Abstract
Worldwide, cervical cancer is the second most common malignancy in women, and is a major cause of morbidity and mortality. Accurate tumor staging is essential for optimal treatment planning and prognosis. Cervical cancer is staged by clinical examination according to the International Federation of Gynecology and Obstetrics staging system. However, clinical staging has inherent deficiencies in evaluating several parameters that are critical for treatment planning. It is now widely accepted that cross-sectional imaging, and in particular MRI, has an important role to play in the staging of these tumors. MRI is an excellent modality for depicting invasive cervical cancer: it can provide objective measurement of tumor size and provides a high negative predictive value for parametrial invasion and stage IVA disease. MRI and PET/computed tomography (CT) play key roles in identifying recurrent disease. PET/CT is also useful in detecting nodal and distant metastases and in radiotherapy planning. Diffusion-weighted MRI is an emerging imaging technique that is currently being evaluated for the detection of primary and recurrent disease and in the assessment of treatment response.
Keywords
 Medscape: Continuing Medical Education Online
Medscape: Continuing Medical Education Online
This activity has been planned and implemented in accordance with the Essential Areas and policies of the Accreditation Council for Continuing Medical Education through the joint sponsorship of Medscape, LLC and Future Medicine Ltd. Medscape, LLC is accredited by the ACCME to provide continuing medical education for physicians.
Medscape, LLC designates this educational activity for a maximum of
Learning objectives
Upon completion of this activity, participants should be able to:
Describe the accuracy of cervical cancer staging with the FIGO (International Federation of Gynecology and Obstetrics) criteria
Describe recommendations of the FIGO system for cervical cancer staging
Describe the FIGO staging criteria for cervical cancer
Compare the accuracy of different modalities for cervical cancer staging
Identify treatment options for different stages of cervical cancer
Worldwide, cervical cancer is the second most common cancer in women (after breast cancer), with more than half a million new cases diagnosed in 2005 [1]. In the UK, there were 2873 new cases in 2006 (8.5 per 100,000 of the population) with 949 deaths (age-standardized mortality of 2.4 per 100,000) [101]. The peak incidence occurs between the ages of 30 and 40 years [101]. During the last several decades, cervical cancer incidence and mortality rates have declined substantially in Western countries following the introduction of screening programs, which detect preinvasive disease on a cervical smear test [2]. However, the survival rates for patients who present with invasive cervical cancer have not improved over the same period.
Cervical cancer remains one of the most common cancers in women in developing countries, including Central and South America, parts of Africa and South Central Asia [101]. The most important etiological factor is exposure to human papillomavirus, particularly types 16 and 18. The introduction of the vaccine against human papillomavirus 16 and 18 is likely to have a major impact on disease prevention [3]. The most common histological type is squamous cell carcinoma, which accounts for approximately two-thirds of all cervical cancer cases. Adenocarcinoma and adenosquamous carcinomas account for 10–25% of cases [101]. Most patients with invasive cervical cancer are asymptomatic at its early phase, although patients with advanced disease typically present with abnormal vaginal bleeding.
In this article, we review the International Federation of Gynecology and Obstetrics (FIGO) staging classification of cervical carcinoma and discuss the role of diagnostic imaging in primary staging and in the detection of recurrence, with a particular emphasis on MRI. The role of MRI in the assessment of disease response to therapy is currently being investigated and is beyond the scope of this review.
International Federation of Gynecology & Obstetrics staging system & treatment
The most widely used staging classification of cervical carcinoma is that of the FIGO staging system. The FIGO staging system has undergone a number of revisions over the past few decades, most recently in 2009 (Table 1) [4,5]. Unlike the staging of the other gynecological cancers, the FIGO staging of cervical carcinoma is clinical and does not rely on surgico–pathological findings. This enables uniformity of staging for all patients worldwide. This is of particular importance because cervical carcinoma is most prevalent in countries where surgical and diagnostic resources are limited. The clinical assessment may be supplemented by other investigations, such as examination under anesthesia, cystoscopy, sigmoidoscopy and intravenous urography, but these are optional and no longer mandatory under the new revised FIGO staging system [4,5].
International Federation of Gynecology and Obstetrics clinical staging of cervical carcinoma and corresponding MRI findings.

FIGO: International Federation of Gynecology and Obstetrics; SI: Signal intensity.
Clinical staging has inherent deficiencies in evaluating several important parameters, including the assessment of parametrial and pelvic sidewall invasion and the accurate measurement of tumor size (particularly in endocervical or infiltrating tumors). Consequently, it has been recognized by the FIGO staging committee that clinical staging errors occur with reasonable frequency. When compared with surgical staging, clinical FIGO staging can be erroneous in up to 32% of patients with stage IB disease and up to 65% of patients with stage III disease [6,7].
The accurate pretreatment evaluation of these features is not only important for prognosis but also for determining the appropriate mode of treatment. This can vary between a primary surgical approach in early-stage disease (small tumors up to stage IIA1) and first-line radiotherapy with concurrent administration of platinum-based chemotherapy (chemoradiation) for patients with bulky IB2/IIA2 disease (tumors larger than 4 cm) or locally advanced disease (stage IIB or greater).
