Abstract
A landmark study indicated the possibility of detecting people that were at ultra-high risk (UHR) for developing psychosis in a help-seeking population of young people during a 12 month follow-up study. Patients were between 14 and 30 years old, did not suffer from a previous psychotic episode and showed attenuated psychotic symptoms, brief limited intermittent psychotic symptoms (BLIPS), and trait and state risk factors [1].
In previous research, an association between cannabis use and the onset and course of psychosis and schizophrenia has been found [2–4]. Philips et al., however, found no association between cannabis use and the development of psychosis in a UHR group [5]. It is unlikely that cannabis use causes psychosis as a single factor, but two recent reviews suggested that cannabis is a partial causal factor [2,6,7]. Henquet et al. showed that the association between cannabis use and the onset of psychosis was stronger for people with a predisposition for psychotic disorders [8]. Frequent use of cannabis was positively correlated with higher levels of risk for developing a psychosis.
Furthermore, cannabis use can increase the relapse rate during the early course of schizophrenia and related disorders. Cannabis use has been found to have adverse effects on the positive symptoms of psychosis, visual hallucinations in particular [9,10]. Stefanis et al. investigated the effects of cannabis use in early adolescence on positive and negative symptoms of schizophrenia [11]. They reported that cannabis use correlated positively with both positive and negative symptoms. They also concluded that if the onset of cannabis use started before the age of 16, there was a much stronger effect than if the onset of cannabis use started after the age of 15. These results were independent of the lifetime frequency of cannabis use.
Schizophrenia patients often have impaired cognitive functioning, for example in visual memory, sustained attention and verbal memory [12–14]. A meta-analysis showed a correlation between performance on neurocognitive tasks and negative symptoms [15].
The relationship between cannabis use and cognitive functioning has also been studied. Healthy non-cannabis using controls were compared with heavy cannabis users [16]. Heavy cannabis use was associated with significant impairments in memory retrieval, verbal expre ssion and mathematical reasoning. In another study, long-term cannabis users were compared with short-term users and non-users. A neuropsychological test battery that assessed attention, memory and executive functioning showed that the long-term cannabis users had impairments in memory and attention that endure beyond the period of intoxication and worsen with increasing years of regular cannabis use [17]. In a recent study, the effects of cannabis consumption in schizophrenia patients and healthy controls were evaluated [18]. The results showed that cannabis abuse before the onset of schizophrenia does not necessarily cause deterioration of cognitive functioning in schizophrenia patients.
Pukrop et al. investigated cognitive domains as indicators of vulnerability to psychosis [19]. The results suggested that impairments in verbal abilities and decreased speed of processing may predict conversion to psychosis in a UHR group.
Cannabis use may trigger psychosis in vulnerable people. The present study focused on the cross-sectional relationship among cannabis use, UHR symptoms and neuropsychology. We expected cannabis-using healthy control subjects and UHR subjects to have increased symptomatology and reduced neuropsychological functioning compared to non-using subjects.
Method
The present study was performed at the Amsterdam site of the Dutch Prediction of Psychosis Study (DUPS). That study is a naturalistic study with a major focus on the course of UHR symptomatology. The participating cities are Amsterdam and Utrecht in the Netherlands. The Amsterdam study is performed in the Academic Medical Center (AMC) of the University of Amsterdam.
Subjects
A total of 63 UHR subjects were included in the present study. Inclusion criteria were (i) age between 12 and 35 years; and (ii) informed consent. Subjects were eligible if they had fulfilled one of the following criteria: (i) familial risk plus reduced functioning: individuals with a DSM-IV schizotypal personality disorder or a first-degree relative with a history of any DSM-IV psychotic disorder and a change in mental state or functioning in the patient leading to a reduction of ≥ 30% or more on the Global Assessment of Functioning (GAF) Scale (an interview was carried out with the patient and a close relative, mostly one of the parents, to define the initial baseline of functioning); (ii) showed attenuated psychotic symptoms: presence of at least one of the following symptoms: delusional ideas, suspiciousness, grandiose ideas, hallucinations or disorganized communication, these symptoms should occur at least several times per week for at least 1 week; (iii) showed BLIPS (previous frank psychotic symptoms, such as hallucinations and delusions, with duration < 1 week and spontaneous remission) and (iv) showed basic symptoms (at least two basic symptoms as assessed with the Bonn Scale for the Assessment of Basic Symptoms-Prediction List (BSABS-P) [20]; this scale assesses self-perceived deficits in cognition and perception).
