Abstract
One of the defining features of schizophrenia is impairment in social function, including communicating with others, maintaining employment, and functioning in the community [1], [2]. Social functioning deficits have been reported premorbidly in individuals who later developed schizophrenia [3–5] and have been reported as being consistently detected in the adolescent offspring of an individual with schizophrenia [6–8]. Häfner et al. have reported that when individuals exhibited the initial prodromal symptoms of schizophrenia, their social development was not yet significantly inferior to controls [9], and that individuals with schizophrenia predominantly exhibited social deficits during prodromal and psychotic prephases [10]. Recent clinical studies have suggested that, among patients with schizophrenia, functional performance is independent of psychosis and tends to be resistant to medication once the illness had become chronic [11–13]. These findings highlight the need for early intervention, optimally prior to the onset of illness.
As a result of these findings, early intervention during the prodromal phase of schizophrenia has emerged as beneficial and has been rapidly developed as an important goal during the last decade. Recent studies have reported that high-risk individuals exhibit social skill deficits and impairment of social functioning [14–16], and have identified poor functioning as a potential risk factor for psychosis [17]. In addition, significant relationships have been found between clinical symptoms, neurocognitive functions, and level of psychosocial functioning among youth at ultra-high risk (UHR) for psychosis [18]. Impairment in social cognition has also been detected in UHR individuals [19], but studies have not yet established which factors are associated with social impairment in the prodromal phase of a psychotic disorder. If the underlying factors of social impairment can be identified in the prodromal state, it may be feasible to prevent psychosis earlier and to improve the long-term outcome of schizophrenia.
We hypothesized that social functioning deficit is simultaneously a trait and state marker of risk for schizophrenia and schizophrenia-related disorders, that is, an enduring phenotype in which severity is related to clinical state. To test this hypothesis, we examined social functioning among subjects at UHR for the development of psychosis. Because psychiatric symptoms can negatively influence social functioning in UHR subjects, we concomitantly examined social functioning among subjects at genetic high risk (GHR) who had no definitive subjective and objective symptoms. We theorized that both high-risk groups would have pre-existing heightened vulnerability such as neurodevelopmental abnormalities and genetic contributions, but that only UHR individuals would have experienced distress, depression and anxiety that might be putatively prodromal symptoms. Thus, we anticipated that social functioning would be more impaired in high-risk subjects than healthy controls (HC), and that social impairment might be more severe in subjects at UHR than subjects at GHR. In view of the suggested association between clinical symptoms and psychosocial functioning in youth at UHR, we investigated relationships between possible contributing factors to social impairment and level of social functioning.
Method
Subjects
Between November 2004 and June 2007, we recruited 94 subjects who met the study criteria. High-risk subjects were recruited from the Seoul Youth Clinic (SYC). Our previous article provides more details about SYC and the recruitment procedure [19].
Subjects were classified into three groups: UHR, GHR, and HC. The UHR group consisted of 32 subjects, aged 16–35 years, all of whom met criteria for at least one of the three operationally defined UHR groups according to the Comprehensive Assessment of At-Risk Mental States (CAARMS) instrument, useful for identifying subthreshold psychotic symptoms [20]. Most subjects (n = 28) met the attenuated psychotic symptoms (APS) criterion. Seven subjects met the vulnerability group (VG) criterion, and no subjects met the brief limited intermittent psychotic symptoms (BIPS) criterion. Three subjects fulfilled the APS and VG criteria concurrently. (The APS, BIPS and VG groups were termed the ‘attenuated psychosis group’, ‘BLIPS group’, and ‘Vulnerability’ in the Yung et al. article [20].) Five subjects who met both GHR and UHR criteria were categorized into the UHR group due to the presence of prodromal symptoms. A total of 13 subjects had already received antipsychotics at the time of enrolment.
