Abstract
A recent meta-analysis has concluded that there is now sufficient evidence that cannabis use increases the risk of onset of non-affective psychotic disorders [1]. In addition, the use of cannabis during adolescence may lead to depression [2,3], have a lasting negative impact on neurocognitive functioning [4] and brain developmen [5,6] and may hinder high school completion [7].
Schizophrenia is hypothesized to be a progressive neurodevelopmental disorder [8] and individuals who later develop schizophrenia as well as subjects at ultra-high risk (UHR) for psychosis may be particularly vulnerable to the effects of cannabis during adolescence. To gain more insight into the effects of cannabis use in the prodromal phase of schizophrenia and in the UHR state it would be valuable to examine which subjective effects individuals themselves experience. Assessing subjective effects in UHR subjects may be clinically relevant by providing insight in the relation between cannabis use and clinical symptoms.
Several studies have investigated the subjective effects of cannabis in patients with schizophrenia or related disorder after illness onset [9–13]. One study examined the subjective effects of general substance abuse in schizophrenia [14]. In these studies a significant number of patients reported that cannabis use increased paranoid thoughts [11,13,14] and hallucinations [11,13], while some patients reported a decrease in hallucinations [14]. Many patients reported feeling less depressed and/or anxious after cannabis use [9,10,13,14], while one study also reported an increase in feelings of depression and/or anxiety [11]; in another study both effects were reported equally [12]. Patients in these studies also reported other positive effects, for example feeling happy and relaxed [11,14], cheerfulness [12], experiencing cognitive enhancement [10], becoming more sociable [9] and energetic [13]. One study investigated more objectively the acute effects of cannabis in healthy controls and schizophrenia patients [15]. In both groups i.v. Δ-9-tetrahydrocannabinol (Δ-9-THC; the main psychoactive ingredient of cannabis) increased learning and recall deficits, and positive, negative, and general schizophrenia symptoms. Schizophrenia patients seemed to be more sensitive to induction of psychotic effects by Δ-9-THC.
We are aware of only one study to compare the subjective effects of cannabis in schizophrenia patients to controls [10]. Patients reported feeling less relaxed after cannabis use, and increased psychotic symptoms. No differences were found regarding anxiety or depression. To our knowledge no study has investigated the subjective effects of cannabis in the prodromal phase of schizophrenia retrospectively, or in UHR subjects.
In summary, individuals who later develop schizophrenia and UHR subjects may be particularly vulnerable to the effects of cannabis. Previous studies in adolescents without schizophrenia have found that cannabis may cause psychosis [1], depression [2,3] and impaired neurocognition [4]. Studies in cannabis-using schizophrenia patients, however, have shown that while some patients experience an increase in psychotic and negative symptoms, anxiety, depression, and cognitive impairments, other patients experience positive effects on psychotic and negative symptoms, mood, cognition, and sociability. These positive effects of cannabis use may serve to alleviate illness related symptoms. We hypothesized that (i) some individuals in the prodromal phase of schizophrenia and some UHR subjects experience negative effects from cannabis use (e.g. psychotic symptoms, negative symptoms, mood disturbances, impaired cognition), while others experience positive effects (e.g. fewer negative symptoms, improved mood, better cognitive functioning, increased sociability); and (ii) individuals in the prodromal phase of schizophrenia and UHR subjects experience negative effects, in particular psychotic effects, more often than cannabis-using individuals without major psychiatric illness. We investigated these two hypotheses by examining the subjective effects of cannabis use in the prodromal phase in recent-onset schizo phrenia patients retrospectively and in UHR subjects, in comparison to cannabis-using controls from the general population.
Methods
Participants
Inclusion criteria for this study were: (i) being between 18 and 28 years of age; and (ii) having used cannabis at least 10 times. Patients admitted to the Adolescent Clinic of the Psychiatric Centre of the University of Amsterdam with a recent onset of schizophrenia, schizophreniform disorder, or schizoaffective disorder according to DSM-IV criteria [16], were included. Only male subjects were included, due to low admission rates of female patients in the clinic. Clinical discharge diagnoses were made with the use of all available diagnostic information (clinical interviews of patients and key informants, and all medical records) by a clinical psychiatrist and the principal investigator (B.D.P.; Longitudinal Expert Assessment of Diagnosis procedure [17]).