Exclusion criteria were (i) IQ < 85; (ii) symptoms due to drug use; (iii) symptoms due to an organic etiological factor; and (iv) previous psychotic episode that lasted > 1 week. Regarding exclusion criterion (ii), patients who used cannabis were asked if they had a period in which they did not use cannabis and still experienced symptoms. If not, they were asked to stop using for 1 month. At the end of this period, the patients were reassessed. This was verified with the Composite International Diagnostic Interview 2.1 (CIDI) [21]. Patients who used hard drugs (e.g. cocaine, heroine, Ecstasy, speed) were excluded. Demographics are listed in Table 1.
Subject details (mean ± SD)

†χ2 (3) = 9.92, p = 0.019.
A division was made within the UHR group into cannabis and non-cannabis users. Thirty-four cannabis UHR users were included, of whom 16 patients were using frequently (10 used every day, two used 3–4 days per week, three used 1–2 days per week, one used 1–3 days per month) at the time of testing. The rest of the cannabis users in the UHR group had used cannabis in the past. The last time they used varied from 2 weeks up to > 1 year previously. A total of 45% of them had used almost every day, 11% used 3–4 days per week, 33% used 1–2 days per week, and 11% used 1–3 days per month.
During a 3 year follow-up period, 17 (27%) of the included UHR patients made the transition to psychosis. Seven (41%) of them were cannabis users at the time of testing or in the past. A number of patients had stopped using cannabis before intake. Of the transition group, only four used cannabis at the time of testing. This number was too small to perform statistical analysis upon. The mean interval between inclusion and transition of this group was 12.77 months (range = 3.00–35.00 months, SD = 7.86). The data for the present study were obtained at baseline.
Fifty-eight healthy control subjects were assessed. The inclusion criteria were: (i) age between 12 and 35 years; (ii) not using cannabis or using at least four joints of cannabis per week (four joints per week was set as a criterion to enlarge the chance of finding a difference between the cannabis- and non-cannabis-using group); and (iii) informed consent. Exclusion criteria were: (i) psychiatric disorder in the past or present; (ii) psychiatric family history; and (iii) scoring on the Structured Interview for Prodromal Syndromes (SIPS) [22] or BSABS-P in the UHR range.
The investigation was carried out in accordance with the latest version of the Declaration of Helsinki. The study design was approved by the Medical Ethics Committee of the Academic Medical Centre. Informed consent of all participants was obtained after the nature of the procedures had been fully explained.
Demographics are listed in Table 1. Age and premorbid IQ were comparable between the four groups. Sex distribution was not. Cannabis users were mainly male (χ2 (3) = 9.92, p = 0.019).
Materials
Clinical interviews
The SIPS is a comprehensive diagnostic tool designed specifically for the assessment of the whole spectrum of prodromal signs and symptoms [22]. The scale is composed of 19 items (five positive, six negative, four disorganization, four general symptoms), each of which is given a score of 0–6 according to defined criteria. A score between 3 and 5 on the positive symptoms indicates attenuated psychotic symptoms and a score of 6 indicates a psychotic state. A score of 3–5 on the positive items indicates a UHR state and makes the subject eligible for the study.
The BSABS-P contains 17 selected self-perceived disturbances in cognition and perception that were found to be predictive for a transition to psychosis over a 10 year period [20]. Each basic symptom is given a score of 0–6 according to frequency of occurrence. A score of ≥ 3 on at least two of the first nine items was necessary for inclusion. The first nine items were found to be most predictive of psychosis in previous research [20].
CIDI section J and L was used to assess drug use. The CIDI is a well-validated instrument to determine an ICD-10 or a DSM-IV diagnosis [21]. This questionnaire assesses the use, amount, onset, frequency and duration of cannabis use. There is a policy in the Netherlands that the use and possession of soft drugs is tolerated. Therefore, data from the CIDI can be considered to be reliable.
Neuropsychological test battery
All subjects were assessed with a neuropsychological test battery composed of the following tests: The Finger-Tapping Test (FTT) assesses motor speed [23]. The Continuous Performance Test (CPT) is a well-known measure of vigilance, which refers to sustaining attention on a stimulus or a certain dimension of that stimulus over a period of time [15]. The Dutch version of the California Verbal Learning Test (CVLT) is a verbal memory task that assesses the verbal short-term as well as the long-term verbal memory [23]. The Dutch version of the National Adult Reading Test gives an indication of premorbid IQ [24]. The Verbal Fluency Test (VF) assesses structured searching in semantic memory [23].
