Abstract
Treatment options for carcinoma of the cervix are guided by tumor stage, and include radical surgery, in cases where the tumor is confined to the cervix, or concurrent chemotherapy and radiotherapy. In those cases treated with chemoradiation, the ability to monitor the response to treatment in order to adapt the management plan during its course may be beneficial. This approach has the potential to offer an individualized treatment plan, allowing for differences in behavior between tumors to be addressed early, rather than a ‘one size fits all’ treatment approach. This article aims to review the use of evolving functional imaging techniques including diffusion-weighted MRI, dynamic contrast-enhanced MRI, and PET as tools for the evaluation of response to treatment of uterine cervical carcinoma.
Keywords
The use of functional imaging techniques in the context of measuring treatment response in cancer patients holds great potential. They offer a combination of physiological, morphological and metabolic information. Treatment of cervical cancer is primarily guided by tumor stage at the time of presentation [1]. Functional imaging techniques may be superior to conventional anatomic imaging in assessing treatment response, and may demonstrate the effects of treatment at an earlier stage. The pitfalls of conventional imaging in evaluating disease after treatment are well established. Namely, postoperative or radiotherapy changes can mask or mimic residual and recurrent disease [2] and there may be a time lag before an objective response (tumor shrinkage) [3]. In these cases, the use of functional imaging as a predictive biomarker of response may be advantageous. An important goal in modern-day cancer management is to move towards individualized patient treatment plans. As not all tumors of the same type will follow the same course of disease, it would be advantageous to subcategorize cervical tumors based on their specific characteristics and the expected response pattern that each subgroup displays. Tumor heterogeneity is seen, not just from patient to patient, but between primary and secondary deposits, and even within the same primary tumor mass [4]. The significance of intratumoral heterogeneity has become important as it may highlight groups of treatment-resistant cells, towards which it is now possible to direct targeted therapy or biopsy [5]. Being able to accurately predict a tumor's behavior would allow a patient to receive the most appropriate treatment for their specific case, rather than a generic treatment. Prolonged ineffective treatment is undesirable, both to the patient, in terms of consequential morbidity, and to the healthcare system. It would be preferable to have an early assessment which offered a window of opportunity to optimize or alter the treatment plan in those patients who are not undergoing a satisfactory response. Alterations could be made to chemotherapy regimes, changes to radiotherapy dose or of the targeted volume. In addition, consideration for entry into clinical trials or the use of experimental techniques may be considered.
This article aims to highlight some of the functional imaging techniques that are showing promise in the management of cervical cancer, including dynamic contrast-enhanced (DCE)-MRI, diffusion weighted (DW)-MRI and F-18-fluorodeoxyglucose (FDG)-PET or FDG-PET-CT.
DCE-MRI
Dynamic contrast-enhanced-MRI represents the assessment of signal intensity changes over time during the injection of intravenous gadolinium. There is an inevitable trade-off between spatial resolution and temporal resolution which will affect the type of dynamic acquisition. The term DCE-MRI is used interchangeably in the literature to mean both the semiquantitative assessment of enhancement of tissues following intravenous gadolinium on MRI over a certain time period with spatial resolution being favored; this technique is widely used for observational, qualitative assessments of tissue enhancement; and quantitative dynamic MRI using mathematical modeling for pharmacokinetic analysis which requires very rapid data acquisition, with protocols favoring high temporal resolution at the expense of spatial resolution. Quantitative DCE-MRI is not currently a widely used clinical tool and remains predominantly a research tool.
Comparison of standard dynamic MRI with quantitative dynamic contrast-enhanced MRI.

DCE: Dynamic contrast-enhanced; EES: Extracellular–extravascular space; ROI: Region of interest; W: Weighted.
Dynamic contrast-enhanced-MRI provides information regarding tumor vasculature, by means of evaluating the transit of intravenous gadolinium from vessels to the tissue with leakage from capillaries into the extracellular–extravascular space (EES) and eventual rediffusion into the venous system. All vascularized tissue will show some enhancement. Neoplastic tissue is expected to show relatively more enhancement, as demonstrated by a greater increase in signal intensity following contrast administration. This is due to pathological architechture, and increased permeability of tumor vessels. Resultant changes to tumor vascularity as part of an early treatment response should be reflected by a change in enhancement patterns.