Owing to the previously mentioned short-comings of the clinical staging system, the FIGO staging committee has, for the first time, encouraged the incorporation of cross-sectional imaging (MRI/computed tomography [CT]) into the evaluation and treatment planning of patients with cervical cancer, if these resources are available [5].
The role of imaging in cervical cancer
Imaging plays a significant role in the evaluation of tumor size, detection of parametrial invasion and assessment of the involvement of the pelvic sidewall and adjacent organs, as well as assessment of nodal involvement and distant metastasis. Although nodal involvement is not part of the FIGO staging classification, nodal stage has marked prognostic implications and may alter the extent of radiotherapy fields.
MRI
Over the past few decades, the role of MRI in gynecological oncology has evolved. MRI is now a widely used imaging modality in the initial staging of primary cervical cancer. It has been given an appropriateness criteria rating of 8 (out of 9) by the American College of Radiology (ACR) in the staging of invasive cervical cancer [102]. The overall staging accuracy of MRI ranges from 77 to 90% [8–13]. In single-institution studies, MRI performs better than CT in the depiction of parametrial invasion and overall staging accuracy [11,13–15]. A recent prospective multicenter study undertaken by the ACR Imaging Network (ACRIN) and the Gynecologic Oncology Group (GOG) compared MRI with CT in patients with early invasive cervical cancer. This study demonstrated that MRI was equivalent to CT for overall preoperative staging. However, MRI performed significantly better for visualization of the primary tumor and detection of parametrial invasion. In addition, CT had much greater interobserver variability when compared with MRI [16]. With the widespread use of multidetector CT, it may be that, in the future, CT will regain a greater role but, at present, MRI remains the superior imaging modality.
Owing to its superior soft tissue delineation and multiplanar capability, MRI is ideal for the delineation of cervical tumors and is now an integral part of local staging for patients with cervical cancer in the UK. It is important for determining the feasibility of uterus preserving surgery, monitoring patient response and detecting recurrence, demonstrating the complications of the disease itself and of treatment and in planning radiotherapy. Furthermore, it has also been demonstrated to be cost effective by limiting or eliminating the need for further diagnostic tests or surgical procedures [15,17].
MRI technique
Imaging technique and patient preparation are critical to ensuring high-quality diagnostic images. The patient should be fasted for 4 h prior to the study in order to limit artifacts owing to small bowel peristalsis. An antiperistaltic agent should be administered to the patient to further reduce peristalsis [18]. It is helpful if the bladder is partially full at the time of the scan. Patients are imaged in the supine position using a surface phased-array coil. This provides higher signal-to-noise ratio than a body coil, with increased spatial resolution and reduced imaging time [19]. An anterior presaturation band is used to reduce breathing motion artifacts. Presaturation pulses above and below the imaged volume reduce intravascular signals from pelvic vessels.
The standard imaging sequences used in our institution are summarized in Table 2. We believe that these sequences will suffice to answer all the relevant clinical questions. Additional sequences, including short T1 inversion recovery, T1, T1 plus fat saturation and T1 plus fat saturation post-gadolinium, have demonstrated no advantage over the high-resolution T2 images [20–23]. Dynamic multiphase contrast-enhanced MRI (DCE-MRI) is a T1-weighted sequence, which produces a volumetric run of images at a stationary level in the body whilst the effect of intravenous contrast is observed. The initial image is precontrast, with subsequent images up to 4 min postcontrast. There are two instances in which DCE-MRI may prove useful. First, it may improve detection of small tumors with a depth of stromal invasion between 3.1 and 5.0 mm, demonstrating a reported sensitivity of 92% compared with 23% with T2-weighted MRI [24]. Small tumors enhance avidly in the early dynamic phase compared with the slight enhancement of the cervical epithelium and stroma. Second, it may help in distinguishing recurrent tumors from radiation fibrosis [25].
MRI sequences for imaging in cervical cancer.

FOV: Field of view.
MRI anatomy of the uterus & cervix
In a woman of reproductive age, the uterine corpus typically has three distinctive zones on T2-weighted spin-echo sequences (Figure 1). Central high signal intensity corresponds to the endometrium and any fluid in the endometrial cavity. The junctional zone, a band of low signal intensity between the endometrium and outer myometrium, corresponds to the inner third of the myometrium. The outer myometrium has an intermediate signal intensity, between that of the endometrium and junctional zone on T2-weighted sequences. The normal cervix is seen as a cylinder on sagittal images and as a round structure on axial oblique images where the typically low signal intensity ring represents the cervical stroma and the high signal intensity represents the cervical canal lined by the endocervical mucosa (the plicae palmatae) and containing secretions.

Anatomy of the uterus and cervix.
Tumor staging on MRI
The FIGO staging system can be readily applied to MRI appearances (Table 1), although there are a number of pitfalls that may be encountered (Table 3).