The GHR group was composed of 32 subjects aged 14–33 years. All had a family history of schizophrenia; a subject with GHR was defined as having at least one first-degree relative with schizophrenia. VG in the UHR group differed from the GHR group because they also had a recent deterioration in functioning in addition to genetic risk. Presumed GHR participants were assessed using the Family Interview for Genetic Studies to investigate family history of psychiatric disorders and degree of genetic loading for schizophrenia [21]. Results indicated that one subject was the monozygotic twin of a schizophrenia patient and seven subjects had two first-degree relatives with schizophrenia as follows: two parents with schizophrenia (n = 1), one parent and one sibling with schizophrenia (n = 3), and two siblings with schizophrenia (n = 3). The remaining 24 subjects had one first-degree relative with schizophrenia, such as one parent with schizophrenia (n = 8) or one sibling with schizophrenia (n = 16).
The HC group included 30 age- and IQ-matched subjects. All were healthy adults aged 19–27 years, with no history of psychiatric disorders or treatment, nor any first–third-degree relatives with a psychiatric disorder. These subjects were recruited via Internet advertisement or the social networks of hospital staff.
All participants were subject to the following exclusion criteria: (i) known history of psychotic illness for longer than a week, substance abuse or dependence, neurological disease, and brain injury; (ii) evidence of medical illness, which can manifest as psychiatric symptoms and affect social functioning; and (iii) intellectual disability (IQ < 70). All subjects provided written informed consent, including parental consent for those <18 years of age. This study was approved by the Institutional Review Board at Seoul National University Hospital.
Assessments
At study intake all potential subjects participated in an intensive clinical interview with two experienced psychiatrists who used the Structured Clinical Interview for DSM-IV Axis I disorders (SCID) [22] to determine past and current psychiatric illnesses. Potential high-risk subjects were assessed using the CAARMS to ensure that intake criteria were met, and the CAARMS was scored by adding the intensity rating scores; in addition, a modified 24-item version of the Brief Psychiatric Rating Scale (BPRS) (rating items 1–7) [23] and the Positive and Negative Syndrome Scale (PANSS) [24] were used to measure psychotic features. Other scales were also used to assess psychological symptoms such as anxiety and depression, which could be prodromal symptoms; these included the Scale for the Assessment of Positive Symptoms (SAPS) [25], the Scale for the Assessment of Negative Symptoms (SANS) [26], the Hamilton Rating Scale for Depression (HAM-D) [27], the Hamilton Rating Scale for Anxiety (HAM-A) [28], and the Yale–Brown Obsessive Compulsive Scale (Y-BOCS) [29]. All subjects also completed the Global Assessment of Functioning (GAF) [30] and the Korean version of the Wechsler Adult Intelligence Scale (K-WAIS) [31] to estimate IQ.
Social Functioning Scale
Social functioning was assessed using the Social Functioning Scale (SFS), a 79-item questionnaire that has been proven as a reliable, valid, and sensitive measure of social functioning for outpatients with schizophrenia [32]. The SFS measures abilities or performance in seven areas: (i) social engagement/withdrawal; (ii) interpersonal behaviour; (iii) prosocial activities; (iv) recreation; (v) independence–competence; (iv) independence–performance; and (vii) employment/occupation. We used the Korean version of the SFS. Subjects answered each question by selecting one of four possible responses. Because the seven subscales have differing means and variances, each scale was standardized and normalized using a T transformation to a mean of 100 and a standard deviation of 15, using the unemployed schizophrenia group as the reference population [32].
Statistical analysis
All analyses were done using SPSS 12.0K for Windows (SPSS, Chicago, IL, USA). We conducted three group comparisons of numerical variables, such as age, using a one-way analysis of variance (ANOVA). If Levene's test indicated an unequal variance across the three groups (p < 0.05), we performed Welch's ANOVA. We also used post-hoc tests, or Tukey's multiple comparison tests, to determine specific group differences. We used the Kruskal–Wallis test to compare parental socioeconomic status (SES), and used Pearson's χ2 test or Fisher's exact test to compare differences in the proportions of categorical variables, such as gender, among the three groups. We compared scores on psychological symptom scales between the UHR and GHR groups using Welch's t-test. For the UHR group, we performed Spearman's correlation analysis to identify associations between SFS scores and five possible contributing factors (i.e. duration of prodromal symptoms and the four CAARMS subscale scores). We used Bonferroni corrections to correct for multiple (five) comparisons, and set the alpha at p < 0.01 for two-tailed analysis.