The UHR patients were recruited from a naturalistic, longitudinal study programme related to the Adolescent Clinic. Inclusion criteria for these patients were defined according to international criteria [18]: attenuated psychotic symptoms (e.g. odd beliefs, paranoid ideation) or brief psychotic moments with spontaneous remission in less than 1 week; and/or a decline in functioning in the past year (30% reduction in Global Assessment of Functioning scale) plus a genetic risk (first-degree relative with schizophrenia-like disorder or a schizotypal personality disorder); or two ‘basic symptoms’ (cognitive, perceptual, emotional and social disturbances [19]).
The control group was composed of cannabis users recruited from a coffee shop (a shop in which cannabis can be bought and smoked legally) in Amsterdam. In addition, cannabis-using university students from the Amsterdam area were recruited according to a ‘snowball sampling’ method [20].
The exclusion criterion for the UHR subjects was a current or past psychotic disorder. The exclusion criterion for controls was the presence of delusions or halluci nations while not being intoxicated with a substance.
Measurements in recent-onset patients
Sociodemographics, onset of symptoms and substance use
The Inventory for the Retrospective Assessment of the Onset of Schizophrenia [21] was used to assess educational level prior to the first psychotic episode, date of onset of the first psychotic symptom(s) and of the first psychotic episode. The first psychotic episode was defined as the first time that the patient experienced psychotic symptoms for at least 1 week, during most part of the day. In addition, an estimate of the lifetime number of cannabis cigarettes smoked and lifetime frequency of hard drug use was made and lifetime diagnoses of alcohol abuse were determined.
Subjective effects of cannabis
A list of acute effects of cannabis was compiled from previous studies on subjective effects of cannabis [11–13] or substances in general [14] in schizophrenia patients. The most commonly reported effects in the domains of psychotic symptoms, negative symptoms, mood, cognition and sociability were selected. A list of chronic effects of cannabis relating to prodromal signs of schizophrenia was composed from the literature on long-term effects of frequent cannabis use in subjects without major psychiatric illness [7,22–24].
The questionnaire consisted of dichotomous rating (yes or no for each effect of cannabis). It was emphasized to the recent-onset patients that the effects should concern cannabis use prior to the first psychotic episode. To this purpose, it was first explained to patients what was meant by psychotic symptoms. Only when consensus was reached between the rater and patient on when the first psychotic episode had started was the list of effects presented to the patient. Psychotic effects were scored when cannabis intoxication acutely (i) caused psychotic symptoms or (ii) enhanced preexisting psychotic symptoms. To differentiate between these two in recent-onset patients, the date of onset of psychotic effects of cannabis was noted, and a timeline was made to assess whether the onset of the first psychotic symptoms had occurred during intoxication with cannabis.
Measurements in UHR subjects
Data (i.e. sociodemographics, amounts of substance use, and subjective effects of cannabis) were collected using the patient interview. Both current and past effects of cannabis were scored.
Measurements in controls
Data (i.e. sociodemographics, the occurrence of psychotic symptoms, amounts of substance use, and sub jective effects of cannabis) were collected using the patient interview. Both current and past effects of cannabis were scored. When controls reported to have experienced a psychotic symptom, they had to indicate whether that symptom occurred during substance use (yes, no, or sometimes).
Procedure
This study was approved by the local and national Medical Ethics Committees and written informed consent was obtained from all participants. Recent-onset patients were interviewed by a research staff member who was not involved in the treatment (B.P. or P.deK.) after the clinical condition had stabilized. First, the two raters each interviewed three recent-onset patients in the presence of the second rater, and then scored the six interviews independently of each other. No discrepancies between the two raters were detected during this procedure and therefore interrater reliability was not tested statistically.
