Abstract
The prevalence of depression in pregnancy has been shown to range between 7.4% and 12.8% across the three trimesters [1]. Antenatal depression is one of the strongest predictors of postnatal depression [2]. Evidence also suggests that antenatal depression and anxiety are associated with an increased risk of pregnancy and neonatal complications and both antenatal and postnatal depression may impact on child health and development [3–6]. The growing awareness of the impact of maternal depression has highlighted the importance of identifying and effectively treating pregnant women for depression.
Treatment options for depression in pregnancy include psychological interventions and pharmacotherapy. For a variety of clinical reasons pharmacotherapy may be the most effective treatment for a woman or may be required in conjunction with psychological intervention. Pharmacotherapy may also be required due to the clinical concerns and severity of the depressive illness such as associated suicidality or psychotic symptoms [7]. It may also be required when there are significant biological symptoms of depression such as severe appetite or sleep changes or psychomotor retardation, which are felt to place the woman at physical risk [7]. In addition, women who are already being treated with antidepressant medication who discontinue their medication in pregnancy have been shown to have an increased risk of relapse; this risk was shown to be 68% compared to 26% who remained on their pre-conception dose of antidepressant medication [8].
Since this increased awareness among clinicians of the importance of treating maternal depression there has been a sharp increase in rates of treatment of antenatal depression, including the prescription of antidepressant medication in pregnancy. A study across the USA found that there was an increase in antidepressant prescription from 2% in 1994 to 7.6% in 2005 and, of this, 6.2% in 2005 were selective serotonin re-uptake inhibitors (SSRIs) [9]. Despite increasing prescription rates, clinical recommendations regarding the management of depressed pregnant women and infants who were exposed to antidepressant medication are based on an inadequate evidence base.
All antidepressants cross the placenta, thus exposing the developing fetus during pregnancy [10]. Placental passage studies for the SSRIs have shown variable concentrations, although all were much lower than maternal doses [10]. The concentration did vary, however, between different medications, with one study showing sertraline being lower than fluoxetine, and it has been noted that there is also individual variability in cord: maternal drug concentration ratios [11].
Antidepressant exposure in utero has been associated with a number of neonatal and developmental outcomes. These include a possible association with malformation, increased risk of persistent pulmonary hypertension of the newborn [12], serotonin neonatal discontinuation symptoms [13,14] and motor development effects [15]. The literature on effects on the newborn infant of antenatal antidepressant exposure is growing. Using a sample of almost 120 000 mothers and infants drawing on population-based linked health data on neonatal outcomes, Oberlander et al. found that SSRI-exposed infants had significantly lower birthweight and gestational age than non-exposed infants [16]. They also found an increased proportion of neonatal respiratory distress, jaundice, and feeding problems in the exposed group. Both Wen et al. and Kallen also found an association between SSRI in utero exposure and low birthweight and preterm delivery [17,18]. Several other studies, however, have failed to find this association, finding instead that those neonates exposed to antidepressants in utero were no different in gestational age at delivery and birthweight from controls [19–21]. Ericson et al., in examining the effect of early pregnancy exposure to antidepressants, found an association between early exposure and preterm delivery but not low birthweight [22]. Of the studies that found an association between exposure and prematurity and low birthweight, none followed up their sample beyond delivery to examine if this is a transitory effect or sustained.
This study assessed birth outcomes and re-assessed these at 1 month postpartum in order to investigate whether there are any sustained effects on newborn growth and development associated with in utero exposure to antidepressant medication.
Methods
This study recruited 27 women who were treated with an SSRI, Serotonin and noradrenaline reuptake inhibitors (SNRI) or Noradrenergic and specific serotonergic antidepressants (NaSSA) for depression during their pregnancy between June 2004 and July 2005. Private psychiatrists, and medical staff at the Mercy Hospital for Women, Melbourne, Australia, referred pregnant women being treated with an antidepressant medication. All subjects receiving antidepressant medications who were enrolled in the study were diagnosed as having major depression according to an independent psychiatric evaluation and found to require pharmacotherapy. A matched control group of 27 pregnant women not taking antidepressants was recruited prospectively via antenatal appointments at the Mercy Hospital for Women. The study was approved by the Mercy Hospital for Women Research Ethics Board. A written informed consent statement was obtained from each participant.
