Abstract
Introduction
Due to its good wearing properties, cotton fabric is one of the main materials used in clothing.
1
However, cotton fabrics are liable to mildew as microorganisms, which reproduce easily under humid conditions and secrete fiber enzymes and acid, can spoil and discolor cotton fiber. At present, many bio-based antibacterial finishing agents are used to treat cotton fabrics. These include ε-polylysine and prodigi-osin, chitosan, modified chitosan, and nano- chitosan, and various types of plant-based antibacterial finishing agents, including honeysuckle, aloe,
In this study,
Experimental
Materials
Bleached, pure woven cotton fabric (100 g/m2) was used.
Preparation of Vegetable Dye
Fresh
Cotton Fabric Treatment
Direct Dyeing
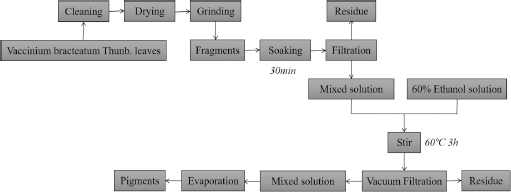
The technological process of vegetable dyestuff abstraction.
Direct dyeing was used to conduct the single factor test. The dyeing conditions were as follows: liquor ratio (LR) of 1:40, dyeing time of 40 min, dyeing temperature of 80 °C, and the concentration of dye solution was 6% owf. Only one variable was changed during each dyeing experiment, with the other variables remaining fixed. Each individual experiment was repeated three times, and the results were averaged. The dyeing process is shown in Fig. 2. A Mettler Toledo AL204 Electronic Balance (Mettler Toledo International Trading, Shanghai, China) was used for weighing and a HH-4 Ther-mostat Water Bath (Jiangsu Changzhou Ronghua Instrument Manufacture Co. Ltd) was used for dyeing the fabrics.
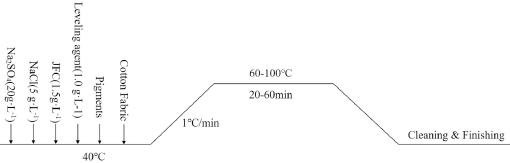
The prescription and flow of direct dyeing.
Pretreatment with Chitosan
To improve dyeing, the cotton fabric was pretreated with chitosan. Since chitosan is insoluble in water, acetic acid was used as solvent. The concentration of chitosan and acetic acid were 20 g/L and 100 g/L, respectively, and the LR was 1:40. The chitosan pretreatment process is shown in Fig. 3, followed by direct dyeing.
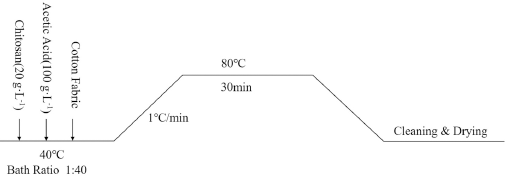
The process of pre-treatment with chitosan.
Pre-Mordanting with FeCl3
Firstly, the cotton fabric was wetted at 40 °C for 10 min, then the FeCl3 mordant was added for pre-mordant treatment. The concentration of mordant was 20% and the LR was 1:30. The process flow is shown in Fig. 4. The pre-mordanted fabric was then dyed.
Antibacterial Activity
Part 3 of GB/T 20944-2008 provides for quantitative tests and evaluation methods for the determination of antibacterial properties of textiles by oscillation method, on the basis of which, quantitative determinations for antibacterial properties of dyed cotton fabrics were conducted.
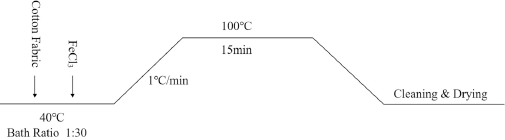
The process of pre-mordant with FeCl3.
There are three main reagents need to be prepared in advance to test the antibacterial property of the fabric, with the recipes given in Table I.
