Abstract
Background:
Hypertensive disorders of pregnancy are associated with longer term cardiovascular risk. Understanding if depression or antidepressant use in pregnancy is associated with HDP is important in identifying those potentially vulnerable to poorer health in later life. This study examines if depression and antidepressants are associated with HDP.
Methods:
In all, 815 pregnant women were recruited within an Australian pregnancy cohort study at less than 20 weeks of pregnancy, all undertook the Structured Clinical Interview for the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, and were assigned to four groups for this paper: those with unmedicated depression meeting criteria for current depression (n = 97), those taking selective serotonin reuptake inhibitors in early pregnancy (n = 101), those taking serotonin and noradrenaline reuptake inhibitors in early pregnancy (n = 31), and those without depression or taking antidepressant medication (control; n = 586). Women were then assessed again following birth. Hypertensive disorders of pregnancy were diagnosed according to the Society of Obstetric Medicine in Australia and New Zealand Guidelines.
Results:
Use of serotonin and noradrenaline reuptake inhibitors (SNRIs) (adjusted risk ratio = 9.10, 95% confidence interval = [3.82, 21.67]) and unmedicated depression (adjusted risk ratio = 3.11, 95% confidence interval = [1.32, 7.35]) were independently associated with significantly higher risk for developing hypertensive disorders of pregnancy compared to controls. Selective serotonin reuptake inhibitors (SSRIs) use did not confer any increased risk. Higher doses of SNRIs, but not selective serotonin reuptake inhibitors, were associated with significantly higher risk for developing HDP (adjusted risk ratio = 4.83, 95% confidence interval = [1.50, 15.58]).
Conclusions:
Our findings suggest that those with depression in pregnancy and/or on an serotonin and noradrenaline reuptake inhibitor should have closer surveillance for the development of hypertensive disorders of pregnancy. These findings support treatment of depression in pregnancy, however, also the consideration of class of antidepressant.
Background
Hypertensive disorders of pregnancy (HDP) are one of the more common complications of pregnancy and are associated with short- and long-term complications for both the woman and her offspring, including a greater risk of later maternal cardiovascular disease (Ghulmiyyah and Sibai, 2012; Khosla et al., 2021; Tooher et al., 2017). HDP are regarded as often uncovering cardiovascular vulnerability for an individual that is of significance to predicting poorer later life health (Khosla et al., 2021). Therefore, understanding any relationship between HDP and depression, may also identify individuals within the intensive health screening, that typical antenatal care provides, who are at ongoing risk beyond pregnancy for poorer mental and physical health. However, a better understanding of this relationship may also reveal opportunities for early intervention and secondary prevention of later life health complications.
Hypertensive disorders that occur in pregnancy include pre-existing essential hypertension (chronic hypertension or hypertension with onset prior to 20 weeks) as well as HDP, which include pregnancy-induced hypertension (PIH, also known as gestational hypertension) and pre-eclampsia/eclampsia, both with onset from 20 weeks of gestation onwards. HDP are associated with complications in pregnancy including pre-term birth and fetal growth restriction, and confer increased long-term maternal cardiovascular risk (Melchiorre et al., 2020). Pre-eclampsia, in particular, is characterised by multi-organ involvement, usually proteinuria and renal disease; however, it can also be associated with hepatic, cardiovascular and haematological sequelae as well as fetal growth restriction (Lowe et al., 2014). Pre-eclampsia, if severe, can also lead to the Hemolysis, Elevated Liver Enzymes and Low Platelet count (HELLP) syndrome. This condition is usually characterised by elevated lactate dehydrogenase, aspartate aminotransferase (AST) and alanine aminotransferase (ALT) and low platelet count (ACOG practice bulletin no. 202, 2019), as well as being associated with placental abruption and eclampsia. Pre-eclampsia and eclampsia are a leading cause of maternal morbidity and mortality worldwide (Ghulmiyyah and Sibai, 2012). Given the severity of these disorders, it is important in managing women with depression to understand whether either depression or antidepressants may increase the risk of HDP. This is especially important when weighing the risks and benefits for antidepressants when treating moderate to severe depression.
Both reviews and meta-analyses have examined the relationship between antidepressant use in pregnancy and HDP. For instance, a systematic review from 2017 found an adjusted relative risk of 1.28 to 1.53 for antidepressant use compared to those not taking antidepressants across the 7 identified studies (Uguz, 2017). However, the review highlighted the methodological limitations across all studies including not accounting for ‘confounding by indication’ by not measuring early pregnancy depression, which is also a potential risk factor for HDP. The review was also unable to draw clear conclusions regarding class of antidepressants (Uguz, 2017). In guiding prescribing recommendations, it is important to examine any differences in HDP risk for specific classes of antidepressants. Selective serotonin reuptake inhibitors (SSRIs) and serotonin and noradrenaline reuptake inhibitors (SNRIs) are both commonly prescribed classes of antidepressant but may have differences in risks specifically for HDP. For instance, SNRIs outside of pregnancy, are associated with elevated blood pressure, which may be attributable to the action of inhibiting the reuptake of noradrenaline, whereas SSRIs are not associated with such elevations (Benkert et al., 1996; Rudolph and Derivan, 1996). In addition, although the recent systematic review was unable to report on classes of antidepressants there have been limited reports of SNRIs being associated with HDP in previous studies (Bernard et al., 2019; Newport et al., 2016).