Results
Baseline characteristics and psychological symptoms
Table 1 shows baseline characteristics for all three groups. The two high-risk groups had significantly lower educational levels than the HC group, and GAF scores were significantly lower in the UHR group than in the GHR and HC groups. The three groups did not differ significantly in any baseline variables other than educational levels and GAF scores.
Baseline characteristics
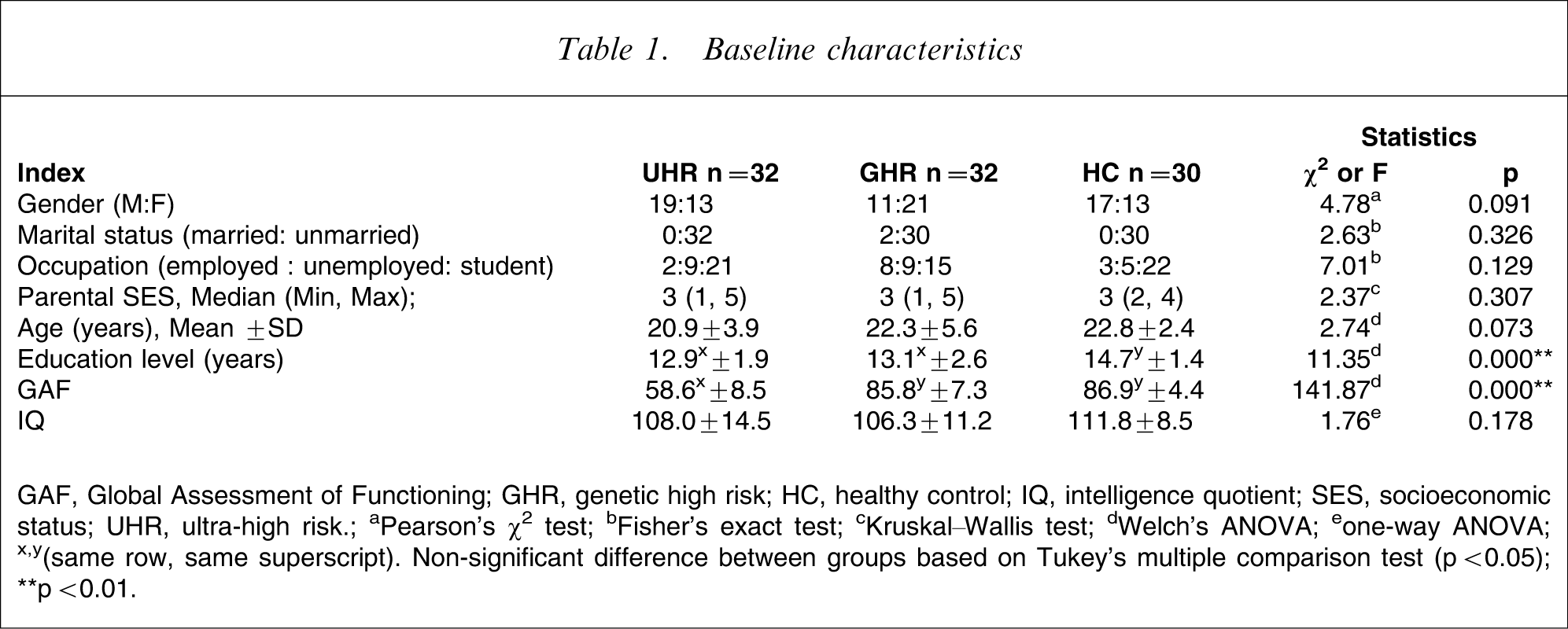
GAF, Global Assessment of Functioning; GHR, genetic high risk; HC, healthy control; IQ, intelligence quotient; SES, socioeconomic status; UHR, ultra-high risk.
aPearson's χ2 test; bFisher's exact test; cKruskal–Wallis test; dWelch's ANOVA; eone-way ANOVA; x,y(same row, same superscript) Non-significant difference between groups based on Tukey's multiple comparison test (p < 0.05); ∗∗p < 0.01.
