Abstract
Jute is a renewable, biodegradable, and lignocellulosic natural fiber traditionally used for making packaging material. Due to the environmentally-friendly nature of this fiber, it is now used for making diversified and value-added products including upholstery, furnishing textiles, handicraft items, soft toys, and even apparel. Jute fabric was dyed with natural dyes extracted from manjistha, annatto, ratanjot, and babool by a pre-mordanting method. Dye extraction conditions were standardized and applied on pre-mordanted jute fabric. There was a substantial improvement of color yield, levelness of dyeing, and washfastness properties of naturally dyed jute fabric after double pre-mordanting using bio-mordants and inorganic mordants. Lightfastness (moderate to good), crockfastness (very good to excellent), and UV protection (very good) ratings were determined, as well as antimicrobial activity.
Introduction
The lignocellulosic golden fiber, jute, is an annually renewable resource. The fiber is conventionally used for making hessians and sackings used as packaging materials. Due to stiff competition in the packaging sector from synthetic packaging material, ∼25% of jute product fibers are used for making value added and diversified products including furnishing, upholstery, technical textiles, and, to some extent, fashion garments.
Chemical processing of jute and its coloration have become an important activity in the jute processing sector for improving its look and feel. The major components of jute are cellulose, hemicelluloses, and lignin. The cellulosic constituent of jute fiber, which is ∼60% of its weight, is primarily responsible for jute fiber coloration. Synthetic dyes are mainly used for jute fabric coloration and several processes has been standardized.1–6 Use of synthetic dyes for jute fabric coloration have a highly negative impact on the environment, for dyers as well as end users. Therefore, an endeavor was made to replace synthetic dyes with natural dyes, with some review, as well as application-oriented work, performed on jute fiber.7–9 Natural dyes, though eco-friendly in nature, have some limitations such as availability, color yield, reproducibility of shade, and technical problems (e.g., limitation of shade, non-standardized processes, and inadequate fastness properties). Some work has been done on the use of bio-mordants and inorganic mordants, and their combinations, for improving washfastness, lightfastness, and level dyeing properties of naturally-dyed jute fabric. 10
Sun protection fabrics are designed to absorb or reflect the sun's UV radiation as a means of protecting the skin from damage. An ultraviolet protection factor (UPF) value is used to measure UV protection provided by a fabric. The safest protection from ultraviolet (UV) radiation exposure is offered by textile accessories such as hats and shade structures such as umbrellas. UV-protective properties of textiles depend on their chemical composition, physical structure (e.g., porosity, weight, and thickness), as well as the presence of dyes, pigments, and/or other auxiliaries that have the ability to reduce or prevent transmission of harmful solar UV radiation. 11
Textile materials based on cellulosic and lignocellulosic fibers provide perfect conditions (e.g., large receptive surface area, suitable temperature, moisture, oxygen, and nutrients) for the growth of microorganisms. Therefore, there is an urgent need for effective means to control and/or inhibit microbiological growth to protect both textiles and textile wearers. 12 Natural dyes have produced good UV protection properties on different textile fibers including wool, silk, cotton, and synthetic fibers, 10 but little work has been done on jute fiber materials.
In this work, natural dyes have been extracted from four common natural dye sources (manjistha, annatto, ratanjot, and babool). Bleached jute fabric was mordanted by a double mordanting process using bio-mordant and inorganic mordant. Pre-mordanted jute fabric was dyed with natural dyes, and the color yield, fastness properties, and levelness of the dyeing were studied. To make the process more eco-friendly, trisodium citrate was used instead of Glauber's salt as exhausting agent for jute fabric dyeing using manjistha. A study was also done to reveal the effect of mordanting, as well as natural dyeing, for attaining better protection against harmful UV radiation and microorganisms. Should the dyes themselves be inherently UV resistant and antimicrobial, protective textiles could be produced at no additional cost.
Experimental Procedure
Materials
Substrate
A plain weave greige jute fabric with the following specification was used.
Warp: 60 ends/dm (Count, 260 tex)
Weft: 52 ends/dm (Count, 256 tex)
Fabric mass: 250 g/m2 (65% relative humidity at 27 °C)
Chemicals
The following chemicals of analytical grade were used in these experiments: hydrogen peroxide (30%), trisodium phosphate, sodium carbonate, acetic acid, sodium silicate, Glauber's salt, and hydrochloric acid supplied by Merck Specialties Pvt. Ltd. and Ultravon JU (non-ionic surface active agent) supplied by Hindustan Ciba-Geigy Ltd.
Mordants
Two types of mordants were used in the experiments: Bio-mordants myrobolan (Terminila chebula) and pomegranate (Punica granatum), and inorganic mordants ferrous sulfate (FeSO4·7H2·) and potash alum (hydrated salt of potassium aluminum sulfate, K2S·4A12(S·4)3.24H2·).