Another systematic review and meta-analysis from 2020 has reviewed 44 studies that included 61.2 million pregnancies to examine the association between depression and pregnancy hypertension including HDP (Shay et al., 2020). However, of the identified studies, only 17 included women with depression diagnosed before 20 weeks’ gestation (thus, before the diagnosis of HDP), and only 3 studies used a diagnostic measure rather than a symptom measure or screening tool for depression. This review (Shay et al., 2020) found that when depression and anxiety was measured prior to the development of HDP after 20 weeks’ gestation, that there was a significantly increased risk of developing HDP with a relative risk of 1.27 (95% confidence interval [CI] = [1.07–1.50]). Despite the increasing use of antidepressants to treat depression in pregnancy this specific review did not consider antidepressant treatment. Therefore, despite recent systematic reviews and meta-analyses on HDP, depression and antidepressants, there is still need for a study with prospective design, with clear well-characterised measures of both current depression and antidepressant exposure, both measured in early pregnancy, as well as with stringent characterisation of HDP and inclusion of important covariates.
The aim of the current study was to examine the risk of developing HDP among women with unmedicated depression or exposed to antidepressants during pregnancy in a prospective pregnancy cohort. Specifically, we hypothesise that antidepressants will be associated with a higher relative risk, even after controlling for empirical covariates of HDP (maternal age, body mass index [BMI] and parity) and depression compared to women with unmedicated depression as well as compared to healthy control women. We also hypothesise that class of antidepressants, specifically SNRIs, and higher doses of these agents will be associated with a higher additive risk of developing HDP.
Methods
Sample and design
Data from 832 women recruited in the Mercy Pregnancy Emotional Wellbeing Study (MPEWS) were included for this paper (see Supplementary Figure S1 for Cohort flow chart). MPEWS is a multi-site, longitudinal, prospective cohort of women recruited prior to gestational week 20 (i.e. early pregnancy), ranging between 5 and 20 weeks (M = 15.69, standard deviation [SD] = 2.64) (Galbally et al., 2017). Of these, seven women had essential hypertension (including those diagnosed with new onset hypertension at less than 20 weeks of gestation) and were excluded from analysis. In addition, 10 women who reported antidepressant agents that were neither an SSRI nor an SNRI were also excluded. Of the remaining 815 women, 464 (56.9%) were recruited in Victoria (Mercy Hospital for Women) between September 2012 and May 2017, 169 (20.7%) from Perth (Fiona Stanley Hospital and King Edward Memorial Hospital) between June 2017 and December 2018, and 182 (22.3%) from rural regions of Western Australia (Goldfields, South West, and Midwest regions) between October 2018 and February 2020. For the purposes of this study, women were categorised into the following exposure groups: women taking SSRIs in early pregnancy (n = 101), women taking SNRIs in early pregnancy (n = 31), women who met criteria for current depressive disorder in early pregnancy at recruitment according to the diagnostic measure Structured Clinical Interview for Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV) (SCID-IV) but did not use antidepressants (i.e. unmedicated depression; n = 97), and control women who were neither on antidepressant treatment nor had current depressive disorder (i.e., control n = 586). After recruitment at less than 20 weeks of gestation, women were followed up prospectively during the third trimester and after birth. Further details of the MPEWS protocol are published elsewhere (Galbally et al., 2017). Women from the MPEWS cohort were included in this sample if they still were participating at delivery and had pregnancy, obstetric, and neonatal data collected from their hospital records.
Measures
Mental health
At recruitment, we used the SCID-IV mood disorders schedule (First et al., 1997) to identify the presence of DSM defined mood disorders. The Edinburgh Postnatal Depression Scale (EPDS; Cox et al., 1987), a 10-item scale with total score ranging 0–30, was also administered at recruitment during early pregnancy as a measure of depressive symptom severity.
Antidepressant use
Antidepressant type, usage, and dosage during early pregnancy (⩽20 weeks) were self-reported by women, as well as obtained from hospital records at birth. As most participants were on sertraline, doses of other antidepressants were converted to a sertraline-equivalent dosage (SED) using a conversion chart (Galbally et al., 2017; Procyshyn et al., 2015).
Hypertensive Disorders of Pregnancy (HDP)
HDP included in this study were subdivided into PIH and pre-eclampsia and were extracted from hospital medical records and confirmed by a medical record entry from an obstetrician. HDP was defined by applying Australian Guidelines (Lowe et al., 2014). Specifically, PIH was defined as a new onset in pregnancy of a systolic blood pressure ⩾ 140 mmHg and a diastolic blood pressure ⩾ 90 mmHg with onset after 20 weeks of gestation. Pre-eclampsia was defined as onset of hypertension after 20 weeks of gestation combined with signs of involvement of other maternal organ systems, commonly this is renal affection with proteinuria, but it can also include hepatic, neurological, and/or haematological manifestations in addition to fetal growth restriction (Lowe et al., 2014).
