Abstract
Although cognitive deficits are recognized as a core feature in schizophrenia, their evolution over the course of the illness is still debated. Longitudinal studies of cognition in patients after a first episode of psychosis (FEP) provide extremely useful information, in that they include an adequate and realistic baseline measure of cognitive performance, while at the same time minimizing the effect of confounding variables associated with chronicity. The aim of this systematic review was to summarize findings of studies assessing the longitudinal course of neuropsychological deficits in patients with FEP for durations of at least one year. Overall, the neuropsychological deficits that are present following a first episode of psychosis appeared to remain stable over time for periods of up to ten years, the only possible exception being verbal memory deficits, where there is some evidence of further deterioration over the long term. However, further studies are needed to confirm this conclusion, especially in the (somewhat inconsistently defined) domain of executive function. Improvements in psychopathology appear to positively influence the course of cognitive deficits, although the effects of antipsychotic medication are not as clear.
Cognitive deficits are a core feature of schizophrenia, and patients with schizophrenia demonstrate deficits in most domains of cognitive functioning compared to healthy control subjects, suggesting a generalized cognitive impairment [1–3]. Cognitive deficits exist early in the course of the illness [3], with patient performance ranging from one to two standard deviations below that of healthy individuals [4–6]. There is evidence that these cognitive deficits are present already before illness onset [7–10], probably even since childhood [11–13] or early adolescence [14].
Cognitive skills have been shown to be associated with the functional status of patients with schizophrenia, and they have been reported to have a greater influence on functional outcomes than positive or negative symptoms [15,16]. Although this latter point has been debated, and indeed might not apply to stabilized, community dwelling patients [17–21], it is generally accepted that baseline cognitive assessment is related to subsequent community functioning and skills acquisition [22–25]. Even more interestingly, changes in neurocognitive ability over time might be related to change in social outcome; in a longitudinal study that addressed the association between neuropsychological and functional status in patients with schizophrenia, change in functional outcome was associated with change in overall cognitive performance, memory, digit symbol substitution, and executive functioning [26].
The course of cognitive deficits and the accompanying psychosocial dysfunction in schizophrenia across the lifespan is still unclear, with some research groups suggesting that cognitive dysfunction deteriorates further, whereas others report stability or even an improvement in some cognitive functions [27,28]. Literature reviews of longitudinal studies investigating the trajectory of neurocognitive deficits across the lifespan in schizophrenia have concluded that neurocognitive deficits evident upon illness onset appear to be stable, at least in community dwelling outpatients [27,29]. In a recent meta-analysis of longitudinal studies of cognition in schizophrenia [28], patients with schizophrenia showed a significant improvement in most cognitive tasks. However, for some cognitive variables, for which an estimation of test-retest effects was available in control subjects (logical memory, Wisconsin card sorting test, trail making test-A, verbal fluency letters), greater improvement was found in controls compared to patients with schizophrenia. Only on the Stroop colour-word test did patients show a greater improvement than controls. The above study highlights the importance of contrasting the time course of cognitive deficits in patients against that of control subjects. Performance on neurocognitive measures is known to be affected by several illness-independent factors (such as familiarity with the procedures, practice effects, age, etc.); hence the reported stability or even improvement in patients may still represent a deficit when compared to the commensurate changes in healthy individuals. Conversely, without the presence of a control group, it cannot be concluded whether a decline in patient performance reflects a neurodegenerative process rather than normal age-related changes in cognition.
The delineation of the longitudinal course of cognitive deficits in patients with schizophrenia may have important implications for theories of pathophysiology as well as for the treatment and outcome of the illness. For one, it could settle the debate between those that view schizophrenia as a neurodegenerative disorder, and those that conceptualize it as a developmental neural insult that remains stable over time [27,28]. Moreover, cognitive deficits that are found to remain stable over time or, if not, their course is predictably influenced by identifiable non-genetic factors, might serve as potential endophenotypes [28]. Finally, the identification of those cognitive deficits that are fixed and those that are most sensitive to change would facilitate the evaluation of interventions, pharmacological treatments or cognitive rehabilitation programmes, aiming at the improvement of cognitive functions. If schizophrenia is characterized by a gradual worsening of some cognitive domains, interventions that stabilize these cognitive deficits would be judged as efficacious whereas, if the cognitive status is static over time, an intervention would need to lead to measurable improvements in cognitive functions in order to be considered as successful [27,28].
One strategy of special relevance when it comes to evaluating the course of cognition over the course of schizophrenia is the longitudinal assessment of first episode (FEP) patients. Such studies provide an adequate and realistic baseline measure of cognitive performance; moreover, they minimize the effect of confounding variables associated with chronicity (such as institutionalization, as well as long-term treatment effects and disease processes). The aim of the current review is to summarize evidence from longitudinal studies investigating the long-term course of cognitive deficits in patients with FEP.
Materials and methods
A PubMed search was conducted twice (last search: 15 January 2010) using the syntax (‘first episode’) AND (‘schizophrenia’ OR ‘psychosis’) AND (‘cognit∗’ OR ‘neuropsycholog∗’) AND (‘longitudinal’ OR ‘follow up’ OR ‘course’). The search was limited to studies written in English and published before January 2010. Additional citations were obtained by hand-screening the reference list of all retrieved studies. Studies published solely in abstract form (such as published abstracts from conference proceedings) were excluded. The authors independently examined all the literature information collected and rated the papers regarding the suitability for inclusion; any disagreements were resolved by discussion. Studies were included in the review only if (i) they were original studies of patients with first episode or recent onset psychosis and (ii) the follow up period was at least one year (the latter criterion was used in order to exclude changes in cognition as a result of short-term fluctuations in symptomatology during the period of symptomatic stabilization; moreover, it has been argued [27] that it would be highly unlikely to detect gradually progressive cognitive decline during shorter test-retest intervals). Papers with mixed first episode and chronic patient samples were selected for inclusion only if data for first episode patients were presented separately (see Figure 1 for a detailed description of the selection process). In the case of studies conducted by the same research group with overlapping patient samples, only the study with the largest number of patients was included in the review, unless the studies in question reported data from different time points during the follow up. Thus, one study by Malla et al. [30], two studies by Schuepbach et al. [31,32], one study by Gur et al. [33], one study by Nopoulos et al. [34], one study by Good et al. [35], and three studies by DeLisi et al. [36] and Hoff et al. [37,38] were excluded due to patient sample overlap with studies by Townsend et al. [39], Hill et al. [40], Censits et al. [41], Gold et al. [42], Kopala et al. [43], and Hoff et al. [44], respectively. For the same reason, one study by Addington and Addington [45], two studies by Addington et al. [46,47], and one by Pencer et al. [48] were excluded due to overlap with a study by Addington et al. [49].