Any subjects who concurrently fulfilled the UHR and GHR criteria were classified into the UHR group, so the GHR group's scores were almost normal on the CAARMS and other psychological symptom scales; the UHR group scored significantly higher on these scales, with the exception of Y-BOCS scores. The UHR group scored the following on CAARMS subscales: Positive (5.1±2.9), Negative (11.6±5.6), Disorganized (2.8±2.2), and General (12.4±6.8). On other psychological symptom scales, the UHR group scored as follows: BPRS (39.8±8.9), PANSS (52.8±9.7), SAPS (8.1±6.0), SANS (21.9±13.3), HAM-D (12.9±7.9), HAM-A (10.2±6.8), and Y-BOCS (1.3±4.0).
Comparison of SFS scores among the three groups
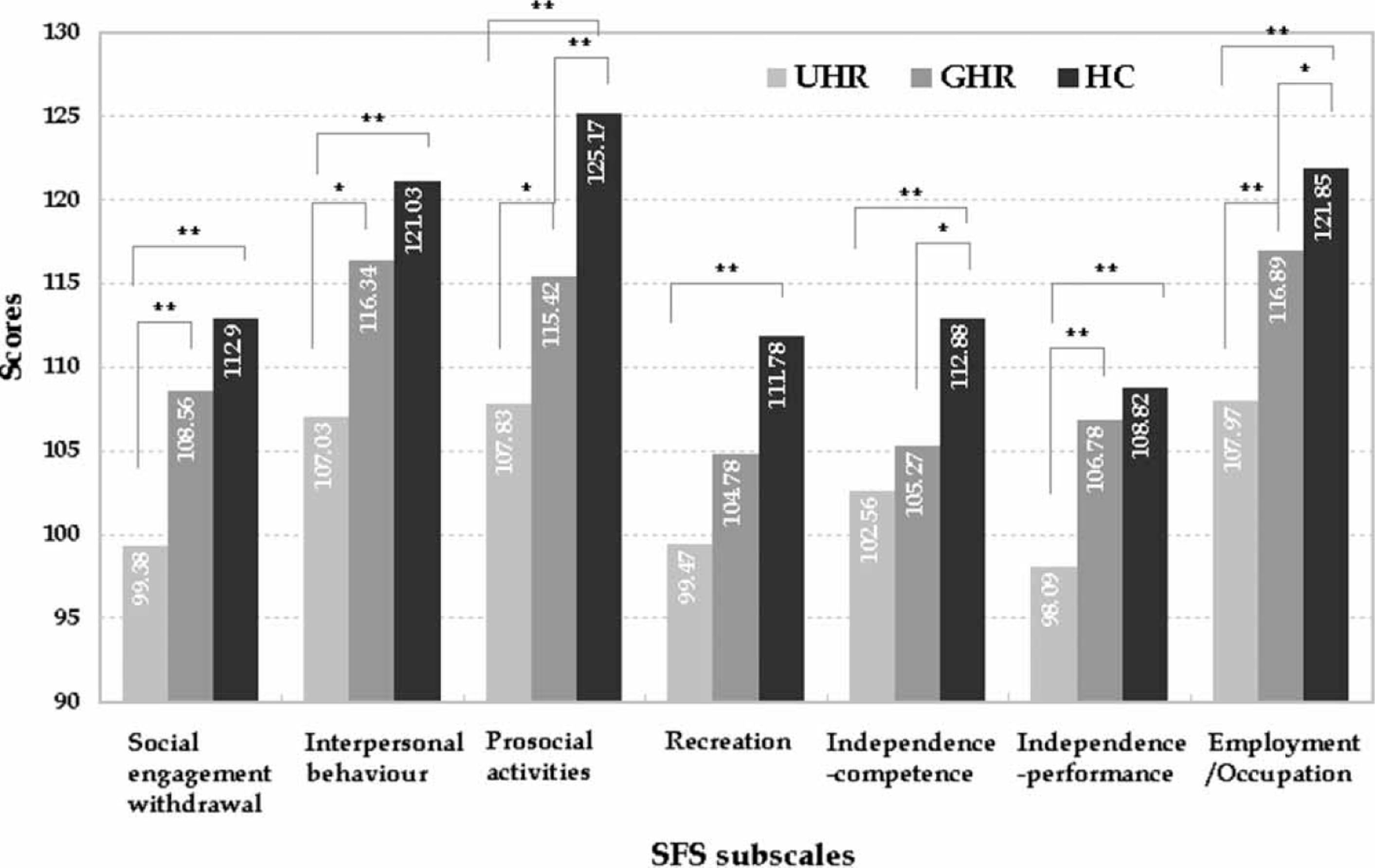
The three groups’ average scores on the SFS differed significantly, as did all subscale scores. Average SFS scores differed on Tukey's multiple comparison test (p < 0.01), with the lowest mean value being in the UHR group and the highest mean value being in the HC (Figure 1). The UHR group scored lower than the GHR and HC groups on three