Abstract
Depression during pregnancy and the postpartum has been found to be associated with a number of adverse outcomes for both mother and infant. These include an increased risk of complications in pregnancy [1], increased maternal mortality [2], the risk of impaired mother-infant interactions [3], and a longer term risk of lowered socio-emotional and cognitive development in children exposed to maternal depression [4–7].
Prospective studies which examined children exposed to untreated depression in pregnancy have found a negative effect on language development in early childhood [7] and a significant effect on adolescent IQ scores [8]. Other studies have focused on the biological outcomes of children that have been exposed to maternal anxiety and found persisting differences in awakening cortisol levels at pre-adolescence compared to control [9]. Whilst the significance of this is unclear, recent work suggests the cortisol levels were normal in infants whose mothers were treated with antidepressants in pregnancy [10]. Furthermore, a study of treatment patterns of depression in pregnancy found that ceasing antidepressant medication in pregnancy significantly increased the rate of relapse from 26% in those who maintain their antidepressants to 68% who cease antidepressants in pregnancy [11]. In contrast, while not specific to depression in pregnancy, a Star∗D-child report found that adequate treatment of maternal depression reduced the risk of developing child psychopathology [12]. This report recommended ensuring maternal depression was adequately managed to improve child outcomes [12].
These overall findings support a potential argument that women who require antidepressant treatment in pregnancy should continue on these medications. This may explain the observed increase in the rate of prescription of antidepressants in pregnancy. A study across the USA found an increase in antidepressant prescription from 2% in 1994 to 7.6% in 2005 [13]. Another study within Tennessee showed that within that state there was an increase in antidepressant use in pregnancy from 5.7% in 1999 to 13.4% in 2003 [14]. In British Columbia a large population-based study of health data from 1998–2001 there was also found to be an increase from 2.3% to 5% in the rate of Selective Serotonin Reuptake Inhibitor (SSRI) use in pregnancy over a 39-month period [15].
All antidepressants cross the placenta and therefore there is foetal exposure to these medications during several sensitive periods of neurological development [16]. The quantity of medication the foetus is exposed to is lower than maternal dose so to date it remains unclear whether the level of exposure is sufficient to affect child development. However, given the number of children now exposed to antidepressants in pregnancy it is important to establish whether there are any potential effects of this exposure on child health and development to allow a balanced assessment of the risks of prescribing antidepressants in pregnancy.
There are currently only three published studies which have tested children exposed to antidepressants in pregnancy beyond 12 months of age using the Bayley Scales of Infant Development [7,17–19]. Two of these studies found no difference in total BSID score. One study found lower scores on psychomotor index of the BSID in exposed children [19].
In this study we used a prospective case control design to examine the developmental outcomes in children exposed to antidepressants in utero and compared those to children not exposed to these medications.
Materials and methods
The original study recruited 27 women who were treated with an SSRI, Serotonin and noradrenaline reuptake inhibitors (SNRI) or Noradrenergic and specific serotonergic antidepressant (NaSSA) for depression at any point across the three trimesters of pregnancy between June 2004 and July 2005. Table 1 presents the medications prescribed at the first trimester for the study group along with dosage range.