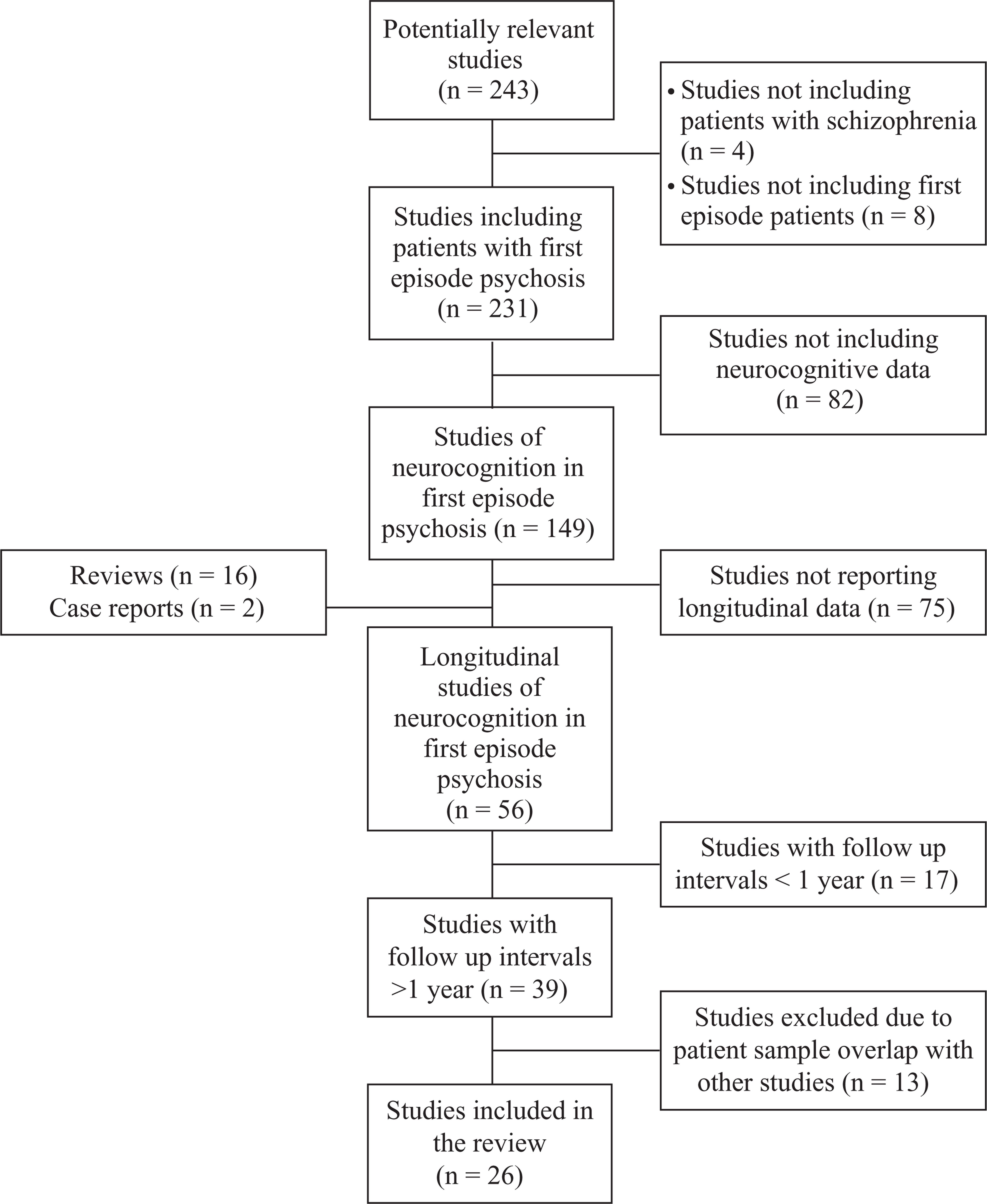
Selection process for the studies included in the review.
Results
In order to facilitate the interpretability of results, retrieved studies were split into two groups, according to the presence or absence of a healthy control group. Studies that did not include a control group are presented in Table 1, whereas those that did are presented in Table 2 and Table 3.
Studies not including a healthy control group
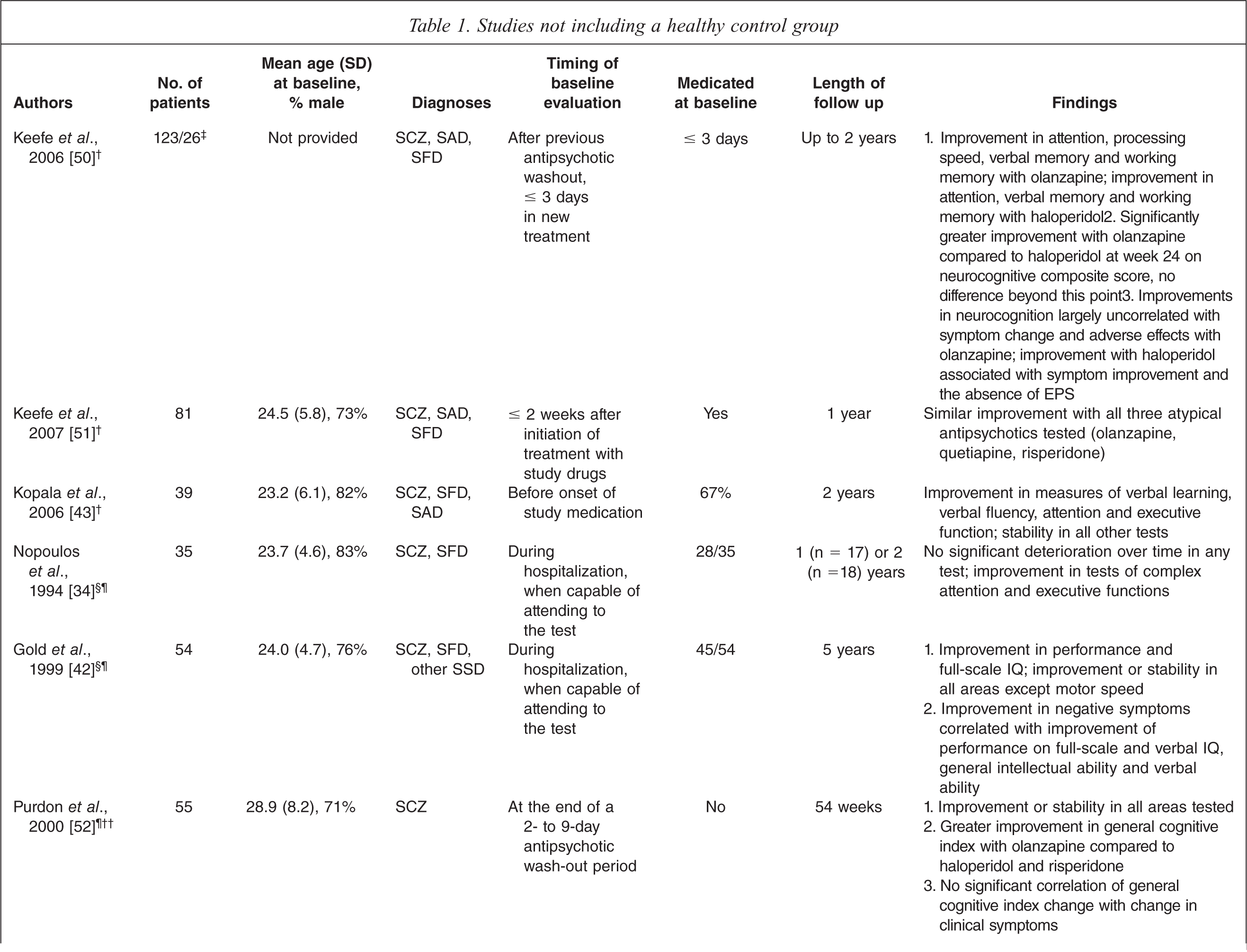
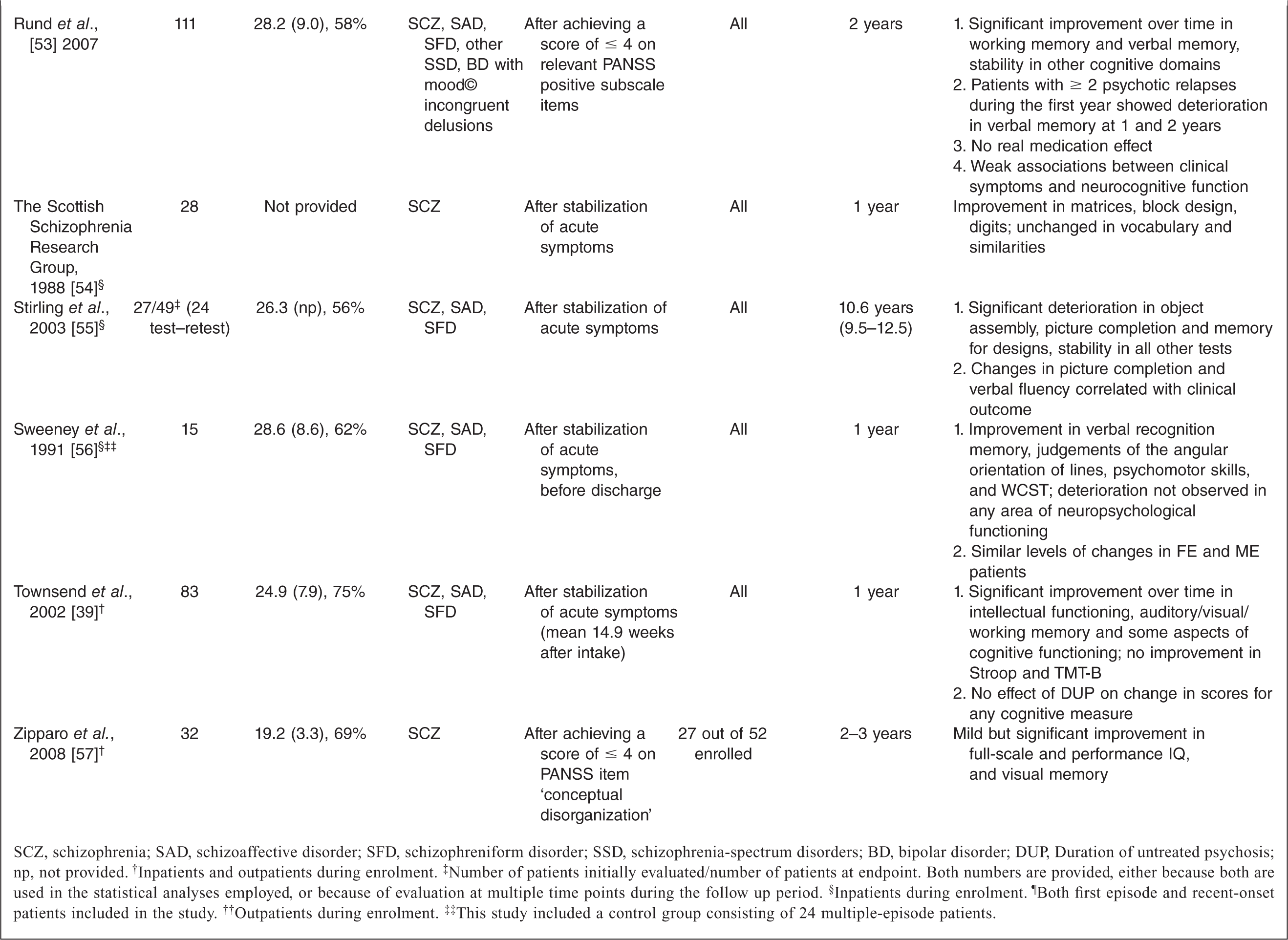
SCZ, schizophrenia; SAD, schizoaffective disorder; SFD, schizophreniform disorder; SSD, schizophrenia-spectrum disorders; BD, bipolar disorder; DUP, Duration of untreated psychosis; np, not provided.† Inpatients and outpatients during enrolment.‡ Number of patients initially evaluated/number of patients at endpoint. Both numbers are provided, either because both are used in the statistical analyses employed, or because of evaluation at multiple time points during the follow up period.§ Inpatients during enrolment.¶ Both first episode and recent-onset patients included in the study.†† Outpatients during enrolment.‡‡ This study included a control group consisting of 24 multiple-episode patients.
