Abstract
Background:
Research suggests that maintaining treatment during pregnancy for women with bipolar affective disorder reduces the risk of relapse. However, one of the key questions for women and clinicians during pregnancy is whether there are implications of exposure to mood stabilizers for longer term child development. Despite these concerns, there are few recent systematic reviews comparing the impact on child developmental outcomes for individual mood-stabilizing agents to inform clinical decisions.
Objectives:
To examine the strengths and limitations of the existing data on child developmental outcomes following prenatal exposure to mood stabilizers and to explore whether there are any differences between agents for detrimental effects on child development.
Method:
Using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines, a rigorous systematic search was carried out of four electronic databases from their respective years of inception to September 2016 to identify studies which examined the effects of mood stabilizers including sodium valproate, carbamazepine, lamotrigine, lithium and second-generation antipsychotics on child developmental outcomes.
Results:
We identified 15 studies for critical review. Of these, 10 examined antiepileptic drugs, 2 studied lithium and 3 studied second-generation antipsychotics. The most consistent finding was a dose–response relationship for valproate with higher doses associated with poorer global cognitive abilities compared to other antiepileptic drugs. The limited data available for lithium found no adverse neurodevelopmental outcomes. The limited second-generation antipsychotic studies included a report of a transient early neurodevelopmental delay which resolved by 12 months of age.
Conclusion:
This review found higher neurodevelopmental risk with valproate. While the existing data on lithium and second-generation antipsychotics are reassuring, these data are both limited and lower quality, indicating that further research is required. The information from this review is relevant for patients and clinicians to influence choice of mood-stabilizing agent in childbearing women. This must be balanced against the known risks associated with untreated bipolar affective disorder.
Introduction
The complexity for clinicians of treating bipolar affective disorder (BPAD) in women in pregnancy is the need for a careful evaluation of the risks and benefits of treatment not only for the mother but also the foetus. Research indicates that if women cease medication, the risk of relapse in pregnancy is high with a longer recovery period (Bergink et al., 2015; Viguera et al., 2000; Wesseloo et al., 2016). Women with severe mental illness including BPAD also have higher risk pregnancies, associated with increased obstetric and neonatal complications (Judd et al., 2014; Nguyen et al., 2012). This may be related to multiple risk factors in addition to mental illness including increased exposure to psychotropic medications, illicit substances, alcohol and tobacco during pregnancy (Brameld et al., 2017; Judd et al., 2014; Nguyen et al., 2012). Additional concerns for women taking medication in pregnancy include the potential for long-term effects on their offspring which must be weighed against the risk of relapse following medication cessation.
Medications used for their mood-stabilizing properties include antiepileptic drugs (AEDs) comprising sodium valproate, carbamazepine and lamotrigine. Lithium is the most commonly prescribed mood stabilizer in BPAD, and more recently, second-generation antipsychotics (SGAs) have been used increasingly as mood stabilizers (Khan et al., 2016). In addition to the treatment of BPAD, many of the mood-stabilizing medications can also be part of treatment for a range of mental illnesses including refractory major depression and psychotic disorders, making the understanding of risks and benefits in pregnancy relevant beyond management of BPAD.
Relapse rates suggest a better course of BPAD during pregnancy if medication is continued. Viguera et al. (2000) found that recurrence rates of BPAD during the first 40 weeks after lithium discontinuation were similar for pregnant (52%) and non-pregnant women (58%). Recurrence rates were much lower for both groups in the year prior to discontinuing treatment (21%). Among those who remained stable over the first 40 weeks after lithium discontinuation, postpartum recurrences were 2.9 times more frequent compared to non-pregnant women (Viguera et al., 2000). Similarly, a recent meta-analysis found that postpartum relapse rates of BPAD were three times higher among those who were medication-free during pregnancy, compared to those using prophylactic medication (Wesseloo et al., 2016). Untreated BPAD during pregnancy may increase the risk of adverse pregnancy and birth outcomes, as well as the neurodevelopment of children (Gentile, 2012). The term neurodevelopment refers to functional outputs of the brain, including motor function, intelligence, socio-emotional functioning and other cognitive skills (Bromley, 2016).
The benefits of treated mental illness must be weighed against the potential risks to the child of medication use in pregnancy. This is a key concern for women in the reproductive age group, especially since 50% of pregnancies are unplanned and the teratogenic effects of mood stabilizers including valproate have been well established (Khan et al., 2016; Meador and Loring, 2016). In addition to anatomical teratogenesis, recent literature including a Cochrane review from 2014 has suggested an increased dose-dependent risk for neurodevelopmental teratogenesis for valproate (Bromley et al., 2014; Meador and Loring, 2016). Specifically, the Cochrane review found a significantly decreased intellectual quotient (IQ) in children exposed to valproate in utero, compared to other AEDs (Bromley et al., 2014). While the teratogenic effects of lithium are known, little is known about the long-term effects of lithium on child neurodevelopment, and despite the expanding use of antipsychotics beyond psychosis to include mood disorders, data about the neurodevelopmental impact of foetal antipsychotic exposure are also limited.