Dyes
Four different types of natural dyes were used in these experiments. The details of the dyes are given in Table I.
Natural Dyes Used for Jute Dyeing

Methods
Scouring
Greige jute fabrics were scoured with sodium hydroxide (4% on weight of fabric (owf)) and non-ionic surface active agent (2 g/L) at boil, keeping the material to liquor ratio (LR) at 1:20. After scouring, the fabric was washed thoroughly in hot water and cold water successively and neutralized with acetic acid (2 mL/L) followed by the usual cold washing.
Bleaching
Bleaching of greige and scoured jute fabrics were done in a closed vessel for 90 min at 80-85 °C, keeping the LR at 1:20 with hydrogen peroxide (20 mL/L), trisodium phosphate (5 g/L), Ultravon JU (2 mL/L), sodium hydroxide (1 g/L), and sodium silicate (10 g/L). The pH of the bath was maintained at 10. After bleaching, the fabrics were washed thoroughly in cold water, neutralized with acetic acid (2 mL/L), washed again in cold water, and finally dried.
Mordanting
Myrobolan
The myrobolan powder (20% on weight of material (owm)) was soaked in water (LR = 10:1) overnight (12 h) at room temperature (RT) to obtain swollen myrobolan gel. This gel was then mixed with a known volume of water and heated at 80 °C for 30 min. This solution was then cooled, and filtered in a 60 mesh nylon cloth. The filtrate was used as a final mordant solution for mordanting at an LR of 20:1. Bleached jute fabric was then treated with the myrobolan solution, initially at 40-50 °C, and the temperature was then raised to 80 °C. The mordanting continued for 30 min. The fabric was then dried without washing to prepare the bleached jute fabric for dyeing or second mordanting with ferrous sulfate or potash alum.
Pomegranate
Pomegranate rind (5% owm) was soaked for 16 h and then boiled for 2 h to extract coloring matter using a 1:10 LR. Carbonate buffer (pH 10) was selected as extraction medium. The extraction temperature was 80 °C. For mordanting, jute fabric was treated in the extracted solution for one hour at 80 °C using a 1:20 LR after dilution with water. Then the fabric was dried without washing to prepare it for bleached jute fabric dyeing or second mordanting with ferrous sulfate/ potash alum.
Ferrous Sulfate
Bleached jute fabric and bio-mordanted (pomegranate/ myrobolan) bleached jute fabrics were mordanted with hydrated ferrous sulfate (2% owm) at a temperature of 80 °C for 30 min, at a 1:20 LR. The mordanted jute fabrics were dried without washing for subsequent dyeing with natural dyes.
Potash Alum
Bleached jute fabric and bio-mordanted (pomegranate/ myrobolan) bleached jute fabrics were mordanted with potash alum (10% owm) at a temperature of 80 °C for 30 min, at a 1:20 LR. The mordanted jute fabrics were dried without washing for subsequent dyeing with natural dyes.
Extraction of Natural Dyes
Respective parts of manjistha, annatto, ratanjot, and babool were dried, ground to powder, and soaked in water for 12 h at three different pH conditions (acidic, alkaline, and neutral). After soaking, the dye solution was boiled for 1 h. The resulting extracts were cooled to RT and filtered to remove the insoluble residues. The resulting filtrate was then used as a stock dye solution for subsequent dyeing experiments.
Dyeing of Control and Pre-Mordanted Jute Fabric
Bleached and pre-mordanted jute fabric samples were dyed with natural dye extracted from manjistha, annatto, ratanjot, and babool under different pH conditions. Color from natural dyes (30% owm) was first extracted and water was added to the extracted solution to adjust the LR at 1:20. Alkaline pH 9-10 was maintained for ratanjot, neutral pH 7 for manjistha and babool, and, for annatto, the first half of the dyeing was carried out under alkaline (pH 9-10) conditions and the second half of the dyeing was carried out under acidic (pH 4-5) conditions. Bleached or pre-mordanted jute fabric was entered in the dyebath containing Glauber's salt (10 g/L), and dyeing continued for 90 min at 80-90 °C. After dyeing, the dyed fabrics were washed thoroughly in cold water and then soaped with 2 mL/L of non-ionic surface active agent at 50 °C for 30 min, followed by usual the washing and drying.
Analysis
Whiteness, Yellowness, and Brightness Indices
The whiteness index (WI) was measured on the Hunter scale, the yellowness index (YI) on the ASTM D1925 scale, and the brightness index (BI) on the TAPPI 452 scale of gray. Scoured and scoured-bleached jute fabric samples were measured using a Spectrascan-5100 spectrophotometer with the relevant software for reflectance measurement.