SFS subscales (social engagement/withdrawal, interpersonal behaviour, and independence–performance), while the two high-risk groups scored lower than the HC group on the independence–competence subscale (Figure 2). The three groups also differed with respect to prosocial activities and employment/occupation subscale scores. Groups differed least in recreation subscale scores. In the UHR group the seven subjects who met the VG criterion had lower average SFS scores (98.71±7.83) than the other subjects in the UHR group. But even after these seven subjects were excluded, the three groups differed significantly in average SFS scores.
Average scores on the SFS. GHR, genetic high risk; HC, healthy control; SFS, Social Functioning Scale; UHR, ultra-high risk. SFS subscale scores. Statistical significance was tested using one-way ANOVA except for ‘prosocial activities’ and ‘employment/occupation’ subscales, which were tested using Welch's ANOVA. ∗p < 0.05; ∗∗p < 0.01, Tukey's multiple comparison test. GHR, genetic high risk; HC, healthy control; SFS, Social Functioning Scale; UHR, ultra-high risk.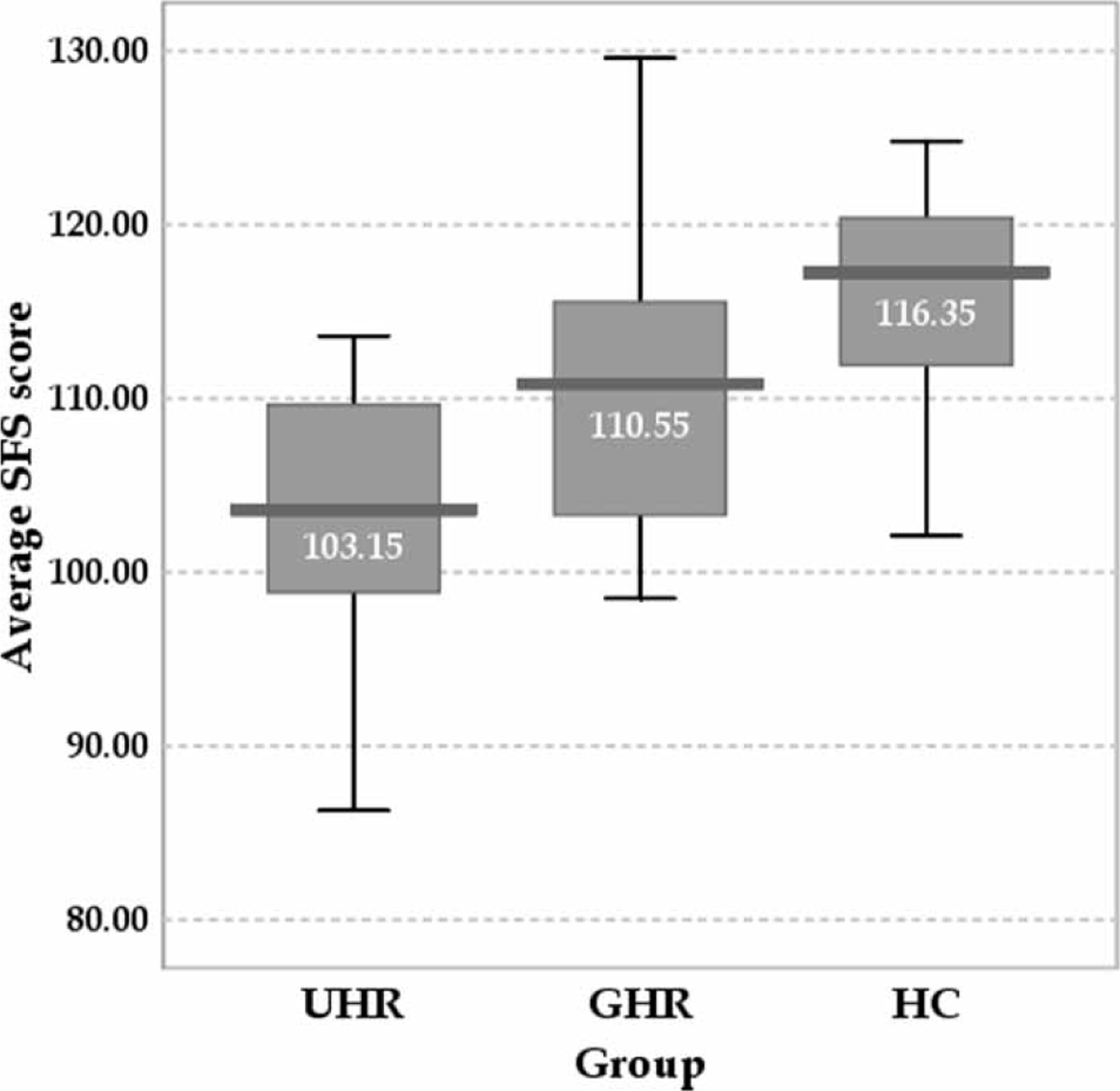
Relationship between duration and severity of prodromal symptoms and SFS scores in the UHR group
To investigate the underlying factors of social dysfunction, we analyzed the relationship between duration and severity of prodromal symptoms and level of social functioning. Prior to this, we confirmed no significant correlations between depression and anxiety symptoms, and level of social functioning. As shown in Table 2, duration of prodromal symptoms was correlated with the interpersonal behaviour and prosocial activities SFS subscales. After Bonferroni correction, however, only the interpersonal behaviour subscale was significantly correlated with duration of prodromal symptoms. The positive CAARMS subscale was not associated with SFS scores, and negative, disorganized and general CAARMS subscales were correlated with one or two SFS subscales. After Bonferroni correction, disorganized and general subscales were correlated only with the independence–competence subscale.