While child neurodevelopment following in utero exposure to AEDs has been examined extensively in the aforementioned Cochrane review, this was in the context of the treatment of women with epilepsy (Bromley et al., 2014). Despite the increasing use of mood stabilizers for treatment of BPAD, there has not been a systematic review examining child developmental outcomes in women using mood stabilizers for treatment of mental illness since 2010 (Galbally et al., 2010a). The same year, a set of guidelines to inform the provision of antenatal care to women taking psychotropic medication was developed (Galbally et al., 2010b). However, there is limited longer term data to inform women and clinicians about the potential impact on childhood development. To our knowledge, this is the first systematic review focusing specifically on mood-stabilizer exposure in utero and longer term child neurodevelopmental outcomes. It is clinically relevant to explore whether the longer term developmental impact of prenatal exposure varies with each individual agent as this may inform both clinical and maternal choice of mood-stabilizing agent in the perinatal period including pre-conception planning.
Method
This review will address the following objectives:
To examine the strengths and limitations of the current research on child developmental outcomes following prenatal exposure to mood stabilizers.
To explore whether there are any differences between agents for detrimental effects on childhood development following exposure in utero.
This review followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (Moher et al., 2009). Strategies to identify eligible studies included a systematic search of four electronic databases including MEDLINE, PubMed, EMBASE and PsycINFO, a search of the grey literature including a hand search of the references of all included articles. The aforementioned Cochrane review for AEDs covered the time period from 1946 to May 2014; therefore, this review will examine child developmental outcomes for AEDs from June 2014 to September 2016. The research for lithium and SGAs will be covered from the receptive years of inception of each electronic database to September 2016. The following search terms were used: pregnancy, mood stabilizer, anticonvulsants, antimanic agents, antiepileptic drugs (AEDs), lithium carbonate, antipsychotic agents, atypical antipsychotic, psychotropic drugs, neuroleptic drugs, prenatal exposure delayed effects, offspring, child development, child development disorders, neurodevelopment, neurodevelopmental disorders, developmental disabilities, delayed development, cognitive impairment, intellectual development disorder, neurobehavioural manifestations, disorders of higher cerebral function, neurocognitive disorders, neuropsychology and intelligence. Boolean operators were used with care. The search was conducted twice with assistance from library staff to ensure inclusion of all relevant articles. All citations were initially reviewed by title, then by abstract and screened according to the following inclusion and exclusion criteria.