Statistical analyses
Prior to fitting the regression models to address the hypotheses, we present sample rates, attributable risk (AR) and unadjusted risk ratio (RR) for developing HDP associated with early pregnancy SSRI and SNRI use and unmedicated depression with antidepressants. To calculate unadjusted RR associated with depression, SSRI and SNRI use, rates of PIH, pre-eclampsia and HDP observed in the control group were used as the reference for comparison.
To address the first hypothesis, a generalised log-binomial regression model was fit using HDP as the target and predicted by the SSRI, SNRI and unmedicated depression groups. In this model, the control group was also used as the reference for the comparison of risk. We only modelled HDP as the outcome given the smaller rates of PIH and pre-eclampsia separately. Empirically supported covariates (i.e. age, obesity, parity) and data-identified covariates of both the exposures and HDP were included in this model to estimate adjusted risk ratios (aRRs) of HDP associated with antidepressant classes and unmedicated depression relative to control. To address the second hypothesis, we fit a second log-binomial regression model using only women taking antidepressants during pregnancy to test the interaction between antidepressant class (SNRI compared to SSRI) and SED on the risk of developing HDP. In this model, we also adjusted for depressive symptoms in early pregnancy. All analyses were conducted using SPSS version 26 and significance was assessed using an alpha level of .05 or less.
Results
Sociodemographic and other key characteristics
Key sociodemographic and other characteristics of the 815 women by exposure group are presented in Table 1, including testing of all pairwise comparisons using a Bonferroni-adjusted p-value (p < 0.008). The women were predominantly identified as Oceanic and/or European, with no significant difference between groups. Although women were almost exclusively in a married, de facto or stable relationships, significantly more women in the control group reported this relationship status compared to women in the SNRI group. Significantly more women in control group were nulliparous prior to the current pregnancy compared to women in the SSRI group. Significantly more women taking SSRIs reported smoking during pregnancy compared to the controls, and significantly more women in both antidepressant exposure groups had a BMI in the obese range (⩾30 kg/m2) at recruitment compared to the control group. There were no differences in using assisted reproduction between the groups. Table 2 displays a summary of antidepressant use and dose, including SED, during early pregnancy (i.e. up to gestation week 20). There were 10 women taking an antidepressant as well as an antipsychotic, and a further 9 women taking only an antipsychotic.
Sample sociodemographic and other key characterises by exposure group (N = 815).
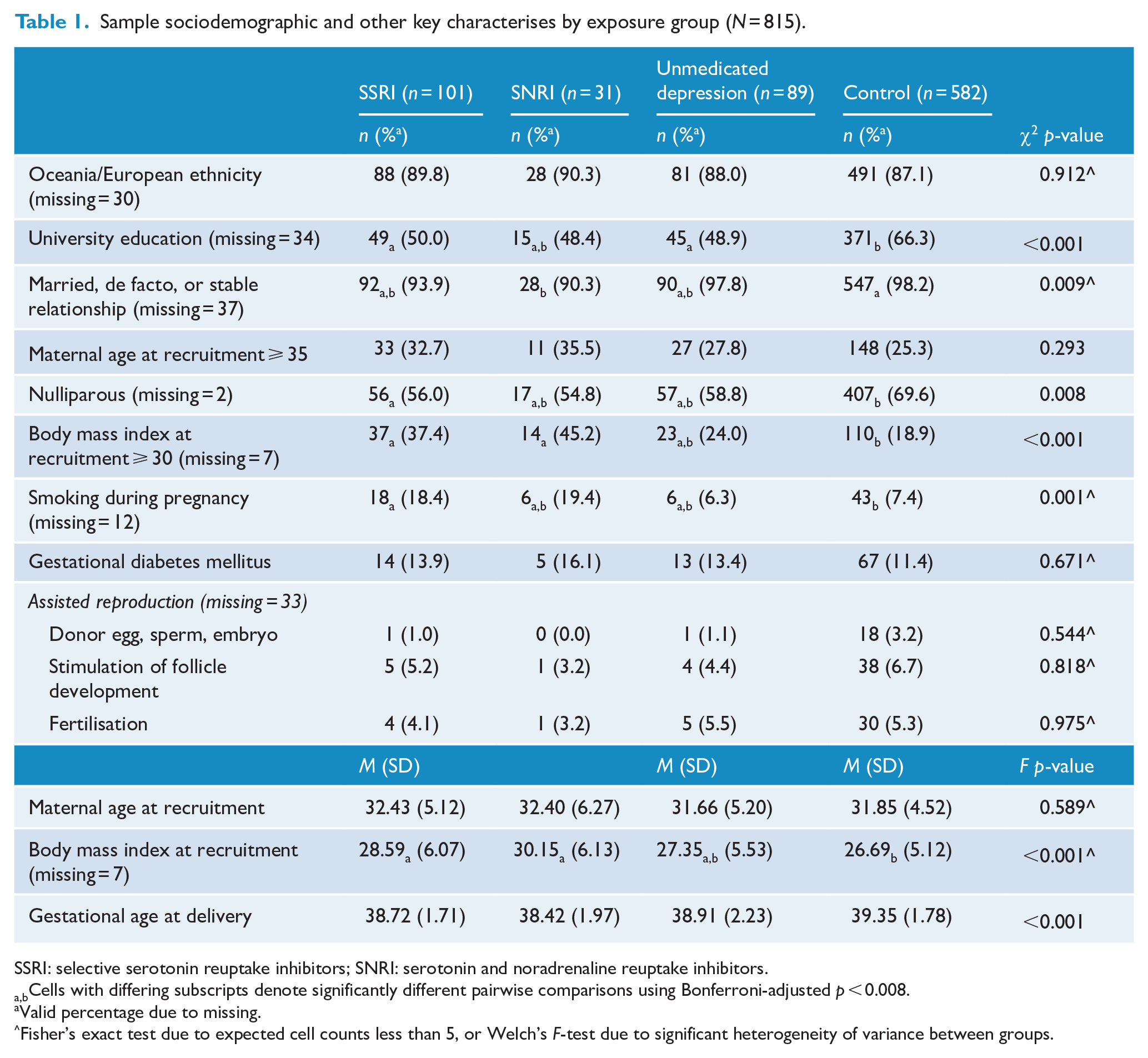
SSRI: selective serotonin reuptake inhibitors; SNRI: serotonin and noradrenaline reuptake inhibitors.