Procedure
Prior to their referral to the AMC, all patients sought help for various complaints at local health services and other health services. The patients were referred to the DUPS project for a second opinion because the referring clinician suspected a psychotic development. After their referral to DUPS, putative UHR patients were invited for a first interview with a psychiatrist and a psychologist. In this approximately 2 h face-to-face interview, subjects were asked about their lifetime history of complaints, family history of psychiatric disorders, drug and medicine use. Subsequently, in a standardized order, the SIPS and BSABS-P were administered.
Simultaneously, in another interview the parents or guardians were asked about the lifetime development of their child. The SIPS and BSABS-P were scored and each subject was discussed in a staff meeting. When considered at ‘ultra-high risk’, the patients were asked to sign a written informed consent before participating in the DUPS project. In this naturalistic longitudinal cohort study, subjects were followed up for 3 years. They were referred back to their referring mental health institution. Some received treatment, others were only monitored. Patients, their parents or caregivers and the referring mental health institutions were asked to contact the DUPS project in case of increasing symptoms. In addition they were seen for a SIPS interview at 9, 18 and 24 months and interviewed by telephone at 36 months.
Statistical analysis
To compare differences in severity of symptomatology and neuro-psychological dysfunction between the four groups, ANOVAs were used. Two-tailed post-hoc t-tests were used to investigate differences between the cannabis and non-cannabis group. Spearman correlations were used to investigate the relationship between frequency of cannabis use and severity of symptomatology and neuropsycho-logical dysfunctioning in the cannabis-using UHR and control group combined.
Results
Significant differences were found between the four groups on the SIPS for the positive (F 3,117 = 103.17, p < 0.0001), negative (F 3,116 = 53.02, p < 0.0001), disorganization (F 3,117 = 34.33, p < 0.0001) and general symptom scores (F 3,116 = 60.23, p < 0.0001), and also in the BSABS scores (F 3,111 = 49.66, p < 0.0001; Table 2).
UHR symptomatology (mean ± SD)

BSABS, Bonner Scale for the Assessment of Basic Symptoms-Prediction List; SIPS, Structured Interview of Prodromal Syndromes; UHR, ultra-high risk.
Independent t-tests showed no significant differences within the UHR group. In the control group three items of the SIPS showed a significant difference. The cannabis control group had significantly more positive symptoms (t 56 = 3.18, p = 0.003), more disorganization symptoms (t 56 = 2.71, p = 0.009) and more general symptoms (t 56 = 2.37, p = 0.025) than the non-cannabis control group. Independent t-tests showed that the cannabis control group also had significantly more BSABS symptoms than the non-cannabis control group (t 55 = 2.58, p = 0.013).
Table 3 describes the mean and standard deviation of the scores on the items of the BSABS and SIPS that showed significant differences between the groups.
Individual SIPS and BSABS items (mean ± SD)

BSABS, Bonner Scale for the Assessment of Basic Symptoms-Prediction List; SIPS, Structured Interview of Prodromal Syndromes.
The basic symptom ‘photopsia’ (simple moving or fixed white, bright or coloured hallucinations in form of flashes, stars, flames, circles, triangles or very strong, blinding light) was significantly different between the four groups (F 3,115 = 11.84, p < 0.0001). Post-hoc analysis showed that the cannabis UHR group had significantly more photopsia than the non-cannabis UHR group (t 59 = 2.26, p = 0.023). Furthermore the basic symptom ‘thought interference’ was significantly different between the four groups (F 3,117 = 11.90, p < 0.0001). Post-hoc analysis showed that the cannabis UHR group had significantly more thought interference than the non-cannabis UHR group (t 61 = 2.18, p = 0.032). Also, within the control group the cannabis users had significantly more thought interference than the non-cannabis users (t 56 = 2.41, p = 0.020).
We found a significantly different score between the four groups on the SIPS items ‘trouble with focus and attention’ (F 3,117 = 32.12, p < 0.0001), ‘perceptual abnormalities/hallucinations’ (F 3,117 = 20.69, p < 0.0001), ‘dysphoric mood’ (F 3,116 = 58.16, p < 0.0001), and the BSABS item ‘thought blockages’ (F 3,117 = 14.54, p < 0.0001). Independent t-tests showed that the cannabis-using control group had significantly more trouble with focus and attention (t 56 = 2.99, p = 0.004), more perceptual abnormalities/hallucinations (t 56 = 2.36, p = 0.023), more dysphoric mood (t 56 = 2.04, p = 0.048), and more thought blockages (t 56 = 2.34, p = 0.024) than the non-cannabis-using control group.