Antidepressant medications prescribed to the study group at first trimester
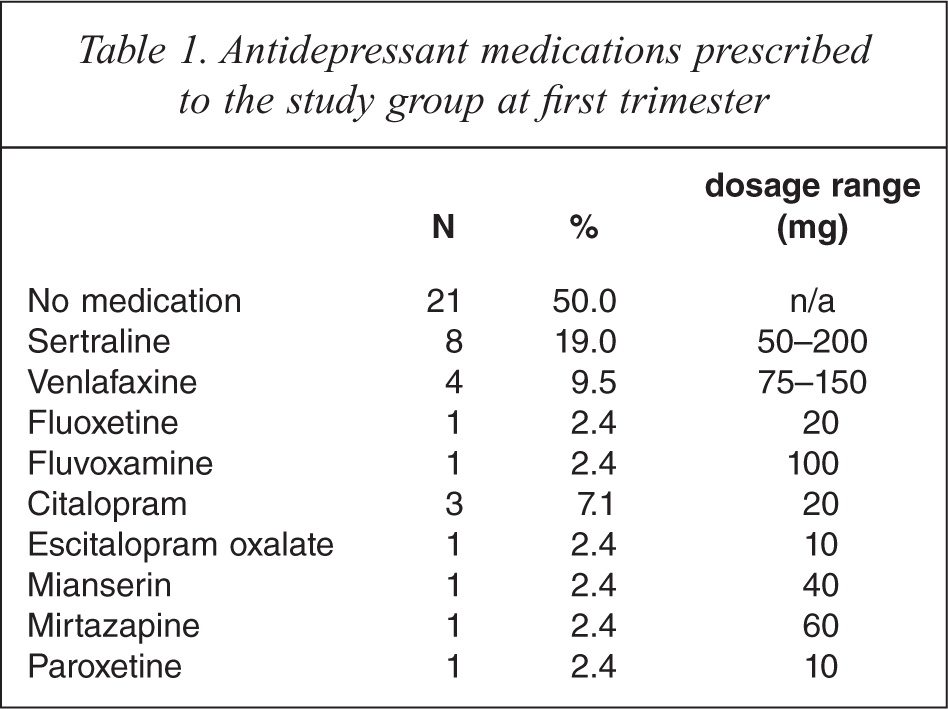
Over the three trimesters nine women increased their medication dose while three reduced their dose. Medical staff at the Mercy Hospital for Women and private psychiatrists referred pregnant women being treated with an antidepressant medication. All subjects receiving antidepressant medications who were enrolled in the study were diagnosed as having major depression according to an independent psychiatric evaluation and found to require pharmacotherapy. A matched control group of 27 pregnant women not taking antidepressants was recruited prospectively via antenatal appointments at the Mercy Hospital for Women. At this point in the study there were 22 mothers and 22 children in the exposed group and 19 mothers and 19 children in the non-exposed due to attrition over time since the original recruitment. The study has been approved by the Mercy Hospital for Women, Human Research Ethics Committee. A written informed consent statement was obtained from each participant.
Women were excluded from the study on the grounds of substance dependence, intellectual disability, serious physical illness and psychiatric illness requiring acute in-patient admission. Inclusion criteria included English proficiency.
Measures
Maternal characteristics and birth outcomes
A purpose designed questionnaire was used to assess demographics, reproductive and medical history, alcohol consumption, tobacco and recreational drug use, prescribed medications, maternal stress and coping. At each assessment (pregnancy, within one week of birth, one month postpartum and 18–35 months postpartum) details concerning the time and duration of exposure to antidepressants and the dose of the antidepressant drug were recorded. Within one week of delivery information about delivery methods, gestation at delivery, APGAR scores, birth outcomes and perinatal complications were collected. At one month postpartum the mother was questioned about the course of her pregnancy, verification of the duration, dose and type of antidepressant treatment during pregnancy and postpartum and postnatal complications. In addition breast feeding data was obtained after delivery and at one month. At 18–35 months postpartum changes in demographic data such as marital status and employment, ongoing pregnancy/delivery complications, childcare, stress/coping, cigarette, alcohol and drug use, mental health service usage, prescribed medications, antidepressant usage and dosage, and medical conditions were all assessed.
Maternal depression
The Beck Depression Inventory, second edition (BDI-II) was used to assess depression in all participants in pregnancy, after delivery, at one month postpartum and at 18–35 months [20]. The BDI-II is a 21-item self-report instrument intended to assess the existence and severity of symptoms of depression. The BDI-II has been reported to be highly reliable as a measure of depression with established construct validity and an established ability to differentiate depressed from non-depressed patients. The BDI-II has coefficient alphas of 0.92 for outpatients and 0.93 for the college students. Test–retest reliability one week apart showed a correlation of 0.93. The original BDI has been used successfully as a treatment outcome measure for pharmacotherapy and has been shown to be a sensitive measure of therapeutic change [21].