Dyeing Evaluation
Color strengths were determined by calculating K/S (surface color strength) values and L*, a*, b* values (color coordinates) from data at a given λmax (nm) value obtained from a Spectrascan-5100 spectrophotometer. The measurement conditions were angle of observer (10°) and illuminant (D65), with speculate radiation included. The relative color strength (K/S value) of different natural dyed jute fabrics were measured by the light reflectance technique using the Kubelka-Munk equation (Eq. 1).
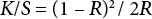
K is the coefficient of absorption, S is the coefficient of scattering, and R is the reflectance. R values of dyed fabrics were measured on a Spectrascan-5100 spectrophotometer. Color values L* (lighter/darker), a* (redder/greener), and b* (yellower/bluer) were also measured by the Spectrascan-5100 using CIELab color space coordinates.
For levelness of dyeing evaluations, K/S values were evaluated at 10 different positions of the fabric sample, and their CV% was determined.
Colorfastness
Washfastness of the dyed samples were determined as per IS:3361-1979 in a Launder-O-Meter. Washfastness ratings were evaluated by the computer color matching system.
Crockfastness (dry and wet) of the dyed samples were determined as per IS:766-1984 using a crockmeter and relevant gray scales.
Lightfastness of the dyed samples were determined as per IS:2454-1984 using a Shirley MBTF Microsal Fade-O-Meter and blue wool standards.
Ultraviolet Protection Factor (UPF)
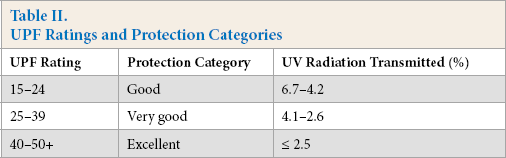
UV transmission analysis of greige, scoured, bleached, mordanted, and various dyed samples were performed using a Labsphere UV Transmittance Analyzer (Model-UV 2000S) with AATCC Test Method (TM) 183-2004. Mean UPF values were reported as an average of nine readings (Table II).
UPF Ratings and Protection Categories
Antimicrobial Activity
Antimicrobial activity against two Gram-positive bacteria (Bacilus subtilis and Staphylococus aureus) for all of the mordanted and natural dyed jute fabrics were tested quantitatively by AATCC TM 100-1999.
Results and Discussion
The present jute research has focused on determining the proper mordanting technique and natural dye selection, as well as optimal application condition for attaining natural dyeing with very good color yield, fastness properties, uniform dyeing, and good protective properties against UV radiation and two test bacterial strains. Results obtained, along with their appropriate discussion, follows.
Greige jute fabric was scoured and bleached with hydrogen peroxide by the conventional process. Jute fabric weight loss was 3.5% and 4.6 %, respectively, after scouring and bleaching. The fabric became absorbent and white after this treatment. Optical properties of greige, scoured, and bleached jute fabrics were evaluated and given in Table III.
Effect of Pretreatment on Optical Properties of Jute Fabric
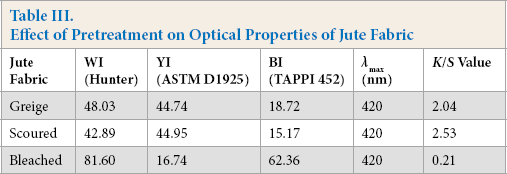
Scouring made the fabric darker, with reductions in whiteness and brightness (Table III). There was a substantial improvement of whiteness and brightness, with reduction of yellowness, after conventional bleaching. These fabrics were then used for dyeing with natural dyes.
Four different natural dyes were used for coloration of jute fabric (Table I). For optimal use of natural dyes, standardization of the extraction conditions from the dyes’ sources and proper conditions of dye application onto the fabric is essential. For extraction condition optimization of natural dyes, all dye sources were first soaked in acidic, alkaline, and neutral conditions for certain durations. The soaked material (for 12 h) was then boiled for 2 h until the color present in the source material came out and dissolved in water. The dye solution was then filtered. The extracted dye solutions were used for bleached jute fabric dyeing with each color in three different media (i.e., alkaline, acidic, and neutral). The dyed fabrics were subsequently evaluated for optical and fastness characteristics. The results are tabulated in Table IV.