Data analysis
The first hypothesis of the present study was that some patients in the prodromal phase of schizophrenia-like disorder as well as UHR subjects experience negative subjective effects from cannabis, while others experience positive subjective effects. To this order, the frequencies of positive and negative subjective effects were counted per subject group. The second hypothesis was that patients in the prodromal phase of schizophrenia-like disorder and UHR subjects more often experience negative subjective effects from cannabis than cannabis-using controls from the general population. For this second hypothesis, between-group differences (recent-onset patients vs UHR subjects vs controls) in endorsement of subjective effects were analysed on logistic regression. Because level of education was significantly different between the three groups, and some effects of cannabis were related to educational level, binary logistic regression was used to correct for educational level for these effects. For this purpose level of education was categorized in high (college bachelor or master level) and low (low- or medium-skilled training, or high school graduate level). No Bonferroni correction was applied because the effects of cannabis were hypothesized a priori to differ between recent-onset patients, UHR subjects and controls. Furthermore, to differentiate between psychotic effects de novo or enhancement of pre-existing psychotic symptoms, the percentage of recent-onset patients experiencing their first psychotic symptoms during cannabis intoxication was calculated as described in the methods section. Continuous data were analysed using one-way analysis of variance (post-hoc pairwise comparisons: Tukey's honestly significant difference test) or the Kruskal–Wallis test for non-normally distributed data (post-hoc pairwise comparisons: Mann–Whitney U-test). Categorical data were analysed with the χ2 test. Level of significance was set at p<0.05 (two-sided). All data analyses were performed using SPSS version 14 (SPSS, Chicago, IL, USA).
Results
Subject characteristics
Five controls reported experiencing delusions and/or hallucinations while not being intoxicated with a substance, and were excluded. Fifty-two recent-onset patients were included. Forty-four were diagnosed with schizophrenia, six with schizoaffective disorder and two with schizophreniform disorder (Table 1). Seventeen UHR subjects were included. Seven UHR subjects were diagnosed with a depressive disorder, and five with other diagnoses (attention-deficit–hyperactivity disorder, post-traumatic stress