The characteristics of the tumor vascularity may be assessed by means of analyzing the changes in signal intensity within the tumor over time (time–intensity curves) and in addition, pharmacokinetic analysis may be undertaken, usually using a multicompartment mathematical model [6]. Using such pharmacokinetic modeling, quantitative parameters may be measured, including volume transfer constant (Ktrans) between blood plasma and the EES and the rate transfer function (Kep) from the EES to the blood plasma. The EES fractional volume may also be calculated (ve). Pharmacokinetic analysis reflects an overview of the whole tumor vascular microenvironment and the measurement of the area under the gadolinium curve may be used for this purpose. Many factors, including microvessel density, capillary permeability, tumor oxygenation and interstitial fluid pressure, are involved in the delivery of treatments, and are known to affect the response and outcome [7]. Measurement of tumor vascular parameters has enormous potential in better understanding the nature of the tumor.
In patients with cervical cancer, the precise time points for DCE-MRI examination in relation to the patients' treatment have varied between authors. However, the main time points of interest are pretreatment, early during the course of treatment (at approximately 2 weeks following the initiation of chemoradiation) and post-therapy (from 0 to 8 weeks following completion of therapy). For longitudinal studies evaluating response to therapy, it is vital that the same imaging parameters are used each time the patient is imaged in order to assess change.
Pretreatment & early during treatment
A baseline pretreatment DCE-MRI gleans information regarding the underlying characteristics of the tumor, and may act as a predictive biomarker of how the tumor will be expected to behave. Early work by Mayr
Associations have been found between enhancement patterns and histological features: higher perfusion measurements correlate with increased microvessel density [10], and faster rate of enhancement with higher tumor grade [11]. Yamashita
Early assessment
As a tool in early assessment of tumor response, studies have shown that tumors with good enhancement postcontrast, or an increase in signal intensity within the first 2 weeks of treatment, predicted better tumor regression (as calculated from volumetric analysis on MRI) [17]), as well as more favorable local recurrence rates [8]. Yuh
The use of early imaging during the course of chemoradiation has not been widely implemented into clinical practice, with many centers imaging at the end of treatment or following an interval to allow for continued tumor shrinkage following radiotherapy. However, there is now compelling evidence to consider DCE-MRI as an early monitor of response. In the context of a clinical trial, DCE-MRI could be used to select or triage nonresponders to undergo novel therapy within a much shorter time frame, rather than waiting for morphological evidence of poor response.
Postcompletion
The routine follow-up of patients who are disease free following completion of radical surgery is predominantly clinically based, with imaging being reserved for patients with clinically suspected recurrence. Determining final response and identifying nonresponders, who may benefit from alternative treatment, conventionally occurs at 3 and 6 months post-treatment, using standard MRI. If there is radiological suspicion of residual disease at the 6-month scan, a targeted biopsy is performed and the patient is considered for exenteration. In studies where post-therapy DCE-MRI has been performed, contrast-enhanced images have been shown to be superior to unenhanced scans in differentiating between neoplastic tissue and post-therapy changes. Enhancement of a suspicious area within the first 90 s had a sensitivity of 91% and specificity of 67% in differentiating suspected recurrent tumor from benign post-treatment fibrosis [19,20]. Appropriate patients may be offered salvage treatment at this juncture. In one small study of women scanned before and after a course of radiotherapy, post-treatment early enhancement was a poor prognostic factor [11]. Hawnaur
DW-MRI
Diffusion weighted-MRI is a specialized functional imaging technique which is particularly sensitive to the microscopic motion of water molecules and allows for noninvasive characterization of biological tissues based on their water diffusion properties

Response assessment using diffusion-weighted imaging: pretreatment.

Response assessment using diffusion-weighted imaging: post-treatment.