Pitfalls of MRI for staging cervical cancer.

FIGO: International Federation of Gynecology and Obstetrics.
Stage I
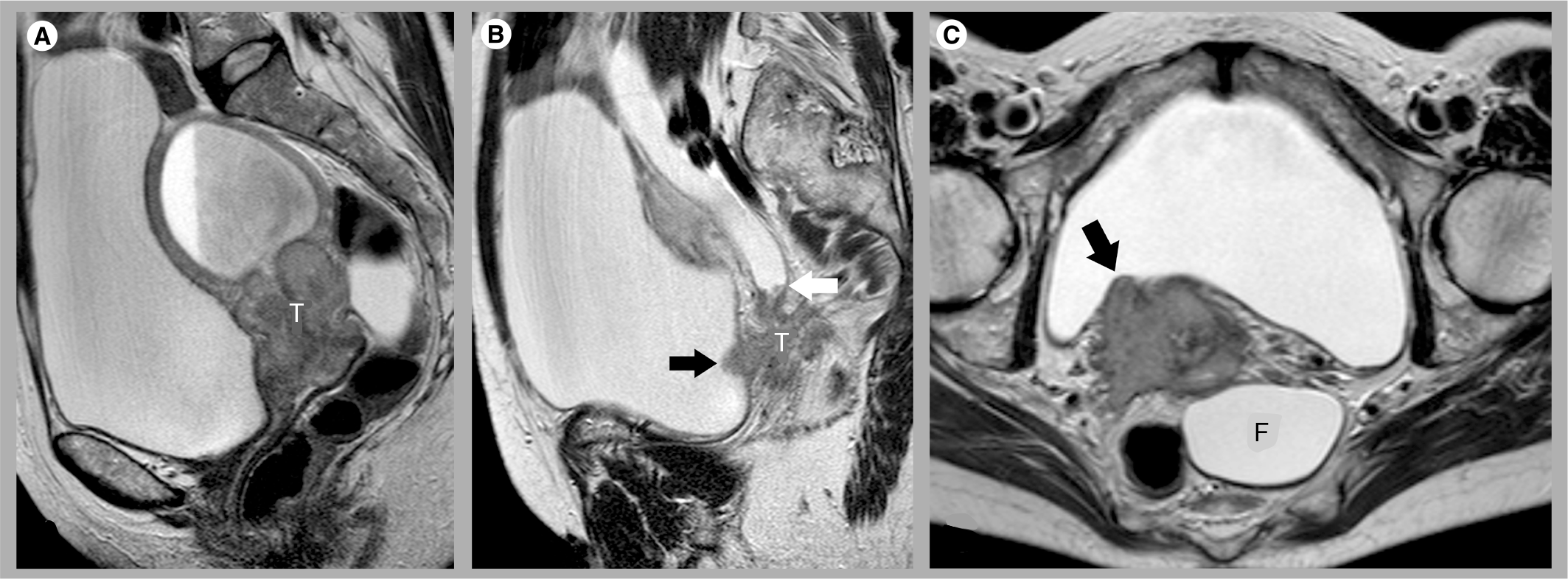
Stage I tumors are confined to the cervix. Stage IA is defined as a microinvasive tumor and cannot be detected on T2-weighted images. Stage IB tumors (confined to the cervix) typically appear as a hyperintense or intermediate signal-intensity mass relative to the low signal intensity of the cervical stroma on T2-weighted images. In young patients, in whom the cervix may have an intermediate signal intensity, the delineation of the tumor may be more challenging. Once tumor is assigned to stage IB, size needs to be taken into account as a crucial determinant of management. The size criterion of 4 cm distinguishes a stage IB1 (Figure 2) from a IB2 (Figure 3) tumor, and the latter is associated with an increased prevalence of nodal involvement [26]. Clinical evaluation is a poor estimate of actual tumor size, especially when the tumor primarily occurs in the endo-cervix. MRI has been shown to be highly accurate in the assessment of tumor size, assessing tumors within 5 mm of the surgical size in 70–90% of cases [8], with an overall accuracy of 93% [9]. In a recent retrospective study of 150 surgically staged patients, carried out in our own institution, 95% of tumors greater than 10 mm in size were within 8 mm of histologic size on MRI [27].
Stage IB1 cervical cancer.

Stage IB2 cervical cancer.
Stage II
In stage II, the tumor has grown beyond the cervix but does not involve the pelvic sidewall or the lower third of the vagina. In stage IIA, the tumor infiltrates the upper vagina without parametrial invasion. On MRI, this is detected as a loss of the low signal intensity of the normal vaginal wall, in contiguity with the tumor mass (Figure 4). MRI is highly sensitive (86–93%) in the depiction of vaginal infiltration [9]. Large tumors may cause a diagnostic challenge by stretching the vaginal fornices and falsely suggesting invasion. Some centers use ultrasound gel to distend the vagina in order to better demonstrate the vaginal fornices [28]. However, MRI of vaginal involvement is less crucial since it is relatively easy for the clinician to detect vaginal involvement on clinical examination.