Optical Properties of Jute Fabric Dyed with Natural Dyes at Different pH Values
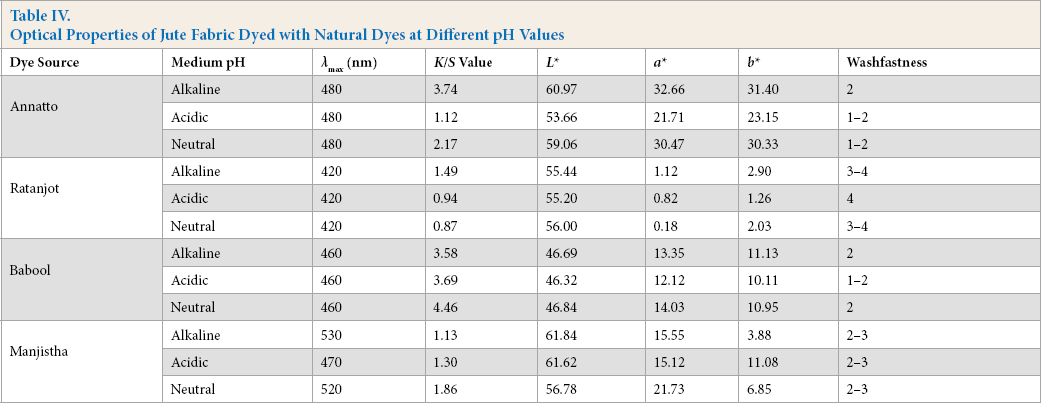
The λmax values of each these dyes were different and they produced various shades on bleached jute fabric (Table IV). Washfastness of jute fabric dyed with ratanjot and manjistha was moderate to good. However, washfastness of jute fabric dyed with annatto and babool extracts was low.
The following conditions were found to be optimal for the various natural dyes with respect to extraction and application on bleached jute fabric.
Annatto—Alkaline medium
Ratanjot—Alkaline medium
Babool—Neutral medium
Manjistha—Neutral medium
To improve the dye affinity, as well as fastness properties, of naturally dyed jute fabric, bleached jute fabrics were first mordanted with a test bio-mordant, and then further mordanted with three different concentrations of a test inorganic mordant. All the mordanted jute fabrics were evaluated for optical properties and these results are tabulated in Table V.
Effect of Double Mordanting on Optical Properties of Jute Fabric
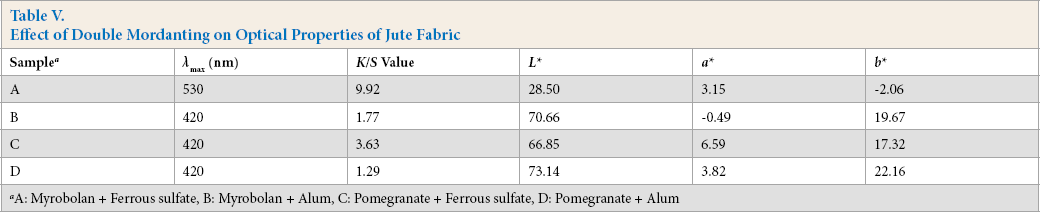
A: Myrobolan + Ferrous sulfate, B: Myrobolan + Alum, C: Pomegranate + Ferrous sulfate, D: Pomegranate + Alum
In all cases, bio-mordanted jute fabric further treated with ferrous sulfate produced a dark, reddish mordanted jute fabric. For potash alum treated, bio-mordanted jute fabric, it became lighter and yellower, with less redness. For double mordanting, due to supportive and additive color interactions, K/S values increased for bio-mordant plus subsequent ferrous sulfate treatment, whereas K/S values decreased for bio-mordant plus subsequent alum treatment due to complementary or opposite color interactions.
Double-mordanted jute fabric samples were subsequently dyed with natural dye extracted from manjistha, annatto, ratanjot, and babool. All dyed samples were then evaluated with the computer color matching system. The evaluation results are tabulated in Table VI.