When DW gradient pulses are applied to a conventional magnetic resonance sequence [23], the signal can be made sensitive to the level of localized water diffusivity that can be quantified as the apparent diffusion coefficient (ADC). Following successful anticancer treatment, alterations in cell density owing to necrosis and apoptosis cause significant changes in water diffusion, and therefore the ADC, which are detectable by DW-MRI. In addition, these changes in water mobility occur well before macroscopic indicators of response such as tumor size or volume [24].
The motion of water molecules in tissues can be assessed by applying DW gradients to T2-weighted sequences. This process incorporates two additional intense and balanced magnetic field gradients, compared with a conventional spin-echo imaging sequence, and is placed symmetrically about a focusing 180° pulse. Although these additional paired gradients have balanced and opposing magnitude, they differ in duration and amplitude. The b-value, measured in s/mm2, indicates the degree of DW, which can be varied by altering the gradient amplitude, time duration of individual gradient pulses or the time interval between both pulses. Parameters such as ADC derived from DW-MRI are attractive as potential imaging biomarkers of response as it is a reproducible physical constant, which is independent of magnetic field, scanner and operator as well as sequence or acquisition parameters. In addition, the technique is noninvasive, does not require exogenous contrast agents, does not use ionizing radiation, yet is quantitative, can be easily incorporated into routine patient evaluations and can be obtained relatively rapidly.
As a biomarker of therapy response, the use of DW-MRI has been investigated in several preclinical or animal model studies [25], which have further encouraged its more recent use in clinical studies. In the majority of malignant tumors, successful treatment is reflected by increases in ADC values. Although most available reports involving DW-MRI have been in brain malignancies, increases in ADC values following treatment have also been demonstrated in studies involving extracranial tumors. For instance, Pickles
Diffusion weighted-MRI has not yet been fully explored in cervical tumors as a response biomarker. Reports have highlighted its diagnostic value, for instance, Naganawa
In a prospective study evaluating DW-MRI as a biomarker of early response in women receiving concurrent chemoradiotherapy for advanced cervical cancer, ADC values, as well as the change in ADC after only 2 weeks of therapy, was found to significantly correlate with clinical as well as conventional MR response. (ADC values: p = 0.009, ρ = 0.568 for clinical response and p = 0.048, ρ = 0.448 for MR response; change in ADC: p = 0.03, ρ = 0.48 for clinical response and p = 0.01, ρ = 0.56 for MR response) [29]. Chen
Liu
The prognostic significance of pretherapy ADC values seems to stem from the relationships between necrosis and high tumor cellularity, and poorer patient outcomes. Necrosis is related to hypoxia-mediated radio resistance as well as the selection of more aggressive tumor clones and causes increases in ADC values. Also, necrotic tumors are frequently found to be hypoxic, as well as acidotic and poorly perfused, resulting in reduced sensitivity to radiotherapy and chemotherapy.