Prodromal symptoms and SFS correlations in the UHR group
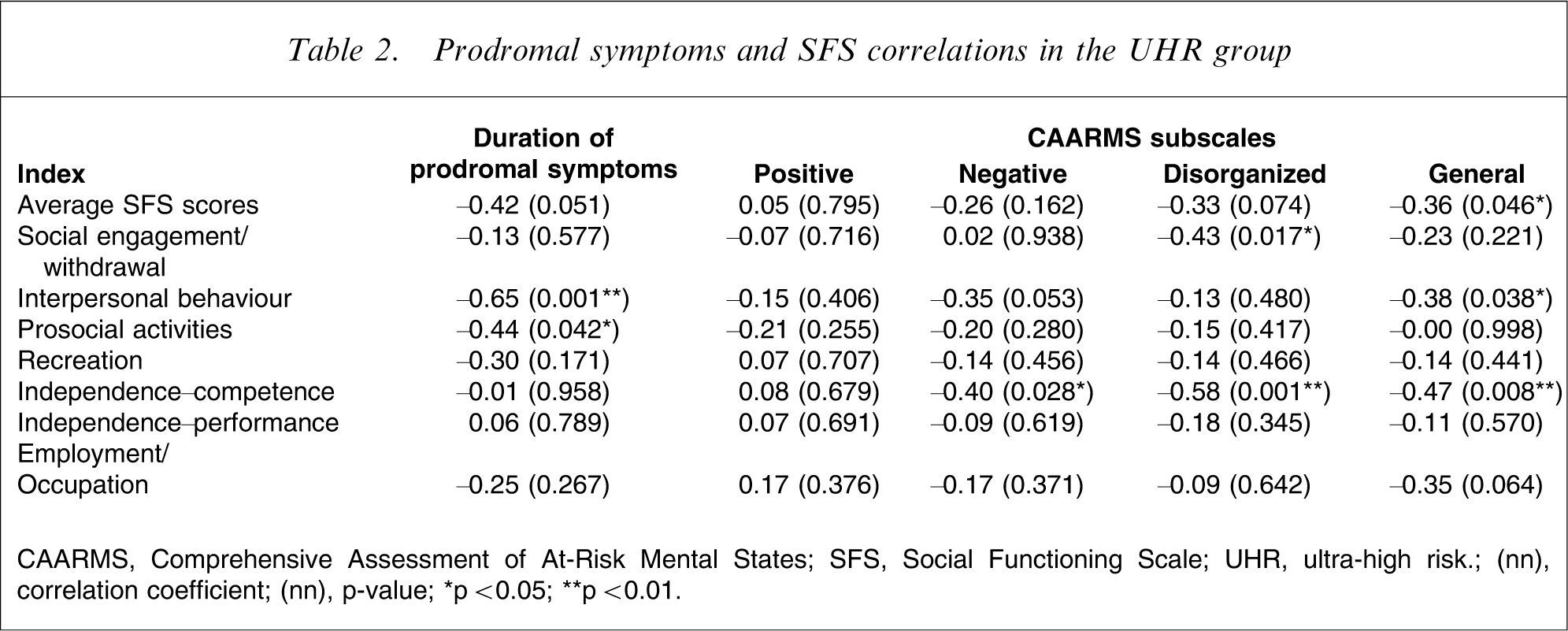
CAARMS, Comprehensive Assessment of At-Risk Mental States; SFS, Social Functioning Scale; UHR, ultra-high risk. (nn) correlation coefficient; (nn) p-value; ∗p < 0.05; ∗∗p < 0.01.
Discussion
To the best of our knowledge this is the first investigation comparing social functioning in subjects at clinical high risk for psychosis with genetically vulnerable subjects and healthy controls. As we anticipated, individuals at high risk for schizophrenia had significantly impaired social functioning, and UHR individuals exhibited more social dysfunction than GHR individuals. In the UHR group, duration of prodromal symptoms was related to impaired interpersonal behaviour. Consistent with previous findings, positive symptoms were not associated with social functioning. Negative symptoms, a well-known contributor to social dysfunction in patients with schizophrenia, were not significantly correlated with social functioning in UHR individuals. Instead, disorganized and general symptoms were strongly correlated with the independence–competence SFS domain.
The Age, Beginning, Course (ABC) study of schizophrenia reported that negative symptoms and functional impairment occurred following depressive symptoms [33]. Many retrospective studies have found social isolation to be one of the most frequently reported prodromal symptoms [34–36]. These findings suggest that in the UHR group, social functioning declined during the prodromal phase. Based on reports of poor social adjustment during childhood and adolescence among individuals who later developed schizophrenia, we postulate that social functioning of UHR subjects was impaired before the initiation of the prodromal phase, and further declined during this phase.