Table 4 describes the mean and standard deviation of the neuropsychological test results in the four groups. The four groups performed significantly differently on the CVLT (F 3,109 = 5.34, p = 0.002) and the VF test (F 3,109 = 4.83, p = 0.003). The non-cannabis-using controls performed significantly better on the CVLT (t 56 = −3.38, p = 0.001) and the VF test (t 56 = −2.47, p = 0.017) than the cannabis-using control group. No significant differences were found between the cannabis-using and non-using UHR groups.
Neuropsychological test scores (mean ± SD)
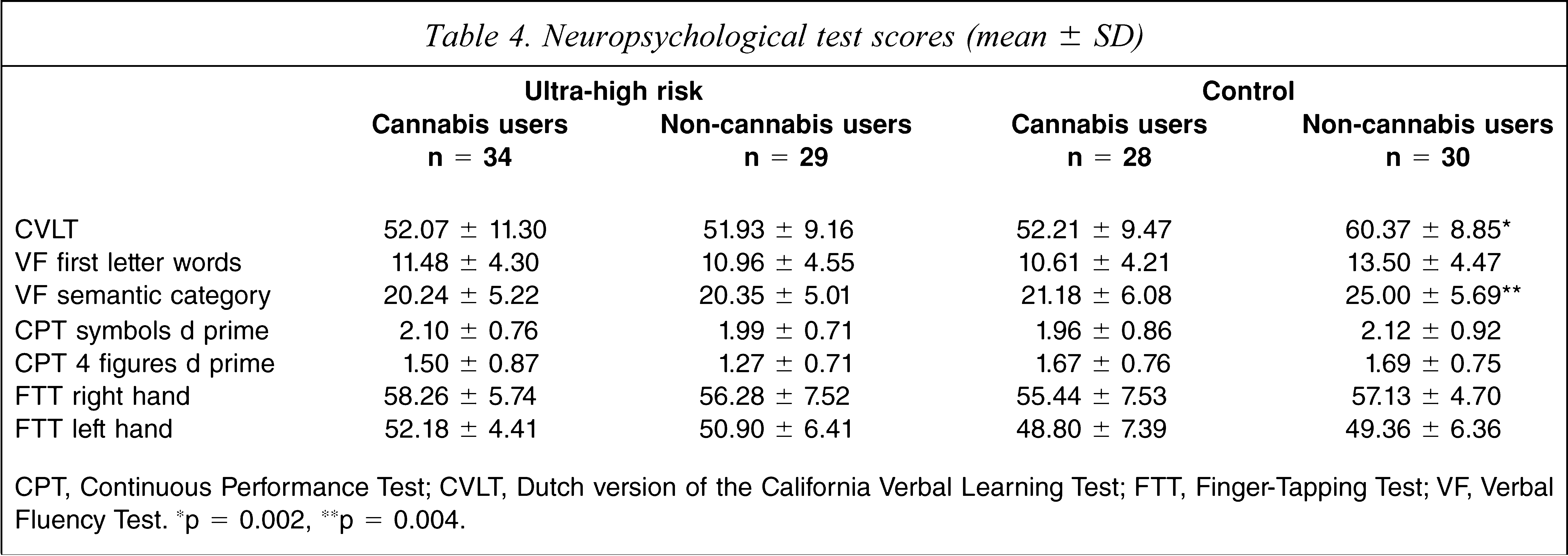
CPT, Continuous Performance Test; CVLT, Dutch version of the California Verbal Learning Test; FTT, Finger-Tapping Test; VF, Verbal Fluency Test. ∗p = 0.002, ∗∗p = 0.004.
Frequency of cannabis use correlated with the SIPS items ‘unusual thought content’ (ρ = 0.258, n = 59, p = 0.049) and ‘suspiciousness’ (ρ = 0.299, n = 59, p = 0.021) in the cannabis UHR and control group combined. Furthermore, more frequent cannabis use was related to increased severity of the BSABS symptom ‘micropsia/macropsia’ (objects are perceived as smaller or bigger than they really are; p = 0.325, n = 59, p = 0.012), as well as ‘diplopsia’ (objects are perceived twofold or threefold, lopsided or crooked; ρ = 0.288, n = 59, p = 0.027). The same relationship was found for the BSABS symptoms ‘changes in the perceived intensity or quality of acoustic stimuli’ (ρ = 0.481, n = 59, p < 0.0001) and ‘loss of automatic skills’ (ρ = 0.355, n = 59, p = 0.006). We found no significant relationship between frequency of cannabis use and any neuropsychological test.