Stage IIA cervical cancer.
In stage IIB, there is extension of tumor beyond the cervical stroma and into the parametrial tissues. The radiological sign for parametrial invasion is breach of the low signal intensity ring of cervical stroma, as seen on oblique axial high-resolution images (Figure 5). Additional features include a spiculated tumour-parametrium interface, soft tissue extension into the parametria and encasement of the periuterine vessels [29]. The accuracy for the detection of parametrial invasion on MRI ranges from 88 to 97%, sensitivity ranges from 44 to 100% and specificity ranges from 80 to 97% [8,10,21,27,30,31]. Preservation of the low signal intensity ring virtually excludes parametrial invasion with a negative predictive value of 94–100% [21,27,29]. However, the positive predictive value of disruption of the stromal ring is lower because it can be difficult to differentiate peritumoral edema from parametrial invasion, particularly in large tumors. It is the ability to rule out parametrial invasion on MRI with a high negative predictive value which is of the most importance clinically, by helping to confidently identify those patients suitable for radical surgery.

Stage IIB cervical cancer.
Stage III
In stage IIIA, the tumor extends down to the lower third of the vaginal wall (Figure 6). Where the tumor results in obstruction of one or both ureters, or if the tumor reaches the level of the vessels along the pelvic sidewall, the tumor has extended into the ‘surgical’ pelvic sidewall, consistent with stage IIIB disease.

Stage IIIA cervical cancer.
Stage IV
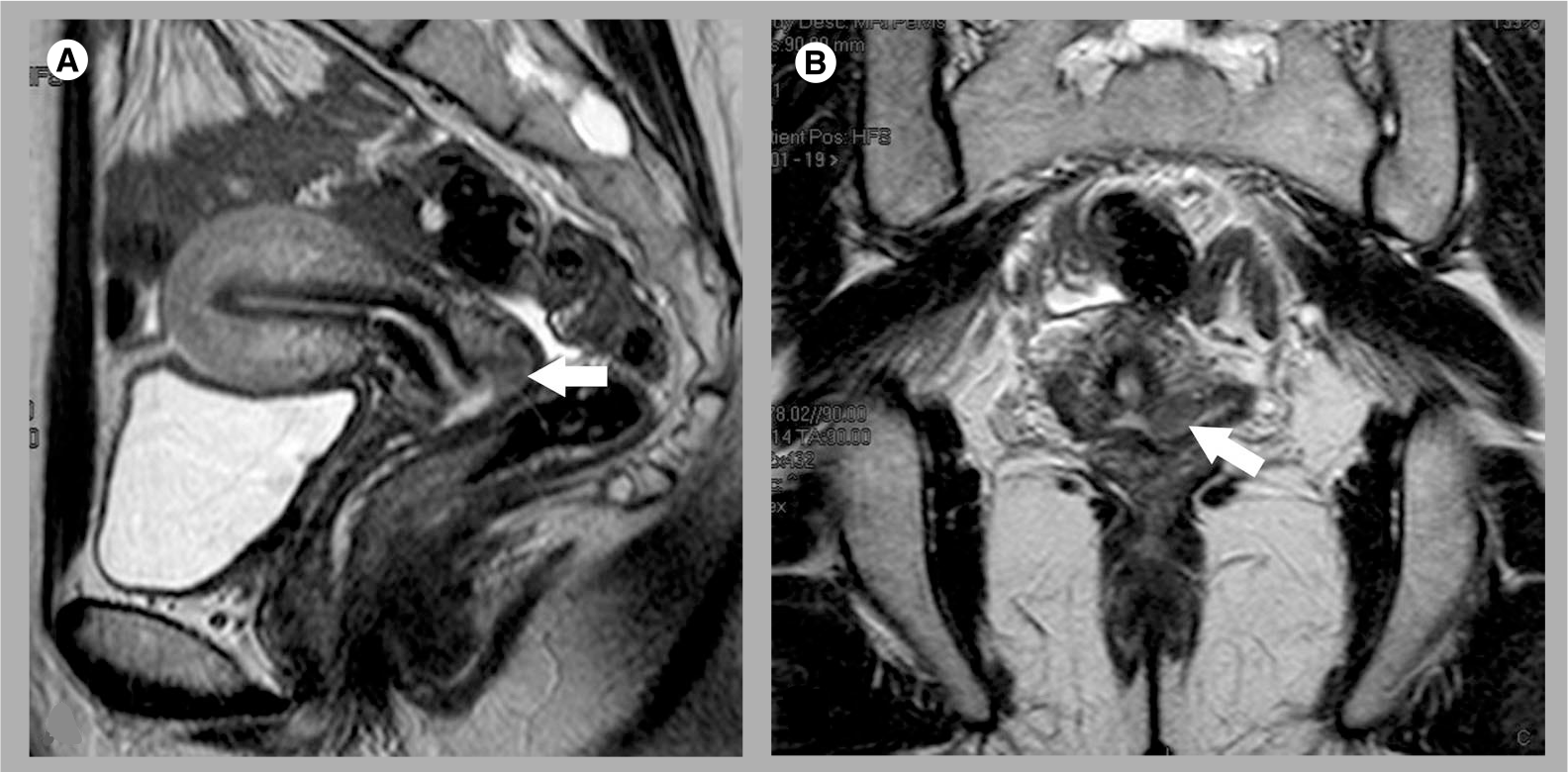
In stage IVA, the tumor invades the bladder or rectal mucosa. MRI findings suggestive of bladder invasion include disruption of the normal low signal intensity bladder wall, nodularity and irregularity of the wall, masses protruding into the lumen or the presence of bullous edema (Figure 7). Direct infiltration of the rectal wall is less common owing to the intervening pouch of Douglas and rectovaginal septum. Rectal invasion usually occurs via the uterosacral ligaments [32]. The reported sensitivity of MRI in the evaluation of bladder or rectal invasion is 71–100%, and the specificity is 88–91% [9,13,17]. If a low threshold for reporting bladder and rectal invasion is adopted, MRI has been found to have a very high negative predictive value of 100% [17], thereby negating the need for invasive cystoscopic or endoscopic staging.