Inclusion criteria
Studies met inclusion criteria if they were original research; English language; prospective and retrospective data; human studies; any exposure during pregnancy to valproate, carbamazepine, lamotrigine, lithium or SGAs; and measured child neurodevelopmental outcomes using standardized neuropsychological methods, clinical examination or academic assessments at 6 months of age and older.
Exclusion criteria
Exclusion criteria comprised the following: review articles, theoretical discussions, case studies and studies in non-pregnant women. Data regarding use of AEDs not used for mood stabilization such as phenytoin were excluded. Exposure to newer AEDs including levetiracetam and topiramate, which are not indicated for treatment of mood disorders in Australia as per Royal Australian New Zealand College of Psychiatrists (RANZCP) guidelines, were excluded (Malhi et al., 2015).
There were a total of 1624 articles identified from all databases after removal of duplicates. In all, 1163 articles were excluded based on title and then a further 437 articles were excluded based on review of abstract. When there was uncertainty regarding inclusion of an article, this was screened by two reviewers. A total of 24 full-text articles were selected. Nine articles did not meet inclusion criteria and were removed, leaving a total of 15 articles for critical review (see Figure 1). The best available National Health and Medical Research Council (NHMRC) level of evidence available was III-2 (controlled cohort studies). The studies were rated according to the Newcastle–Ottawa Quality Assessment Scale (NOS), included in Tables 1–3 (Wells et al., 2014).
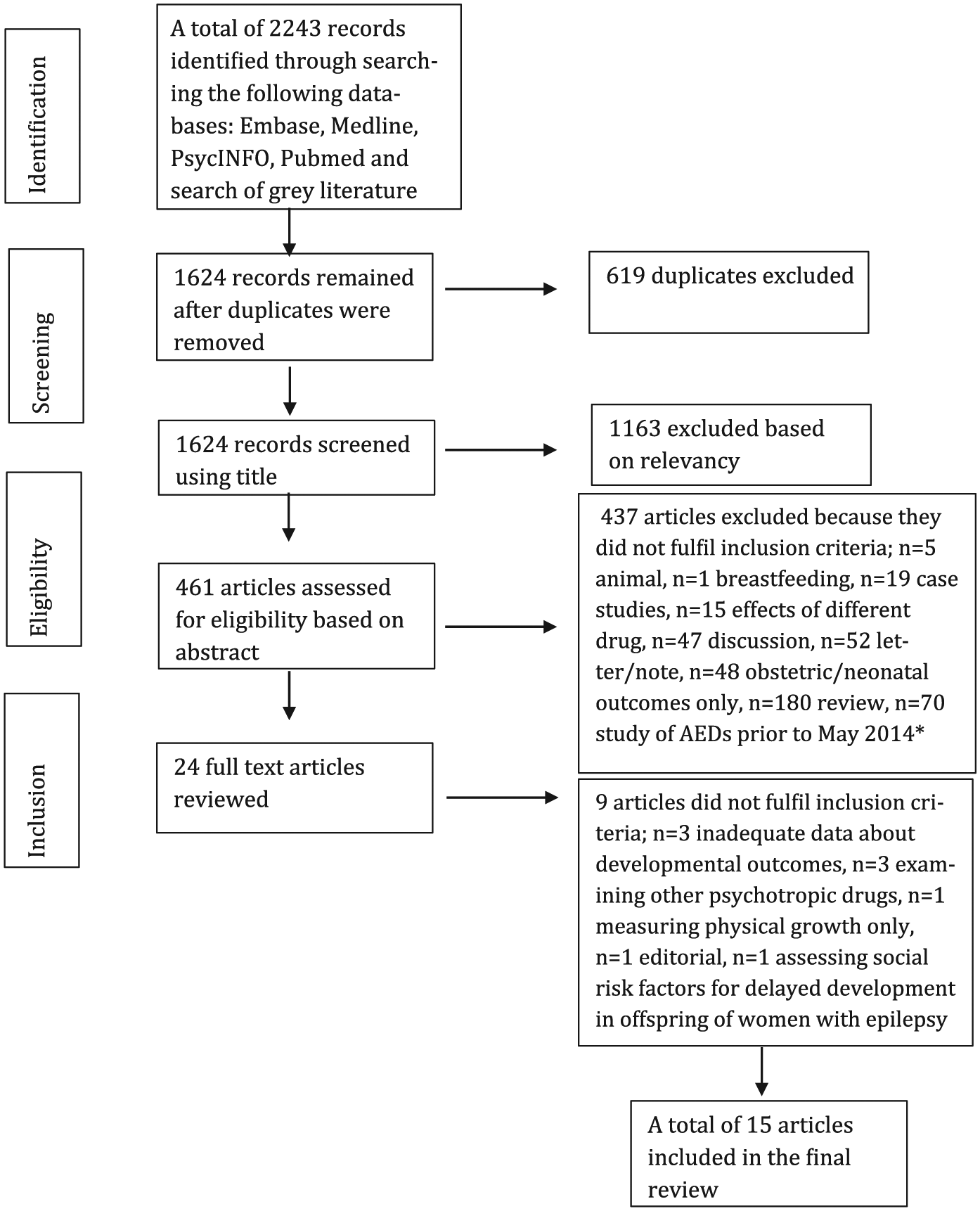
Flowchart of systematic literature review.
Summary of studies exploring AED exposure in pregnancy and child neurodevelopmental outcomes.
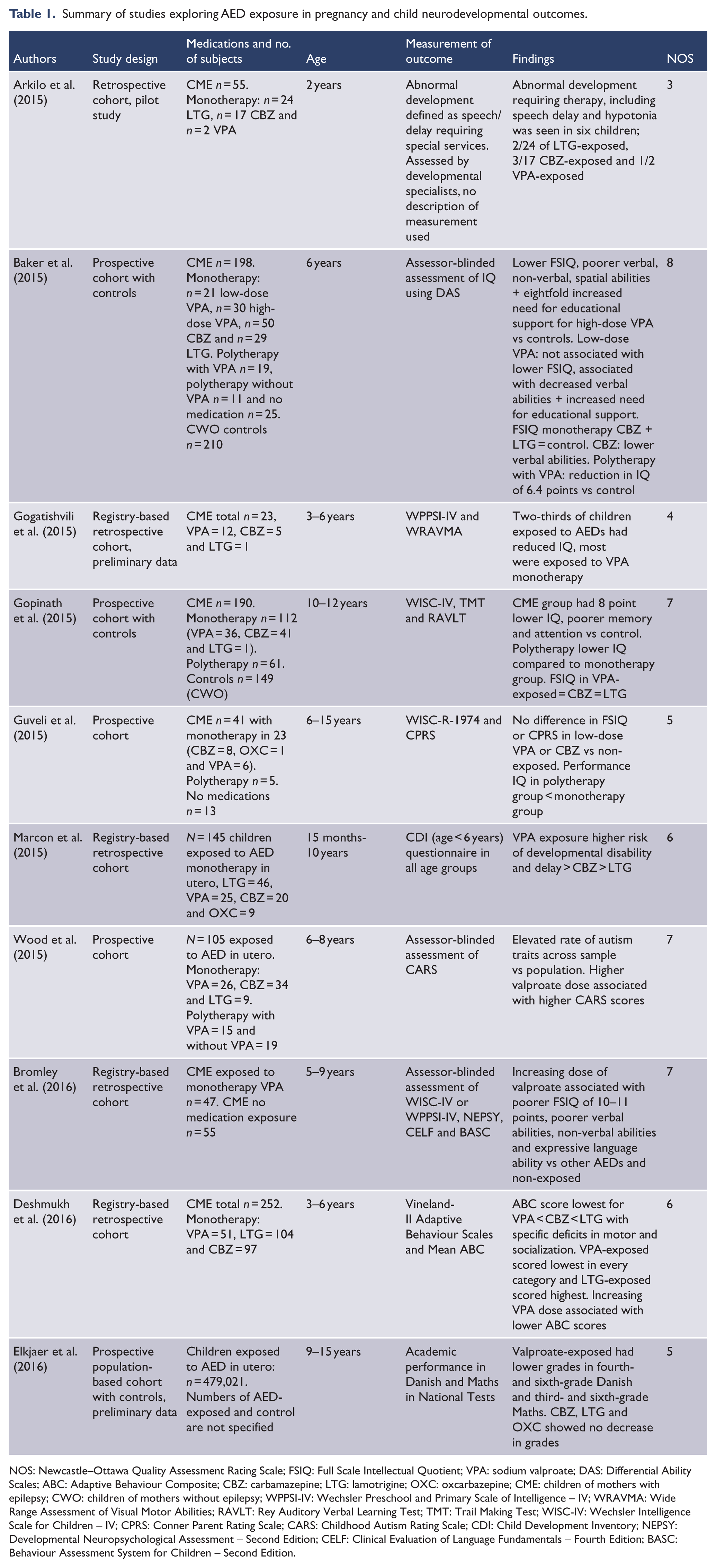
NOS: Newcastle–Ottawa Quality Assessment Rating Scale; FSIQ: Full Scale Intellectual Quotient; VPA: sodium valproate; DAS: Differential Ability Scales; ABC: Adaptive Behaviour Composite; CBZ: carbamazepine; LTG: lamotrigine; OXC: oxcarbazepine; CME: children of mothers with epilepsy; CWO: children of mothers without epilepsy; WPPSI-IV: Wechsler Preschool and Primary Scale of Intelligence – IV; WRAVMA: Wide Range Assessment of Visual Motor Abilities; RAVLT: Rey Auditory Verbal Learning Test; TMT: Trail Making Test; WISC-IV: Wechsler Intelligence Scale for Children – IV; CPRS: Conner Parent Rating Scale; CARS: Childhood Autism Rating Scale; CDI: Child Development Inventory; NEPSY: Developmental Neuropsychological Assessment – Second Edition; CELF: Clinical Evaluation of Language Fundamentals – Fourth Edition; BASC: Behaviour Assessment System for Children – Second Edition.