Effect of Double Mordanting on Dyeing Behavior of Naturally Dyed Jute Fabric
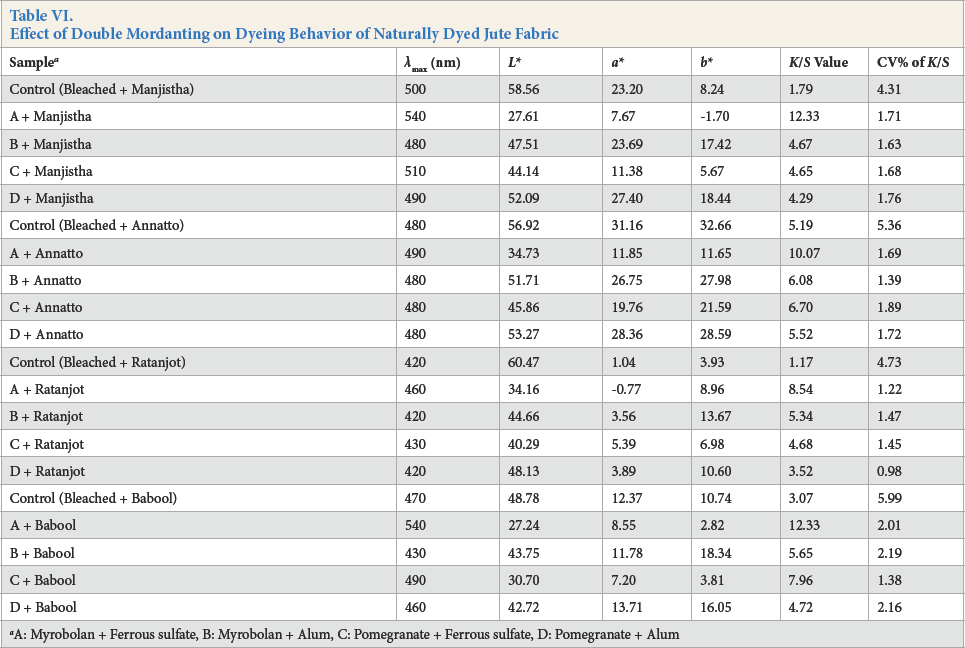
A: Myrobolan + Ferrous sulfate, B: Myrobolan + Alum, C: Pomegranate + Ferrous sulfate, D: Pomegranate + Alum
Double mordanting of jute fabric and subsequent dyeing with natural dyes produced substantial improvements in dyeing color yield and levelness over single mordanted and natural dyed jute fabric (Table VI). Mordanting using the bio-mordant and ferrous sulfate combination, followed by dyeing using natural dyes, resulted in changes in hue of the original color. Color yield produced in these cases were also maximal. For the ratanjot dyeing, the b* value (yellowness) was much higher than the a* value (redness), producing a yellow tone. But, for manjistha dyeing, the a* value was higher than the b* value, indicating a reddish tone. For the other two dyes, pure color (occurring when a* and b* values were similar) was produced.
Analysis of Table VII indicates that double mordanting produced further improvement in washfastness of the naturally-dyed jute fabric compared to single mordanted and naturally-dyed jute fabric. Vegetable mordant, containing tannic acid (acts as a mordant to combine with metallic salt to anchor natural dyes), resulted in improvements of color yield and washfastness. Lightfastness and crockfastness properties of naturally-dyed jute fabric after single mordanting or double mordanting were identical.
Effect of Double Mordanting on Fastness Properties of Naturally Dyed Jute Fabric
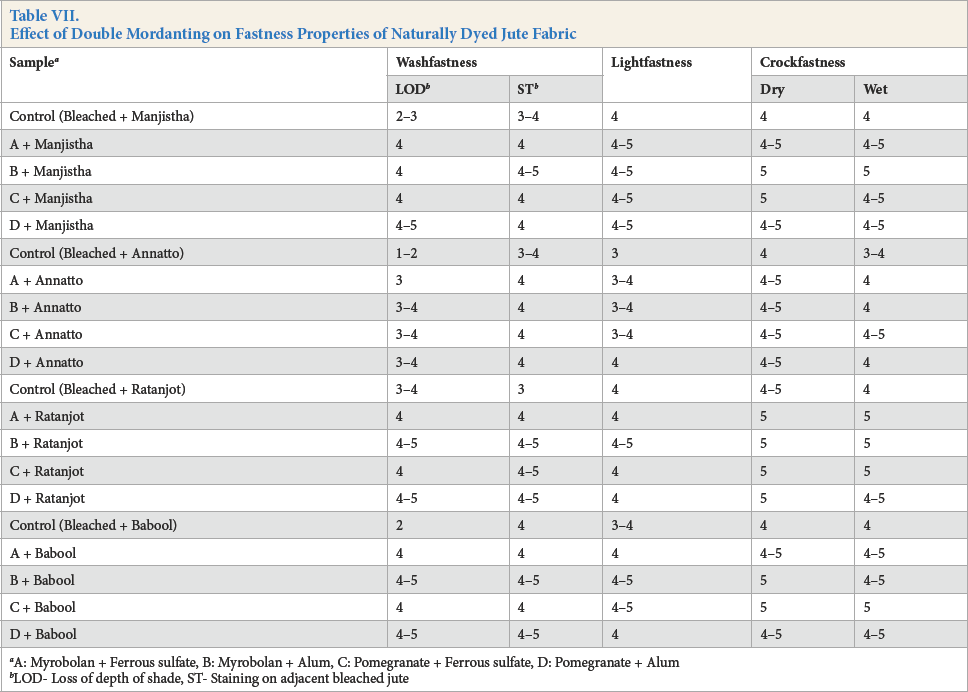
A: Myrobolan + Ferrous sulfate, B: Myrobolan + Alum, C: Pomegranate + Ferrous sulfate, D: Pomegranate + Alum
LOD- Loss of depth of shade, ST- Staining on adjacent bleached jute
UV-protection properties of undyed jute fabrics can be explained in terms of fiber composition and fabric construction. Plain weave greige, scoured, and bleached hessian fabric gave poor UV protection, with very high transmission of both UV-A and UV-B radiation (Table VIII).