Women were excluded from the study on the grounds of substance dependence, intellectual disability, serious physical illness and psychiatric illness requiring acute inpatient admission. Inclusion criteria included English proficiency.
Measures
Maternal characteristics
A purpose-designed questionnaire was used to assess demographics, reproductive and medical history, drug use, maternal stress and coping. During the initial consultation, during pregnancy, a detailed medical, genetic, and obstetric history was obtained from each mother. We also collected information about alcohol consumption, tobacco and recreational drug use, and maternal medical care. Details concerning the time and duration of exposure to antidepressants and the dose of the antidepressant drug were recorded. After delivery a set of nine questions was used to assess for serotonin discontinuation symptoms in the neonate. At this interview the mother was questioned about the course of her pregnancy, verification of the duration, dose and type of antidepressant treatment during pregnancy, maternal illnesses, and perinatal and/or postnatal complications. Information about delivery methods and perinatal complications was collected.
Maternal depression
The Beck Depression Inventory–second edition (BDI-II) was used to assess depression in all participants in pregnancy, at delivery and at 1 month postpartum [23]. The BDI-II is a 21-item self-report instrument intended to assess the existence and severity of symptoms of depression. The BDI-II has been reported to be highly reliable as a measure of depression, with established construct validity and an established ability to differentiate depressed from non-depressed patients. The BDI-II has coefficient alphas of 0.92 for outpatients and 0.93 for college students. Test–retest reliability 1 week apart showed a correlation of 0.93. The original BDI has been used successfully as a treatment outcome measure for pharmacotherapy and has been shown to be a sensitive measure of therapeutic change [24].
Neonatal characteristics
Information about neonatal birth outcomes was collected from the mother and this information was verified with hospital birth records. In addition, information on birthweight, length, head circumference and Apgar scores was collected. Data on weight, length and head circumference were collected at 1 month based on the records of the universal maternal child health nurse check that is carried out for children born in Victoria. The date on which these measures were taken was also recorded. Data on symptoms consistent with neonatal serotonin discontinuation were also collected and have been previously reported [25].
Results
Data screening
One participant in the medication group had a very low gestational age and significant complications and was an outlier. This participant's data were eliminated from further analysis. Data collected at the 1 month period included those for three participants who missed their 1 month appointment but presented at 60 days, and another participant who presented for a postnatal check up at 14 days. These four participants’ data were eliminated from the ANOVA analysis of 1 month outcomes. After such adjustments, skew and kurtosis were within acceptable levels for univariate and multivariate analyses.
Demographics
Table 1 lists characteristic of the medicated and control groups. Gender of infants was 12 girls in the study group (24%) and 17 girls in the control group (34%), with 12 boys in the study group (24%) and nine boys in the control group (18%). A χ2 test did not indicate a significant difference in the distribution of genders between groups (χ2(1) = 0.66, p = 0.42).
