Abstract
Background:
Inguinal lymph node (LN) metastasis and particularly the number of metastatic lymph nodes (NMLN) represent a determinant prognostic factor in vulvar squamous cell carcinoma (VSCC). However, the NMLN may be related to the number of removed LNs. Therefore, the lymph node ratio (LNR) reflects not only the burden of LN involvement but also the quality and extent of lymphadenectomy.
Objectives:
To investigate the value of the LNR and the count of LN on overall survival (OS) and recurrence-free survival (RFS).
Design:
This study is a retrospective, longitudinal, institution-based study.
Methods:
This study included 192 patients treated for VSCC at the Salah Azaiez Institute between 1994 and 2022. Clinical, pathological, and evolutionary data were reported. Survival curves were generated by the Kaplan–Meier method, and predictive factors of outcome were analyzed using Cox proportional hazards models.
Results:
Surgery consisted of a radical vulvectomy, hemivulvectomy, and pelvic exenteration in, respectively, 96.4%, 2.1%, and 1.6% of cases followed by adjuvant radiotherapy in 38.5% of cases. LN dissection was bilateral in 88.5% of cases. LNR = 0, LNR = 0–0.2, and LNR ⩾0.2 were recorded in, respectively, 64.7%, 22.1%, and 13.2% of cases. With a mean follow-up time of 35 ± 42.06 months, the 5-year OS was 52.5% and the 5-year RFS was 55.8%. On multivariate analysis, the independent prognostic factors of OS were the LNR (hazard ratio (HR) = 5.702; 95% confidence interval (CI) = 2.282–14.245; p < 0.0001), Federation of Gynecology and Obstetrics (FIGO) stage (HR = 2.089; 95% CI = 1.028–4.277; p = 0.042), and free margins (HR = 2.247; 95% CI = 1.215–4.155; p = 0.01). Recurrences were recorded in 37.5% of cases. Independent prognostic factors of RFS were the LNR (HR = 2.911; 95% CI = 1.468–5.779; p = 0.002), FIGO stage (HR = 1.835; 95% CI = 1.071–3.141; p = 0.027), and free margins (HR = 2.091; 95% CI = 1.286–3.999; p = 0.003).
Conclusion:
Surgical margin, FIGO stage, and LNR represent the independent prognostic factors of survival and LNR showed superior prognostic predictive accuracy compared with the revised 2021 FIGO staging system for predicting OS and RFS in VSCC.
Introduction
Vulvar cancer ranks as the fourth most common gynecologic malignancy, accounting for 5%–8% of all gynecologic cancers, and usually affects post-menopausal women with a median age of 68 years. 1 Despite the contribution of the sentinel lymph node (LN) technique in reducing postoperative complications and lymphedema in patients with node-negative vulvar squamous cell carcinoma (VSCC), radical vulvectomy combined with complete inguinofemoral lymphadenectomy represents the standard surgical approach for patients with LN involvement.
Numerous independent prognostic factors have been extensively outlined in the literature. Nevertheless, potentially pertinent data present a diverse and occasionally conflicting landscape. Nonetheless, the historical consensus remains that nodal status stands as the foremost independent prognostic determinant for survival and recurrence in VSCC.2,3 The 5-year overall survival (OS) rate for patients with positive LN (PLN) varies between 30% and 58.5%, while it increases within the range of 64.7% to 90.9% for those without LN involvement.4,5 Moreover, the number of metastatic lymph nodes (NMLN) serves as a significant prognostic factor in evaluating survival for vulvar cancer, and it was integrated into the 2009 International Federation of Gynecology and Obstetrics (FIGO) staging system. However, previous studies have indicated that a high NMLN does not necessarily translate to an elevated risk of death, and survival outcomes among patients with an identical count of PLNs may exhibit noteworthy disparities. 6 Consequently, it is worthwhile to explore whether patients with an equivalent NMLN but varying number of retrieved lymph nodes (NRLN) experience comparable survival outcomes.
Therefore, the lymph node ratio (LNR), defined as the ratio of the NMLN to the total NRLN, represents a reliable parameter reflecting not only the burden of LN involvement but also the quality and extent of inguinofemoral lymphadenectomy. In fact, numerous studies have underscored the LNR as an independent indicator of survival outcomes in solid tumor malignancies, encompassing breast, esophageal, gastric, and colorectal cancers.7 –9 Additionally, a high LNR has been associated with unfavorable survival in gynecologic cancers such as cervical, ovarian, and endometrial cancers.10 –12 However, the prognostic value of the LNR in VSCC remains unclear.
This cohort study aimed to examine the significance of both the LNR and the count of LN in predicting outcomes among patients with VSCC.
