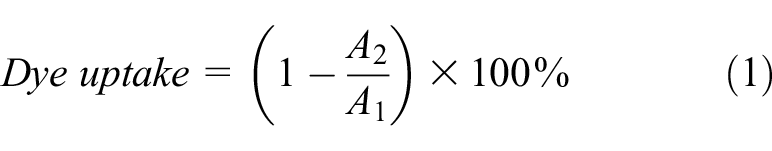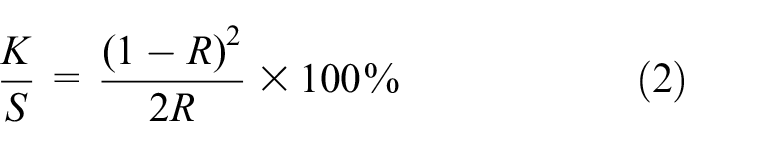Abstract
This preliminary research discusses the extraction of colorants from pumpkin peel as vegetable dyes and the potential dyeing capability of silk and cotton fabrics. For pumpkin extracts, thermal gravity analysis has verified the range of temperature for application, while a combination of UV–vis spectra and high-performance liquid chromatography analysis confirmed the main colored components and structural characteristics. It was found that the extracts of this vegetable dye have excellent thermal stability as well as stability in neutral and acid conditions. With the objective of achieving the greatest absorbency rate of extracted solution, the following optimum extraction conditions were obtained: 100% ethanol, an extraction temperature of 70°C, an extraction time of 60 min, and a material-to-liquor ratio of 1:10. To attain the highest dyeing uptake rate and K/S values, the optimum dyeing profiles with meta-mordanting for silk fabrics were found to be 95°C, 90 min, pH 5.5, and a liquid ratio of 1:60. The optimum dyeing profiles with meta-mordanting for cotton fabrics were 95°C, 90 min, and a liquid ratio of 1:60. Nearly all colorfastness results for silk and cotton fabrics met the basic requirement of the China National Standard, except a few.
Introduction
The interest in the use of natural colorants from plant sources for color has grown in recent years. This increased demand for plant-based colorants, particularly for textile applications, is due to the market’s appeal to find alternative sustainable natural compounds to synthetic dyes and chemicals, at least partially. Many plants and animal/insect sources have been identified for the extraction of color and their diversified use in textile dyeing. Colorants extracted from natural materials could generate moderate and graceful shades on different types of textiles and are being investigated as functional agents for highly active textile surfaces having properties such as deodorization, antioxidant, antimicrobial, and ultraviolet (UV) protection. 1
Native to North America, pumpkins (Cucurbita) are one of the oldest domesticated plants, having been cultivated as early as 7500 to 5000. Nowadays, the name is most used for cultivars of Cucurbita pepo, but some cultivars of Cucurbita maxima, Cucurbita argyrosperma, and Cucurbita moschata with similar red-yellowish appearance are also sometimes called “pumpkin.” 2 World production of pumpkins was 27.6 million tons in 2018, with China alone accounting for one third of the total production. 3 Pumpkins are widely grown for commercial use and food, aesthetics, and recreational purposes. Understandably, pumpkin has majorly been consumed as food; however, the by-products such as peel and pulp account for about 20%, which might be repurposed as natural colorants. A prized traditional vegetable, namely C. maxima, is known for its deep yellow to orange coloration. Thereby, it is assumed that the combination of natural fiber and pumpkin colorants would be ideal for the manufacture of eco-friendly clothes with dependable color qualities. The color of pumpkin is due to carotenoid and its derivative lutein combination, while chemical formulas of these two compounds are shown in Figure 1. According to the United States Department of Agriculture, carotenes (α- and β-carotene, 0.72 mg) are terpenoids without oxygen and lutein zeaxanthin (0.15 mg), which are mainly responsible for orange and/or red colors. 4
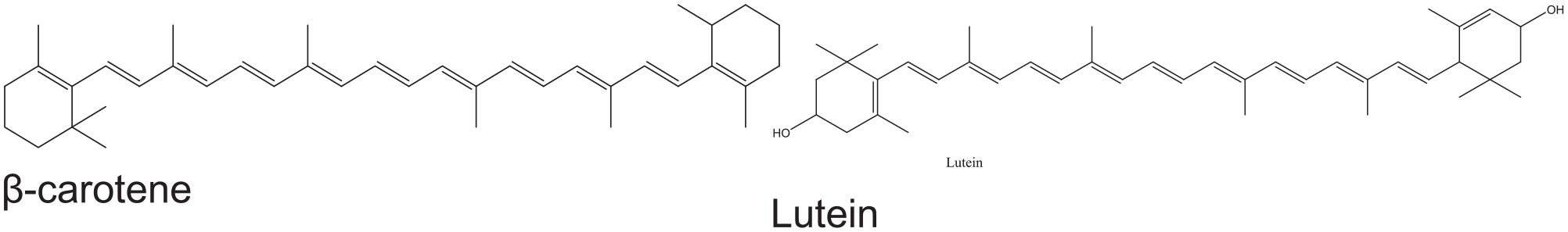
Structural diagram of β-carotene and lutein.