Child development
Children were assessed by trained and experienced neuropsychologists with the Bayley Scales of Infant Development, third edition (BSID-III) [22]. The neuropsychologists were blinded as to whether the children they assessed were exposed or not exposed to antidepressants in pregnancy. The BSID-III assesses development across five domains: cognitive, language, motor, socio-emotional and adaptive and is suitable for use in 1-42 months. The clinical normative values are based on 1700 children tested. The BSID-III is regarded as the reference standard in the assessment of infant and toddler development [23].
Results
Maternal and child socio-demographic characteristics
Demographic characteristics for study and control groups have been extensively reported in Galbally et al. and Lewis et al. [24,25]. The groups were compared in terms of the number of smokers and number using alcohol in pregnancy (χ2 = 1.3, p = 0.52; χ2 = 0.3, p = 0.59 respectively), age at delivery (t(52) = 0.38, p = 0.7) and years of education (t (52) = 1.21, p = 0.23) and the fact that there were no statistically significant differences suggests that the groups were well matched. Participants using illicit substances were excluded from the study. It is also of note that as a result of variation in the time of testing a significant difference was found between medicated and control groups in the time of testing at the 18 month time point. The control group was measured at a mean of 23.09 (SD 3.82) months and the medicated group at 28.53 months (SD 6.22) (t (28.98) = − 3.31; p = 0.003). However, scaled scores have been used in all analyses which have been corrected for such age differences. The scores compared were therefore standardized scores which compared the individual score to population norms, the raw score was not used in the analysis and this therefore meant the direct comparison of groups was possible despite this difference. The mean difference between the groups was approximately 5 weeks which is small with regard to a child's motor development at this age, and despite the medicated group being the older group a trend in lower motor scores was found.
Neonatal birth outcomes
The mean gestational age at delivery for exposed infants was 38.86 weeks (SD 1.32) and in the non-exposed group was 39.86 weeks (SD 1.14). The birth weight in exposed infants was 3773.6 g (SD 547.83) and in the non-exposed group 3671.19 g (SD 540.88). Neither the mean gestational age and nor birth weight in either group fell within abnormal clinical parameters. There were no statistically significant differences in APGAR scores at 1 min and 5 min between exposed and non-exposed infants.
Development in exposed compared to non-exposed children
Table 2 provides the mean differences for children's performances on the Bayley's Scales of Infant Development between study and control group children. Differences between the groups were found on the motor subscale with a moderate Cohen's effect size d = 0.47 for fine motor and d = 0.43 for gross motor. However, this failed to reach a significant level of p < 0.05 instead reaching p = 0.07 and p = 0.09 respectively. For expressive communication, at a power of 0.8 using a one-tailed significance test (95% CI) a minimum of 91 participants would be required for each group for this effect to reach statistical significance. So too, using the same parameters, significance for fine motor and gross motor would require a minimum sample size of 56 and 67 per group respectively.