Material and methods
Patients
Data of patients diagnosed with primary vulvar cancer and surgically treated at the Salah Azaiez Institute of Oncology of Tunisia between 1994 and 2022 were retrospectively reviewed. Inclusion criteria were as follows: histopathologically proven VSCC and surgical treatment with or without lymphadenectomy. Exclusion criteria were: Patients with Vulvar cancer (VC) and histopathology other than Squamous cell carcinoma (SCC), those who underwent surgery at another center, patients treated with palliative chemotherapy/RT, a history of other malignancies, unexplained mortality, and unknown variables. Based on these criteria, a total of 192 cases of VSCC were included in this study.
The surgical procedure consisted of vulvar excision combined with surgical verification of regional LN status. Vulvar surgery consisted of hemivulvectomy, radical vulvectomy, or exenteration, depending on the size tumor and the coexistence of dystrophic and precancerous changes. Regional inguinofemoral lymphadenectomy was performed unilaterally or bilaterally. In the case of lateral tumors, defined as a lesion that does not cross the midline, and whose medial edge is located more than 10 mm from midline structures, only ipsilateral lymphadenectomy was performed. Sentinel lymph node biopsy (SNB) was performed in tumors smaller than 4 cm in case of negative clinical and ultrasound findings in the inguinofemoral lymphatic region; bilateral systemic lymphadenectomy was performed in case of positive sentinel node.
Adjuvant treatment either concurrent chemoradiation or radiation alone of the groins and pelvis was performed if more than one inguinofemoral LN was positive, if a metastasis measured more than 10 mm in diameter, or if extracapsular spread had been noted. Adjuvant radiotherapy to the vulva was recommended for patients with positive margins or close surgical margins (defined as tumor-free margin <8 mm based on histology), and it was considered for patients with more than one of the following risk factors: advanced tumor stage, large tumor size, deeper invasion and lymphovascular space invasion (LVSI). Neoadjuvant chemotherapy was administrated in patients with unresectable vulvar tumors that otherwise have required extensive surgery.
After treatment, patients were followed by gynecologic oncologists every 3 months for the first year, every 4 months for the second year, every 6 months from the third to the fifth year, and every year thereafter. Computed tomography scans were performed when clinically indicated.
The demographic and clinical data were searched from the medical records including age, underlying disease and comorbidities, risk factors, menstruation status, preoperative physical examination, treatment type, and postoperative complications.
Pathological reports were retrospectively analyzed for tumor size, depth of invasion, tumor grade (1: well-differentiated; 2: moderately differentiated; or 3: poorly differentiated/undifferentiated), LVSI, and perineural invasion (PNI), presence of intraepithelial neoplasia (VIN) and surgical margin. For tumor staging and classification, the revised 2021 FIGO stage classification was used. 1 The details of groin node status were reported including the number of retrieved and metastatic LN and the presence of extracapsular node invasion. The number of retrieved (NRLN) was subdivided in two groups: <12 LN and ⩾12 LN, and the NMLN was subdivided into four groups: zero, one, two, and three or more. The LNR was stratified into the following three risk groups, according to LNR as previously published in the report from Gynecologic Oncology Group (GOG) protocol #37 13 : 0.0, 0.0–0.2, and >0.2.
In the case of recurrent disease, its localization (local/groin/distant) and characteristics were analyzed. We have followed the STrengthening the Reporting of OBservational studies in Epidemiology (STROBE) Guidelines when preparing the manuscript.
Statistical analysis
Continuous normally distributed variables were expressed by their mean and standard deviation. Not normally distributed variables were expressed as medians, and their interquartile (IQR) ranges and categorical variables were expressed as n (%).
To compare the continuous variables with normal distribution, we used the t-test. In case of not normally distributed, we used the Mann–Whitney test. To compare the categorical variables, we used the chi-square or Fisher test if the assumption for the first was not complied.
OS was calculated from the date of surgery to the date of death or last follow-up. Recurrence-free survival (RFS) was defined as the interval from the date of the end of treatment to the earliest appearance of locoregional or distant relapse.
The RFS and specific OS rates were obtained using the Kaplan–Meier methods. In the case of comparing subgroups, we used the log-rank test. For the estimation of risk in the univariate or multivariate analysis, we used the Cox regression, expressing the hazard ratio (HR) and 95% confidence interval (CI). Statistical significance was considered to be at p < 0.05. Analyses were performed using IBM SPSS 18.0.