Stage IVA cervical cancer.
Stage IVB indicates distant metastatic disease outside of the true pelvis. Although pelvic node metastases do not change the FIGO stage, paraaortic or inguinal metastases are classified as stage IVB (Figure 8).

Stage IVB cervical cancer.
Nodal staging on MRI
Within the pelvis, cervical cancer spreads first to the parametrial nodes, then to the obturator and iliac nodes. Although not incorporated in the FIGO staging system, the presence of lymph node metastases has significant prognostic and treatment consequences. The 5-year survival for node-positive patients is 39–54%, compared with 67–92% in patients without nodal involvement [33,34]. In early-stage tumors, the involvement of any node is important since it excludes curative surgery changing the treatment to chemoradiotherapy. In higher-stage tumors, the detection of nodes is important for planning the extent of radiotherapy treatment fields.
Size is the main criterion used to diagnose a nodal metastasis, and the most widely quoted threshold above which a node is considered malignant is 10 mm in short axis diameter. Other criteria used include a round shape (for nodes with a short axis diameter of between 8 and 10 mm) [35], central necrosis, soft tissue of the same signal intensity of the tumor within the node and extracapsular extension of the tumor beyond the nodal capsule [36]. The presence of visible necrosis within the node, seen as pockets of high T2 signal intensity, has a positive predictive value of 100% for nodal involvement [37].
Using standard size criteria, the sensitivity for detecting nodal metastases on MRI is low, ranging from 29 to 86%. This is due to the inability to detect micrometastases in normal-sized nodes. New techniques are being developed to improve the assessment of lymph node involvement. 18F-fluorodeoxyglucose (FDG)-PET/CT has proved useful for lymph node staging (see later). Lymph node-specific MRI contrast agents, such as ultrasmall superparamagnetic iron oxide particles, have been demonstrated to increase the sensitivity for the detection of nodal metastases with no loss of specificity [38]. In one study, Rockall
Diffusion-weighted imaging (DWI), now readily usable in the pelvis, achieves image contrast by evaluating the random motion of water molecules within tissues. It yields information on tissue cellularity, microcirculation and cell membrane integrity. DWI appears to be of value in both identifying and in characterizing abnormal tissue. The images generated should be looked at in conjunction with the apparent diffusion coefficient (ADC) map and the T2-weighted images (Figure 9). DWI has many favorable points: the additional scan time is relatively short and intravenous contrast is not needed, meaning it can be applied to patients with renal impairment and integrated into routine scanning protocols.

Stage IIB cervical cancer.
Chen
They demonstrated no significant difference in the ADC values between normal and infiltrated nodes [40]. By contrast, another study demonstrated that the ADC value of metastatic nodes was lower than that of benign nodes. The same study demonstrated that the addition of the DWI sequence increased the sensitivity for identifying lymph nodes, meaning the information could be used as a map to aid surgical planning, avoiding extensive lymphadenectomy and refining radiotherapy fields [41].
There are some pitfalls in the use of DWI and ADC values. As yet, there is no defined ADC value that is diagnostic for neoplastic tissue. Imaging parameters will change from center to center, making standardization of the imaging process difficult. It should be remembered that some normal tissues can present low ADC values; for example, benign lymph nodes, peripheral nerves, endometrium and bowel tissue. On the other hand, both well differentiated tumors and poorly differentiated necrotic tumors may have high ADC values. Readers should also be aware of the concept of T2 shine-through.
This refers to hyperintensity on T2-weighted images influencing the DWI, which is seen as high signal on high b-value images, along with a bright area on the ADC map. It highlights the need to assess the DWI in correlation with the ADC map [42]. DWI in the pelvis clearly requires further research before interpretation of these images can be made confidently. Preliminary anecdotal evidence at our institution suggests there is additional information to be gained.