Summary of studies exploring lithium exposure in pregnancy and child neurodevelopmental outcomes.
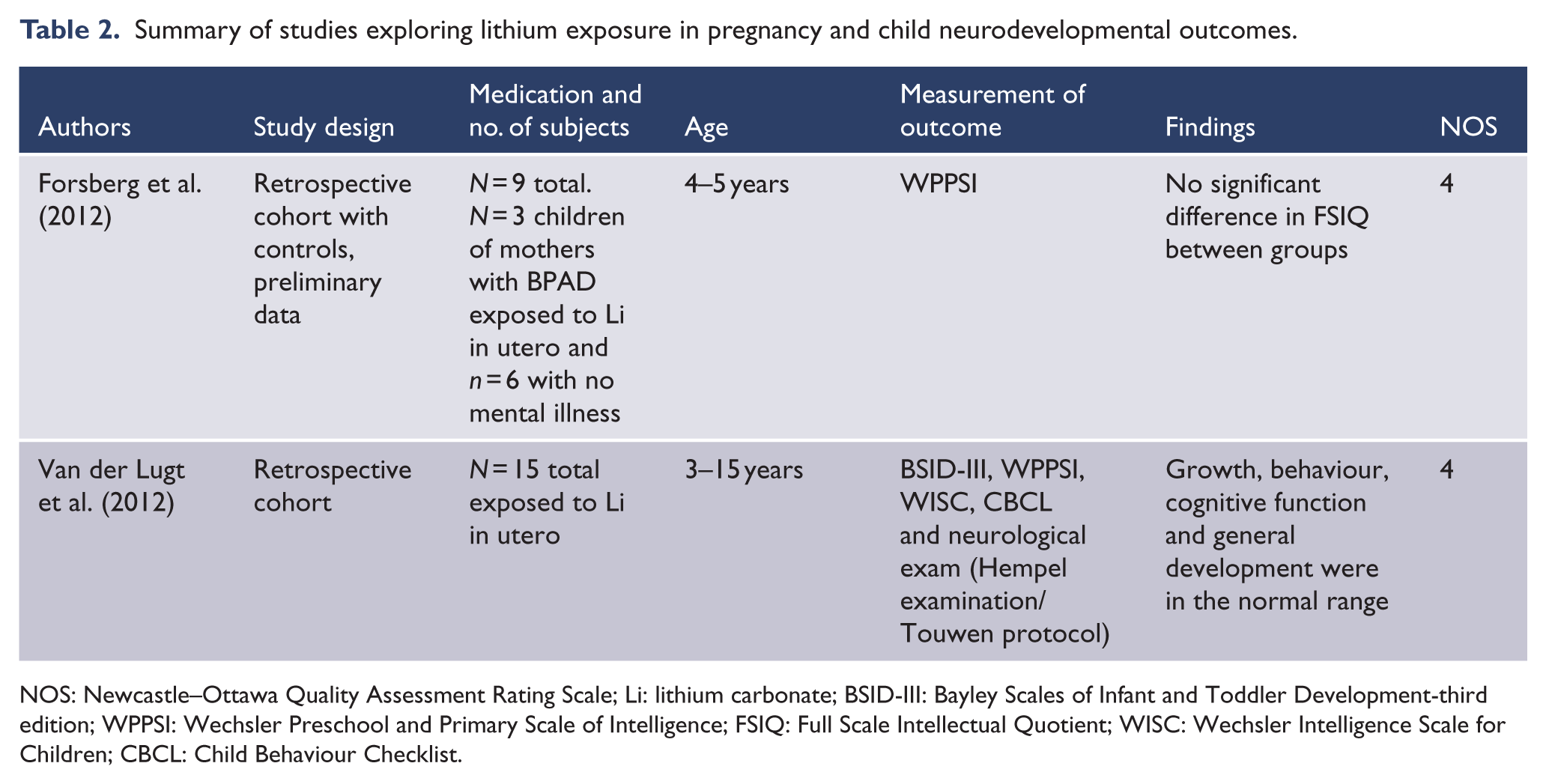
NOS: Newcastle–Ottawa Quality Assessment Rating Scale; Li: lithium carbonate; BSID-III: Bayley Scales of Infant and Toddler Development-third edition; WPPSI: Wechsler Preschool and Primary Scale of Intelligence; FSIQ: Full Scale Intellectual Quotient; WISC: Wechsler Intelligence Scale for Children; CBCL: Child Behaviour Checklist.