Results
Patient characteristics
Between 1994 and 2022, a total of 192 women underwent surgical treatment for VSCC. The characteristics of the patients are listed in Table 1. Three patients underwent neoadjuvant chemoradiation therapy for locally advanced tumors with a residual disease compatible with complete vulvar and LN surgery. Radical vulvectomy was performed in 185 (96.4%) cases and hemivulvectomy in four cases. Inguinal lymph node dissection (LND) was performed in 190 patients and was bilateral in 88.5% of cases. We omitted LND in two cases staged FIGO IA. Isolated SNB was performed in eight patients (4.2%). Of all patients who underwent inguinofemoral lymphadenectomy (n = 182), 179 patients underwent primary inguinofemoral lymphadenectomy, whereas three patients underwent inguinofemoral lymphadenectomy subsequent to SNB procedure during the learning curve (with standard inguinofemoral lymphadenectomy after Sentinel lymph node (SLN) procedure) in two cases and because of positive sentinel LN in one case.
Clinical, surgical, and histological characteristics of patients.

CT: chemotherapy; LND: lymph node dissection; LNR: Lymph node ratio; LNS: lymph node status; LVSI: lymphovascular space invasion; N−: negative lymph node; N+: positive lymph node; NE: not evaluated; NMLN: Number of metastatic lymph node; NRLN: Number of retrieved lymph node; PNI: perineural invasion; R0: free tumor margin ⩾8 mm; RT: radiation therapy; RTCT: radiochemotherapy; VIN: vulvar intraepithelial neoplasia.
Postoperative complications were recorded in 77 cases (40.1%). Medical complications were dominated by thromboembolic accident in 4 cases (2.1%) and urinary infection in 2 cases (1%). Early surgical complications were dominated by wound infection recorded in 19 cases (9.9%), wound dehiscence in 15 cases (7.8%), hematoma in 3 cases (1.6%), and rectal fistula in 2 cases (1%). Surgical revision was necessary in 10 cases (5.2%): 3 cases of wound infections, 3 cases of wound dehiscence, 2 cases of bleeding, and 2 cases of rectal fistula. Late surgical complications were dominated by lymphoedema in 19 cases (9.9%) and lymphocyst in 15 cases (7.8%). The histological data are summarized in Table 1. Adjuvant radiation therapy was indicated in 75 cases (39.1%). The target was the vulva in 31 cases (41.3%), the groin in 5 cases (6.7%), and both the vulva and groin in 38 cases (50.7%).
The predictive factors of LN involvement:
On univariate analysis, the predictive factors of LN involvement were age younger than 70 years (41.8 versus 26.2% in patients aged more than 70 years; p = 0.027), the presence of LVSI (100% versus 39.8%; p = 0.037) and PNI (80% versus 35.4%; p = 0.001), the tumor size exceeding 40 mm (45.8% versus 28.8%, p = 0.017), and T stage (34.2% in pT1 versus 38.5% in pT2 and 66.7% in pT3; p = 0.048). Multivariate analysis revealed that independent factors of Lymph node metastasis (LNM) were the presence of PNI (OR = 0.298, 95% CI = 0.174–0.686; p = 0.001) and the tumor size (OR = 0.199; 95% CI = 0.021–0.379; p = 0.029).
Relapses and RFS
The mean follow-up period was 35 ± 42.06 months. During follow-up, 120 (62.5%) patients were in clinical remission and 72 (37.5%) had recurrence with 73.6% (n = 53) of relapses occurring in the first 2 years. Among the 72 women with recurrent disease, 54 patients (28.1%) had an isolated locoregional recurrence, 11 (5.7%) had an isolated distant relapse, and 7 (3.6%) presented both local and distant recurrence. A local recurrence in the vulva was recorded in 38 patients (19.2%) and a groin recurrence in 35 patients (12%). Predictive factors for locoregional recurrence are detailed in Table 2.
Factors associated to groin and vulvar recurrence.