Ultraviolet Protection Properties of Greige, Scoured, and Bleached Jute Fabric
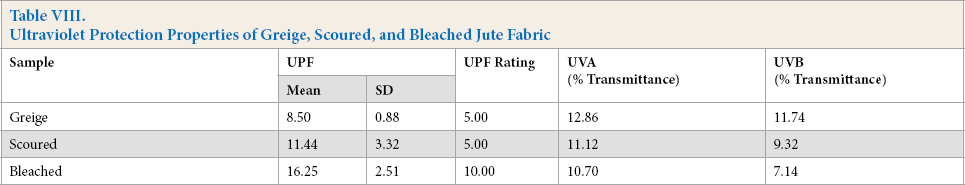
Double mordanting of jute fabric resulted in further improvement of UPF rating and UV radiation transmission (between 4.2% and 6.7%) (Table IX). Therefore, UV protection was good for all double-mordanted jute fabric. Double mordanting imparted pale to medium color to the jute fabric, which blocked and/or absorbed harmful UV radiation.
Effect of Double Mordanting on Ultraviolet Protection Properties of Dyed Jute Fabric
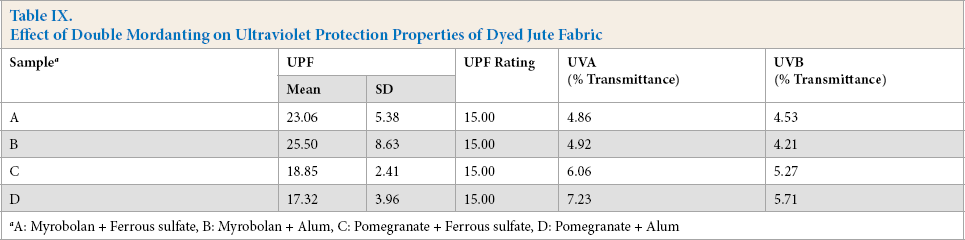
A: Myrobolan + Ferrous sulfate, B: Myrobolan + Alum, C: Pomegranate + Ferrous sulfate, D: Pomegranate + Alum
Double mordanting of jute fabric before natural dyeing was accompanied by improvement in UV-protection properties irrespective of mordants used (Table X). A very good UPF rating was produced by manjistha, annatto, and babool dyed jute fabric after double mordanting. Maximal UV protection in these test fabrics occurred when the myrobolan-ferrous sulfate pretreatment was used. Improvement in UV protection was mainly attributed to the synergistic effect of mordant and natural dyes, thereby maximizing UV protection and minimizing UV transmittance.
Effect of Double Mordanting and Natural Dyeing on UV Protection Properties of Dyed Jute Fabric
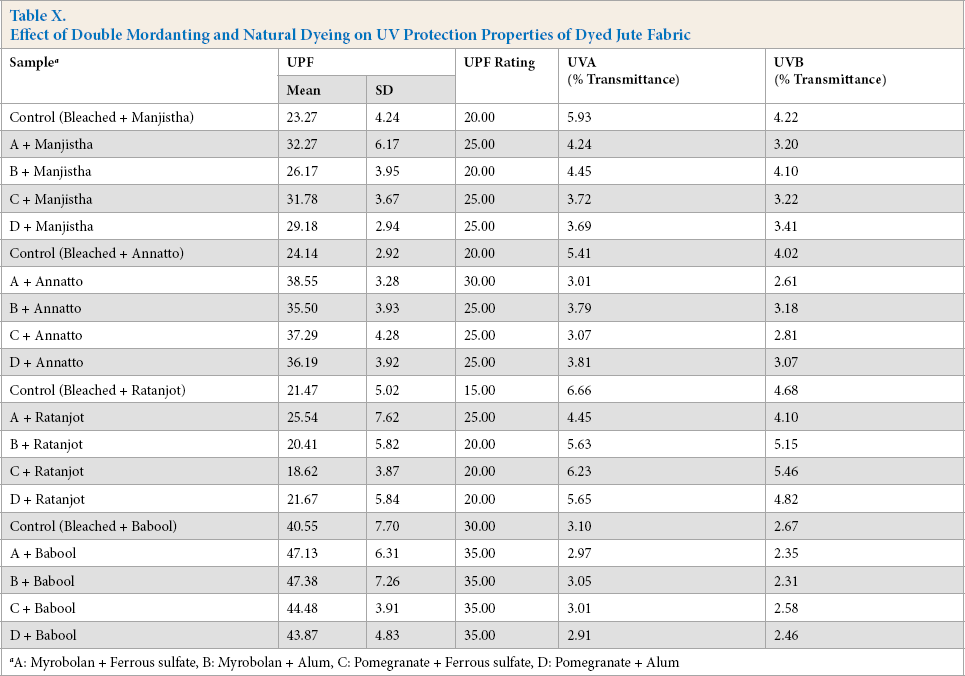
A: Myrobolan + Ferrous sulfate, B: Myrobolan + Alum, C: Pomegranate + Ferrous sulfate, D: Pomegranate + Alum
The K/S value of the dyed fabric is a measure of the color depth. Since pre-mordanting using bio-mordant and inorganic mordant combinations produce higher K/S values, the UV radiation transmission was less, resulting in higher UPF values. The degree of protection imparted after dyeing is a function of the colorant concentration in the fabric. 13 In addition, darker colors provided better protection due to higher UV absorption, as in the case of the bio-organic and ferrous sulfate double mordanted natural dye combination. Therefore, good protection against UV radiation, from dark hues or high colorant concentrations in the fabric, has to be provided.
