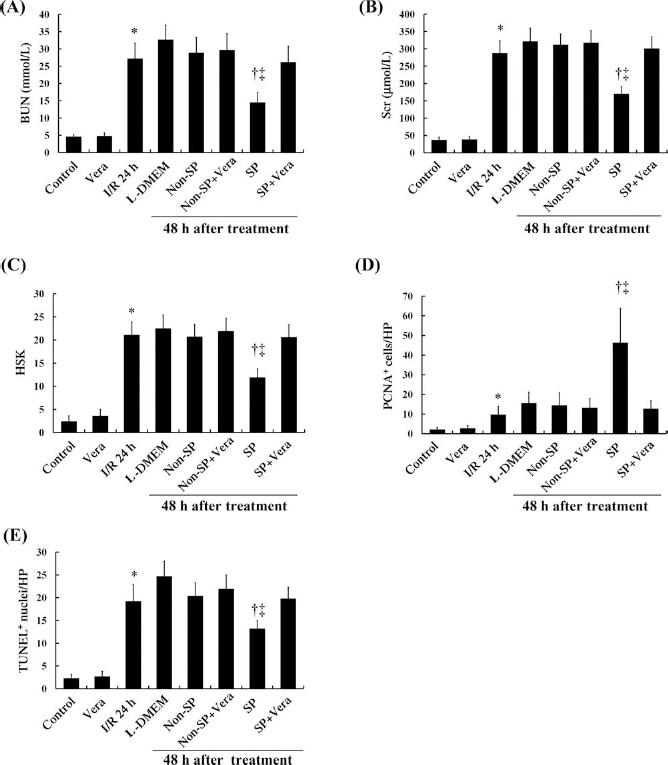
Role of ABCG2 on the therapeutic efficacy of kidney SP cells for the treatment of renal I/R injury. Blood urea nitrogen (BUN; A) and serum creatinine (Scr; B) levels were measured in bilateral renal ischemia/reperfusion (I/R) mice that received SP cells, non-SP cells or vehicle (L-DMEM), treated with or without verapamil (50 μM). (C) The histological score for the kidney (HSK) in bilateral renal I/R mice that received SP cells, non-SP cells, or vehicle-treated with or without verapamil (50 μM) was calculated. (D, E) SP-treated mice showed a significantly earlier increase in the number of proliferating cells (D), and this simultaneously reduced the number of apoptotic cells compared with non-SP-treated mice (E). Pretreatment with verapamil significantly impaired the improved therapeutic potential of SP cells. Vera, verapamil; PCNA, proliferating cell nuclear antigen; TUNEL, terminal transferase-mediated dUTP nick-end labeling. *p < 0.05 versus Control; †p < 0.05 versus L-DMEM; ‡p < 0.05 versus Non-SP.
Next, we sought to determine the effect of the BCRP1/ ABCG2 inhibitor, verapamil, on the therapeutic potential of the kidney SP cells. Compared with control, verapamil treatment alone showed no effect, as assessed by BUN, Scr, and histological examinations (Fig. 2). Pretreatment with verapamil did not alter the effects of non-SP cells, but it significantly impaired the improved therapeutic potential of SP cells (Fig. 2).
Hypoxia Activates ERK Phosphorylation in Kidney SP Cells In Vitro and In Vivo
We sought further evidence for the involvement of the MEK/ERK pathway in the kidney SP cells under H/R and I/R. Western blot analysis revealed that H/R caused a significant increase in ERK phosphorylation in the kidney SP cells, but not in the non-SP cells (Fig. 3A). Similarly, in the kidney SP cells taken from mice after renal I/R, phospho-ERK levels were observed to be increased in the ischemic (left) kidney compared with the nonischemic (right) kidney, whereas total ERK levels remained stable (Fig. 3B). There were no statistically significant differences in phospho-ERK levels in the non-SP cells between the ischemic (left) and nonischemic (right) kidneys at day 3 after surgery (Fig. 3B). These results reveal that hypoxia increases ERK phosphorylation levels in the kidney SP cells but not in the non-SP cells in vitro and in vivo.