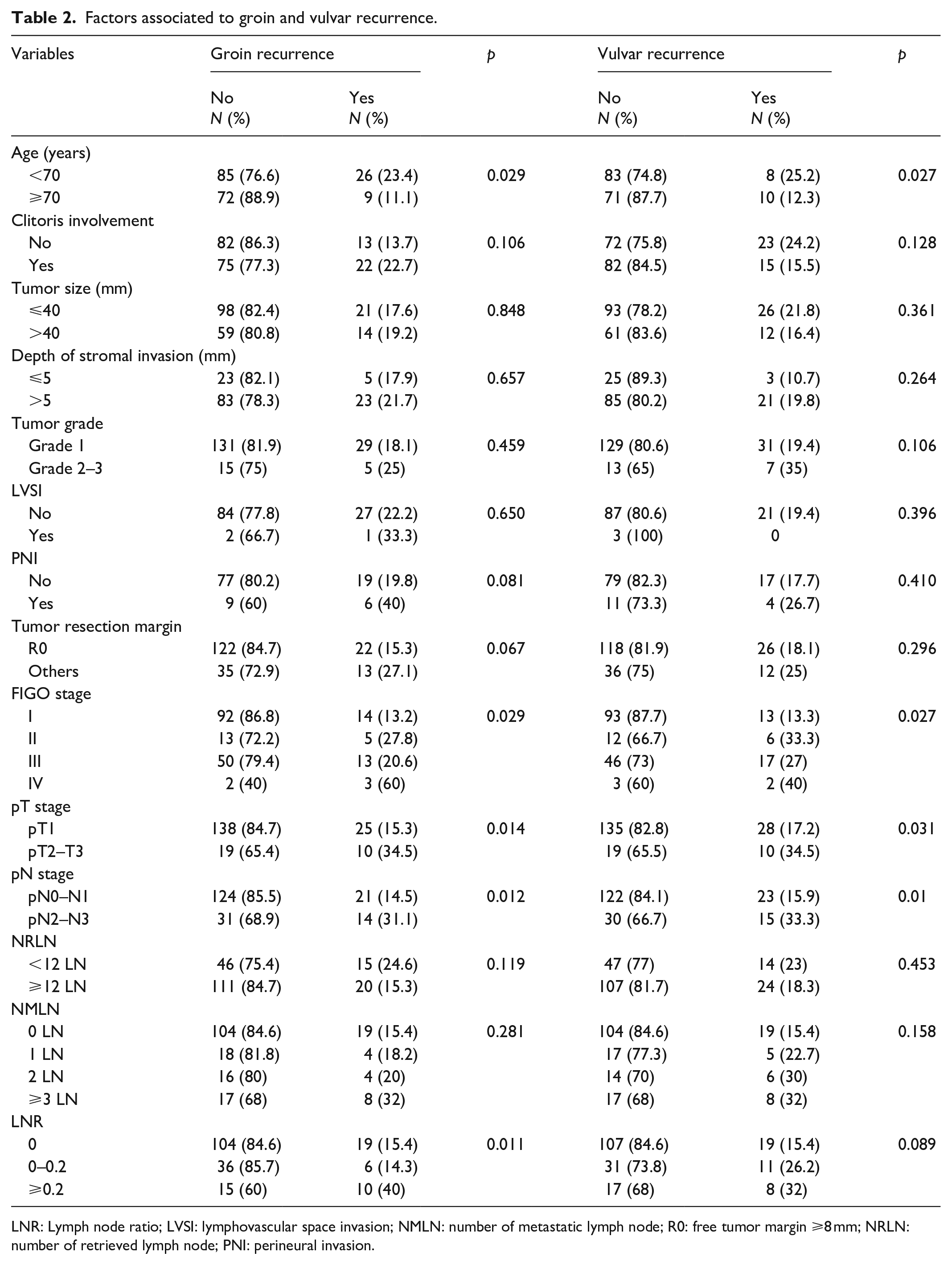
LNR: Lymph node ratio; LVSI: lymphovascular space invasion; NMLN: number of metastatic lymph node; R0: free tumor margin ⩾8 mm; NRLN: number of retrieved lymph node; PNI: perineural invasion.
The RFS rate was 55.8% at 5 years and 36.4% at 10 years. On univariate analysis, no significant correlation was found between the RFS and the age, the clitoris invasion, the tumor size and grade, the depth of invasion, and the presence of LVSI (Table 3). However, the 5-year RFS was significantly decreased with urinary meatus invasion (41.7% versus 56.6%; p = 0.047), advanced FIGO stage (p < 0.0001) (Figure 1), advanced pT stage (83.3% versus 57.4% versus 43.6% versus 33.3% in pT1a, pT1b, pT2, and pT3, respectively; p = 0.03) (Figure 2), the presence of PNI (20.8% versus 60.8%; p = 0.001) and invaded resection margins (41% versus 60.7%; p = 0.003) (Figure 3).
Univariate analysis of prognostic factors for overall survival and RFS in vulvar cancer.

LNR: Lymph node ratio; LNS: lymph node status; LVSI: lymphovascular space invasion; N-: negative lymph node; N+: positive lymph node; NMLN: Number of metastatic lymph node; NRLN: Number of retrieved lymph node; PNI: perineural invasion; R0: free tumor margin ⩾8 mm.

Kaplan–Meier survival curves of RFS (a) and OS (b) according to the FIGO stage.

Kaplan–Meier survival curves of RFS (a) and OS (b) according to the stage pT.

Kaplan–Meier survival curves of RFS (a) and OS (b) according to the surgical margin.