Summary of studies exploring SGA exposure in pregnancy and child neurodevelopmental outcomes.
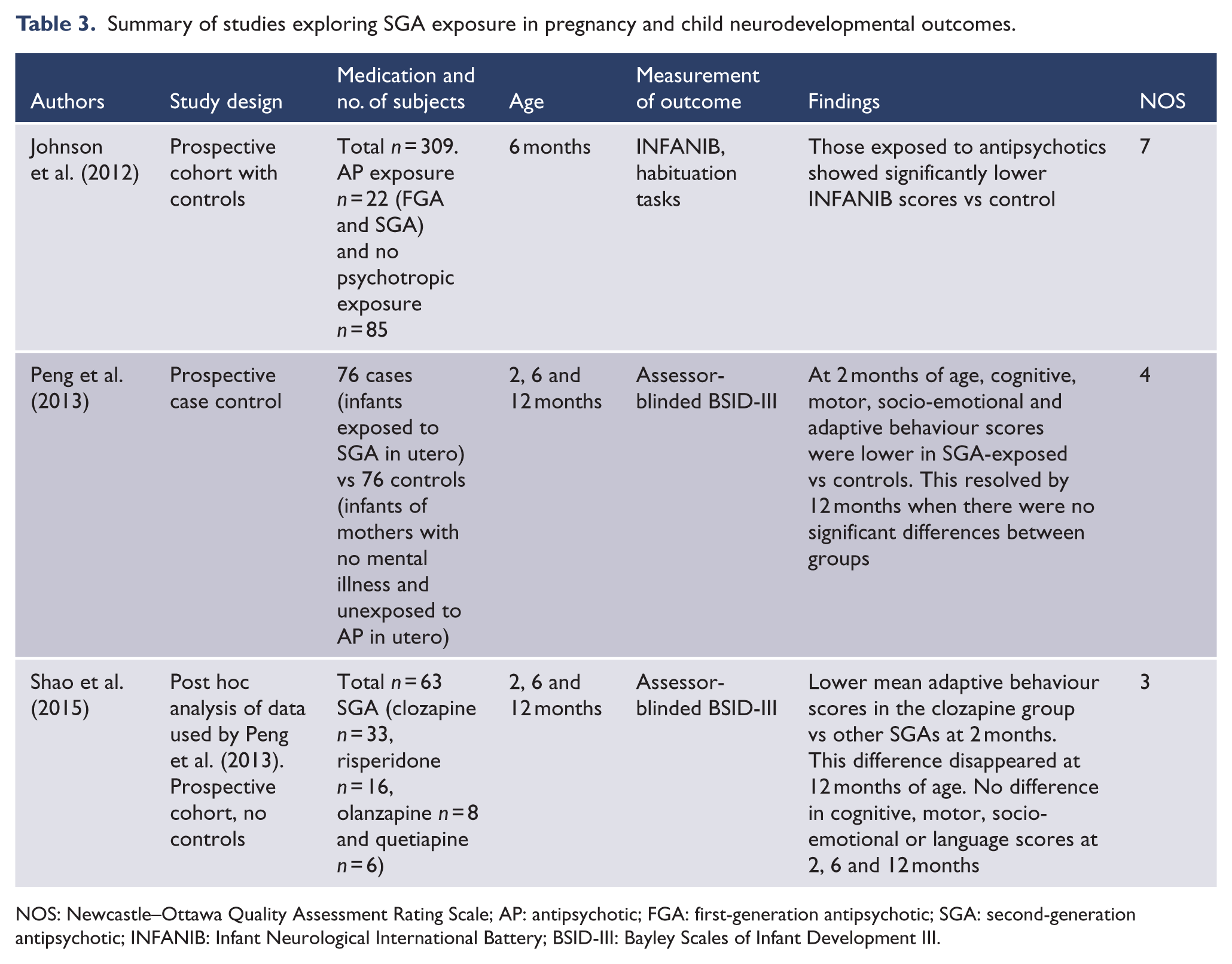
NOS: Newcastle–Ottawa Quality Assessment Rating Scale; AP: antipsychotic; FGA: first-generation antipsychotic; SGA: second-generation antipsychotic; INFANIB: Infant Neurological International Battery; BSID-III: Bayley Scales of Infant Development III.
Results
Of the 15 studies identified, 10 examined AEDs. There were very few lithium and SGA studies, with two and three in each group, respectively. Overall, these were of lower quality than the AED studies according to NOS scores. Two other relevant studies were identified including the Australian National Register of Antipsychotic Medication in Pregnancy (NRAMP) study and the follow-up study of children from the Lithium Baby Registry (Kulkarni et al., 2014; Schou, 1976). Neither study found any adverse neurodevelopmental outcomes. However, they were excluded from this review as the outcome measure was based solely on maternal self-report on child outcomes.
AEDs
Studies included three prospective cohorts with controls, two prospective cohorts without controls, one retrospective cohort and four registry-based retrospective cohorts. These are described in Table 1. All studies were in women with epilepsy and included valproate. Nine studies included carbamazepine and eight included lamotrigine. Four studies examined dose–response relationships (Baker et al., 2015; Bromley et al., 2016; Deshmukh et al., 2016; Wood et al., 2015). Four studies examined AED monotherapy alone (Arkilo et al., 2015; Bromley et al., 2016; Deshmukh et al., 2016; Marcon et al., 2015), while four studies compared monotherapy and polytherapy (Baker et al., 2015; Gopinath et al., 2015; Guveli et al., 2015; Wood et al., 2015). Two studies did not specify the use of monotherapy or polytherapy (Elkjaer et al., 2016; Gogatishvili et al., 2015).