disorder, identity disorder and anxiety disorder not otherwise specified). Fifty-two controls were included. Mean age did not differ significantly between the groups. Nine controls (17%) were university students, 43 were coffee shop recruits. Controls had a significantly higher level of education than recent-onset and UHR subjects (χ2(2)= 19.3, p <0.001). In recent-onset patients mean age of onset of the first psychotic symptom was 18.8 years (SD = 4.5), and of the first psychotic episode 21.0 years (SD = 3.2). Mean duration between the start of the first psychotic episode and the interview was 77.7 weeks (SD = 65.3). In all recent-onset patients and UHR subjects lifetime cannabis use was much greater than lifetime hard drug use. In all but five controls lifetime cannabis was use much greater than hard drug use. The UHR subjects had used fewer cannabis cigarettes in their lifetime than the recent-onset patients and the controls (χ2(2) = 7.9, p <0.02). There were no significant differences in history of alcohol abuse between the patient groups and the controls.
Subject characteristics
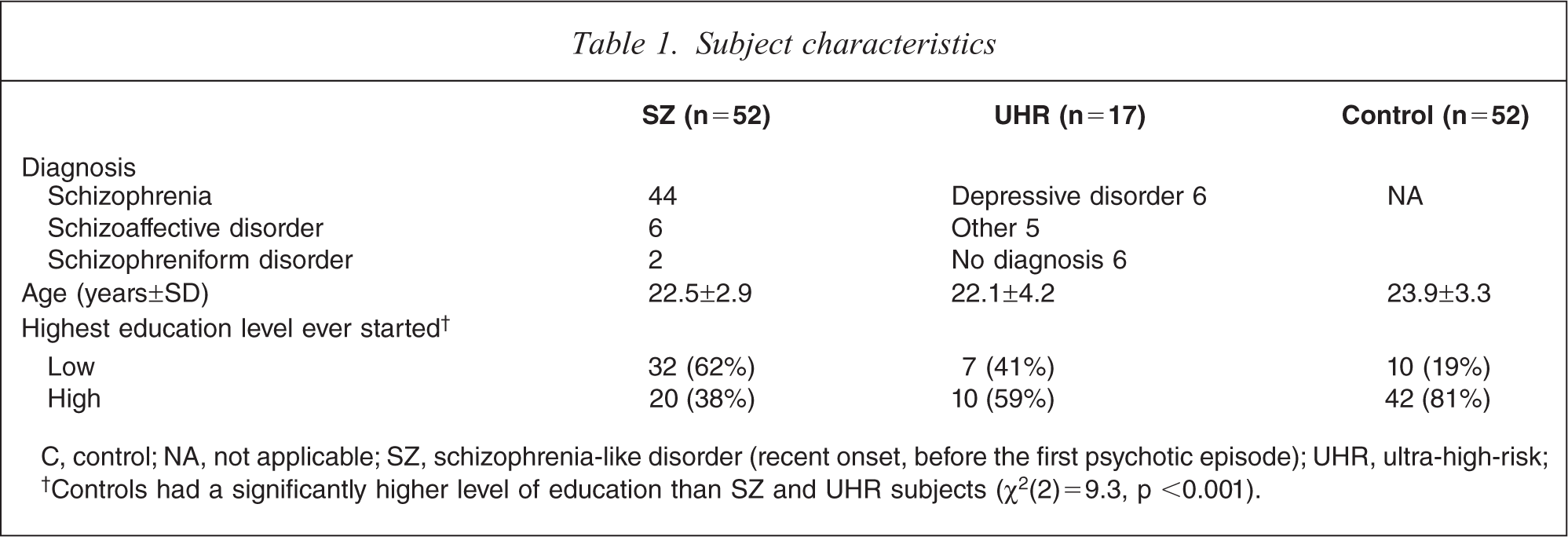
C, control; NA, not applicable; SZ, schizophrenia-like disorder (recent onset, before the first psychotic episode); UHR, ultra-high-risk; †Controls had a significantly higher level of education than SZ and UHR subjects (χ2(2)=9.3, p <0.001).
Subjective effects of cannabis use
The complete results regarding acute and long-term effects of cannabis use in the three groups are listed in Table 2. The following effects differed between both patient groups and the controls. Both recent-onset and UHR subjects reported feeling more depressed (recent-onset: odds ratio (OR) = 20.9, 95% confidence interval (CI) = 2.6–170.2, df 1, p = 0.004; UHR (n= 15: data missing for two UHR subjects): OR=21.7, 95% CI = 2.2–210.3, df=1, p = 0.008;), anxious (recent-onset: OR=7.2, 95% CI = 1.9–27.8, df= 1, p = 0.004; UHR: OR=10.1, 95% CI = 2.2–46.8, p = 0.003), and suspicious (recent-onset: OR=16.2, 95% CI =.3–61.8, df=1, p<0.001; UHR: OR= 11.0, 95% CI = 2.4–50.4, df=, p = 0.002) immediately after cannabis use compared to controls. In addition, recent-onset patients also reported increase of other psychotic symptoms during cannabis intoxication: unusual visual experiences (OR=4.9, 95% CI=1.5–15.9, df=1, p = 0.009), confusion (OR=4.8, 95% CI = 1.5–15.1, df=1, p = 0.007) and acoustic hallucinations (OR=14.5, 95% CI=1.7–121.2, df= 1, p = 0.01) during cannabis intoxication. In 36.5% of the recent-onset patients the very first psychotic symptoms occurred during cannabis intoxication.