18-FDG-PET
PET is a molecular imaging technique that uses radio-labeled molecules to image biological processes
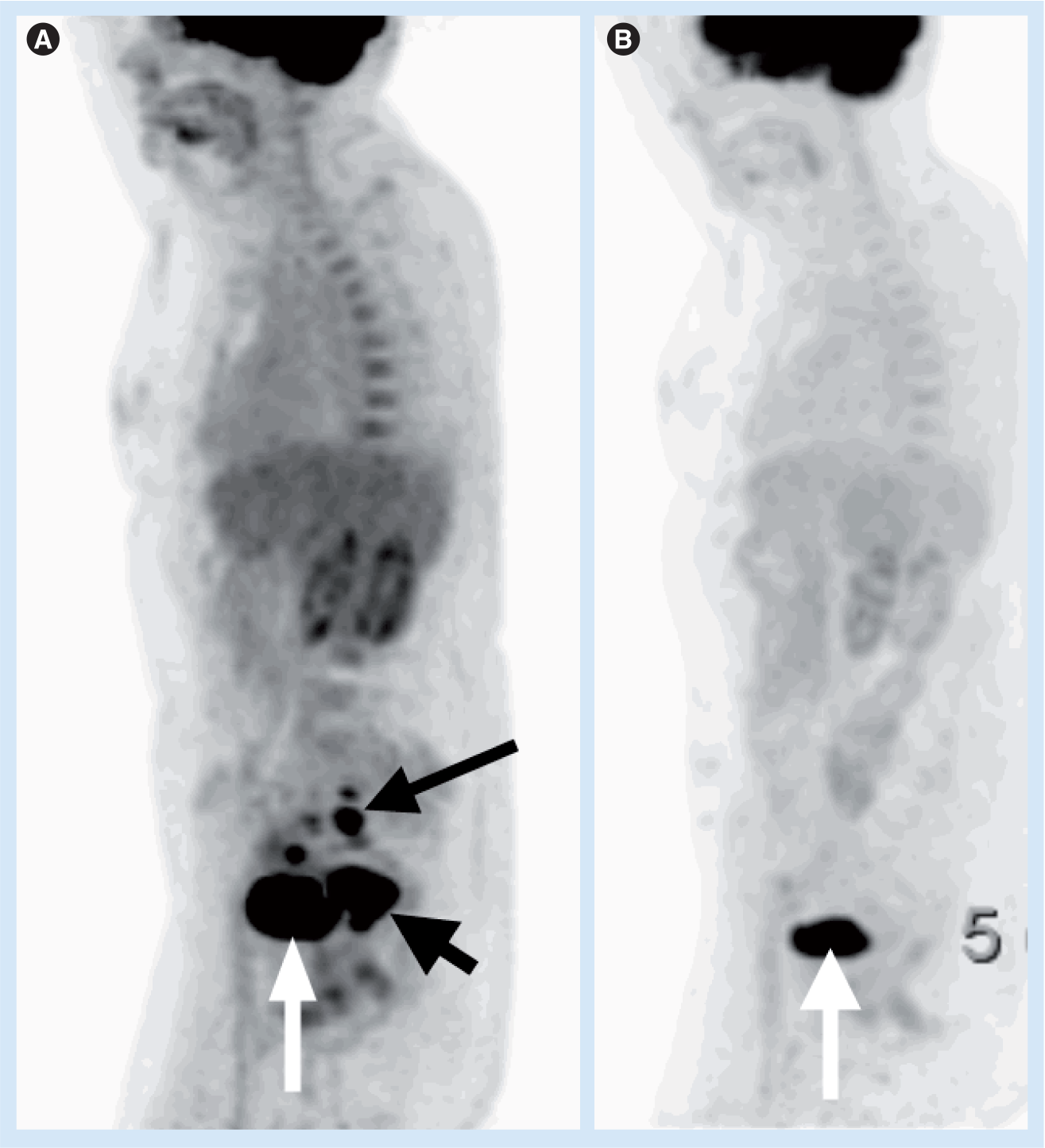
18-fluorodeoxyglucose-PET.

Axial MRI and PET/computed tomography imaging in a patient with stage 2B adenocarcinoma.

Axial MRI and PET/computed tomography imaging in the same patient†, 4 months post completion of treatment.
However, FDG is not entirely specific for malignant cells and a high uptake can be seen in inflammatory or proliferating cells and in metabolically active normal tissues. The development of hybrid FDG-PET/CT scanners, allows integration of the morphological data from CT with the functional images from FDG-PET. This improves localization of abnormal FDG uptake compared with FDG-PET alone.
Changes in glucose uptake and metabolic activity within a tumor may occur soon after the start of therapy, before changes in tumor size are apparent. Thus, changes in metabolic activity have the potential to allow detection of early functional response. There have been numerous reports demonstrating the value of FDG-PET in evaluating treatment response and predicting survival in patients with cervical cancer [32–35]. Treatment response evaluation can be performed during the course of therapy with the potential to individualize the therapeutic plan, or in the immediate post-treatment setting, where it can be used for prognostication.