Cells with differing subscripts denote significantly different pairwise comparisons using Bonferroni-adjusted p < 0.008.
Valid percentage due to missing.
Fisher’s exact test due to expected cell counts less than 5, or Welch’s F-test due to significant heterogeneity of variance between groups.
Summary statistics for antidepressant use before gestational week 20 (n = 132).
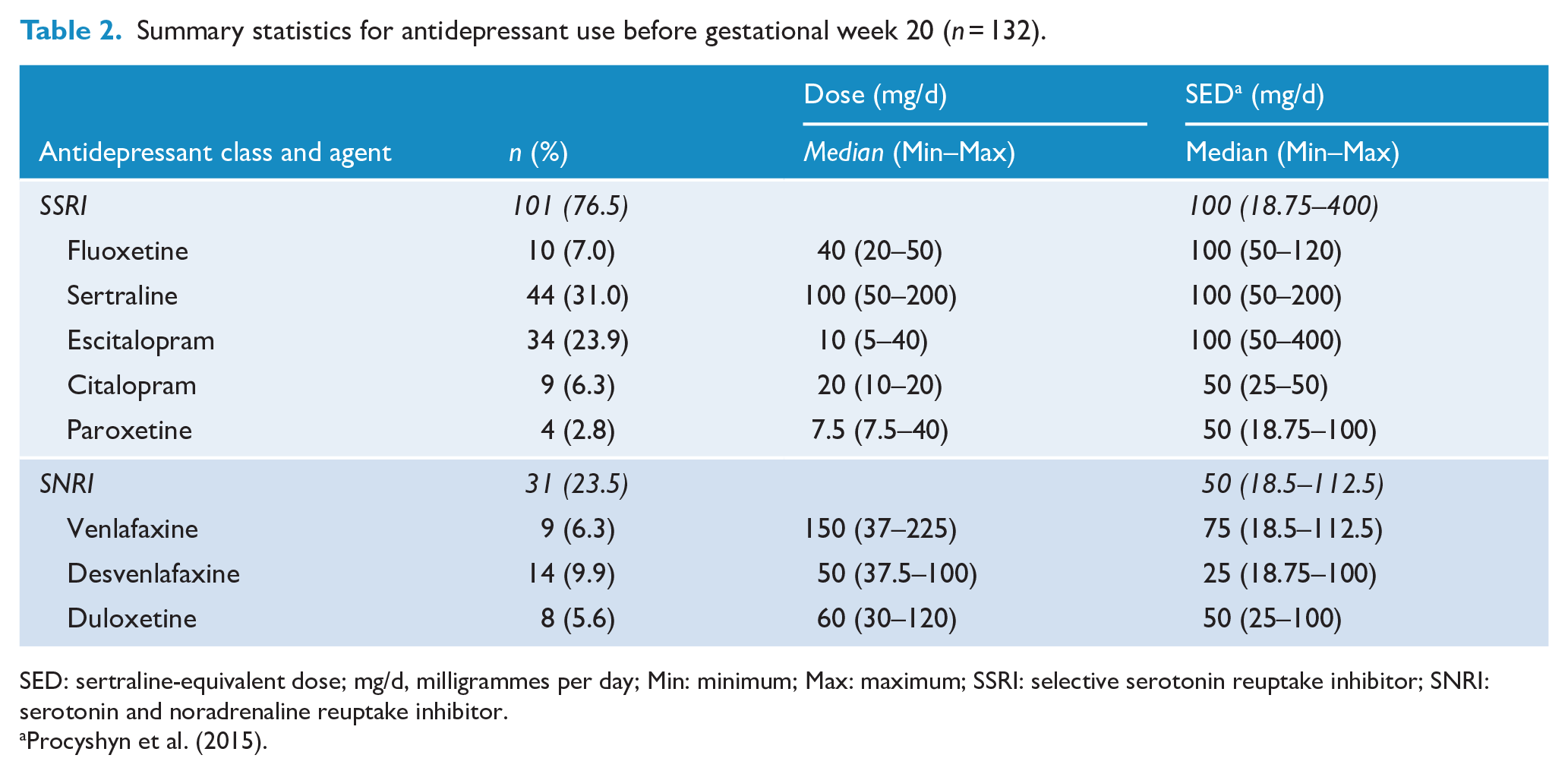
SED: sertraline-equivalent dose; mg/d, milligrammes per day; Min: minimum; Max: maximum; SSRI: selective serotonin reuptake inhibitor; SNRI: serotonin and noradrenaline reuptake inhibitor.