The presence of node invasion significantly reduced the 5-year RFS (p < 0.0001), particularly in the presence of more than three invaded nodes (30.7% versus 48.8% and 39.1% in the case of two and one invaded nodes respectively; p < 0.0001) (Figure 4) and the 5-year RFS was significantly correlated to the pN stage (p < 0.0001) (Figure 5).

Kaplan–Meier survival curves of RFS (a) and OS (b) according to the NMLN.

Kaplan–Meier survival curves of RFS (a) OS (b) according to the stage pN.
The subgroup analysis of patients with PLN did not reveal a significant variation in the rate of RFS based on the NMLN (43.8% with one PLN, 39.1% with two PLN nodes, and 30.7% with more than three PLN, p = 0.546)
We found no significant difference in the 5-year RFS according to the number of retrieved LN (38.2% in patients with an NRLN < 12 versus 64.9% in those with a NRLN ⩾ 12; p = 0.092). However, the subgroup analysis revealed that a NRLN ⩾ 12 was significantly associated to a better 5-year RFS compared to NRLN < 12 in patients aged more than 70 years old (21.3% versus 8.6%; p = 0.001), in stage pT1 (70.1% versus 38.9%; p = 0.016), patients without LNM (81% versus 46.5%, p = 0.032), patients with three or more LNM (33.6% versus 0%, p = 0.027), in case of R0 resection (74.8% versus 36.6%; p = 0.005) and in the absence of LVSI (64.9% versus 28.7%, p = 0.046) (Figure 6).

Kaplan–Meier survival curves of RFS (a) and OS (b) according to the LN ratio.
Survival curves revealed that the LNR was inversely proportional to RFS, and the 5-year RFS rates were 65.8%, 45.7%, and 20.5% for a ratio of 0, 0–0.2, and ⩾0.2, respectively (Figure 7).

Kaplan–Meier survival curves of RFS according to the number of retrieved LN in the subgroups of patients aged ⩾70 years, stage T1, stage N0, NMLN ⩾3 LN, R0 resection, and without LVSI.
The results of multivariate Cox regression analyses revealed that the independent prognostic factor of RFS were the LNR (HR = 2.911, 95% CI = 1.468–5.779; p = 0.002), FIGO stage (HR = 1.835, 95% CI = 1.071–3.141; p = 0.027), and free margins (HR = 2.091, 95% CI = 1.286–3.999; p = 0.003) (Table 4).
Multivariate analysis of prognostic factors for OS and RFS in vulvar cancer.

HR: hazard rate, 95% CI: confidence interval, LNR: lymph node ratio.
Predictive factors of OS
The OS rate was 52.5% at 5 years and 32.9% at 10 years. On univariate analysis, no significant correlation was found between the OS and the age, the clitoris invasion, the tumor size and grade, and the depth of invasion (Table 3). However, we found a significant decrease of the OS with urinary meatus invasion (30% versus 53.4%; p = 0.006), advanced FIGO stage (p < 0.0001) (Figure 1), advanced pT stage (75% versus 53.5% versus 46% versus 0% in pT1a, pT1b, pT2 and pT3 respectively; p = 0.015) (Figure 2), the presence of PNI (20.8% versus 60.8%; p = 0.001), the presence of LVSI (0% versus 51.9%; p < 0.0001), and invaded resection margins (36.3% versus 58.3%; p = 0.002) (Figure 3).
The presence of node invasion significantly reduced the 5-year OS (p < 0.0001), particularly in the presence of more than three invaded nodes (Figure 4) and the 5-year OS was significantly correlated with the pN stage (64.2% versus 44.1% versus 27.2% versus 0% in stage pN0, pN1, pN2, and pN3 respectively; p < 0.0001) (Figure 5).
The subgroup analysis of patients with PLN observed a degradation in OS rates based on the NMLN without reaching statistical significance (45.8% with one PLN, 32.5% with two PLN, and 19.5% with more than three PLN, p = 0.059).However, compared with patients with a single PLN, the 5-year OS of patients with more than three PLN was significantly impaired (p = 0.045). There was a trend toward decreased OS in patients with more than three PLN compared with those with two PLN (p = 0.068), whereas no difference could be demonstrated regarding OS between the groups with one and two affected LN (p = 0.899).
We found no significant difference in the 5-year OS according to the NRLN (37.6% in patients with a NRLN <12 versus 57.8% in those with an NRLN ⩾12; p = 0.976). Survival curves revealed that the LNR was inversely proportional to OS, and the 5 years OS rates were 64.2%, 43.4%, and 11.6% for a ratio of 0, 0–0.2, and ⩾0.2 respectively (Figure 7).