Because sex was significantly different between the groups, we used gender as a covariate in a MANOVA to investigate the influence of this variable in the results. No significant influence was found in the reported results.
Discussion
The present results showed that cannabis-using UHR subjects have more basic symptoms than non-using UHR subjects. Healthy cannabis users showed more subclinical UHR, basic symptoms and more neuropsychological dysfunctions than non-cannabis users.
When we performed analyses on the BSABS items, photopsia and thought interference were more prevalent in the cannabis-using UHR group compared to the non-cannabis-using UHR group. Of 63 BSABS symptoms, thought interference was one of the most predictive for schizophrenia in the study by Klosterkotter et al. [20]. Thought interference may be related to cannabis use and may be a precursor of thought disorder in schizophrenia.
In the control group, cannabis users had more trouble with focus and attention than non-cannabis users. In addition, they experienced more dysphoric mood as assessed with the SIPS. The BSABS items thought interference as well as thought blockages, were found to be significantly more severe in the cannabis-using control group compared with the non-cannabis-using subjects.
Cannabis users who used more frequently, experienced more unusual thought content and suspiciousness as assessed on the SIPS. They also experienced more micropsia/macropsia, diplopsia, changes in perceived intensity or quality of acoustic stimuli and loss of automatic skills, as assessed on the BSABS. These results suggest a correlation between frequency of cannabis use and UHR symptoms. The found relationship between frequency of cannabis use and these BSABS symptoms suggests that frequent cannabis use is related to changes in visual information processing. This result is consistent with the reported adverse effects of cannabis use on visual hallucinations [9,10]. The aforementioned results correspond to the experiences in clinical practice. Sensory changes, suspiciousness and unusual thought content (e.g. extreme philosophical ideas) are characteristic acute effects of cannabis and are often reported by cannabis users.
In the control group, cannabis users performed worse on the CVLT than the non-cannabis users, even at the level of the UHR group. Furthermore, the cannabis users in the control group performed worse on the verbal fluency categories than the non-cannabis users. Neuropsychological performance was not significantly different between the cannabis users and non-users in the UHR group.
Cannabis-using control subjects performed worse on a test of verbal learning and memory and on the verbal fluency test than non-cannabis-using controls. This result is consistent with the literature on this subject. That we found no difference in the UHR patient group may be due to milder cannabis use in some of the UHR patients. Not all patients in the UHR cannabis group used at least four joints per week at the time of testing, whereas this was an inclusion criterion in the control cannabis group. Cannabis use seems to reduce the score of healthy controls matched for age and premorbid IQ on a verbal memory test to the level of UHR patients.
Approximately half of the UHR cannabis users (18/34) were classified on the basis of previous pattern of use whereas the controls were classified on the basis of current use. We could not control for the use of the UHR group but we could control for the use of the control group. We chose a minimum of four joints per week for the controls to enhance the chance of finding a difference between the cannabis and non-cannabis group. Many patients stopped using cannabis before intake. Because of the difference in cannabis use, post-hoc tests were performed within the UHR and control group separately. Therefore within these groups cannabis use was equivalent, making inferences about the effect of cannabis on subclinical UHR symptoms and neuropsychological functioning possible.
Future research should focus on larger groups of UHR subjects with more frequent cannabis use than that in the present study. Furthermore, the quantity of cannabis use should be studied. Future research should also focus on the long-term effects of cannabis use on UHR symptoms. It would be interesting to investigate whether mild UHR symptomatology and neuropsychological dysfunction disappears when controls stop using cannabis. Controls that continue to use cannabis may have an increased risk for developing psychosis. We will follow up this control group.
Meta-analyses of epidemiological studies show that cannabis can have an adverse effect when subjects have a predisposition for developing a psychosis. The present results suggest that frequent cannabis use is a risk factor for developing subclinical UHR symptoms and impaired neuropsychological functioning in healthy adolescents. Based on these results one may argue that adolescents should be informed about the risks of using cannabis.
Footnotes
Acknowledgements
This study was supported by the Dutch Prediction of Psychosis Study (grant 2630.0001) from the Netherlands Organization of Health Research and Development (ZONMW).