Hypoxia activates ERK1/2 phosphorylation in vitro and in vivo. (A) Western blot analysis was performed to detect activation of extracellular-signal regulated kinase (ERK) 1/2 in the kidney SP and non-SP cells treated with or without H/R. β-Actin was used as a control. *p < 0.05 versus non-H/R. (B) Western blot analysis was performed to detect activation of ERK 1/2 in the SP and non-SP cells separated from the ischemic (I/R) and contralateral (non-I/R) kidneys of unilateral renal ischemia/reperfusion (I/R) injury mice. β-Actin was used as a control. P-ERK, phosphorylated ERK; T-ERK, total ERK. *p < 0.05 versus non-I/R.
The MEK/ERK Pathway Is Involved in ABCG2 Expression in Hypoxic Kidney SP Cells
To confirm the involvement of the MEK/ERK pathway in ABCG2 expression, we showed that MEK expression in kidney SP cells was upregulated by transfection with the recombinant sense expression vector pcDNA3.1-MEK and downregulated by pretreatment with U0126, a specific MEK inhibitor. Western blot analysis of whole-cell lysates from the kidney SP cells treated by H/R or/and with overexpression of MEK showed an upregulation of ABCG2 expression and ERK activity. Cells pretreated with U0126 (10 μM/L) showed significant inhibition of H/R- or MEK-induced upregulation of ABCG2 protein expression and ERK phosphorylation levels (Fig. 4A). Similarly, ABCG2 mRNA was upregulated in cells overexpressing MEK and inhibited by U0126 (Fig. 4B). Taken together, these data confirmed that hypoxia-induced ABCG2 expression was regulated by the MEK/ERK pathway in kidney SP cells.
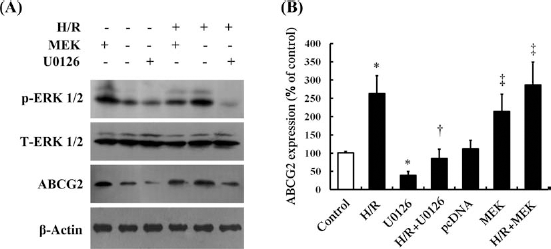
Effects of MEK on H/R-induced ABCG2 expression in kidney SP cells. (A) Western blot analysis was used to evaluate the levels of ERK 1/2 phosphorylation and ABCG2 expression. H/R and mitogen-activated protein kinase/extracellular signal-regulated kinase kinase (MEK) overexpression by plasmid cytomegalovirus promoter DNA 3.1 for MEK (pcDNA3.1-MEK) increased ABCG2 expression. U0126, a specific MEK inhibitor, inhibited H/R-induced ABCG2 expression and ERK 1/2 phosphorylation. β-Actin was used as a control. (B) RT-PCR was used for the analysis of ABCG2 mRNA levels. The kidney SP cells exposed to H/R or overexpressing MEK showed increased ABCG2 mRNA expression levels. U0126 inhibited the H/R-induced increase in ABCG2 mRNA levels. Nontreated cells were used as a control. *p < 0.05 versus Control; †p < 0.05 versus H/R; ‡p < 0.05 versus pcDNA. pcDNA: empty vector.
The MEK/ERK Pathway Prevents Kidney SP Cells From H/R-Induced Cytotoxicity
We wanted to further determine the mechanism by which the MEK/ERK pathway protects against H/R-induced cytotoxicity in kidney SP cells. As expected, U0126, a MEK-specific inhibitor, enhanced H/R-induced cytotoxicity in the kidney SP cells (Fig. 5). In contrast, the cytotoxicity induced by H/R was significantly decreased in the kidney SP cells overexpressing MEK compared with the cells transfected with an empty vector (Fig. 5). Importantly, the decreased cytotoxicity of the kidney SP cells with MEK overexpression was reversed by pretreatment with verapamil (Fig. 5), suggesting that the MEK/ ERK pathway regulates the role of ABCG2 in protecting kidney SP cells from H/R-induced cytotoxicity.