Hypertensive disorders of pregnancy by exposure group
Of the 815 women in this MPEWS sample, 25 (3.1%) developed PIH and 21 (2.6%) developed pre-eclampsia; together, 46 women (5.6%) had developed HDP. Table 3 displays rates and unadjusted RR of PIH, pre-eclampsia and HDP for the unmedicated depression, SSRI and SNRI groups each compared to the control group. Overall, both SNRI exposure and unmedicated depression were associated with significantly higher unadjusted risk for developing HDP.
Observed rates and unadjusted relative risk (RR) of pregnancy-induced hypertension and pre-eclampsia, and hypertensive disorders of pregnancy (i.e. pregnancy-induced hypertension and pre-eclampsia combined) by exposure group (N = 815).
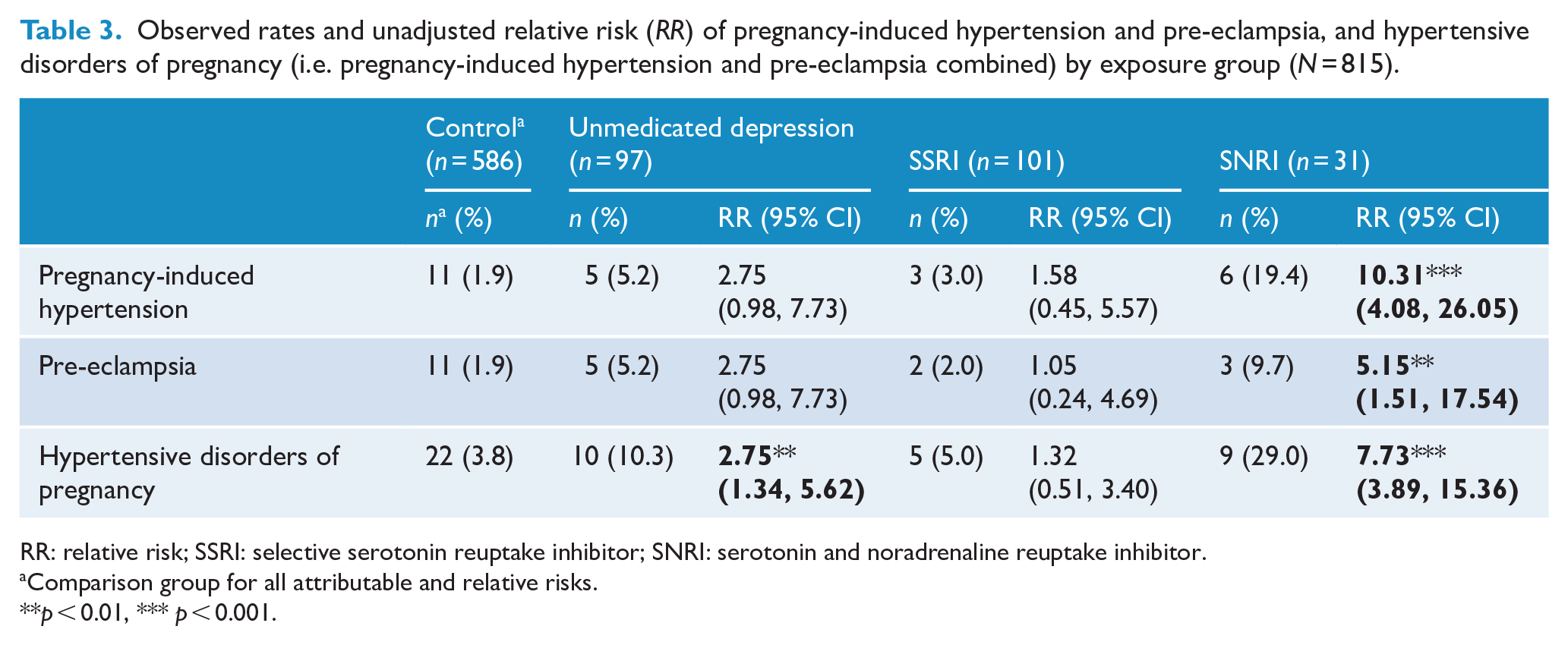
RR: relative risk; SSRI: selective serotonin reuptake inhibitor; SNRI: serotonin and noradrenaline reuptake inhibitor.
Comparison group for all attributable and relative risks.
p < 0.01, *** p < 0.001.