Subject details (n = 54)
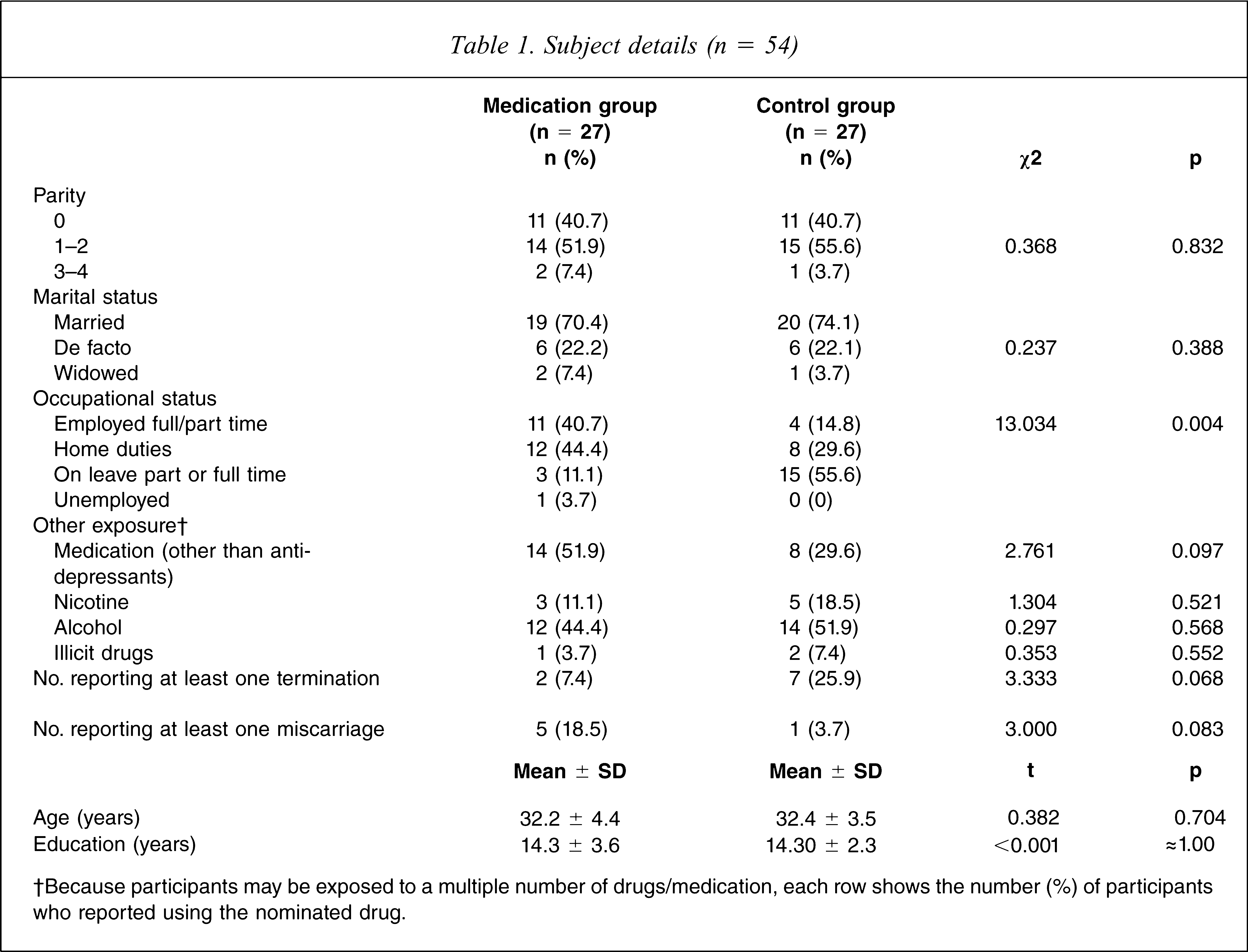
†Because participants may be exposed to a multiple number of drugs/medication, each row shows the number (%) of participants who reported using the nominated drug.
Infant outcomes
Differences in birth and 1 month outcomes are presented in Table 2, along with significance tests and effect sizes.