The results of multivariate Cox regression analyses revealed that the independent prognostic factor of OS were the LNR (HR = 5.702, 95% CI = 2.282–14.245; p < 0.0001), FIGO stage (HR = 2.089, 95% CI = 1.028–4.244; p = 0.042), and free margins (HR = 2.247, 95% CI = 1.215–4.155; p = 0.01) (Table 4).
Discussion
In our study, we evaluated predictive prognostic factors for survival and recurrence in VSCC, focusing on nodal status defined by the NMLN, the NRLN, and the interaction between these two parameters, expressed as the LNR. In analyzing the results of the current study, we must underline some clinical aspects of this series. Approximately 42% of patients were aged more than 70 years, distribution of disease stage involved 32.8% with stage III and 38% of tumors were >4 cm, and 35.9% of patients had LN metastases. We were able to identify several prognostic factors for survival and recurrence, namely, the presence of PNI and LVSI, pT stage and LN involvement, and consequently the FIGO stage, surgical resection quality and LNR. In our study, we demonstrated that a high LNR exceeding 0.2 was associated with a 31.8% reduction in OS rate and a 25.2% reduction in RFS rate, with a significant increase in the risk of groin recurrence. Furthermore, in multivariate analysis, this parameter exhibited a prognostic value superior to that of FIGO stage and surgical margins.
Numerous independent prognostic factors have been extensively outlined in literature. Nevertheless, potentially pertinent data present a diverse and occasionally conflicting landscape. Nonetheless, the historical consensus remains that nodal status stands as the foremost independent prognostic determinant for survival and recurrence in VSCC.2,3 The 5-year OS rate for patients with PLNs varies between 30% and 58.5%, while it increases within the range of 64.7%–90.9% for those without LN involvement.4,5 Beyond the presence of LNM, the NMLN stands as a crucial predictor in evaluating survival rates for VSCC and was incorporated into the 2009 International FIGO staging system bringing major modifications from original 1988 staging system, especially the definition of stage III cancer. 14 A meta-analysis including fourteen retrospective studies, revealed distinct 5-year OS and progression-free survival (PFS) rates according the NMLN : 84.5% and 86.6% for patients without affected nodes, 58.5% and 48% with one PLN, 47.4% and 40.3% with two PLNs, and 30.1% and 27.6% with more than three PLNs, respectively. 15
Nevertheless, several past studies have indicated that a higher NMLN does not necessarily correlate with an elevated risk of recurrence and mortality. In our study, although univariate analysis had shown that a high NMLN was associated with a significant reduction in 5 years OS and RFS, multivariate analysis did not identify this parameter as an independent prognostic factor. Moreover in patients with PLN, there was no significant variation in the rate of RFS and OS based on the NMLN.
In the study of Woelber et al. including 157 patients, the 2-year disease-free survival (DFS) was significantly compromised in all patients with PLN (88% in node-negative patients; 60%, 43%, and 29% in patients with 1, 2, and ⩾2 affected LN, respectively; p < 0.001). However, no significant difference in DFS was observed among the various node-positive subgroups (p = 0.430), and the multivariate analysis revealed that the impact of the NMLN varied significantly depending on the adjuvant treatment received, 16 which was in line with our results. Tabbaa et al. 17 had conducted a retrospective study of 468 cases that were staged according to both the FIGO 1988 and FIGO 2009 classification systems, demonstrating that among the node positive cases, the FIGO 2009 classification results in slight differences in cancer-specific survival (CSS), primarily between intra- and extracapsular disease and not according to the NMLN and LNM diameter.
Additionally, our study did not observe a substantial elevation in the risk of groin or vulvar recurrence, consistent with the results from the Vulcan trial involving 1535 cases of VSCC indicating that the NMLN serves as an independent factor for distant recurrences but not for local recurrence (HR = 1.10, 95% CI (1.2–1.21); p = 0.83). 2
The prognostic value of LN count in vulvar cancer is significant and is often used as a key factor in determining the stage and prognosis of the disease. However, the exact number of LNs that need to be examined for accurate staging and prognosis can vary based on individual cases and guidelines. Additionally, significant diversity appears in individual anatomical structures, variability in surgical approaches, and in laboratory methods employed for node retrieval and assessment. 18 In our study, the median number of nodes removed was 14 LNs.