Prescriptions of Tree Main Reagents
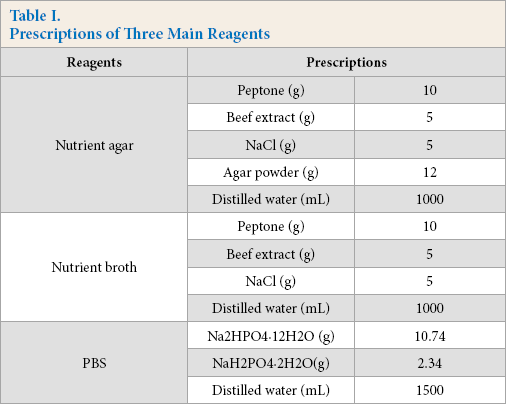
Bacterial Suspension
The preserved
The process of preparation for bacterium suspension.
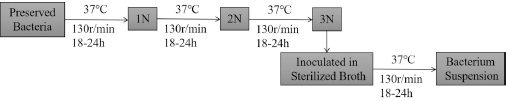
Bacterial Treatment of Samples
Each sample fabric (0.75 ± 0.05 g) was cut into 5 × 5 mm fragments. Each fabric was then added into a triangular flask containing phosphate buffer solution (PBS, 0.03 M, pH = 7.2 ± 0.2, 70 mL) and sterilized at 121 °C for 20 min. The
The process of preparation for sample solution with bacteria.
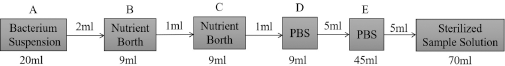
Preparation of Dilution Method
Bacteria were cultivated by the dilution method and the sample solution respectively diluted 101 ∼ 108 times. Taking different diluted multiple sample solutions (50 μL) respectively directly into three sterile nutrient agar culture medium, they were then all put in an environment at constant temperature and humidity (37 °C, 60% RH, 18–24 h) for cultivating. At the specified time, the petri dish was taken out to select the appropriate dilution factor area for calculation. The specific process is shown in Fig. 7.
The process of preparation for dilution method.
Performance Tests of Dyed Fabrics
K/S Value
The Kubelka-Munk dyeing depth equation established a certain function relationship between the absorption coefficient
Antibacterial Property
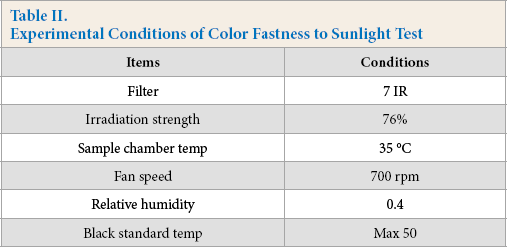
The antibacterial property could be represented by the antibacterial rate as calculated using Eq. 1.
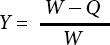
Color Fastness
Color fastness to washing was determined according to GB/T 3921.1-2008 Color Fastness to Washing Test for Textiles: Test 1. The specific experiment procedure was as follow: The dyed cotton fabric sample was cut to a fixed size (100 × 40 mm), which should be stitched together with the same size interlining and weighed (1.567 g). The bath ratio was set as 1:50, with 5 g/L soap fakes (0.3925 g) and 2 g/L of anhydrous sodium carbonate (0.157 g) as detergent. The test specimen, detergent and steel ball of the stainless steel cup were all installed on the main shaft of the SW-12 Wash Fastness Tester (Dongguan SafTonny/Tomy Co. Ltd.), heating the water in the cabinet after reaching 60 °C, and driven by motor spindle rotate at a speed of 40 rpm for 30 min. Then the sample was rinsed and dried thoroughly. This operation was to simulate the daily household cleaning or commercial washing five times.
Results were determined by observing the color change of samples after washing, and evaluating the discoloration and staining grade with the Gray Scale for Evaluating Staining (AATCC).
The color fastness to friction was determined according to GB/T3920-2008 Textile Color Fastness Test to Friction. The dyed cotton fabric sample was cut to a fixed size (200 × 50 mm) and the Y571B Friction Fastness Tester (Dongguan BLD Instrument Equipment Co. Ltd.) was used to test the crock fastness of dyed cotton fabrics. The number of times of friction application was set at 10, and the dry and wet friction of the fabric were tested successively. Results were obtained by observing the color change of samples after rubbing, and evaluating the discoloration and staining grade with the Gray Scale for Evaluating Staining.