Studies including a healthy control group
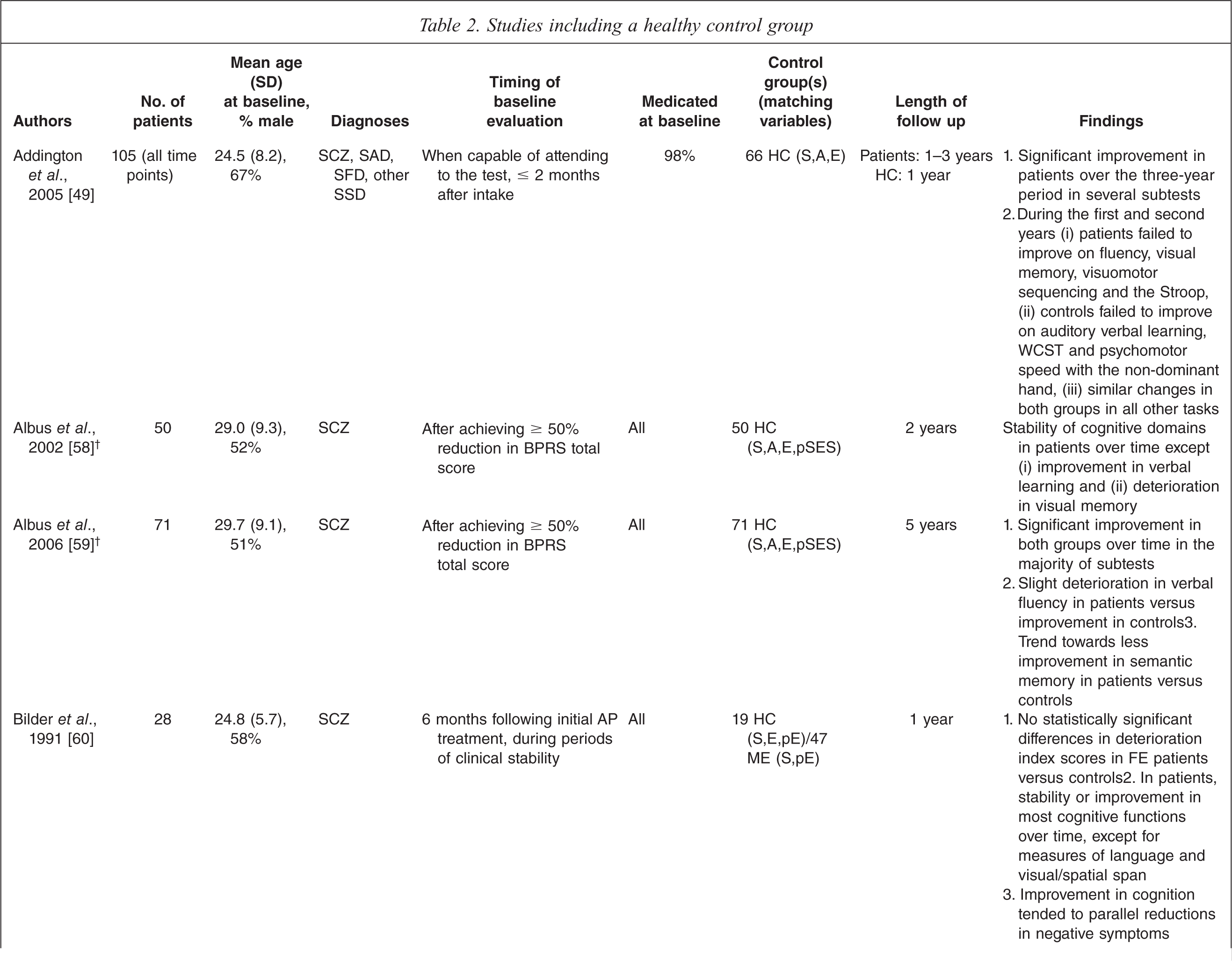
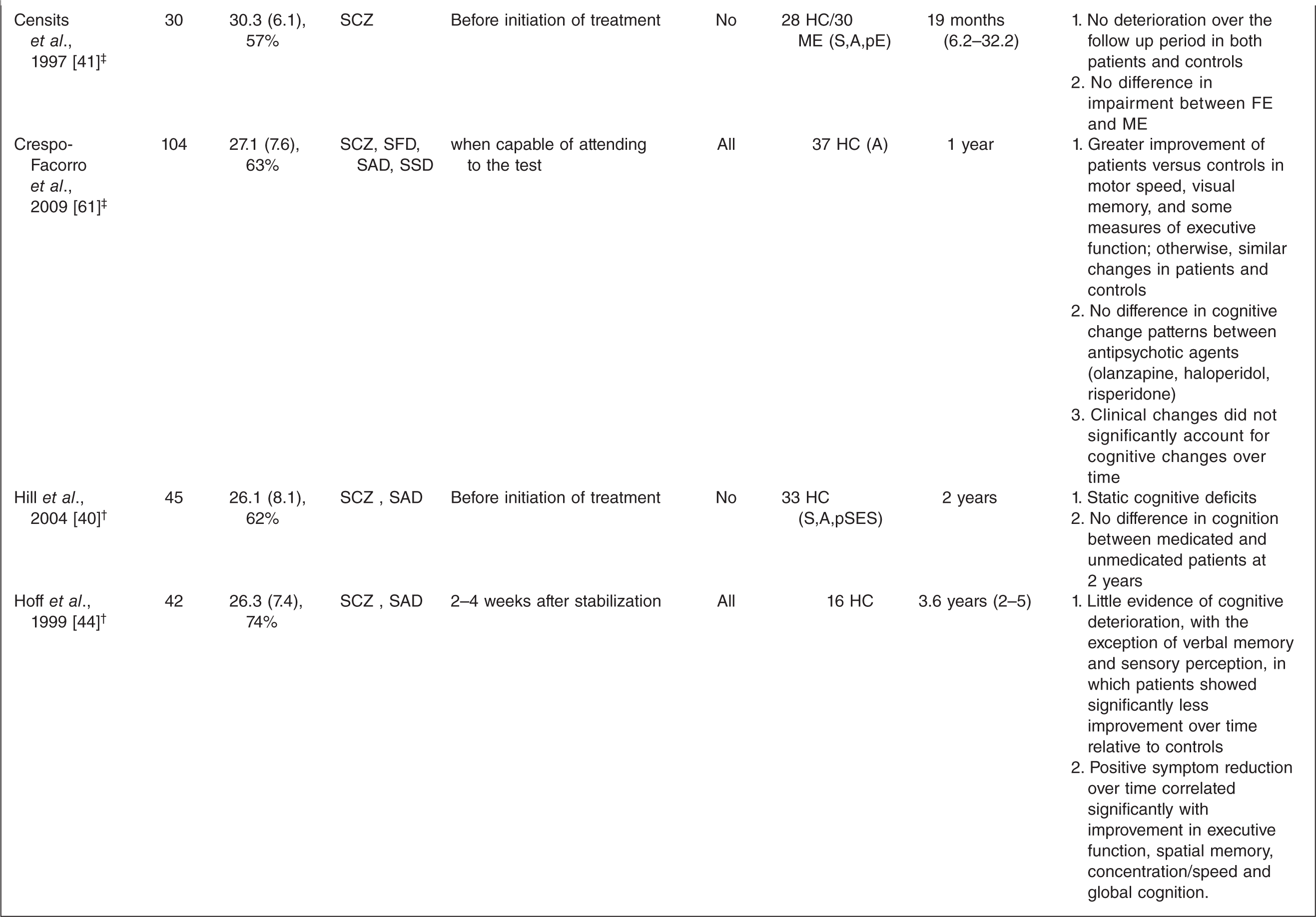
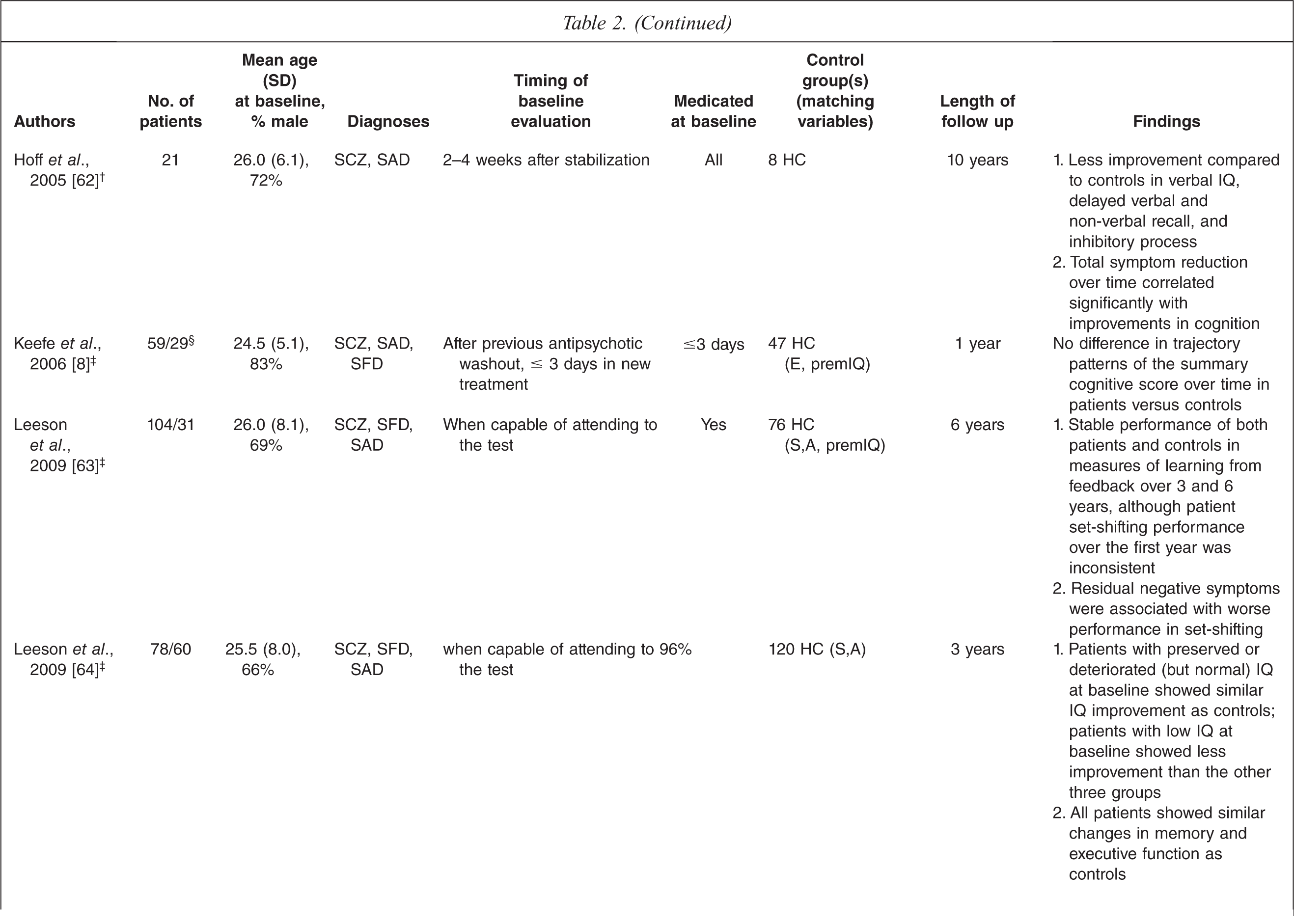

SCZ, schizophrenia; SAD, schizoaffective disorder; SFD, schizophreniform disorder; SSD, schizophrenia-spectrum disorders; AD, affective disorders; BD, bipolar disorder; FE, first episode patients; HC, healthy controls; ME, multiple episode patients; S, gender; A, age; E, education; premIQ, premorbid IQ; pE, parental education; pSES, parental socio-economic status.† Inpatients during enrolment.‡ Inpatients and outpatients during enrolment.§ Number of patients initially evaluated/number of patients at endpoint. Both numbers are provided, either because both are used in the statistical analyses employed, or because of evaluation at multiple time points during the follow up period.