Subjective effects of cannabis in male users
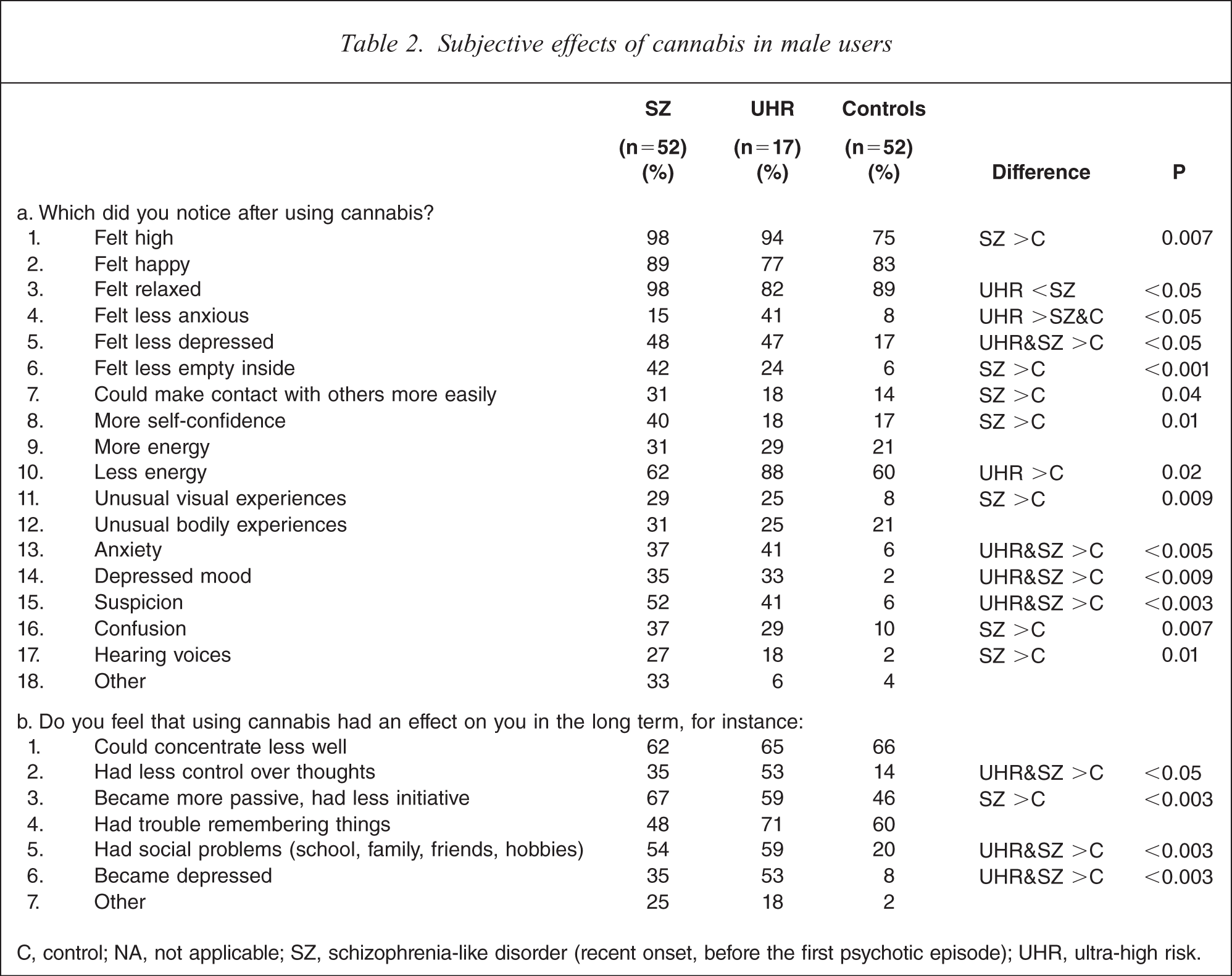
C, control; NA, not applicable; SZ, schizophrenia-like disorder (recent onset, before the first psychotic episode); UHR, ultra-high risk.
The two patient groups also reported feeling less depressed immediately after cannabis use (recent-onset: OR = 4.4, 95% CI= 1.8–10.9, df=1, p = 0.001; UHR: OR=4.2, 95% CI=1.3–14.0, df=1, p = 0.02). Of all subjects, 15 individuals (12.4%) reported less as well as more depressed feelings after cannabis use; 12 individuals (9.9%) reported to experience both less and more energy, and eight individuals (6.6%) reported both less and more anxiety. Mixed depressive effects were experienced most often by the recent-onset patients (25% of the recent-onset patients experienced mixed depressive effects). Mixed effects of energy and anxiety were most often reported by the UHR subjects (23.5% and 17.6% of the UHR subjects, respectively).
Long-term effects of cannabis use reported more often by both patient groups were: less control over thoughts (recent-onset: OR= 3.4, 95%CI = 1.3–9.1, df=1, p = 0.01; UHR: OR=7.2, 95%CI = 2.1–25.0, df=1, p = 0.002), social problems (recent onset: OR=4.6, 95% CI=1.8–11.9, df=1, p = 0.001; UHR: OR=5.8, 95% CI = 1.8–19.3, df=1, p = 0.004), depression (recent onset: OR=6.4, 95% CI = 2.0–20.5, df=1, p = 0.002; UHR: OR=13.5, 95% CI = 3.3–54.5, df=1, p <0.001).