We observed no significant differences in survival rates or groin recurrence rates based on the NRLN. However, our subgroup analysis indicated that retrieving more than 12 LNs was associated with an increased 5-year RFS rate among patients with nodal metastases, especially when the NMLN was ⩾3 LN. These findings consisted with those in the study of Baiocchi et al. involving 158 patients with VSCC who underwent bilateral inguinofemoral lymphadenectomy showing that a higher NRLN did not impact PFS significantly (p = 0.92). Additionally, there was no substantial difference in exclusive groin recurrence between patients with <12 LN (28.6% of recurrences) versus ⩾12 LN (23% of recurrences) (p = 0.74). However, they noted that resection of fewer than 12 LN in cases of node-positive disease negatively impacted PFS (20.8% versus 52.8%; p = 0.003). 19 Furthermore, in the study of Wu et al. 20 involving 703 patients diagnosed with node-positive VSCC, the prognostic relevance of the NRLN was definitively established. Their findings suggested that extracting more than 6 LNs improved the 3-year CSS rates (48.9%, 65.9%, 73.1%, and 67.3% for patients with 1–6, 7–10, 11–16, and 17–45 RLNs, respectively; p < 0.001).
Therefore, we sought to determine if the extent of lymphadenectomy as determined by the NRLN impacts OS and the RFS in patients with node-negative disease through the potential removal of such micrometastatic disease. Following the analysis of this subgroup, we did not identify any influence of the NRLN on OS. Nevertheless, an NRLN of <12 LNs resulted in a 34.5% decrease in the 5-year RFS rate. In the study of Gadducci et al., 21 among the 47 women who underwent bilateral inguinal-femoral lymphadenectomy with histologically proven negative nodes, groin recurrence occurred in three out of the 25 (12%) patients who had ⩽15 removed nodes compared to none of the 22 (0%) patients who had >15 nodes removed (p = 0.2368). These findings were consistent with our results, suggesting that removing more LNs reduces the potential for LN micrometastases, thereby decreasing the risk of relapse. Additionally, the extensive study of Courtney Brooks et al. 22 encompassing 1030 cases of node-negative VSCC revealed that harvesting more than 10 LN in stages II and III of FIGO had a significant impact on 5-year OS (p = 0.04 and p = 0.03) and 5-year RFS for stage III (p = 0.02). These findings suggest the necessity of a comprehensive inguinofemoral LND in patients with advanced-stage node-negative vulvar carcinoma. More recently, Panici et al. 23 assessed the prognostic role of nodal staging in patients over 75 years old, indicating that the removal of fewer than 10 LNs during unilateral inguinal dissection and fewer than 15 LNs during bilateral dissection had a negative impact on survival (p < 0.006 and p < 0.0001). These findings are consistent with our study, where retrieval of over 12 LNs resulted in a 12.7% increase in the 5-year RFS rate among patients aged over 70 years. These results imply that advanced age does not justify a reduction in the extent of LN surgery.
These mixed data regarding the prognostic value of the NMLN and the NRLN, as well as the interaction between these two parameters, establish the LNR as a reliable means to enhance the discriminative prognostic value of nodal status in assessing survival and recurrence. The LNR, defined as the ratio of the NMLN to the total NRLN, represents a reliable parameter reflecting not only the burden of LN involvement but also the quality and extent of inguinofemoral lymphadenectomy. In our study, independent prognostic factors of survival and recurrence were the FIGO stage, surgical margins, and LNR. However, LNR showed superior prognostic predictive accuracy compared with the revised 2021 FIGO staging system.
Research on the prognostic implications of LNR in VSCC patients is relatively rare. In 2009, Kunos and his team were the first to introduce the LNR as prognostic assessment tool based on an analysis of the GOG protocol #37 study. 13 In their study, they reported that LNR >20% was correlated with an increased risk of relapses (58% versus 36%, p = 0.03) and cancer-related death (58% versus 33%, p = 0.02) compared to patients with an LNR ⩽20, which was in line with our finding.
Basing their approach on Kunos’s risk stratification, Polterauer et al. 24 examined the significance of LNR in 745 cases of VC from a multi-institutional European registry (VULCAN study). They observed that a high LNR (>20) correlated with lower rates of RFS and OS. This identification allowed for the categorization of patients into different risk groups for determining the need for adjuvant treatment. Their findings highlighted the potential benefits of adjuvant therapy in patients with higher LNR values, suggesting that LNR might offer greater utility than nodal positivity in the selection of suitable candidates for adjuvant radiation therapy following VC surgery.