The most consistent finding across all studies was a dose–response relationship with valproate, with doses above 800–1000 mg associated with poorer neurodevelopmental outcomes. Carbamazepine was not found to be associated with lower IQ in any of the studies. It was found to be associated with lower verbal abilities in one study (Baker et al., 2015). No adverse effects on IQ or specific cognitive skills were found for lamotrigine compared to other AEDS or controls. AED polytherapy was found to carry an increased risk, but these medication regimens included AEDs not used in the treatment of mental illness (Baker et al., 2015; Gopinath et al., 2015; Guveli et al., 2015).
The measurement of Full Scale Intellectual Quotient (FSIQ) was carried out in five studies, using Differential Ability Scales, Wechsler Preschool and Primary Scale of Intelligence – IV (WPPSI-IV) or Wechsler Intelligence Scale for Children – IV (WISC-IV). Other studies used a range of methods to examine adaptive behaviour, socialization, motor skills, neurological function, autism traits and academic grades which are listed in Table 1. Developmental specialists performed clinical assessments in one study, with no specification of the measurement used (Arkilo et al., 2015).
Baker et al. (2015) reported that children exposed prenatally to >800 mg/day of valproate had an IQ score that was 9.7 points lower than unexposed children derived from the general population. Additionally, the risk of IQ < 85 was eight times higher in children born to women treated with high doses of valproate (>800 mg/day) than in those born to control women (adjusted relative risk (RR) = 8.6, 95% confidence interval [CI] = [3.1, 18.8],
Additional findings of decreased verbal, non-verbal and expressive language abilities were found with increasing doses of valproate (Bromley et al., 2016). Deshmukh et al. (2016) found specific deficits in motor skills and socialization for valproate compared to lamotrigine and carbamazepine. Furthermore, high valproate dose in utero was a significant predictor of increased rates of autistic traits after controlling for several confounders including polytherapy and maternal IQ in a recent Australian study (Wood et al., 2015).
Lithium carbonate
Two studies were identified, including a retrospective cohort with controls and a retrospective cohort. These studies are summarized in Table 2 (Forsberg et al., 2012; Van der Lugt et al., 2012). Both studies were conducted in 2012 and showed no adverse effects on cognitive development at ages 5 and 15 after in utero exposure to lithium. Both studies were limited by small size and retrospective design.
The first was a preliminary study by Forsberg et al. (2012) that examined the FSIQ of nine participants at age 4–5 years. Three of these children had been exposed to lithium in utero and six were unexposed. No significant difference in FSIQ between groups was found.
The second was a study by Van der Lugt et al. (2012) whereby neurocognitive and behavioural tests were conducted in 15 children aged between 3 and 15 years exposed to lithium in utero and found that their neurodevelopmental outcomes were within normal limits. Given the wide age group, age-specific cognitive tests were conducted by a psychologist including Bayley Scales of Infant and Toddler Development – Third Edition (BSID-III) for children aged 16–30 months and WPPSI/WISC for older children. Neurological development was assessed on physical examination. The study lacked a control group, and four of the mothers had been taking other psychotropic medications in pregnancy.
SGAs
Three studies were identified, including a prospective cohort study with controls, a prospective case control and a prospective cohort which was a post hoc analysis of the case-control study. These are summarized in Table 3 (Johnson et al., 2012; Peng et al., 2013; Shao et al., 2015). Overall, the studies found a transient neurodevelopmental delay which resolved by age 12 months.
Johnson et al. (2012) compared 22 mother–infant pairs exposed to first-generation antipsychotics (FGAs) and SGAs to a control group of 85 infants with no psychotropic exposure. Women in both groups took other medications, and there was a combination of FGA and SGA use in the exposed group. Antipsychotic exposure based on the mother’s report included FGA-only in 9 participants, SGA-only in 12 (aripiprazole, olanzapine, quetiapine, risperidone and ziprasidone) with 3 of these exposed to more than one SGA and both FGA and SGA in 1 participant. The outcome was based on scores on the Infant Neurological International Battery (INFANIB) at 6 months of age.
Infants exposed to antipsychotics showed significantly lower INFANIB scores compared to unexposed infants, after controlling for significant co-variants using multiple regression analysis. Maternal depression during pregnancy was associated with less efficient habituation, suggesting a potential impact of both prenatal exposure and maternal psychiatric illness. Moderately lower neuromotor scores for SGA-exposed versus FGA-exposed infants were not statistically significant, and small sample size prevented isolated analysis of the impact of antipsychotic monotherapy.
A Chinese case-control study compared 76 infants of mothers with schizophrenia exposed to SGA in utero to 76 control infants whose mothers had no history of mental illness or antipsychotic use in utero (Peng et al., 2013). For the mothers in the exposed group, treatment groups included clozapine (
A post hoc analysis using data from the above study examined 33 infants exposed to clozapine in utero to 30 infants exposed to other SGAs (risperidone, olanzapine and quetiapine; Shao et al., 2015). There was no control group. The study found that infants exposed to clozapine had lower mean adaptive behaviour scores at 2 and 6 months compared to infants exposed to other SGAs. No statistically significant differences were found between the groups in any cognitive domain at 12 months.