FDG-PET/CT
The use of FDG-PET is now well established in cervical cancer since most cervical tumors are FDG avid. Adenocarcinomas, which usually have a low FDG uptake, are an exception [43]. FDG-PET/CT can be used at the time of presentation for staging and prognostication, to monitor response, to detect recurrence and to plan radiotherapy. However, FDG is not entirely specific for malignancy, and there are a number of pitfalls in using this radiotracer (Box 1).
In the context of primary tumor staging, PET/CT plays a valuable role in the evaluation of lymph node metastases. Nodal metastases are frequent in patients with advanced disease (i.e., FIGO stages IIB to IVB) and FDG-PET has been demonstrated to have a high specificity for the detection of nodes in this group of patients (Figure 10) [44]. Prospective studies have found sensitivities of75–100% and specificities of 87–100% [45–48]. FDG-PET also improves initial staging in cases of advanced disease by demonstrating unexpected sites of disease beyond the pelvis or retroperitoneum, such as supraclavicular nodal metastases [44]. In patients with advanced disease at presentation, PET or PET/CT has been found to alter management in a significant number of patients [49].

Stage IIB cervical cancer with recurrence.
By contrast, the value of FDG-PET in early-stage disease (i.e., FIGO stages I to IIA) is questionable. Many studies have reported low sensitivities for the detection of nodal metastases, ranging from 25–73%. Chao
Recurrent disease
Recurrent cervical cancer is defined as local tumor regrowth or the development of distant metastases at least 6 months after the lesion has regressed [50]. Residual disease is that which is evident within 6 months of primary treatment. The proportion of women treated for invasive cervical cancer who die from recurrent or residual disease is approximately 30% [51]. The average rate of recurrence is that approximately two-thirds of cases recur within the first 2 years following initial treatment, and 90% of cases recur by 5 years [52]. A retrospective study at our own center concluded that 70.4% of recurrences occurred within 1 year of starting treatment [53]. Recurrence following primary surgery is in the region of 27% [54]. Rates following fertility-sparing radical trachelectomy are favorable, with a figure of 4% quoted [55]. One small study following-up patients who were treated with concomitant chemoradiotherapy demonstrated a recurrence rate of 32% [56]. However, a direct comparison of the two treatment modalities should not be drawn because the stage of disease will determine which therapeutic path the patient will take; those with early disease having surgery and those with more advanced disease being managed with chemoradiotherapy.
There are some salvage treatment options available that have been demonstrated to prolong survival, particularly when the recurrence is detected at an early stage. Just as primary staging dictates the initial management, location of the recurrent disease and initial therapy will determine the course for subsequent treatment. Local recurrent disease in the vaginal cuff or pelvic sidewalls (Figure 11) may be treated with chemoradiotherapy, if this has not been given previously. In patients who have already received chemoradiotherapy, central recurrence limited to the cervix and/or vagina (Figure 12) may be treated with exenteration. Distant metastatic disease may be treated with chemotherapy. It is imperative to accurately identify those patients deemed suitable for such radical steps as pelvic exenteration, which is associated with considerable morbidity. Up to a third of women who elected to undergo exenteration were deemed unsuitable at the time of surgery, owing to the fact that the extent of disease was more advanced than was realized in the preoperation work-up [57].
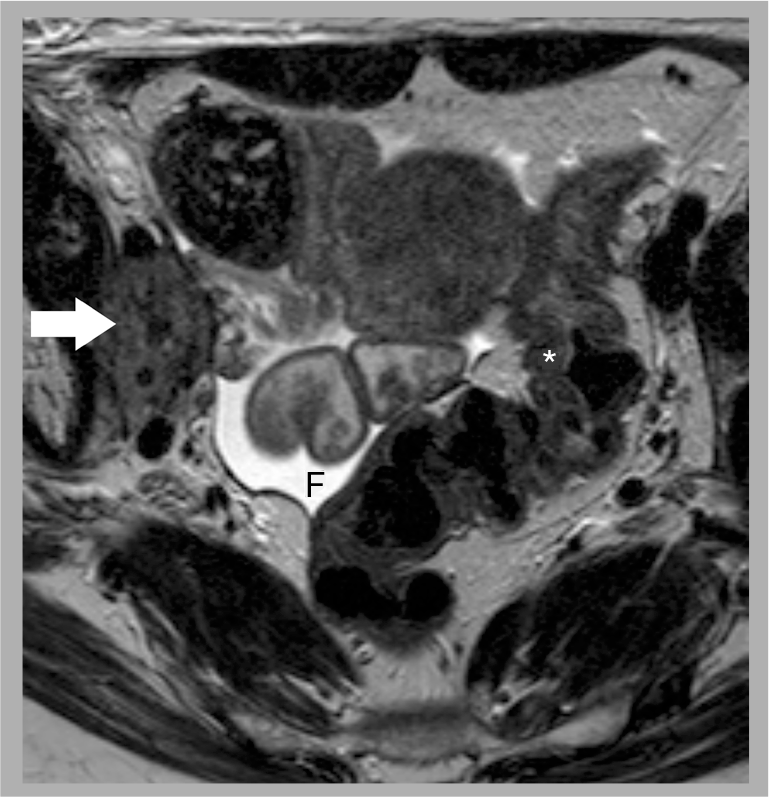
Pelvic sidewall recurrence following chemoradiation for cervical cancer.