Monitoring treatment response after chemoradiation
Complete metabolic response at the end of chemoradiation has been shown to be a good predictor of survival [36–38]. In a retrospective study of 152 patients by Grigsby
These results have subsequently been validated in a prospective cohort study by the same group [37]. In this study, 92 patients were imaged with FDG-PET between 2 and 4 months (mean 3 months) after completion of chemoradiation. Post-therapy FDG-PET showed a complete metabolic response in 65 patients (70%), a partial metabolic response in 15 (16%), and progressive disease in 12 (13%). The 3-year progression-free survival rates were 78, 33 and 0%, respectively (p < 0.001). The hazard ratio (HR) for risk of recurrence based on the post-therapy metabolic response showing progressive disease was 32.57 (95% CI: 10.22–103.82). A partial metabolic response had an HR of 6.30 (95% CI: 2.73–14.56). These data suggest that the 3-month post-therapy FDG-PET can provide valuable long-term prognostic information, which may be important in selecting patients for salvage therapy.
Monitoring response after radiotherapy
Extent of tumor regression during the course of radiotherapy is an important predictor of survival [40]. Lin
Recently, Schwarz
Monitoring response to chemotherapy
These data relating to the use of FDG-PET in monitoring response to chemotherapy alone are limited to a few case reports. Dose
Conclusion & future perspective
As advances in treatment options available for cancer management develop, there is a preference to move away from planning treatment based only on their pretreatment stage towards a more patient-specific approach. It is therefore appropriate that as therapeutic options become more adaptable and complex, imaging techniques are developed to allow improved stratification of patients into the most appropriate therapeutic plan both before and during the course of treatment.
Recent publications suggest that functional imaging will have an important role in assessing treatment response in the future. Where DCE-MRI is concerned, pretreatment and early assessment scans appear to be of most value. Early assessment in DWI also shows promise. However, many studies have only included small numbers of women, and there is substantial variability in both the technique and interpretation of imaging data. FDG-PET/CT following treatment has an important role in predicting patient outcome.
For these modalities to be incorporated into clinical practice, it is imperative that the imaging and postprocessing protocols become standardized, and that the results are shown to be reproducible between centers. Only following verification in multicenter trials will it be possible to incorporate these imaging modalities into mainstream cancer management. At the present time, it remains to be shown whether detection of early treatment response will lead to any benefit in long-term outcomes.
Looking prospectively, multiparametric imaging, combining the information provided by DW-MRI, DCE-MRI and FDG-PET/CT may be found to be of value [45]. This will potentially identify different aspects of the tumor biology. This information has the potential to direct best treatment, depending on the tumor behavior.
Executive summary
Functional imaging techniques to assess treatment response in cervical cancer include dynamic contrast-enhanced (DCE)-MRI, diffusion weighted (DW)-MRI and 18-fluorodeoxyglucose (FDG)-PET/computed tomography.
They offer a combination of physiological, morphological and metabolic information.
Functional imaging can show changes in tumor biology during the course of treatment before morphological changes are apparent.
Differences between DCE and dynamic perfusion MRI can be evaluated looking at acquisition, measurable parameters, advantages and disadvantages.
DCE-MRI achieves an overview of the tumor microenvironment by means of contrast delivery and uptake in tissues.
DCE-MRI pretreatment and early during the course of treatment seem to predict how certain tumors will behave. Post-treatment scans can be useful in distinguishing between residual/recurrent disease and benign post-therapy changes.
DW-MRI uses the random motion of water molecules to convey information about the cellularity of a tissue. Changes in the cellularity are reflective of a response to treatment.
Positive responses to treatment are represented by an increase in the apparent diffusion coefficient of a tissue.
Increased tracer uptake is seen in metabolically active tissues, including cervical cancer.
Fusion of FDG-PET with computed tomography images allows for better localization of areas of increased uptake.
Post-treatment assessment is a good prognostic indicator.
Functional imaging techniques show good potential in the role of assessing tumor response to treatment. This will hopefully lead to a more individualized way of managing patients with cervical cancer.
Multiparametric imaging may help to better decipher tumor biology and guide management.
Further work is required to develop robust protocols that will be reproducible between centers.
Footnotes