There are few preliminary research works on the extraction of pumpkin colorants. Properties and recent research advances in the application of plant-derived colorants in food, cosmetics, and textile materials, including carotenoids, were reported. 5 Lyudmila Rodionova carried out experiments on a different carotenoid pigments contained in pumpkin fruits. The quantitative characteristics of carotenoids were established, and the predominant carotenoid pigments for the studied pumpkin varieties were specified. 6 For pumpkin, an increase in temperature up to 45°C would cause softening of the plant tissue, accelerate the molecular movements and penetration of solvent into the plant material, enhance the solubility and diffusivities of the pigments and colors into the solvent, and increase the extraction yield. 7 Sharma determined the effects of green extraction techniques on carotenoids, polyphenols, and antioxidant activities of pumpkin pulp and peel. Innovative green extractions (ultrasound and microwave-assisted extractions) synergized with corn oil were compared with conventional extraction. 8 The results proved to be advantageous in terms of improving extraction efficiency; reducing extraction cost, time, and energy; and enhancing the yield. An ultrasound-assisted extraction (UAE) method was observed to have the highest efficiency. In another attempt by Feng, UAE was used to extract trans lutein and total trans carotenoids from the pumpkin peel. Five optimized extraction factors (solvent type, ultrasound frequency, power, solvent-to-sample ratio, and ultrasonic time) were obtained. 9 Kulczyński’s study confirmed the high diversity of pumpkin cultivars in terms of their antioxidant activity, which is more affected by the type of cultivar than by the pumpkin species. 10 Plant-associated strains of the species Paenibacillus polymyxa and Pseudomonas chlororaphis, originally isolated from pumpkins, were tested for their production of volatiles for antimicrobial effect on clinically relevant pathogens which were applied on textiles. 11 Karthikeyan further studied antimicrobial effects and dyeing of pumpkin peel. The natural dye from pumpkin skin was extracted, and the chemical dye effluent from the textile industry was collected. A comparative analysis on the degradation and decolorization of these two dyes in Kirk’s medium supplemented with glucose, Bacillus licheniformis and Aspergillus niger showed that the maximum microbial growth and degradation were achieved. 12 In Vershinin’s investigation, carotenoids and their derivatives have many other interesting biological functions, including specific coloration forms in plants, spectral filtering, and acting as a visual pigment as a chromophore in bacteriorhodopsin photosynthesis. 13 In addition, there were studies which investigated the potential of low-cost adsorbents using pumpkin seed powder.14,15
Despite so many studies on carotenoids, studies focusing on their coloring and coloration properties are in the minority. 16 For dyeing application to textile materials, there were, to the best knowledge of us, no published articles in this area. Therefore, the objective of this study was to investigate whether vegetable colorants extracted from pumpkin peel, after application to silk and cotton fabrics, would be able to provide sufficient colors to natural protein and cellulosic fiber with reasonable colorfastness. This research will bring new knowledge in this area.
Experimental
Material, Equipment, and Chemicals
Pumpkins in this study were purchased from a supplier in Jinan, China. The name is “red (or gold)-skin pumpkin” for a direct translation of Chinese, and it is a local variety of Rouge Vif d’Etampes, C. maxima Duch. species under Cucurbita L. genus. The performance properties of the greige silk and cotton fabrics, obtained from Huzhou Silk Company of Zhejiang province, are described in Table S1 in the Supplemental Information file. Standards of β-carotene and lutein were purchased from Yuanye Bio-Technology (Shanghai, China). The high-performance liquid chromatography (HPLC)-grade solvents such as methanol and acetonitrile were purchased from Energy Chemical (Anhui, China).