The extent of improvement in the UV protection property using natural dyes is governed by the chemical structure of the dyes, their absorption characteristics in the UV region, and the extent of interaction and complexation with the pre-mordanted substrate, as well as the ability to block or absorb hazardous UV radiation. 14 Improvement in UPF was determined by the dyeing regime (i.e., double mordanting following the dyeing > dyeing only > double premordanting > bleached fabric > greige fabric) reflecting the synergistic effect of the mordant and dye, as well as the complex formation between the dye and the mordanted substrate, giving an ability to block UV transmission and to absorb harmful radiation. 12
Antimicrobial activities against Gram positive bacteria of all mordanted and naturally dyed jute fabrics were tested quantitatively by AATCC TM 100-1999. The antibacterial activity was expressed as percent reduction in bacterial count (RBC%) and is given in Table XI
Effect of Mordant and Natural Dye on Antimicrobial Activity
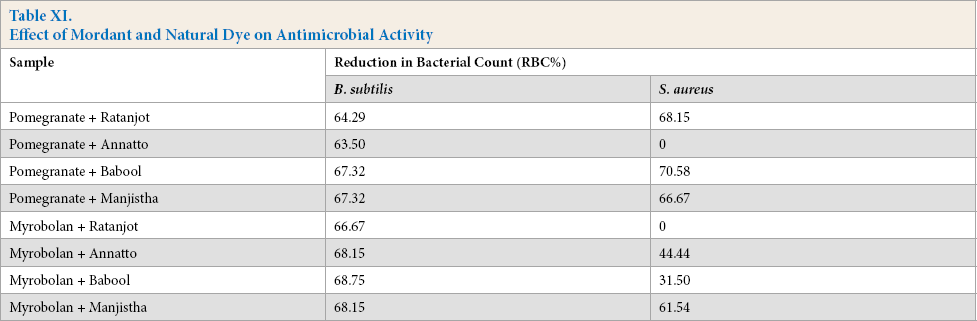
All bio-mordanted jute fabric, after dyeing with natural dyes extracted from all plant extracts tested, produced satisfactory protection against two Gram positive bacteria. This may be due to the presence of active antimicrobial components (i.e., anthraquinone and other phenolic groups present in the natural dye molecule).
To make the process of natural dyeing of jute fabric more environmentally-friendly, eco-friendly trisodium citrate has been used as exhausting agent in place of sodium sulfate and a comparative study was done. The jute fabric was scoured, bleached, and both single and double mordanting was done, followed by subsequent dyeing with natural dye extracted from manjistha. Lightfastness of greige and scoured jute fabric was 4-5, whereas lightfastness of bleached jute fabric was 2-3. For optimization of citrate concentration, three different concentrations (1,2, and 3 g/L) were used for dyeing the bleached jute fabric with manjistha. The dyed fabric was evaluated and results given in Table XII. Citrate concentration of 2 g/L was found to be optimal.