Birth and 1 month infant growth outcomes
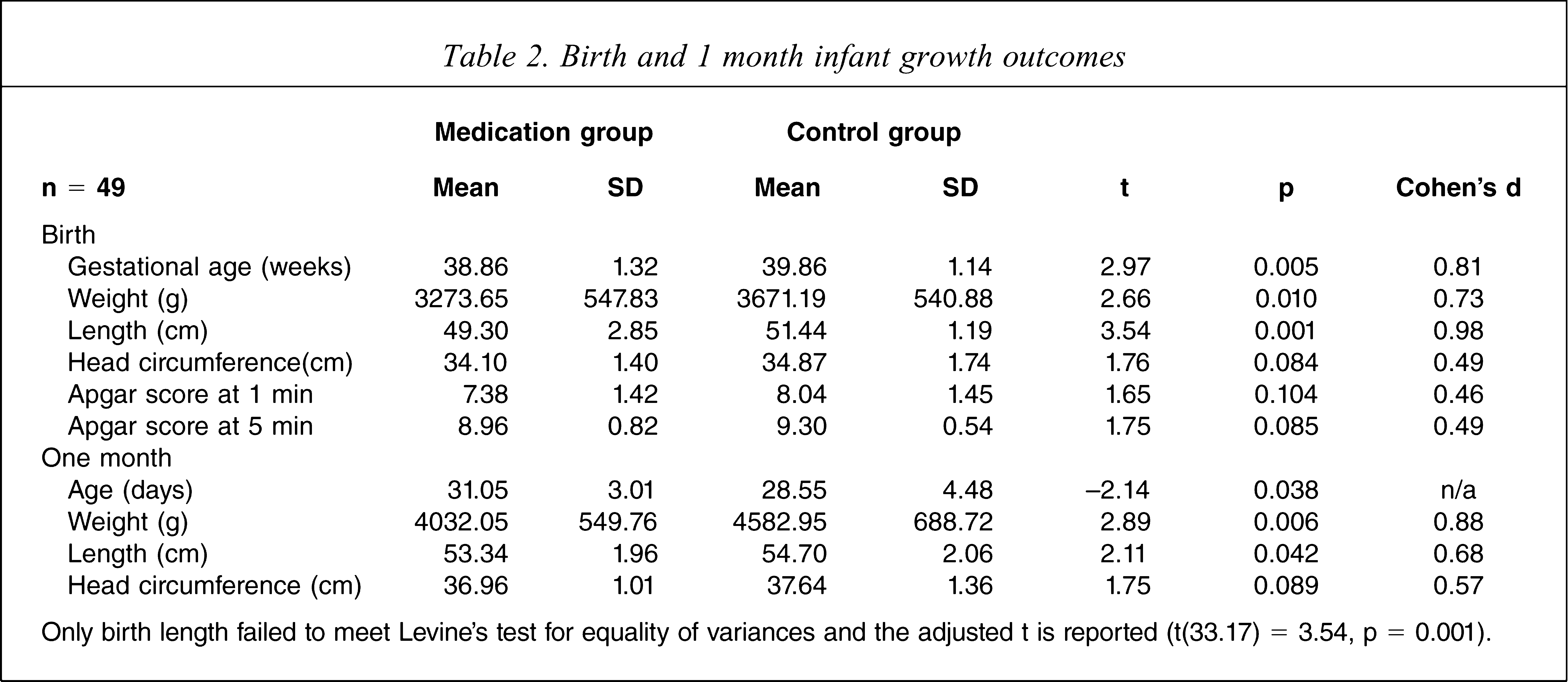
Only birth length failed to meet Levine's test for equality of variances and the adjusted t is reported (t(33.17) = 3.54, p = 0.001).
Further analysis was undertaken to determine the number of neonates from each group who fell within a clinical range for lowered birthweight (<2500 g) and reduced gestational age (<37 weeks). Four neonates were below 2500 g in the medication-exposed group, while none was below that weight in the control group. A Peto odds ratio (OR) calculation was used because one score was zero, therefore OR = 8.33 (95% confidence interval (CI) = 1.11–62.67) for being in the clinical range for low birthweight if exposed to antidepressant medication in utero. Four neonates were <37 weeks gestation in the medication-exposed group, while one was <37 weeks in the control. The OR for being in the clinical range for prematurity was OR = 4.52 (95%CI = 0.47–43.41).
It is to be noted that despite removal of outliers in the time at which 1 month measurements were taken, the groups remained significantly different in that the medication group was, on average, measured 2.5 days later than the control group. This difference was taken into consideration by performing subsequent MANCOVAs with birth outcomes at 1 month as dependent variables and adding age of measurement as a covariant. The results of these analyses suggest that at 1 month postpartum weight (F = 8.68, p = 0.005), length (F = 5.57, p = 0.023) and head circumference(F = 5.40, p = 0.025) were all significantly lower in the medicated group, even after controlling for the variance in time of measurement.