Performance on Bailey's Scales of Infant Development (scales scores) and tests of difference for control and medication exposed children at 18–35 months of age
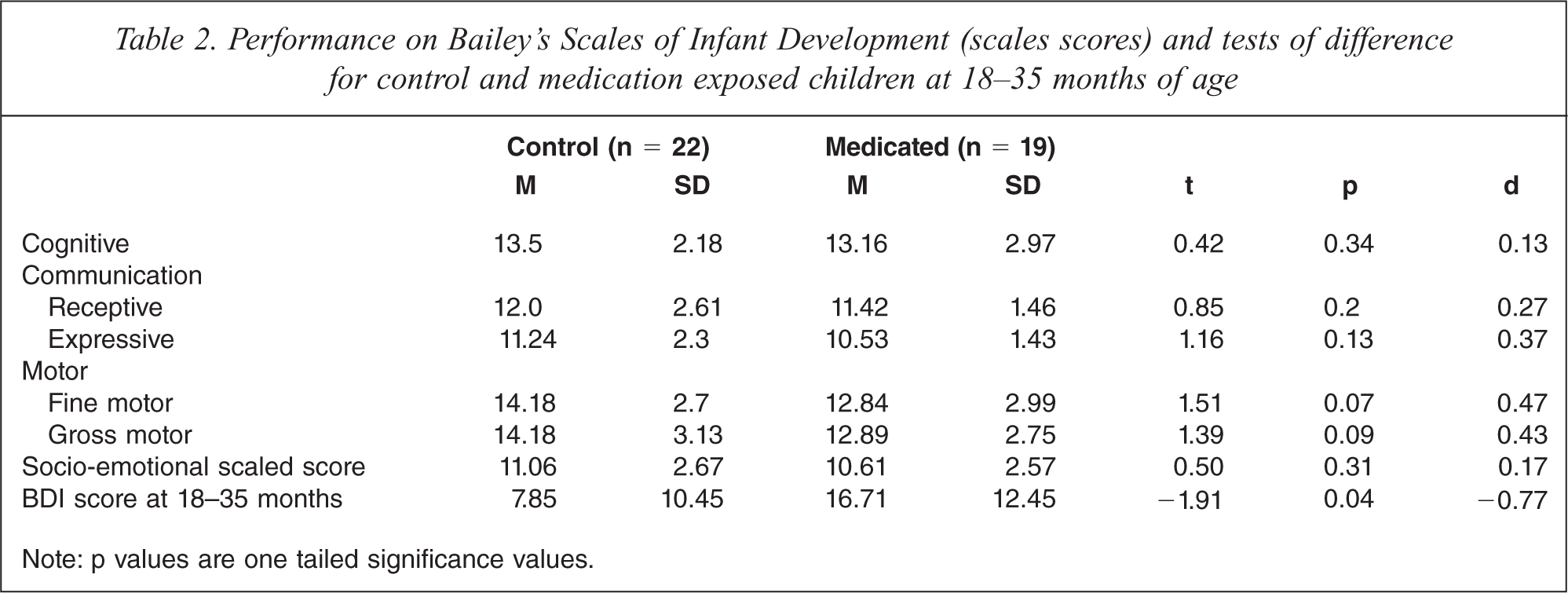
Note: p values are one tailed significance values.
Pregnancy and postpartum maternal depression and child development
Table 3 reports a correlation coefficient for depression scores at four time-points between pregnancy and 18–35 months post partum in relation to child neuro-cognitive performance at 18–35 months. The only significant association was found for higher expressive language performance and increased depression rating in pregnancy. Given the high number of analyses and the lack of any specific prediction for this particular finding, this is not likely to be of any real significance. All these findings suggest that the level of maternal depression across the pregnancy and postnatal developmental period has not had a significant impact on child neuro-cognitive development as measured at 18–35 months of age.
Pearson's correlations for Maternal BDI scores from pregnancy to 18–35 months and child neurodevelopmental outcomes on the BSID at 18–35 months

∗p < 0.05; ∗p < 0.001.
Discussion
The findings of this study support previous findings of Casper et al. of potential subtle motor development effects in children exposed in pregnancy to antidepressants [19]. While both this study and Casper et al. are limited by small numbers, Casper et al. had n = 31 for exposed children and n = 13 for control children; both have shown a moderate to large effect size between children exposed compared to those not exposed in the area of motor development. Although it should be noted that the range of scaled scores found in this study did not meet the clinical criteria for delayed development.
In trying to understand the findings from both this study and Casper et al. it is important to note that the serotonin system within the central nervous system exercises a regulatory function on muscle tone and motor output which may indicate that the developing motor system is vulnerable to SSRI exposure [19,26,27]. During in utero development the serotonin system plays an important role in brain maturation including effects on other neurotransmitter systems [28]. A study of pregnant guinea pigs, where therapeutic levels similar to humans were used, found the only change in offspring was an increased pain threshold perhaps reflecting a central nervous system effect, although no other alteration to birth outcomes or startle response was noted [29]. In addition, a study of mice indicated that those exposed to fluoxetine had altered emotional responses as adults [30]. Antidepressants given to neonatal rats has shown effects on the developing brain including an alteration in serotonergic pathways and a reduction in dendritic development in the hippocampus [31,32]. However, the relevance of these findings in animal studies to human brain development is as yet unclear.