Direction of patient performance over time compared to that of control subjectsa,b
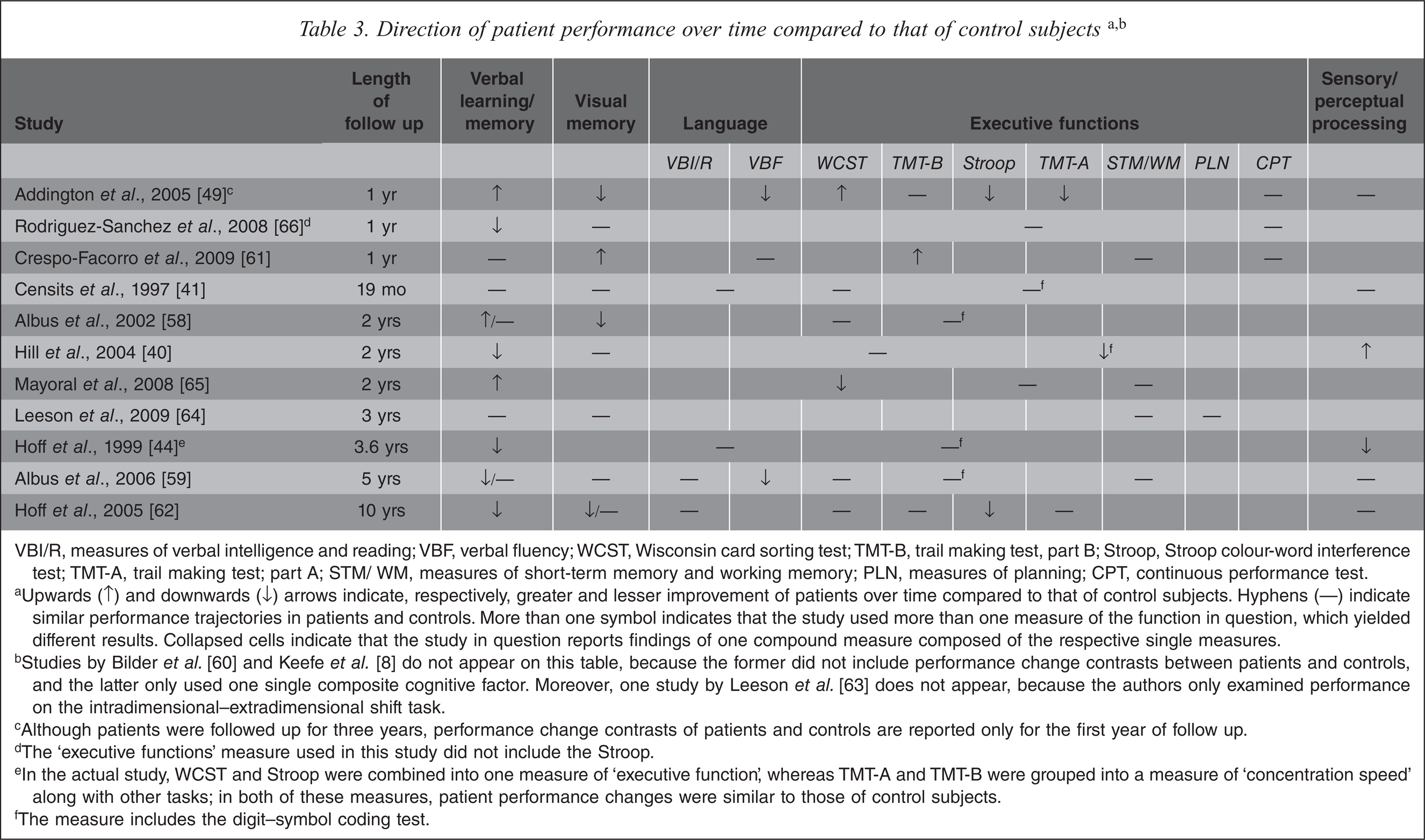
VBI/R, measures of verbal intelligence and reading; VBF, verbal fluency; WCST, Wisconsin card sorting test; TMT-B, trail making test, part B; Stroop, Stroop colour-word interference test; TMT-A, trail making test; part A; STM/ WM, measures of short-term memory and working memory; PLN, measures of planning; CPT, continuous performance test.
aUpwards (↑) and downwards (↓) arrows indicate, respectively, greater and lesser improvement of patients over time compared to that of control subjects. Hyphens (—) indicate similar performance trajectories in patients and controls. More than one symbol indicates that the study used more than one measure of the function in question, which yielded different results. Collapsed cells indicate that the study in question reports findings of one compound measure composed of the respective single measures.
bStudies by Bilder et al. [60] and Keefe et al. [8] do not appear on this table, because the former did not include performance change contrasts between patients and controls, and the latter only used one single composite cognitive factor. Moreover, one study by Leeson et al. [63] does not appear, because the authors only examined performance on the intradimensional–extradimensional shift task.
cAlthough patients were followed up for three years, performance change contrasts of patients and controls are reported only for the first year of follow up.
dThe ‘executive functions’ measure used in this study did not include the Stroop.
eIn the actual study, WCST and Stroop were combined into one measure of ‘executive function’, whereas TMT-A and TMT-B were grouped into a measure of ‘concentration speed’ along with other tasks; in both of these measures, patient performance changes were similar to those of control subjects.
fThe measure includes the digit–symbol coding test.
Studies not including a control group
Virtually all studies that do not include a control group report stability or improvement in all neuropsychological areas tested for intervals of 1 to 5 years. The only exception is a study by Gold et al. [42], whose patients worsened over time only in motor speed (finger tapping). As regards the evolution of deficits over longer periods of time, the only study that investigated this question [55] found deterioration in some visuospatial tasks (object assembly, picture completion and design memory) in patients after a mean follow up duration of approximately ten years.
Studies including a control group
In studies that include a healthy control group, cognitive changes in patients for the most part parallel those observed in healthy control subjects over time periods of one to ten years. However, in some studies patients failed to improve as much as controls, or even deteriorated, in some of the tasks used to assess performance. Results grouped by cognitive domain are listed below.
The majority of studies that included assessment of executive functions and attention [40,41,44,59, 61–64,66] found no differences in the trajectory patterns of first episode patients compared to controls, or, in one case [61], improvement in patients compared to controls. Only three studies deviated from this pattern: Mayoral et al. [65] found that, in contrast to matched controls, adolescents with a first episode of psychosis showed no changes in a composite measure of executive function over time. Addington et al. [49] also reported diminished improvement in patients compared to controls only in some (trail making test part A, Stroop colour-word test), but not other (trail making test part B, Wisconsin card sorting test) measures of executive function. Similarly, Hoff et al. [62] only found differences between patients and controls on the inhibition trial of the Stroop colour-word test.
Regarding language abilities, four studies indicated a similar pattern of evolution in patients compared to controls [41,44,61,62]. On the other hand, Addington et al. [49] reported a failure of their patient sample to improve in letter fluency as much as controls, and Albus et al. [59] found a slight deterioration of patients in semantic fluency, in contrast to an improvement exhibited by control subjects on the same task.
Findings are extremely variable in the case of verbal memory and learning, with some studies [40,44,59,62,66] indicating less improvement or even deterioration in patients compared to controls on some measures of verbal memory, while others report no difference [41,58,59,61,62,64] or even greater improvement in patients compared to healthy control subjects on at least some measures of verbal memory [49,58,65].