Effect of Glauber's Salt and Trisodium Citrate on Optical Properties of Manjistha Dyed Jute Fabric
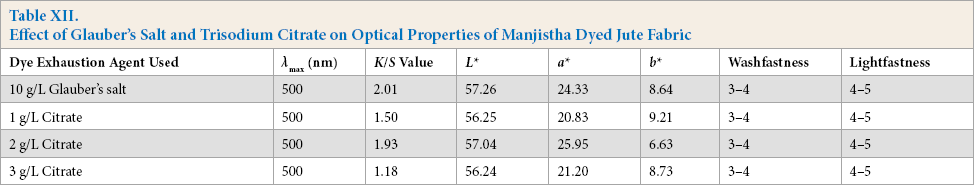
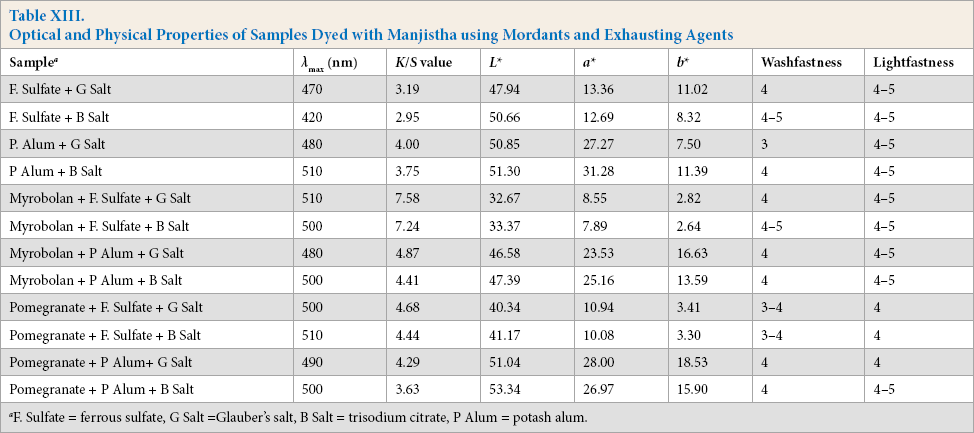
Dyed samples using both mineral salt and citrate were evaluated for optical and physical properties and the results are given in Table XIII.
Optical and Physical Properties of Samples Dyed with Manjistha using Mordants and Exhausting Agents
F. Sulfate = ferrous sulfate, G Salt =Glauber's salt, B Salt = trisodium citrate, P Alum = potash alum.
Analysis revealed that depth of shade of the natural dyed sample produced by using citrate was slightly less than that produced using Glaubers salt as exhausting agent (Table XIII). But there was improvement of washfastness and lightfastness for samples using citrate.
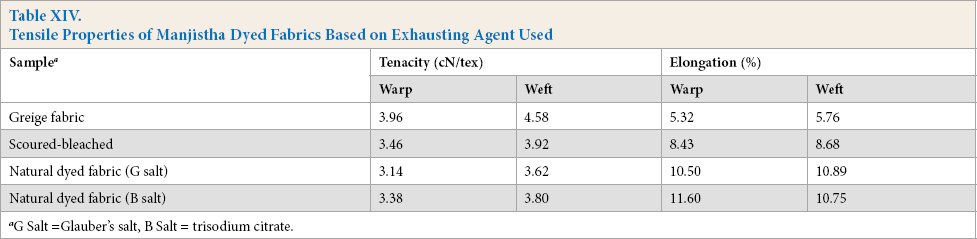
Tensile properties of dyed samples produced using either Glauber's salt or citrate was evaluated, compared, and given in Table XIV.
Tensile Properties of Manjistha Dyed Fabrics Based on Exhausting Agent Used
G Salt =Glauber's salt, B Salt = trisodium citrate.
It is clear from Table XIV that tensile strength retention was greater if natural dyeing was carried out using citrate as exhausting agent compared to Glauber's salt.
Conclusions
Premordanting with sequential treatment of bio-mordant followed by inorganic mordant resulted in substantial improvement of uniformity and levelness of dyed jute fabric using natural dyes extracted from manjistha, annatto, babool, and ratanjot. Double mordanting produced high K/S values after natural dyeing— good to excellent washfastness ratings were achieved. Natural dyes gave moderate to good lightfastness properties. Crock-fastness was very good to excellent irrespective of the dyeing method.
Greige, scoured, and bleached jute fabrics had no UV protection properties. UPF values of the naturally-dyed jute fabrics were good. Babool dyeing gave very good UV protection ratings. All naturally-dyed jute fabric produced very good UV protection properties after premordanting with bio-mordant and inorganic mordant combinations. UV protection properties of naturally-dyed jute fabric followed the order babool > annatto > manjistha > ratanjot.
All of these naturally-dyed fabrics showed satisfactory antimicrobial activity against B. subtilis and S. aureus.
The optimized concentration of citrate exhausting agent was 2 g/L for natural dyeing using manjistha. In this case, the depth of shade was slightly less than that when Glauber's salt was used, but washfastness, lightfastness, tensile, and handle properties were better.
Footnotes
Acknowledgement
The authors are thankful to Dr. D. Nag, Director, National Institute of Research on Jute and Allied Fibre Technology, Kolkata, India, for his continuous encouragement and support for this work.