This study has the advantage of having assessments of maternal depression in pregnancy, at delivery, 1 month and 18–35 months postpartum when examining any effect of anti-depressant exposure on child development. This is especially important given the evidence for the potential effect of maternal depression on child development [8]. It is important to note that in contrast to some findings, this study found no correlation between difficulties in language development and maternal depression.
There are only a small number of studies published which have examined neurodevelopmental outcomes in children exposed to antidepressants, and few beyond the first year of life. A recent review identified 12 publications that examined neurodevelopmental outcomes of children following exposure to antidepressants in pregnancy. Ten studies found no adverse findings, although all were quite methodologically diverse, small in numbers, and ranged in age at assessment from birth until 12 years old [33]. The two studies that did identify adverse neurodevelopmental outcomes following in utero exposure to antidepressants both found effects on motor development [19,34].
There are seven published studies which have used the BSID to assess development; however, three of these studies examined children under 9 months of age. The BSID has reduced classification accuracy in younger age groups and reduced test–retest reliability coefficients [35]. For example, at one month of age the test–retest reliability co-efficient for total score is <0.59 compared to 0.9 at 12 months [35]. In addition the BSID has only moderate predictive validity. For instance, in a study of children with congenital heart disease assessed at one year with the BSID and then at 8 years of age with the Wechsler Intelligence Scale for Children, these scores were only modestly associated with many at risk for poor outcomes at 8 years not identified by the BSID at 1 year [36]. Casper et al. had a wide range of ages assessed from 6 to 40 months, as have most published studies in this area, although it should be noted all children in this current study were over 12 months of age. The clear advantage of this current study is the older age of children tested.
This is particularly important when one considers the potential of a delayed effect of in utero exposure on development which may only become manifest as the individual reaches childhood or even adulthood. For instance, the experience of both prematurity and low birth weight shows these in utero and neonatal events can exhibit effects on neurodevelopment that are not apparent in infancy [37]. This indicates the importance of longitudinal studies which follow exposed children beyond infancy and into childhood and adolescence.
The remaining four identified papers which did test children at or beyond 12 months of age using the BSID reflect three separate studies. Two of the three studies found no difference in total BSID score, although they did not report on the subscales including motor subscale for the BSID [7,17,18]. The third study did find differences in psychomotor development index of the BSID, p = 0.02, d = 0.76, and in tremulousness in children exposed, p = 0.08, d = 0.6 [19]. Given the small numbers in most studies in this area it is important that subscale scores and effect sizes are reported in order to be able to detect possible trends which can be explored more accurately in larger studies.
This study indicates the importance of future research which can replicate with larger numbers and older age children exposed to antidepressant medication in utero. The limitations of this study were the relatively small numbers overall and small numbers for each individual antidepressant which therefore did not allow analysis for the effects of individual antidepressants. The small numbers also did not allow analysis for a dose effect. The BSID is a good early screening assessment for development; however, more comprehensive developmental assessment in older children in particular using a specific motor assessment in addition to a global developmental assessment would be more accurate to further explore the findings of this study.
Given the increasing awareness of the importance of detecting and treating maternal depression, a clearer understanding of the impact of psychopharmacological treatments in pregnancy on subsequent child development is crucial. This will allow clinicians the opportunity to give accurate recommendations regarding the relative risks and benefits of this form of treatment for depression in pregnancy and to consider the range of both psychological and pharmacological interventions available in perinatal psychiatry [38].
Footnotes
Acknowledgements
The authors would like to thank the women and children who participated in this study, Yasna Blandin de Chalain, Amanda Williamson, Karen Gough, Gillian Opie.