Discussion
This systematic review appraised the literature available in order to assist clinical decisions about choice of mood-stabilizing agent during pregnancy. Clinical decisions to continue or commence mood-stabilizer medication in pregnancy require weighing a range of factors that are crucial to the effective management of BPAD. These include the risk of relapse should psychopharmacological treatment be ceased and the potential for poorer response if treatment is reinstated such as can be the case with lithium. Additional considerations include if relapse occurs in pregnancy following medication cessation the potential then for the requirement of multiple agents to achieve remission and finally, the implications of length to recovery on maternal and neonatal outcomes if relapse occurs. BPAD can be a chronic mental disorder with remission achieved through maintenance mood-stabilizer treatment. The onset of BPAD is likely to occur during childbearing years, and hence, there is a need for careful consideration and planning around pregnancy as a routine component of clinical care.
The findings of this review support the Cochrane review findings of increased neurodevelopmental risk for valproate and highlight the paucity of literature for other agents. While the results for the lithium and SGA groups are reassuring, this needs to be placed in context of very limited number of studies and methodological limitations with the available studies.
When examining the findings across the classes of mood-stabilizer agents, the strengths of the AED studies were clear with a larger number of studies and participants, and moderate- to high-quality methodological design. This included half of the studies having a prospective design, decreasing the potential for recall bias surrounding teratogen use and confounding variables. The superior methodology and size of the studies allowed for clinically relevant issues such as monotherapy versus polytherapy groups and dose–response relationships to be examined.
These studies also examined a wide range of developmental outcomes extending beyond IQ and assessed socialization, behavioural and motor skills. Limitations of the AED studies include the use of over 15 different types of assessments and measurement of childhood development over a wide age range, decreasing the reliability of the results. In addition, correction for confounding variables was a limitation of the data with, for instance, only four of the AED studies controlling for a key confounder, maternal IQ (Baker et al., 2015; Bromley et al., 2016; Gopinath et al., 2015; Wood et al., 2015).
The most consistent finding across all studies despite varied methodologies was a dose–response relationship with valproate, with high doses associated with poorer global cognitive abilities (Baker et al., 2015; Bromley et al., 2016; Deshmukh et al., 2016). The data suggest that valproate lowers the IQ by 8–11 points, with higher risk at doses >800–1000 mg. These results have significant implications, as IQ is a major predictor of important outcomes across the lifespan, including socioeconomic status and health (Eriksen et al., 2013).
Increasing doses of valproate also appear to be linked with decreased verbal and non-verbal abilities, as well as a higher rate of autism spectrum features (Baker et al., 2015; Bromley et al., 2016; Wood et al., 2015). These results support existing research, which has consistently shown that exposure to valproate presents the highest teratogenic risk of all the AEDs for both malformations and neurodevelopmental outcomes, leading to guidelines enforcing the avoidance of valproate treatment in women of childbearing age (Bromley et al., 2014; Tomson et al., 2015). The association between carbamazepine and lower verbal abilities found in one study requires further investigation (Baker et al., 2015). No adverse outcomes were found for lamotrigine in this review.
Finally, the characteristics of the population in the AED studies require consideration, given that the majority of studies examine women using AEDs for epilepsy. This poses the question of whether these results are generalizable to women prescribed AEDs for treatment of mental illness, such as their use as mood stabilizers. Supporting the validity of drawing on this research is evidence that the neurodevelopment of children born to women with untreated epilepsy has been documented to be comparable to control children (Baker et al., 2015). Additionally, animal data have shown abnormal neurodevelopment including autistic features in the offspring of mothers without epilepsy who were exposed to AEDs during pregnancy (Bambini-Junior et al., 2011). This suggests that information regarding AED use in pregnancy remains relevant to all populations taking AEDs in pregnancy, regardless of the indication, and emphasizes the need for studies in women using these medications as mood stabilizers.
While the two studies examining lithium and child developmental outcomes included a range of assessments including FSIQ, neurological and behavioural development in one study, overall, the quality ratings of the lithium studies were low. This was due to small study size, retrospective design and neither of the studies adjusted for confounding variables or used blinding of child assessments.
Strong aspects of the SGA studies include prospective study design and use of blinding in two of the three identified studies. Weaknesses included lack of adjustment for confounding variables, with only one study attempting to correct for confounders, the young age of infants at follow up and small study size.
The limited data examining neurodevelopmental outcomes for lithium are encouraging in that none of the studies found any neurodevelopmental deficits for lithium-exposed children. The SGA studies found an early transient neurodevelopmental delay at 2 and 6 months, with lower mean adaptive behaviour scores for those exposed to clozapine compared to other SGAs, although reassuringly by 12 months of age there was no statistically significant difference between groups. However, the assessment of 12-month-old infants may not reflect long-term outcomes.
The comparison of findings between the classes of mood stabilizers should also be viewed in the context of quality of available data such as the number of studies and participants, quality of methodological design and age to follow up with considerable variation between classes identified in this review. The clearer findings for the AEDs arise out of a context of well-designed, replicated and longer follow-up studies than is currently available for lithium and SGAs.