A number of additional comparisons were performed for other exposures during pregnancy that might be thought to influence birth outcomes. These included the comparison of the tobacco and alcohol use of the two groups. No significant differences where found for cigarettes smoked per day between medication and control group or the number of alcoholic drinks consumed per week. In a similar fashion, although previous studies have found an association between levels of depression during pregnancy and at birth and child outcomes, the present study found no such associations with depression levels measured using the BDI-II in pregnancy. These were not significantly correlated with the gestational age of the neonate (r = −0.01, p = 0.95), birthweight (r = 0.01, p = 0.97), length (r = 0.22, p = 0.12), or head circumference (r = −0.02, p = 0.87). Similar non-significant associations were found with the BDI-II scores taken at birth.
Growth rates between birth and 1 month
In order to explore the effects of medication exposure for the time interval between birth and 1 month of age, a repeated-measure ANOVA was performed for weight, length and head circumference, respectively.
With respect to weight, the analysis indicated a significant difference in the birthweight of medication-exposed infants as compared to controls (F(1,20) = 4.34, p = 0.05, partial η2 = 0.18) and, as would be expected, a significant change for both groups as an effect of growth over 1 month (F(1,20) = 281.05, p < 0.001, partial η2 = 0.93). The rate of growth, however, for the medication-exposed infants was significantly lower over the 1 month time period, suggesting a decline in weight gain for the medication group. The interaction between time and weight is significant (F(1,20) = 6.27, p = 0.02, partial η2 = 0.24).
A significant difference in length was found between medication-exposed and control infants (F(1,20) = 6.27, p = 0.02, partial η2 = 0.28) and, again a substantial effect of growth in head size over the 1 month period. With respect to growth rate in length, however, there was a small catch-up effect for the medication group. But due its very small magnitude, the interaction between time and head circumference failed to reach significance (F(1,20) = 3.35, p = 0.08, partial η2 = 0.17).
A significant difference in head circumference was found between medication-exposed and control infants (F(1,20) = 4.19, p = 0.05, partial η2 = 0.18) and, again a substantial effect of growth in head size over the 1 month period. With respect to growth rate, however, there was no difference between groups, suggesting a lack of catch-up head growth for the medication group. In other words, no interaction was found between time and head circumference (F(1,20) = 0.28, p = 0.61, partial η2 = 0.014).
In order to analyse the rates of growth while considering the different ages at which the growth measurements were taken, a mean growth rate per day was calculated. For example, regarding weight, this was achieved by subtracting birthweight from the 1 month weight and dividing that weight gain by the infant's age (in days) when the data were collected. Because this method accounts for the variability in time of measurement, the data from those who were weighed at 60 days were re-introduced. The mean rates of growth for weight, length and head circumference are reported in Table 3, together with reports of the standardized means and significance tests of these differences between the medication and control group. It is to be noted that only the rate of weight gain was significantly lower in the medication group.
Mean rates of change over 1 month
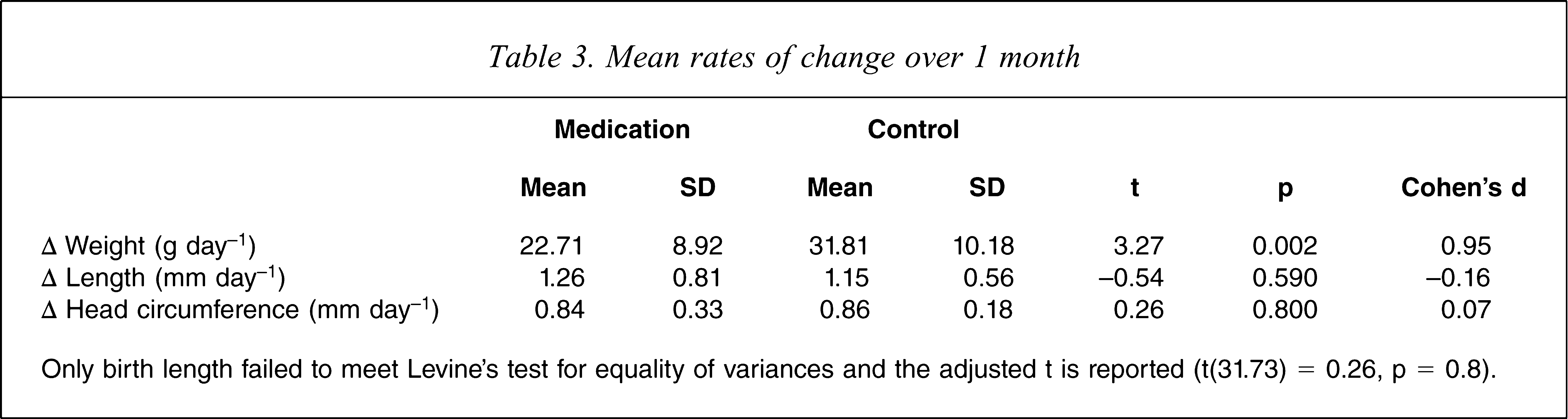
Only birth length failed to meet Levine's test for equality of variances and the adjusted t is reported (t(31.73) = 0.26, p = 0.8).