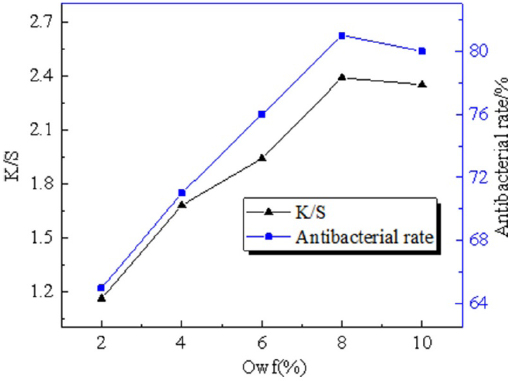
The color fastness to sunlight was determined according to GB/T8427-2008 Textile Color Fastness Test Xenon Arc Lamp to Artificial Light. The Q-Sun Xe-2 Xenon Test Chamber (Q-Lab Corp.) was used and experimental conditions were set as shown in Table II. In this experiment, the fabric sample should be checked once after every 7-8 h of irradiation. If the fading of the fabric sample did not reach level 4, the irradiation would continue. If the fabric sample fades to level 4, the irradiation was removed. Results were determined by observing the color change of the blue card, and evaluating the grade of the lighting fastness.
Results and Discussion
Influences on Dyeing Effect and Antibacterial Property
Bath Ratio
As shown in Fig. 8, the antibacterial property decreased with the increase of the dyeing bath ratio, and the best antibacterial property could be obtained when the bath ratio was 1:20 and 1:30. As a larger bath ratio means a smaller concentration of pigment in the solution, the ability of pigment bonding with the fabric reached saturation when the bath ratio was set at 1:20. If the bath ratio was decreased further, it might lead to uneven dyeing, which might affect the
The process of preparation for dilution method.
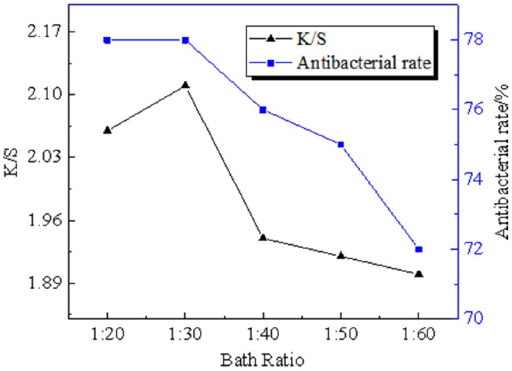
Dyeing Concentration
When the dyeing solution concentration reached 8%, the
The influence of owf on dyeing effect and antibacterial property.
Experimental Conditions of Color Fastness to Sunlight Test
Dyeing Temperature
Fig. 10 shows that with the increase of dyeing temperature, the pigment molecules were fully released, and was conducive to the combination of pigment molecules and fabric. However, when the temperature exceeded 80 °C, the flavonoids in the dye solution decomposed,
12
resulting in a smaller
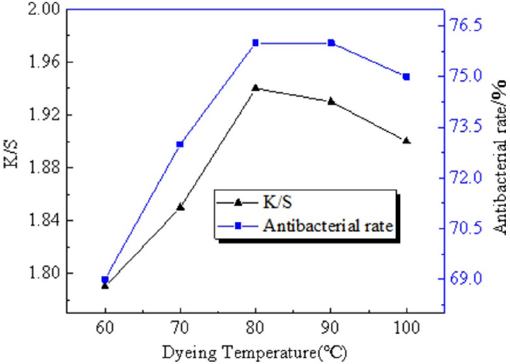
The influence of dyeing temperature on dyeing effect and antibacterial property.
Dyeing Time
The best dyeing effect and antibacterial property was obtained when the dyeing time was 50 min. As shown in Fig. 11, when the dyeing time exceeded 50 min, the dyeing effect remained stable, which means the combination of pigment and fabric reached a balance. Moreover, the pigment could decompose due to the long dyeing time. 13
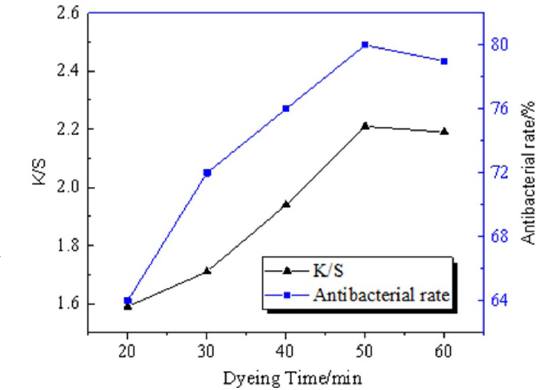
The influence of dyeing time on dyeing effect and antibacterial property.