All studies of high-risk offspring have consistently reported significant social difficulties [37]. Approximately 10% of all first-degree relatives of patients with schizophrenia become psychotic [38] and >80% of the offspring of patients with schizophrenia do not develop any serious mental disorders [39]. Thus, social dysfunction in GHR individuals might be attributable to elevated rates of schizophrenia spectrum disorder among the relatives of patients with schizophrenia [40], [41]. In classical schizophrenia, the familial association of schizophrenia spectrum disorder is known to be caused mainly by genetic factors [42].
Meehl proposed a model in which a ‘schizogene’ produced a neural integrative defect called ‘schizotaxia’, which produces an observable phenotype called ‘schizotypy’ [43], [44]. Depending on environmental and genetic circumstances, schizotypy may involve only minor clinical symptoms or it may involve more severe, decompensated conditions, including schizophrenia [45]. Recently, schizotaxia itself has been proposed as a clinical condition, characterized by abnormalities in brain structure and function, cognition, affect, and social functioning among the non-schizotypal and non-psychotic relatives of patients with schizophrenia [46], [47].
High-risk subjects had relatively low educational levels compared with healthy controls, because the high-risk groups were slightly lower in age, and included more teenage students than the HC group. But an investigation of outpatients with schizophrenia found that years of education were not correlated with social functioning [48]. Thus, it is unlikely that educational level affected social functioning in the present study. Unlike SFS scores, GAF scores did not differ significantly between the GHR and HC groups. Because GHR subjects tend not to seek help, GAF scores (which mainly reflect the severity of psychiatric symptoms), might not be lower among GHR subjects than healthy controls.
In the UHR group, the interpersonal behaviour SFS domain was strongly correlated with duration of prodromal symptoms and partially correlated with general symptoms. This interpersonal behaviour domain assesses number of friends, heterosexual contact, and quality of communication. These correlations could be interpreted to indicate that individuals in the late putative prodromal phase exhibit more general symptoms, with the result that they find it difficult to maintain intimate relationships. The independence–competence SFS domain was related to disorganized and general symptoms in the UHR group. This domain evaluates the ability to perform the skills necessary for independent living. Self-assessment of one's own performance inevitably reflects self-esteem, which might be lower in anxious, depressive persons.
The present study found that duration of prodromal symptoms was partially related to social dysfunction. Previous studies have shown that early intervention, including medication and clinical management, effectively reduces prodromal symptoms [49] and improves the level of functioning [9]. McGorry et al. found that low doses of risperidone with targeted cognitive therapy and social support lowered the likelihood of transitions to psychosis [50]. It is widely known that environmental factors interact with genetic liability in a negative manner to produce disorders in the schizophrenia spectrum disorder [51], [52]. Accordingly, even if the most effective type of intervention for high-risk people is not yet established, early interventions to delay or prevent psychosis may include medication, social support, stress management, and social skill training.
The present study had several limitations; one major limitation was the underlying selection bias: self-selection of participants. We do not know much about the potential subjects who met the inclusion criteria but did not enter the study. In addition, the high-risk subjects were not representative of high-risk individuals demographically, limiting the generalization of our findings. Subjects at UHR are ‘at risk of developing psychotic disorder’, but do not inevitably progress to psychosis [53]. Some subjects at UHR might not be in the actual prodromal phase of schizophrenia, that is, they are false positives. These subjects might be diagnosed later with other psychiatric disorders such as anxiety disorder or mood disorder. Because the SFS is self-reported, depressive symptoms are likely to influence SFS scores. Moreover, the SFS instrument was originally developed to assess social functioning in a schizophrenia population, and is not sufficient to identify social dysfunction among young, pre-psychotic individuals.
In conclusion, the present findings support the hypothesis that impaired social functioning is both a trait and state marker of risk for psychotic disorders including schizophrenia. According to the model proposed by Nuechterlein et al.[54], social impairment seems to be a mediating indicator for vulnerability.
Footnotes
Acknowledgements
This paper was supported by a 21st Century Frontier Research Program grant (M103KV010013-07K2201-01310) from the Brain Research Center, funded by the South Korean Ministry of Science and Technology.