Adjusted risk of hypertensive disorders of pregnancy
In the regression model, the higher unadjusted risks of HDP attributable to SNRI use and unmedicated depression during pregnancy, relative to the comparison group, remained significant. Table 4 displays the focal results of this generalised regression model (complete results of the model are presented in Supplementary Material, Table S1). Following adjustment for both the empirically and data-identified covariates (advanced maternal age [⩾35 years], obesity at recruitment (BMI ⩾ 30 kg/m2), parity, smoking in pregnancy, a university education, relationship status, and early pregnancy depressive symptoms), women taking SNRIs in pregnancy had an 9.10-fold (95% CI = [3.82, 21.67]) increase (in their risk of developing HDP relative to the control group (p < 0.001). Furthermore, women with unmedicated depression in pregnancy, relative to the control group, had a 3.11-fold (95% CI = [1.32, 7.35]) increase in their adjusted risk of developing HDP (p = 0.010).
Results for log-binomial generalised linear regression model predicting HDP (n = 772).
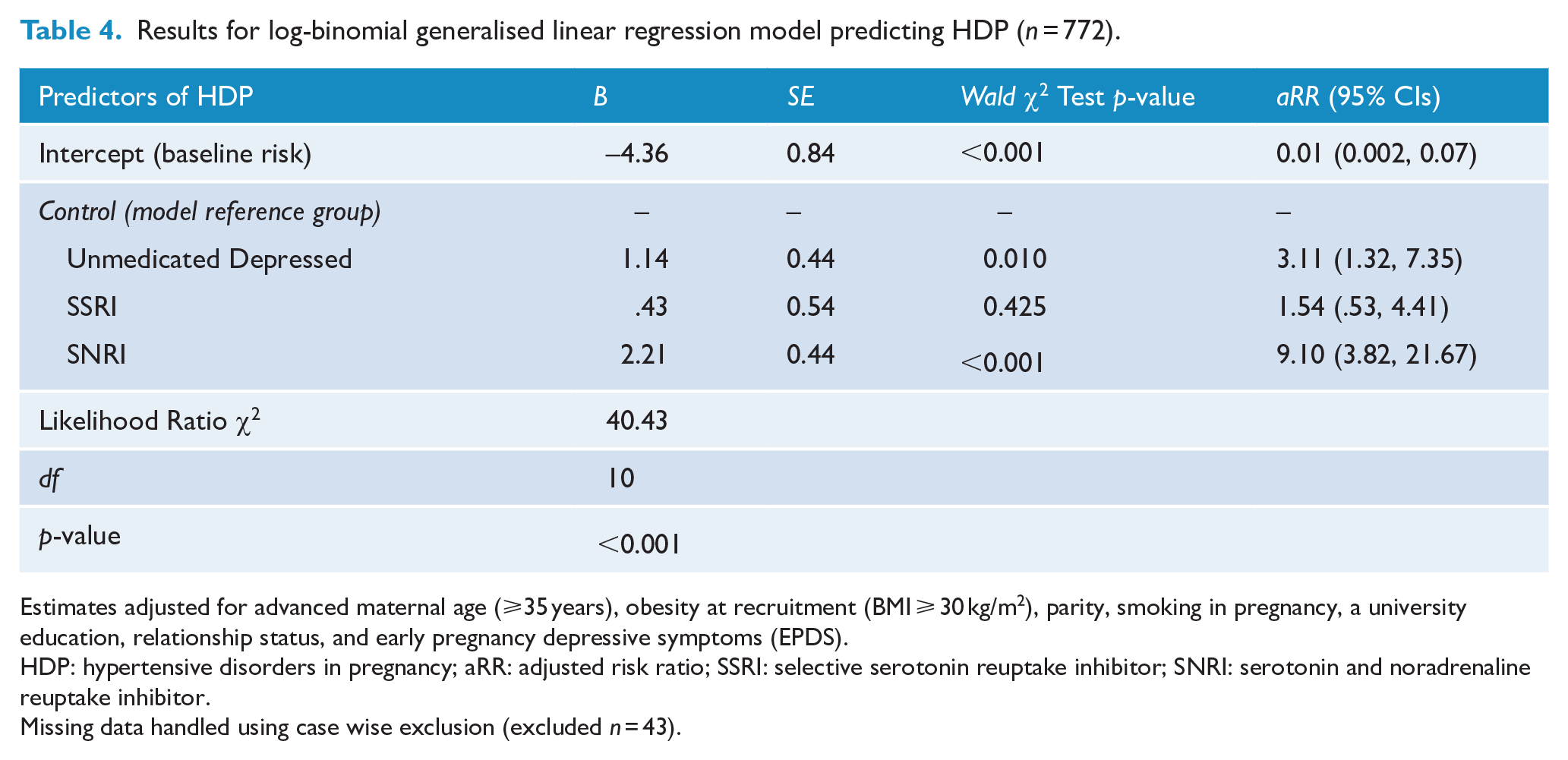
Estimates adjusted for advanced maternal age (⩾35 years), obesity at recruitment (BMI ⩾ 30 kg/m2), parity, smoking in pregnancy, a university education, relationship status, and early pregnancy depressive symptoms (EPDS).
HDP: hypertensive disorders in pregnancy; aRR: adjusted risk ratio; SSRI: selective serotonin reuptake inhibitor; SNRI: serotonin and noradrenaline reuptake inhibitor.