Polterauer et al. 25 subsequently reanalyzed the data of 1047 patients included in the AGO-CaRE-1 study multicenter database. Similarly to the results of the analysis performed in the VULCAN trial cohort and our results, the univariate analysis revealed that 3-year PFS rates were 75.7%, 44.2%, and 23.1% and 3-year OS rates were 89.7%, 65.4%, and 41.9%, in patients with LNRs 0%, N0% b 20%, and ⩾20%, respectively (p < 0.001, p < 0.001). In the multivariable analyses, LNR (HR = 7.75, 95% CI 4.01–14.98, p < 0.001), FIGO stage (HR 1.41, 95% CI 1.18–1.69, p < 0.001), and the patient’s performance status (HR = 1.59, 95% CI 1.39–1.82, p < 0.001) were identified as factors associated with PFS. Moreover, LNR (HR = 12.74, 95% CI 5.64–28.78, p < 0.001) and performance status (HR 1.72, 95% CI 1.44–2.07, p < 0.001) were the only independent parameters associated with OS. Notably, in comparing the two different multivariable models, LNR consistently exhibited a stronger correlation than the NMLN.
In the study by Wu et al., 6 involving 2332 patients with VSCC, the 5-year OS rates were 73.8%, 51.8%, 47.6%, and 23.9% among patients with zero, one, two, and three or more PLN, respectively (p < 0.001). The 5-year OS rates were 73.8%, 54.7%, and 25.2% among patients with LNR of 0, 0.01–0.2, and >0.2, respectively (p < 0.001). Moreover, the multivariate analysis demonstrated that both the NMLN and the LNR were independent prognostic factors for OS, with the LNR showing a superior predictive value (HR: 3.081; 95% CI: 2.128–4.462; p < 0.001) compared to the number of NMLN (HR: 1.18; 95% CI: 1.129–1.233; p < 0.001).
Poddar et al. investigated the role of LNR in relation to recurrence in 111 patients with VSCC. In this study, the RFS rate of patients with an LNR of 0 was significantly higher than that of patients with an LNR of 1–20 or >20 (log rank = 7.61; p = 0.02). 26
More recently, Lei et al. 3 have examined 2166 postoperative patients diagnosed with VSCC. Both univariate and multivariate analyses identified LNR as an independent prognostic factor for OS, alongside race, age at diagnosis, marital status, FIGO staging, and tumor size with the LNR showing a superior predictive value (HR: 3.953; 95% CI: 1.156–13.514; p = 0.028) compared to FIGO stage (HR: 2.692; 95% CI: 0.847–8.558; p = 0.043) which is in line with our finding. Then, they formulated a prognostic nomogram based on these factors and demonstrated that the nomogram based on the LNR showed superior prognostic predictive accuracy for OS with a C-index value of 0.772 (95% CI: 0.752–0.792) compared with the FIGO staging system with a C-index value of 0.676 (95% CI: 0.6540.698).
To our knowledge, this the first and largest Tunisian study reporting the pattern of recurrence and survival factors of relapse of SCCV using the revised 2021 FIGO stage classification. Our findings allow for identifying high-risk patients prone to recurrence, necessitating more intensive treatment or vigilant monitoring. Our focus was on assessing the prognostic significance of LNR, which was identified as an independent prognostic factor for survival and recurrence, with a predictive value surpassing that of the new FIGO classification. This is significant in an era where prognostic classifications for vulvar cancer undergo crucial variations related to the LN status.
The scope of the study is limited due to its retrospective and single-center nature, and consequently, a power analysis for sample size calculation was not performed. Another limitation is that the inclusion involved patients treated over three decades during which several changes in the standards of nodal surgery were introduced with variable numbers of removed LN according to the surgical procedure, finally, another limitation of our study is the lack of assessment of the impact of this risk group stratification based on LNR on the indications for adjuvant treatment and its long-term oncological outcomes. Moreover, given the variability in adjuvant treatment and the heterogeneity within this group, we did not evaluate survival outcomes based on this factor.
Conclusion
Controversy surrounds the utility of the NMLN in predicting survival outcomes and the isolated consideration of the NMLN warrants careful interpretation. A high LNR was associated with poor survival and higher risk of recurrence. LNR provides a more precise categorization of patients compared to the number of positive nodes, regardless of other well-established prognostic factors. Assessing LNR could prove beneficial in grouping patients based on risk levels, aiding in the selection of suitable candidates for adjuvant radiation as the NMLN remains a metric that does not encompass the extent of LND. This aspect should be evaluated in forthcoming clinical trials.
Supplemental Material
sj-docx-1-whe-10.1177_17455057241285396 – Supplemental material for Lymph node ratio as an indicator of nodal status in the assessment of survival and recurrence in vulvar cancer: A cohort study
Supplemental material, sj-docx-1-whe-10.1177_17455057241285396 for Lymph node ratio as an indicator of nodal status in the assessment of survival and recurrence in vulvar cancer: A cohort study by Houyem Mansouri, Ines Zemni, Saida Sakhri, Mohamed Ali Ayadi, Nadia Boujelbene and Tarek Ben Dhiab in Women’s Health
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