In the domain of visuo-spatial memory, the majority of studies did not find any differences between patients and controls in the time pattern of changes [40,41,44,59,66], and in one study [61] patients improved more than control subjects. However, two studies [49,58] reported less improvement or even deterioration in patients compared to controls; in another study [62], patients improved less than controls on one measure of visual memory (visual reproduction – delayed), but similarly to controls on another measure (Benton visual retention test).
The majority of studies that have investigated sensory and perceptual ability reported no differences in the pattern of performance changes between patients and controls [41,49,62], or even greater improvement in patients versus controls [40]; Only one study reported a decreased gain in patients compared to controls over time [44].
Most studies assessing the effect of symptomatic improvement on cognition longitudinally reported significant associations of neurocognitive changes with positive [44,50], negative [50,60], or overall [62,65] symptom reduction. Along a similar line, Stirling et al. [55] found that neurocognitive changes correlated with clinical outcome (in terms of overall functionality and need for service contact). Rund et al. [53] also reported significant associations between verbal learning and working memory with number of relapses during the first (but not the second) year of follow up. However, the postulated link between symptomatic and clinical improvement is not unequivocal, as some studies failed to find any significant associations [52,61].
Medication effects
Studies that assessed the longitudinal course of neurocognition in medicated patients [43,50,51,61] reported significant improvements in neuropsychological performance over time. However, none of these studies included a (non-medicated, or placebo-treated) comparison group. On the other hand, there do not appear to be any differences in cognitive gain between typical and atypical agents [50,61], between various atypical agents [51,61], or even between medicated and unmedicated patients [40,53]; the only exception in this pattern is a study by Purdon et al. [52], who found a relative advantage for olanzapine compared to risperidone and haloperidol.
Regarding the effect of extrapyramidal adverse effects on cognitive change, the overall association appears to be weak or non-existent [52,61]. However, the link is stronger in the case of haloperidol, where improvement of cognitive performance was reported to be significantly associated with the absence of extrapyramidal symptoms in two studies [50,61].
Discussion
The aim of the present review was to summarize existing evidence regarding the course of neuropsychological deficits following a first episode of psychosis.
Starting with the studies that did not include a healthy control group, these generally reported stability or even improvement of cognitive deficits in patients over time. The only notable exception is a study by Stirling et al. [55], who found significant deterioration in tasks assessing visuospatial abilities in patients after a mean follow up period of ten years. However, as pointed out in the Introduction, studies that do not include a healthy control group, although providing important information regarding the absolute time course of cognitive abilities in schizophrenia, tell us nothing about their evolution relative to those of healthy individuals, and thus do not take into account other factors that might affect performance, such as familiarity with the procedures and practice effects, or age-related cognitive changes. This is particularly evident in the above-mentioned study by Stirling et al. [55], who included patients of up to 50 years of age at baseline; thus, due to the long follow up period and the absence of an age-appropriate control group, the possibility cannot be ruled out that their findings might, at least in part, be attributable to age-related cognitive decline in some of their patients.
On the other hand, studies investigating the course of neuropsychological deficits in patients with FEP relative to a healthy control group differ along several dimensions, thus limiting the interpretability of results: For example, there are important differences among studies in patient sampling, with some studies [41,58–60] assessing only patients with schizophrenia, while others include patients with schizo-affective, schizophreniform and other psychotic disorders [8,40,44,49,61–63,66], or even patients with affective disorders with mood-incogruent delusions [65]. Other important methodological differences include (i) patient recruitment strategies (inpatients versus inpatients and outpatients, see Table 2) and the timing of baseline neuropsychological evaluation in patients (before initiation of treatment versus variable time into stabilization of symptoms, see Table 2), (ii) duration of follow up (1 to 10 years, see Table 2), as well as (iii) differences in the specific neuropsychological tests implemented, their categorization, and their use in statistical analyses: for example, Mayoral et al. [65] include measures of verbal fluency in their composite measure of executive function, whereas most other studies assess verbal fluency separately [e.g. 49,59], or as a part of a composite measure of language ability [e.g. 41,44,62]. Another important source of variation in findings may result from differences in recruitment strategies for healthy control subjects. Finally, an additional confounding factor is attrition rate, which ranges widely among studies, from 70–90% in some studies [40,58,59, 65,66] to lower than 50% in others [8,49,62], due to patients being lost to follow up or receiving a different diagnosis. Since attrition tends to increase with time [8,44,49,59,62], studies with longer follow up durations would be more vulnerable to a loss of statistical power ensuing from sample size shrinkage. The matter is further complicated by the fact that attrition rates are not always similar in patients and control subjects [40,44]. A final limitation that should be mentioned is the well-known fact that group studies such as those included in this study may mask individual differences or potential neuropsychological subgroups that are related to sub-syndromes of psychosis [67]. The importance of this point is highlighted by a study by Jashan et al. [68], not included in this review because of the short time of follow up (6 months): In that study there were no differences at the group level between FEP patients and controls in the trajectory of most cognitive functions; however, at the individual level and using a regression-based approach, for each of the domains of executive function, processing speed, working memory and IQ, there was a significant proportion (approximately 20%) of patients who showed less-than-expected improvement.
Despite the above limitations, there are some clearly discernible trends in the findings: First of all, despite some test-retest declines on isolated cognitive measures that differ according to the specific study (see Results), all studies included in this review conclude that the degree of overall cognitive change over time tends to be similar in patients and controls. In this regard, the most stable cognitive domains appear to be executive function, language, and sensory/perceptual abilities, where the vast majority of studies do not find any differences between patients and controls in the time pattern of changes. It should be noted here that this result is best interpreted with caution in the case of executive function, since the concept comprises a variety of separable cognitive skills that depend on different anatomical substrates, such as inhibition, attention, working memory etc. Although an overview of findings presented in Table 3 suggests that, in most cases, these functions or composite measures comprising their combination evolve similarly in patients and controls, further research is clearly warranted to confirm this impression. This is particularly evident in the case of the Stroop colour-word test (Table 3), for which less improvement is reported in patients compared to controls in two studies [49,62]; since these were the only two studies that assessed performance on the Stroop separately from other executive functions, the significance of this finding is doubtful, as it may have been due the lack of correction for multiple comparisons, a possibility that necessitates further research. Similarly, in two [49,59] out of three studies [49,59,61] that applied separate verbal fluency assessments, patients failed to improve as much as controls. Although composite measures of language and measures of verbal intelligence used in other studies do not exhibit different trajectories over time in patients and control subjects, further research into the longitudinal course of verbal fluency would be valuable, as the latter does not only depend on the organization of semantic memory, but also on strategic retrieval and data manipulation processes (i.e. ‘executive’ functions) [69].