Missing data handled using case wise exclusion (excluded n = 43).
In the final log-binomial regression model using only women taking antidepressants during pregnancy (n = 123; see Table S2), SNRI use was associated with a significant 10.37-fold (95% CI = [3.21, 33.49], p < 0.001) increase in the adjusted risk for developing HDP compared to SSRI use when holding SED at 0 (i.e. when using an average SED, Z = 0). The interaction between antidepressant class and SED was significant, suggesting that for SNRIs (aRR = 4.83, 95% CI = [1.50, 15.58], p = 0.008), but not for SSRIs (aRR = 0.57, 95% CI = [0.28, 1.15], p = 0.117), an increase of one standard deviation from the mean in SED (i.e. Z = 1) was associated with a significant additive risk of developing HDP.
Discussion
Our study found that both depression and antidepressant use in pregnancy were associated with greater risk of HDP. In particular, taking an SNRI in pregnancy had the greatest risk with an 9.10-fold increased risk of developing HDP, compared to control women, and this but the risk was also increased compared to women on SSRIs in pregnancy. Furthermore, women with unmedicated depression in pregnancy had a 3.11-fold increase in the adjusted risk of developing HDP relative to the control group. Women with either SNRI use in early pregnancy or unmedicated depression in early pregnancy had an increased risk for both PIH and pre-eclampsia. Of note, increasing dose of SNRIs but not SSRIs was associated with an increased risk of HDP. No other previous study we are aware of has included a depressed unmedicated group who met criteria for a current depressive disorder in early pregnancy; however, we were able to verify and track exposure in detail and we also undertook and included a range of key confounding factors. All recent studies relied on obstetric assessment and record of HDP diagnosis as did our study.
Our sample included 5.7% of women who developed HDP and this is somewhat higher than the 3.5% rate in Australian women who gave birth between 2014 and 2019 (Australian Institute of Health and Welfare, 2020). In part, this may be explained by our over-representation of women with depression, with 29% of our sample with either treated or unmedicated depression. Our findings support the results from a retrospective study of women with severe mental illness in pregnancy, which also found that both depression and SNRI use were associated with HDP (Frayne et al., 2021). However, this previous study did not measure current mental illness and was unable to distinguish between the various hypertensive disorders of pregnancy. In a recent prospective study of 7866 pregnant women, antidepressants, and in particular SNRIs, were associated with an increased risk of pre-eclampsia if used in early pregnancy, but this risk was reduced if medication was stopped by 16 weeks of gestation(Bernard et al., 2019). While that study had an overall larger sample size, the numbers identified taking antidepressants in pregnancy was small with only 57 women on SNRIs and 103 on SSRIs, use was based on hospital records only as was reported depression/anxiety in the unmedicated comparison group and there no was dosage information for antidepressants reported. That study found that SNRI use (n = 57) was associated with an adjusted odds ratio (OR) of 6.46 (95% CI = [2.49, 16.78]) for pre-eclampsia. It also found a significant association, albeit smaller, for SSRI exposure (n = 103) with an OR of 3.09 (95% CI = [1.22, 7.85]). There was no significant association with gestational hypertension (PIH). Furthermore, there was no significant association between non-medicated depression/anxiety (n = 41) and HDP (Bernard et al., 2019).
In contrast, two recent studies have not found an association for SNRI exposure including Yang et al. (2021) and Zakiyah et al. (2018). In the most recent of these, a retrospective study of a Taiwanese health database, the authors were not able to compare to an unmedicated depressed group and although they examined SNRI exposed women they did not compare these to SSRI exposed or to unmedicated depressed women (Yang et al., 2021). They also did not examine subgroups of HDP. The study, though, did find that those with higher doses of antidepressants had a higher risk of HDP. The other retrospective study, of a prescription database, found that use of antidepressants, and in particular SSRIs, was associated with an increased risk of HDP (Zakiyah et al., 2018). However, the authors of that study grouped SNRIs together with other agents such as mirtazapine, trazodone, bupropion and nefazodone into a category of ‘other’ so they were unable to examine SNRIs specifically and they did not have an unmedicated depressed comparison group.
Our findings that SNRIs were associated with HDP is supported by SNRIs being known to cause clinically relevant increases in blood pressure, in contrast to SSRIs (Benkert et al., 1996; Rudolph and Derivan, 1996). This adverse drug reaction is particularly prevalent at higher doses and has also been previously reported during pregnancy (Bernard et al., 2019; Newport et al., 2016). The divergent effects on blood pressure for SNRIs and SSRIs can be explained by their differential mechanisms of action, as SNRIs also inhibit the reuptake of noradrenaline. The resultant noradrenergic stimulation (Stahl, 1998) may increase blood pressure and induce hypertension, including in pregnancy such as in the case of PIH (Stahl, 2005).