Discussion
Negative subjective effects in comparison to controls
Recent-onset patients and, to a lesser extent, UHR subjects reported increased psychotic effects during cannabis intoxication, either de novo or as an increase in pre-existing psychotic symptoms. Whereas recent-onset patients reported multiple psychotic effects of cannabis, the UHR subjects reported only suspicion more often than controls. This difference could be explained by the lower vulnerability to psychosis of the UHR group, which has an estimated psychosis transition rate of approximately 30% in 2½ years [25]). In accordance with the present findings, Green et al. found that schizophrenia patients reported paranoia after cannabis use more often than controls [10]. Cannabis use has also been related to increased psychotic symptoms in subjects with a genetic liability for schizophrenia [26], and cannabis-using UHR subjects may have higher psychosis transition rates [27], although this was not confirmed in another study [28]. The present findings add qualitative evidence to longitudinal studies that suggest that cannabis use increases the risk of developing a psychotic disorder [1]. The relation between cannabis and psychosis may be mediated by effects on dopamine neurotransmission [29]. One case report showed that inhalation of cannabis acutely increased dopamine levels in the striatum, which was accompanied by increased psychotic symptoms [30]. Henquet et al. found that the catechol-O-methyltransferase (COMT, an important enzyme that metabolizes dopamine in the central nervous system) gene moderated the psychotic effects of Δ9-THC [31]. Finally, a longitudinal study found that carriers of the high-activity Val allele of the COMT gene had an increased risk of developing a schizophreniform disorder after adolescent cannabis use [32].
Recent-onset and UHR subjects also reported other negative effects (depressed mood, anxiety) during cannabis intoxication and after long-term use (less control over thoughts, social problems, depression), more often than controls. On a pathophysiological level, depression may follow cannabis use by increasing levels of interferon-λ [33]. Alternatively, depressive effects of cannabis might be caused by a decrease in prefrontal dopamine levels: in animal studies repeated exposure to Δ-9-THC was found to persistently reduce frontal dopamine turnover [34,35], which in turn has been implicated in depression [36]. Finally, depressive effects may also be mediatedby direct effects on the endogenous cannabinoid system [37]. Psychosis-prone individuals may be particularly vulnerable to the effects of cannabis on prefrontal dopamine and endocannabinoid neurotransmission, because these systems have also been implicated in the pathophysiology of schizophrenia [38,39]. Increased anxiety immediately after cannabis use may be related to the increased psychotic effects reported by the recent-onset patients and UHR subjects.
Recent-onset patients and UHR subjects did not regard impaired cognition to be a long-term effect of cannabis use. This is in line with findings of preserved cognitive performance in schizophrenia patients with cannabis abuse compared to patients without cannabis abuse [40].
Positive subjective effects in comparison to controls
UHR subjects, but not recent-onset patients, reported feeling less anxious after cannabis use. We speculate that this is related to the lower amounts of cannabis used by the UHR group compared to the recent-onset and control group. Low doses of cannabinoid receptor agonists have been found to induce anxiolytic-like responses in male rats, whereas a high dose induced anxiogenic-like effects [41]. The modulation of anxiety by cannabinoid agonists found in human and experimental animal studies are, however, complex and often contradictory. Indeed, some of the present subjects experienced mixed effects regarding anxiety (i.e. both increases and decreases). Animal data have shown cannabinoid modulation of anxiety to be dose dependent, bidirectional and dependent on the environmental context, and to interact with other substance use [42]. Mixed effects on energy and depressed mood were also reported by some of the present subjects. Depressive effects of cannabis may also depend on the environmental context [43], as well as on mood state before ingestion of cannabis [44] and the presence of psychological stress [45]. Further research is needed on how psychological, biological and environmental factors determine differences in effects of cannabis between and within individuals.