The dyeing rate of the fabric was positively correlated with the antibacterial property. Therefore, better antibacterial properties can be obtained by increasing the dyeing effect on the cotton fabric.
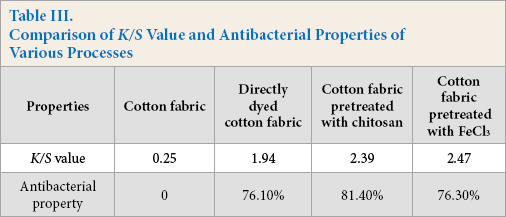
Effect of Various Processes on K/S Value and Antibacterial Property
Directly Dyeing
Table III shows the
Comparison of
Pre-Treatment with Chitosan before Directly Dyeing
It can be seen in Table III that the
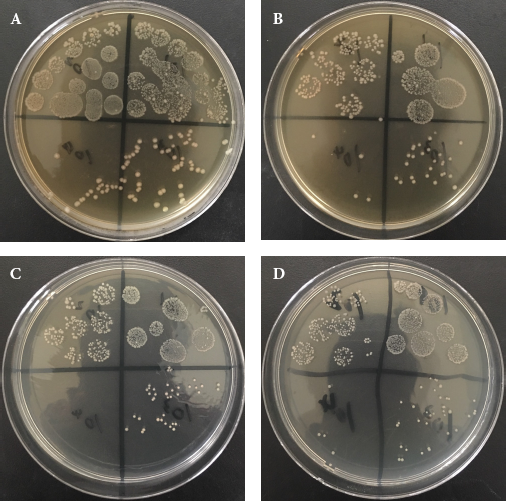
(a) The antibacterial property result of raw cotton fabric, (b) the antibacterial property result of directly dyed cotton fabric, (c) the antibacterial property result of directly dyed cotton fabric with chitosan pretreatment, and (d) the antibacterial property result of directly dyed cotton fabric with FeCl3 pretreatment.
Pre-Mordanting with FeCl3 before Directly Dyeing
Pretreatment with iron mordant (FeCl3) greatly improved the
Color Fastness and Antibacterial Durability
Color Fastness
A dyeing experiment was carried out based on the optimal process of directly dyeing cotton fabric, as previously discussed, to test the
Color Fastness Level of Dyed Cotton Fabric
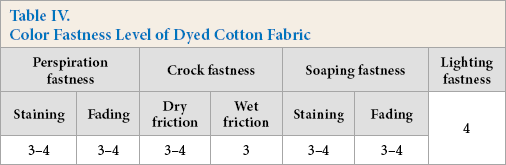
Antibacterial Durability
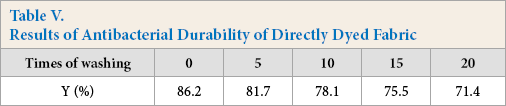
Antibacterial durability was determined in the same manner as the test method for color fastness to washing. It can be seen from Table V that the antibacterial property of the directly dyed cotton fabric was still over 70% after 20 washing cycles, indicating that the pigment has been closely combined with the cotton fabric.
Results of Antibacterial Durability of Directly Dyed Fabric
Conclusions
Optimal dyeing of cotton fabric was obtained when the vegetable dye was extracted from
Both the pretreatment of chitosan and FeCl3 could not only increase the dyeing effect on cotton fabric, but also improve the antibacterial property of the cotton fabric as well. Chitosan had a greater impact on improving the antibacterial property compared to FeCl3, while FeCl3 could better improve the dyeing effect. Chitosan and FeCl3, together with plant dyes made from leaves of
A new process of cotton fabric dyeing was developed, which adds ecological and functional value to cotton fabric, greatly improving the added value of the fabric and providing new ideas for ecological textile dyeing and finishing technology.
Footnotes
Acknowledgments
This work was supported by the Postgraduate Research & Practice Innovation Program of Jiangnan University (Grant No. JNKY19_019) and the Philosophy and Social Sciences Research Project of Universities (Grant No. 2019SJZDA130).