Our finding related to SNRIs and pre-eclampsia is in contrast to a recent study which did not find such an association (Lupattelli et al., 2017). However, there are possible underlying mechanisms by which SNRIs could be associated with pre-eclampsia, given the vasoconstrictive effect of this drug class and the potential impact on placentation (Roberts and Hubel, 2009; Staff, 2019). Both early and late onset pre-eclampsia are considered to arise from poor placentation, with early onset thought to be associated with poor placental perfusion and a growth restricted foetus whereas late onset is more frequently associated with maternal risk factors and not necessarily with foetal growth restriction (Staff, 2019). Whether the use of SNRIs could be associated with vasoconstriction in early pregnancy impacting the development of microcirculation with resultant poor placentation is a matter at this point for speculation (Maaliki et al., 2019; Staff, 2019).
Our findings of an increased risk for HDP associated with current depression in early pregnancy confirms the results from a recent meta-analysis. Within this meta-analysis, only 3 studies identified had utilised a diagnostic measure to confirm current depression and overall across all the 44 studies RR was 1.39 (95% CI = [1.25, 1.54]) (Shay et al., 2020). The meta-analysis did not examine subgroups of HDP such as PIH and pre-eclampsia. Our findings that depression was associated specifically with pre-eclampsia is important to build on this previous meta-analysis and highlight the importance of distinguishing between subgroups of HDP in future research. While previous studies have attempted to consider depression severity this is often as either a proxy to diagnosis (Lupattelli et al., 2017) or measured through number of healthcare visits and/or hospitalisations (Palmsten et al., 2012), in contrast our study has the advantage of both having a diagnostic measure administered in early pregnancy and using the symptom measure the EPDS, which is validated specifically for use in pregnancy.
Finally, our finding of SSRIs not being specifically associated with HDP offers an important finding given the association between depression and HDP. Whereas mild to moderate depression is ideally managed first line with psychological treatment, for women with moderate to severe depression, antidepressant treatments are an important option including in pregnancy (Galbally et al., 2020). These treatments are also supported in a range of national guidelines (Molenaar et al., 2018).
The strengths of this study include its prospective design and the inclusion of a control group of unmedicated depressed women who met current criteria for depression on a diagnostic interview in early pregnancy in addition to a non-depressed control group. Antidepressant use was registered at repeat time points using several parallel methods including self-report, hospital records and drug analyses in blood samples at birth to confirm compliance. The information collected on antidepressant use included information on dose, agent, class and changes in treatment over pregnancy.
Limitations of our study include small groups for individual antidepressant agents making only class comparisons rather than agent comparisons possible. Nevertheless, although we analysed drug exposure by SSRI and SNRI class rather than by individual agent, the drugs included in each of these two groups are very similar from a pharmacodynamic perspective. Another limitation is that we were unable to distinguish early and late pre-eclampsia. We also did not include measures of depressive symptoms beyond 20 weeks of gestation and did not have information on how long women had been on antidepressants prior to becoming pregnant. Although we adjusted for current depressive disorder and depressive symptoms, we were also unable to identify if those who were commenced on an SNRI had a more severe depression prior to treatment. Although there were significantly fewer nulliparous women in the antidepressant group and in the depressed group compared to control women, adjustment was made for this in our regression model.
Our study found that SNRI exposure before gestational week 20 of pregnancy was associated with an increased risk of developing HDP including pre-eclampsia and PIH. However, we also found that unmedicated, current depressive disorders in early pregnancy were associated with an increased risk of developing pre-eclampsia, making the inclusion of a group with current depression in early pregnancy integral to studies examining antidepressant treatment and the risk of developing HDP. Managing depression in pregnancy is complicated by the often conflicting and complex range of recent research findings for risks for both untreated, undertreated and antidepressant treated depression for offspring outcomes and requires an individual personalised approach to decision making (Galbally et al., 2022; Galbally and Snellen, 2020). While psychological treatment can benefit many women, for some, antidepressant treatment is an important option and our findings will assist clinicians to guide individual treatment choices where the risk of HDP is present.
Supplemental Material
sj-docx-1-anp-10.1177_00048674221106915 – Supplemental material for Depression and antidepressant treatment in the development of hypertensive disorders of pregnancy: Results from a prospective cohort study
Supplemental material, sj-docx-1-anp-10.1177_00048674221106915 for Depression and antidepressant treatment in the development of hypertensive disorders of pregnancy: Results from a prospective cohort study by Megan Galbally, Stuart J Watson and Olav Spigset in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The authors would like to thank those who have supported the development of MPEWS including Michael Permezel, Andrew Lewis and Marinus van IJzendoorn. Thank you also to Susan Walker for her advice and support for this study. The authors also thank staff, students and volunteers on the MPEWS study as well as study co-ordinators: Nicole Brooks, Tina Vaiano and Jacinta Holland for their contribution to MPEWS. We are also sincerely grateful to the study participants who have contributed a substantial amount of time to participating in this study. We would like to also acknowledge the support of the Spinnaker Foundation, Telethon Perth Children’s Foundation and WA Country Health Service in supporting this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported through the 2012 National Priority Founding Round of Beyondblue in a 3-year research grant (ID 519240), a 2015 National Health and Medical Research Council (NHMRC) project grant for 5 years (APP1106823), and a Spinnaker Health Research Foundation 2017/2018 Research Award.
Ethics Approval
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008. This project has ethics approval with Mercy Health Human Research Ethics Committee Reference R08/22 and with WA Health South Metropolitan Human Research Ethics Committee Reference 2016-192.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