Recent-onset patients also reported several positive effects (feeling less empty inside and less depressed; being able to make contact with others more easily; having more self-confidence) more often than controls. This may suggest that cannabis was used by some recent-onset patients as self-medication of specific prodromal symptoms. Problems in social interaction and negative symptoms such as flattened affect, which can be difficult to distinguish from depression, are frequently present many years before the overt onset of the illness [46]. Negative symptoms may reflect a persistent deficit in dopaminergic transmission in the prefrontal cortex [39,47], and animal studies have shown that Δ-9-THC acutely increases prefrontal dopaminergic activity [48]. However the fact that the most often reported effects by all three groups in the present study were ‘feeling high’, ‘feeling relaxed’ and ‘feeling happy’ suggests that cannabis use is mainly driven by its positive effects and not by specific UHR states or prodromal states of schizophrenia. Green et al. found that the occurrence of positive affect enhancement after cannabis use correlated with amount and frequency of use in schizophrenia patients [10].
The present findings may be clinically relevant. Because cannabis use increases the risk of developing a psychotic disorder, cannabis use should be a focus of treatment in individuals at UHR of psychosis. It may be preferred to focus on those individuals who experience psychotic effects from cannabis. In addition, self-perceived negative effects on mood and social functioning may act as additional motivators to stop using cannabis. Motivational interviewing, which makes use of individuals’ perceptions of the positive and negative consequences of substance use, has been found to be effective in reducing cannabis use in adolescents [49]. Furthermore, individuals who experience fewer anxiety and depressive symptoms after cannabis use may benefit from help from caregivers for such mood symptoms in order to stop using cannabis.
Limitations
One limitation of the present study is the small sample size of the UHR group. This may have resulted in spurious findings or type two errors, for instance the lower frequency of psychotic and other effects in the UHR group compared to the recent-onset group. A second limitation is that a standardized, validated questionnaire on effects of cannabis use was not used. We are not aware, however, of the existence of a validated questionnaire on the subjective effects of cannabis. We composed a theory-driven questionnaire with a wide range of effects reported in other studies. A limitation of the questionnaire is the dichotomous option of choice between pre-selected effects. In future studies on effects of cannabis use before the first psychotic episode it may be preferable to first ask patients in an open-question what effects cannabis has/had on them. In addition, a Likert scale could help differentiate the relative importance of items. Of note, a study in which patients with a psychotic disorder and controls could generate their own responses has found similar effects as the present study [10]. Further validation of measuring subjective effects of cannabis is needed.
Furthermore, there were some possible sources of bias in the present study. First, there may have been a bias introduced through the selection of controls. Most controls were recruited from a coffee shop, and we did not assess what the main method of acquiring cannabis was for the patients. There may be differences between users who acquire cannabis from a coffee shop and users who acquire cannabis from other sources (e.g. acquaintances). Second, recall bias may have influenced the results in the recent-onset patients because they were asked to recall subjective effects retrospectively. Current mental symptoms can influence memory [50]. Also, the patients had received psychoeducation about cannabis and its negative effects, which may have created recall bias. Recall bias may have been increased by cannabis use, because ongoing cannabis use can result in memory impairments [51]. Cannabis use was minimized during treatment, and to further reduce potential recall bias only patients with a recent onset in a clinically stabilized condition were interviewed. Duration between time of interview and the first psychosis was relatively short (on average 77.7 weeks). Overall, the validity of the present findings in the recent-onset patients was partly confirmed by similar findings in the UHR subjects.
Conclusions
To our knowledge this is the first study on the subjective effects of cannabis in the prodromal phase of schizophrenialike disorder and the UHR state, in comparison with a cannabis-using control group from the general population [10]. Increased negative effects in both patient groups, including psychotic symptoms, suggest heightened sensitivity to some effects of cannabis in the prodromal phase of schizophrenia-like disorder and the UHR state. These findings add qualitative evidence to longitudinal studies that suggest that cannabis increases the risk of developing a psychotic disorder. Increased positive effects were also reported by recent-onset patients, which suggests that cannabis may be used as self-medication prior to illness onset. Further studies in UHR subjects using both objective and subjective measures, especially controlled studies using oral cannabis or i.v. Δ9-THC, can provide further insight into the effects of cannabis in the UHR state. Insight into the subjective effects of cannabis may assist in decreasing cannabis use in subjects at UHR of psychosis.
Footnotes
Acknowledgements
The authors thank Luuk Wouters (Department of Psychodiagnostics, Academic Medical Center, University of Amsterdam) for his expert assistance in the statistical analyses. This study was funded by Grant 28-1241-2 from the Dutch Health Research and Development (ZONMw).