Central recurrence within the cervix following chemoradiation for cervical cancer.
Pitfalls of using 18F-fluorodeoxyglucose-PET.
Physiological uptake occurs in metabolically active normal tissues (i.e., brain, bowel, genitourinary tract, salivary glands, etc.).
Inflammation or infection can be metabolically active.
Adenocarcinomas have a low FDG uptake and may not be detected.
In premeopausal women, physiological uptake in the normal ovaries and uterus can mimic nodal or parametrial disease.
Bladder activity can mask disease in the pelvis.
Focal areas of ureteral activity can mimic nodal disease.
Reactive lymph nodes can be FDG avid.
Necrotic lymph nodes may not be FDG avid.
FDG: 18F-fluorodeoxyglucose.
In the developed world, imaging in the assessment of response will usually begin with MRI. It can be particularly challenging in the first 6 months after chemoradiation to decipher residual disease from post-treatment changes. Where morphological imaging is suboptimal in this differentiation, the evolving paradigm of functional imaging aids diagnosis. These imaging aids include DCE-MRI, DWI and FDG-PET/CT.
Diffusion-weighted imaging is now being evaluated in the assessment of treatment response. In their study, Harry
One group has demonstrated a strong association between metabolic response and patient outcome. Grigsby
Routine follow-up has a relatively low yield in identifying recurrence [60]. Whilst clinical examination is still key, other tests that were traditionally performed at follow-up, such as Pap smears, have not been demonstrated to significantly aid detection of recurrence. There is no routine post-surgical imaging follow-up. This is only undertaken if it is indicated to be a result of clinical symptoms or signs. However, in patients who have received chemoradiation, post-treatment imaging is usually undertaken. If a small area of residual tumor is detected then there is a window of opportunity to offer exenterative surgery. These patients should undergo PET prior to surgery in order to establish there is no distant spread of disease. If, after chemoradiation, the patient has had a good response, they will not receive any routine imaging follow-up. Elit
CT and MRI play key roles in identifying recurrent disease, with both having reasonable sensitivities. On MRI, tumor recurrence appears as a region of intermediate to high T2-weighted signal amid the low signal intensity of the irradiated tissues. MRI is superior in distinguishing fibrosis and scarring from active disease, although the appearances can be indeterminate, particularly within the first 6 months following treatment. Delineation of fistulae is greater on MRI compared with CT.
Dynamic multiphase contrast-enhanced MRI has been assessed as a tool for detecting recurrence. Kinkel
On diffusion-weighted images, hyperintense signal on high
In the context of recurrence, the applications of PET/CT include identifying residual/recurrent disease at the primary site, assessment of nodal disease, detection of distant metastases and radiotherapy field planning [64]. Chung
In a retrospective study of patients suspected of having recurrence on the basis of an elevated serum squamous cell carcinoma antigen, PET/CT detected malignancy in 100% of these patients. The diagnostic accuracy for detecting recurrent cervical cancer was 96.8% (one patient was demonstrated to have a primary lung neoplasm). PET/CT was demonstrated to be more efficient at showing recurrence and displaying more lesions than other imaging modalities used [66].
Mittra
FDG-PET is proficient at restaging localized recurrent cervical cancer. Yen
Executive summary
The most important etiological factor is exposure to humanpapilloma virus, particularly types 16 and 18.
Cervical cancer rates continue to decrease in Western countries but is still the second most common cancer in women worldwide.
Squamous cell carcinoma accounts for approximately two-thirds of all cases.
Adenocarcinoma and adenosquamous carcinomas account for 10–25% of cases.
Cervical cancer is staged using the International Federation of Gynecology and Obstetrics (FIGO) staging system, which is clinical.
Clinical staging has inherent deficiencies in evaluating several parameters that are critical for treatment planning, including parametrial and pelvic sidewall invasion, estimation of tumor size and evaluation of lymph node metastases.
Clinical staging errors occur with reasonable frequency.
Radical surgery is the treatment of choice in young patients with early-stage disease.
Patients with bulky or locally advanced disease are treated with chemoradiotherapy.
Although information derived from cross-sectional imaging (i.e., MRI/computed tomography [CT]) is not included in the FIGO staging system, it can be used for treatment decisions.
Owing to its superior soft tissue resolution and multiplanar capability, MRI is now a widely used imaging modality in the initial primary tumor staging of cervical cancer.
MRI technique and patient preparation are critical to ensure high-quality diagnostic images.
In a woman of reproductive age, the uterine corpus typically has three distinctive zones on T2-weighted spin-echo sequences.
MRI is often normal in microinvasive (stage IA) tumors.