This review identified a number of considerations for design of studies that examine pregnancy exposures and child developmental outcomes. The first is the high rate of change in developmental abilities across childhood and the predictive validity of measurements used to assess neurodevelopmental outcome requiring careful attention in terms of both measures used and age to follow up (Bromley, 2016). Abilities of children under school age and particularly in those younger than 3 years are commonly assessed using the Bayley Scales of Infant and Toddler Development, which requires the infant to demonstrate certain responses following the presentation of cues (Bayley, 2005). For school-aged children, cognition is commonly measured by the Wechsler series assessments and these are the most widely used measure of cognitive function across the lifespan (Wechsler, 1949). While the FSIQ generated by the Wechsler measures is useful to summarize cognitive function in a research setting, it should be considered a global summary of cognitive function. Consideration of index and subtest scores on the Wechsler measures, as well as additional specialized neuropsychological tests, is useful to examine specific cognitive domains, such as executive function, when understanding neurodevelopmental outcomes for children (Galbally et al., 2012, 2015; McCorry and Bromley, 2015). The studies in this review used varied outcome measures, ranging from BSID and Wechsler measures to academic grades, making direct comparisons between studies difficult. Early assessment in infancy should not be interpreted as the final outcome as neurodevelopment is a dynamic process which requires long-term follow up, preferably into adulthood (Bromley, 2016).
Other methodological issues identified include power calculations, blinding, confounding variables and exposure measurements. Generally, studies investigating neurodevelopmental outcomes report power calculations requiring between 40 and 50 participants per group to detect moderate to large effect sizes (Baker et al., 2015; McCorry and Bromley, 2015). Therefore, half of the studies were underpowered, including four of the AED studies, both of the lithium studies and one of the SGA studies. More reliable data are produced when the investigator examining the child’s performance is blinded to whether or not the child has been exposed, which aims to minimize detection bias (McCorry and Bromley, 2015). Blinding was under-utilized, with three of the AED studies, none of the lithium studies and two of the SGA studies using blinded assessments.
Confounding factors including parental education and maternal IQ are well-known predictors of child IQ (Eriksen et al., 2013). In addition, socioeconomic status, breastfeeding, adequate nutrition, concomitant medication use, illicit substance use and gestational age have been shown to be significantly associated with child developmental outcome (Bromley et al., 2016; McCorry and Bromley, 2015). Maternal mental illness itself can negatively impact child development (Johnson et al., 2012). The majority of the studies did not undertake adequate statistical control for confounding factors. In particular, use of multiple medications in exposure groups contributed to confounding factors in the lithium and SGA studies and meant that direct comparison between SGA monotherapy groups was not possible.
The most reliable measurement of exposure involves the collection of serum drug levels to corroborate medication exposure. The next most reliable measurement is prospective medical records. Exposure based solely on retrospective medical records or patient questionnaires is likely to introduce bias. In this review, exposure measurements were largely based on registry records, retrospective medical records or patient questionnaires, making it difficult to establish a causal relationship between mood-stabilizer exposure and child developmental outcomes.
Selection bias is apparent in all studies since women who have concerns about their children are more likely to participate and the use of registries represents a small proportion of women in the community. Cultural differences and external validity require consideration especially for the SGA group, since two out of three studies examined Chinese women with schizophrenia. With regard to pharmaceutical industry sponsorship, there were only three studies that were directly supported (Baker et al., 2015; Deshmukh et al., 2016; Wood et al., 2015) and four studies had authors with a declared conflict of interest, (Baker et al., 2015; Bromley et al., 2016; Johnson et al., 2012; Wood et al., 2015) representing one-third of the total studies. Hence, methodological design of future studies should consider issues of measurement of exposure, selection and recruitment of sample; measurement of key potential confounding variables, ensuring adequate power; and finally, follow up to an age where robust measures of development can be undertaken.
The limitations of this review include assessment of studies in English language only and reliance on the Cochrane review for articles prior to May 2014. Additionally, while the reference lists of all included articles were examined, there was no extensive search of the grey literature beyond that. These limitations aside, the clinical implications of this review are fourfold. First, each mood-stabilizing agent has a different risk–benefit profile for impact on cognitive development. Second, high-dose valproate has been consistently associated with poorer neurodevelopmental outcomes with available data suggesting that lamotrigine may be a preferred treatment to valproate or carbamazepine due to lower risk of adverse outcomes for the child, but further exploration is required to confirm this. Third, while the existing data are reassuring with regard to the neurodevelopmental effects of lithium, further investigation is required to draw definitive conclusions. Finally, there is insufficient long-term data regarding individual SGAs to suggest preference of one agent over another. Therefore, it is imperative that potential risks and benefits from exposure to medication should be compared to the risks of maternal relapse, taking into account the severity of the illness and considering the risk to the infant of having an unwell mother (Galbally et al., 2010a). Care should be taken to discuss these aspects with the patient, her family and with all involved clinicians to devise an appropriate management plan (Galbally et al., 2010a, 2010b).
Conclusion
The findings of this review are consistent with previous research with regard to higher neurodevelopmental risk for valproate. There is a paucity of literature examining the use of lithium and SGAs for longer term child developmental outcomes and further research is required. Since the use of randomized controlled trials is unethical in pregnant women, the gold standard for this population is prospective, controlled observational studies that compare monotherapy groups and utilize blinded, standardized assessments. Preferably, follow up would continue into adulthood to determine whether the impact on children is persistent or transient. Further examination of AED use in women with mental illness is needed, as well as studies that adequately control for confounding factors. Ideally, studies would examine the impact of mental illness on child development by including an untreated control group. However, in women with BPAD, this may not be possible except in large naturalistic studies. Ongoing assessment of the impact of dose regimens and the effect of specific medication monotherapy, particularly for SGAs, is required to enable informed decision-making about mood-stabilizer use during pregnancy.
Footnotes
Acknowledgements
The authors would like to acknowledge assistance from library staff including Marcia Powell (Fiona Stanley Hospital) and Jeanette Longwood (Graylands Hospital).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: One author received grant/research support from Lundbeck.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