Discussion
Neonatal outcomes such as gestational age at birth, birthweight, length, head circumference and early neonatal growth have been shown to be affected by fetal factors such as chromosomal abnormalities, maternal physical and mental illness and in utero exposure to substances such as nicotine and alcohol. The present study found that in utero exposure to antidepressant medications affected gestation at birth and neonatal growth outcomes. Contrary to previous findings, maternal depression reported in pregnancy and at birth did not account for these neonatal outcomes.
Although the present sample size was relatively small, the magnitude of the birth and 1 month effects of in utero exposure was in the moderate–large range by conventional criteria [26]. Findings of lower birthweight, length and earlier gestation age at delivery for exposed neonates were statistically significant and in line with previous studies. Further analysis of the odds of falling into a clinical range suggested that exposed neonates were eightfold more likely to be of clinically significant lowered weight (<2500 g) and 4.5-fold more likely to be premature, although this last effect was not statistically significant. The magnitude of such effects is certainly of clinical significance in routine clinical practice.
One of the unique features of the study design was to facilitate examination of the developmental trajectory of exposed infants. While exposed infants were lighter and smaller, there were also sustained lower growth rates in weight during a 1 month period. For head circumference and length, exposed neonates failed to make any significant recovery after 1 month.
Against predictions, and in contrast to other similar studies, the current study found no association between self-reported depression during pregnancy and the birth outcomes under investigation. Therefore this is an unlikely alternate explanation for these birth outcome findings.
The limitations of the present study include the small sample size, particularly of women on individual antidepressants, which precluded comparisons between individual antidepressants in their effects on neonatal outcomes. In addition, other variables that may also influence fetal and infant growth and development such as maternal and paternal weight and height, pregnancy weight gain and nutritional intake, infant feeding (breast or formula) at 1 month were not recorded. The advantage over many larger studies is that the exact doses and timing of antidepressant exposure have been recorded, whereas some studies such as that by Kallen could not specify timing of exposure in 40% of cases [18]. Given that later pregnancy exposure is of most importance in examining the relationship with birth outcomes and gestational age at delivery, this is an important advantage. In addition, maternal depression was assessed in pregnancy, after delivery and at 1 month, allowing the examination of this as an important potential confounding variable.
Conclusion
The present study has shown that antidepressant exposure in utero may impact on fetal growth, resulting in lower birthweight, smaller birth length and smaller head circumference than in non-exposed neonates. In addition neonates exposed in utero were eightfold more likely to be of clinically significant low birthweight as compared to controls. The present study has shown that this difference does not correct itself by 1 month postpartum, which indicates the importance of follow up of infants exposed to antidepressants in pregnancy to monitor growth beyond the neonatal period. Further studies are required to examine if individual antidepressants carry more or less risk of poorer birth outcomes, and also if these findings at 1 month are sustained into late infancy and toddler stages and associated with other neurodevelopmental child outcomes.
Footnotes
Acknowledgements
The women and children who participated in this study, Helena Sandahl, Neuroscience Research Grant, Pat and Toni Kinsman Postnatal Depression Scholarship, Pfizer, Wyeth and Lundbeck.