Stage IB tumors are confined to the cervix and appear as a hyperintense or intermediate signal intensity mass relative to the low signal intensity cervical stroma.
Stage IIA tumors infiltrate the upper two-thirds of the vagina and are detected as a loss of the normal low signal intensity of the vaginal wall.
Parametrial invasion classifies the tumor as stage IIB, which is identified as breech of the low signal intensity ring of cervical stroma on the oblique axial high-resolution images.
Preservation of the low signal intensity ring virtually excludes parametrial invasion with a negative predictive value of 94–100%.
Stage IIIA tumors extend down to the lower third of the vaginal wall. When the tumor extends to the pelvis sidewall (i.e., the pelvic musculature and iliac vessels) or causes hydronephrosis, it is defined as a stage IIIB disease.
Stage IVA tumors invade the mucosa of the bladder or rectum.
The high negative predictive value of MRI for stage IVA disease may obviate the need for invasive cystoscopy or endoscopy.
Stage IVB involves distant metastatic disease, including paraaortic or inguinal node metastases.
The presence of lymph node metastases does not alter the FIGO stage but has significant prognostic and treatment consequences.
The main criterion for diagnosis of lymph node metastasis is a short axis diameter above 10 mm.
Using standard size criteria, MRI has a low sensitivity (29-86%) for detecting nodal metastases owing to its inability to identify micrometastases in normal-sized nodes.
Lymph node-specific MRI contrast agents, such as ultrasmall superparamagnetic iron oxide particles, increase the sensitivity for detection of nodal metastases with no loss of specificity.
Most cervical tumors are 18F-fluorodeoxyglucose (FDG) avid.
In the context of primary tumor staging, PET/CT plays a valuable role in the evaluation of lymph node metastases.
In patients with advanced disease at presentation, PET or PET/CT has been found to alter management in a significant number of patients.
The value of FDG-PET in early-stage disease (FIGO I to IIA) is questionable.
Approximately a third of women treated for cervical cancer will suffer from recurrent disease, with the majority of those cases occurring within the first 2 years. Radical trachelectomy patients have a lower rate of recurrence of 4%.
Of women treated for invasive cervical cancer, 30% will die of residual or recurrent disease.
Salvage therapies include chemoradiation for patients who have previously been treated with surgery or pelvic exenteration for those who have already undergone chemoradiotherapy.
Functional imaging can be of considerable advantage in assessing treatment responses, particularly in the first 6 months, when residual/recurrent disease can be difficult to distinguish from pretreatment changes. These include dynamic multiphase contrast-enhanced MRI (DCE-MRI), diffusion-weighted imaging (DWI) and FDG-PET/CT.
Only patients who have received primary chemoradiation undergo routine imaging follow-up. MRI and CT are the main modalities utilized in the developed world. FDG-PET also has been found to be effective at identifying recurrence. Further evaluation is needed of the efficacy of PET/CT, DWI and DCE-MRI in diagnosing recurrent disease.
It is likely that the use of functional imaging will develop in the context of cervical cancer.
MRI sequences, including T2-weighted images, DCE-MRI and DWI may become incorporated into standard imaging protocols.
More work needs to be performed in order to establish the use of DWI in the detection of disease and recurrence, and in the assessment of responses to treatment.
Improved imaging techniques will allow for more accurate fusion of images for colocation with radiotherapy planning, including intensity-modulated radiotherapy. This can be enhanced by using flat beds for MRI scanning and careful positioning of the patient.
Use of 18F-fluorothymidine-PET/CT, a marker of cellular proliferation, may be of use in those cervical carcinomas that are not FDG avid, such as adenocarcinomas.
Conclusion & future perspective
Imaging has assumed increased importance in the management of patients with cervical cancer in recent years. MRI is now widely accepted as the most reliable imaging modality for staging and follow-up, and has now been recognized by FIGO as being a helpful adjunct to treatment planning. Excellent image quality is vital for accurate interpretation, and MRI protocols need to be optimized in order to avoid potential pitfalls. In addition to conventional imaging, future protocols may also incorporate functional imaging techniques that integrate both morphological and biological information. These novel imaging techniques offer promise as early indicators of therapy response and may prove to bevaluable in tailoring treatment regimes on an individual patient basis.
 MRI & PET scans for primary staging & detection of cervical cancer recurrence
MRI & PET scans for primary staging & detection of cervical cancer recurrence
To obtain credit, you should first read the journal article. After reading the article, you should be able to answer the following, related, multiple-choice questions. To complete the questions and earn continuing medical education (CME) credit, please go to http://www.medscape.com/cme/futuremedicine. Credit cannot be obtained for tests completed on paper, although you may use the worksheet below to keep a record of your answers. You must be a registered user on Medscape.com. If you are not registered on Medscape.com, please click on the New Users: Free Registration link on the left hand side of the website to register. Only one answer is correct for each question. Once you successfully answer all post-test questions you will be able to view and/or print your certificate. For questions regarding the content of this activity, contact the accredited provider,






Footnotes
CME Author
Editor