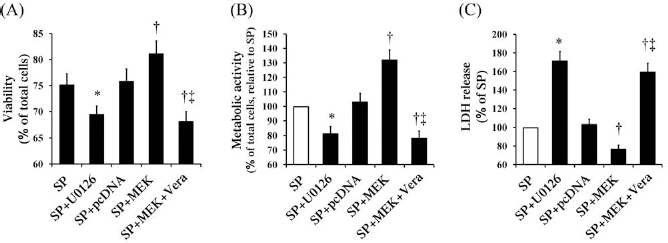
MEK is involved in preventing kidney SP cells from H/R-induced cytotoxicity. The kidney SP cells overexpressed MEK and/or were pretreated with U0126 (10 μM/L) or verapamil (50 μM/L) under the H/R condition. Cell viability, metabolic activity, and membrane damage were determined by PI staining (A), MTT assay (B), and LDH assay (C), respectively. Vera, verapamil. *p < 0.05 versus SP; †p < 0.05 versus SP + pcDNA; ‡p < 0.05 versus SP + MEK.
Discussion
Renal resident stem cells are postulated to participate in recovery after kidney damage. Several studies indicate the presence of different renal resident stem/progenitor cell populations in the renal papilla (21), the S3 segment of the nephron (17), glomeruli (6), tubules (4, 8), interstitium (3), and Bowman's capsule (23) of adult kidneys. SP cells have been found in various types of adult tissues including the kidney and are presumed to be a stem cell-rich population (4, 9). We have identified the existence of SP cells in adult C57BL/6 mouse kidneys, which is consistent with work by Hishikawa et al. (9) and Challen et al. (4). This population contained only low levels of cells that expressed CD45 and c-kit, which are markers that were predominant in the bone marrow SP cells, suggesting that the SP cells are a resident nonhematopoietic cell population.
The SP cells constitutively express ABCG2, which is a member of the ATP-binding cassette transporter family and a very important determinant of the SP phenotype (26). After renal I/R injury, the expression levels of ABCG2 were upregulated (11, 13). We have identified the expression of ABCG2 by RT-PCR in kidney SP cells, but not in non-SP cells. Furthermore, H/R-induced cytotoxicity in SP cells was lower than that in non-SP cells. Verapamil, an inhibitor of BCRP1/ABCG2, was incubated with kidney SP cells in suspension, and it significantly increased H/R-induced cytotoxicity in SP cells, suggesting a role for ABCG2 in protecting kidney SP cells against ischemic/hypoxic injury. In addition, animals infused with SP cells 24 h after I/R had significantly better renal function, lower HSK and apoptotic scores, and higher mitogenic indices than vehicle- or non-SP-treated animals. However, this improved therapeutic potential of SP cells was significantly reduced by pretreatment with verapamil. Thus, we further explored the molecular signaling pathways that might be involved in this phenomenon.
The MAPK signaling pathway transmits extracellular cues into the cell, resulting in a broad range of biological functions (5). MEK/ERK, members of the MAPK pathway, play a role in promoting cellular proliferation and inhibiting apoptosis (5). Our study demonstrated that hypoxia induced ERK 1/2 phosphorylation in kidney SP cells but not in non-SP cells in vitro and in vivo. Furthermore, ABCG2 expression in kidney SP cells was upregulated by H/R or by overexpression of MEK, which preferentially activates ERK 1/2, and was downregulated by the specific MEK inhibitor U0126. These results indicate that hypoxia-upregulated ABCG2 expression is correlated with the upstream signal transduction cascades that include ERK 1/2 activation. Thus, we examined the role of the MEK/ ERK pathway on H/R-induced cytotoxicity. MEK overexpression significantly decreased H/R-induced cytotoxicity in kidney SP cells, while inhibiting ERK with the specific inhibitor U0126 markedly increased cytotoxicity under H/R. In particular, treatment of kidney SP cells with the selective BCRP1/ABCG2 inhibitor verapamil completely reversed MEK overexpression-resistant cytotoxicity. This hypoxia-induced ERK 1/2-dependent ABCG2 expression results in resistance to H/R injury, which partially explains the mechanism by which ABCG2 contributes to cellular protection in kidney SP cells.
Taken together, our study provides, for the first time, evidence that the increased resistance to cytotoxicity induced by H/R of kidney SP cells compared to non-SP cells is associated with the expression of ABCG2, which is regulated by the MEK/ERK signaling pathway. These results indicate potential targets for promoting renal regeneration following I/R injury.
Footnotes
Acknowledgments
This work was supported by grants from the National Nature Science Foundation of China (Nos. 81370016, 81000309, 30900277, and 30901553) and Natural Science Basis Research Plan in Shaanxi Province of China (No. 2009K12-02). The authors declare no conflicts of interest.