In the case of visuo-spatial memory there exists greater variability but, here too, it appears that the time course of performance is generally similar in patients and control subjects. Only a few studies stand out due to different findings; in two of those the failure of patients to improve as much as controls is not unequivocal, either because it applies only to some and not all of the measures used to assess visuo-spatial memory [62], or because it is not found at a later point during patient follow up [58,59]; and in the third [49], no direct comparison is made between patients and controls along the dimension of performance change.
Findings are much more difficult to reconcile in the case of verbal memory and learning, where much larger variability is observed, covering the whole range from the one extreme of greater deterioration to the other extreme of greater improvement in patients compared to controls. Interestingly though, this discrepancy in findings might be attributable to differences in length of follow up: Studies that follow up subjects for relatively short intervals (i.e. up to 3 years) usually report similar changes over time in patients and controls [41,58,61,64], or greater improvement in patients [49,58,65]. Only two studies [40,66] deviate from this pattern, and in one of these [40] the difference between patients and controls is only trend-wise significant and largely due to differences during the first 6 weeks of assessment, i.e. at a time when the acutely ill inpatients comprising the patient sample of the study may not have achieved clinical stability. In contrast, studies with longer follow up durations find less improvement, or deterioration, in patients compared to controls in all [44,62], or some [59] measures of verbal memory.
Thus, with the possible exception of verbal memory, the evolution of neuropsychological deficits in patients after a first episode of psychosis does not appear to differ from the cognitive changes observed in healthy individuals over the same periods of time. This conclusion, although in concordance both with the conclusions of isolated studies included in this review and with the prevalent view of schizophrenia as a ‘static encephalopathy’ (for a recent review, see Kurtz [27]), is in stark contrast with the findings of progressive brain tissue reductions in structural neuroimaging studies of schizophrenia [e.g. 33,36, 57,70–72]. Indeed, two studies that assessed the longitudinal course of both structural and cognitive changes in patients with a first episode of schizophrenia [33,57] both found that, although the two variables significantly correlated with each other, neuropsychological performance remained stable over mean follow up periods of 2 to 3 years, while there were significant decrements in brain tissue over the same period of time. A possible explanation of this discrepancy might be that the tasks used to assess cognitive function are not sensitive enough to detect changes brought about by small reductions in brain volume (see Zipparo et al. [57], for a similar argument). Alternatively, it has been suggested that patients with schizophrenia may develop specific compensatory strategies to perform cognitive tasks [73,74]; it is conceivable that this might mask the pathological decline of cognitive resources in patients during the progression of the illness. On the other hand, since patients with a first episode of schizophrenia usually already score one to two standard deviations below controls in most neuropsychological areas investigated [8,40, 41,58,59,66], it might simply be that the emergence of further neuropsychological deficits with ongoing brain tissue reductions is prevented by a floor effect, i.e. there is not much room for further cognitive deterioration beyond a certain point. This is consistent with the view by Pantelis et al. [72,75], who have argued that schizophrenia is characterized by a ‘developmental arrest’ (i.e. a failure to develop appropriately), rather than deterioration, of certain cognitive functions, due to an early neurodevelopmental insult. A final explanation of this discrepancy of findings between neuropsychological and structural neuroimaging studies relates to the disconnectivity hypothesis [76], according to which cognitive deficits in schizophrenia are not related to localized brain lesions per se, but rather to disturbed connectivity between brain structures. In any case, it is interesting to point out that the most consistent sites involved in progressive brain tissue loss are the prefrontal and medial temporal lobe [57,70–72], on the integrity of which verbal memory is known to depend heavily [77,78]; the intriguing possibility emerges, then, that the trend observed in this review, i.e. for verbal memory to deteriorate more in patients compared to controls during long follow up periods, reflects the ongoing structural changes in the above areas of the brain. If this is true, more focused research is warranted in the domain of executive functions as well, since it has been shown that some measures of executive function (such as the Stroop and verbal fluency tasks) [79,80] are more dependent on the coordination of frontal and more posterior brain areas than others, in which case it would not be meaningful to simply investigate the time course of one single composite measure of ‘executive function’, as in most of the studies included in this review (see Table 3).
Regarding the effects of psychopathology on cognition over time, symptomatic improvement is significantly correlated with improvement in neurocognitive performance in the majority of studies [44,50,60,62]. Although other studies failed to find significant correlations [52,61], the assumption of an association between cognitive and clinical improvement receives additional indirect support from other sources: In one study [65], changes in neuropsychological performance over time in patients with schizophrenia disappeared after covarying with changes in symptomatology, and two other studies found significant correlations between neurocognitive changes and measures of clinical outcome [53,55]. On the other hand, the effects of antipsychotic medication on the course of neuropsychological deficits are not as clear: although significant cognitive improvements are reported with antipsychotic treatment [43,50,51,61], reported differences between typical and atypical agents are at best equivocal [50,52,61], and studies that compare medicated to unmedicated patients do not report any advantage for the former in terms of cognitive improvement [40,53]. Thus, it is possible that the reported beneficial effects of medication on cognitive abilities are simply a result of reduced symptomatology. Further placebo-controlled studies are warranted in order to confirm the time window during which medication effects, if any, are present in first episode psychosis, to assess any differences between various antipsychotic agents, and to explore whether the cognitive changes observed with antipsychotic agents are simply a function of medication-related symptomatic improvement and/or side effects, or whether these agents act in other ways as well, e.g. through their neuroprotective capacities (see, for example, Lieberman et al. [81]).
Conclusion
Evidence available so far suggests that neuropsychological deficits that are present following a first episode of psychosis can be expected to remain stable over time. A possible exception in this pattern might be the domain of verbal memory, where there is some evidence of further deterioration over the long term; since this specific cognitive ability is closely dependent on cortical structures known to show progressive volume reductions during the course of the illness. Further studies are warranted to confirm this observation and/or to identify any subgroups of patients that are more likely to decline over time. Also, as most studies have focused on compound concepts of ‘executive function’, more detailed research is also needed regarding the time course of specific executive functions. This is especially important, since some of these functions (working memory, attention) have been proposed as endophenotypes for use in genetic studies of schizophrenia [82]. Further research is also needed in order to establish a time pattern for other, more complex cognitive abilities that have been associated with social outcomes in schizophrenia, such as facial affect recognition or theory of mind [47,83]. Improvements in psychopathology appear to positively influence the course of cognitive deficits; hence, sufficient and comprehensive treatment of these symptoms is of utmost importance, although further research is needed in order to investigate specific medication effects.
Footnotes
Acknowledgements