Extraction
The UAE method was adopted with slight modification from a study by Song and Sharma with the same ultrasonic device.8,9 Briefly, the pumpkin peel was first chopped into small pieces. Then, pumpkin peel pieces were dried in an oven at 60°C for 4 h before grinding. The pigments in the pumpkin peel were extracted using ethyl alcohol. The extraction was carried out in an ultrasonic cleaner (KQ-250 GVDV, Kunshan Ultrasonic Instruments Ltd. Co., Kunshan, China), with a frequency of 45 kHz and a maximum input power of up to 200 W. The standard ultrasonic mode was applied. During the sonication and extraction processes, the sample container was kept in the water bath at a defined temperature monitored by a temperature circulator. Specifically, dried pumpkin peel powder was mixed with an ethyl alcohol solvent (CH3CH2OH, 99.7%, A.R., Shanghai) in a 200 mL triangular flask. After sealing, this flask containing the mixture was immersed in the ultrasound bath and fixed well in the same position during sonication for a defined period. Variables with known principal effects were selected, including extraction time, extraction temperature, solvent concentration, and solvent-to-sample ratio. The extracted liquid was filtered first and then put into a rotary evaporator. The concentrated dye paste was obtained with a yield of 63.69% (paste weight vs dry pumpkin powder weight) and ready for further processing. Furthermore, 10 g of concentrated pumpkin dye paste was dispersed with water in a volumetric flask with 250 mL capacity, which was used as master solution for textile dyeing on cotton and silk fabrics.
Dyeing
A typical dyeing profile for natural dyeing is as follows: the fabric versus liquor ratio was 1:40, the heating rate was 1°C/min from 40°C, and holding time for mordanting was 30 min at 95°C and for dyeing was 60 min at 95°C. 17 The generic process is (Mordanting) and (Dyeing) > Washing > Soaping > Washing > Drying. For cotton fabrics, to achieve better color properties, a chitosan agent was used to pre-treat cotton fabric before dyeing. Glacial acetic acid (CH3COOH, 99.7%, A.R., Shanghai) was used to adjust the pH of the dyeing solution. Ferrum mordant (FeSO4, 99.7%, A.R., Shanghai) has been used in the following dyeing experiment research for both fibers since it generally worked well with the brown category of natural dyes. Preliminary experiments with alum, ferrum, and copper mordants suggest that only ferrum mordants exhibited the favorable color and absorbency to pumpkin dyes at certain concentrations; therefore, during the mordanting process of this research, the concentration of ferrum mordant was kept constant at 7.5%.
Measurement and Evaluation
The dyeing profiles (parameters) and colorfastness of the silk and cotton fabric samples were measured to identify the characteristics of the extracted pumpkin colorants in this study.
Dye absorbance, within the UV–visible range of the spectrum, for extraction liquid and dye uptake were measured with a UV–vis spectrophotometer (759s UV–vis, Shanghai LINGGUANG Tech. Ltd., China; SN759S21016). The rate of dye uptake refers to the concentration percentage of dye on the fiber compared with the concentration percentage dye in the original dye solution. The dye update was calculated using equation (1):
where A1 refers to the absorbance of dyeing liquid before dyeing; A2 refers to the absorbance of dyeing liquid after dyeing. Dye absorbance was measured at 455 nm.
Thermogravimetric analysis (TGA) was conducted using the NETZSCH TG instrument (SN:209F3).
The K/S value is a function of the color depth of a dyed sample. The higher the value of K/S, the deeper is the color. The K/S value can be calculated using equation (2):
where R refers to reflectance data of color samples.
A color spectrophotometer (datacolor 800 with QC software) was used to measure color reflectance data and K/S values. The colorfastness to washing, wet and dry crocking, and sunlight were measured in accordance with the Chinese National Standards Test Method: GB/T 12490-2014: “Color fastness to domestic and commercial laundering,” GB/T 3920-2008: “Color fastness to rubbing,” and GB/T 8427-2008: “Colorfastness to artificial light: Xenon arc,” respectively. The evaluation was based on the American Association of Textile Chemists and Colorists (AATCC) Evaluation Procedure 1—2012: “Gray Scale for Color Change” and AATCC Evaluation Procedure 1—2012: Gray Scale for Color Staining.
Results and Discussion
TGA and Spectral Analysis of Pumpkin Dyes
TGA is a method of thermal analysis in which the mass of a sample is measured over time as the temperature changes. As is shown in Figure 2(a), the pumpkin peel extract compound maintains its physical and chemical integrity below 100°C, and there is a significant mass loss between 120°C and 200°C. Since natural textile dyeing usually takes place where the temperature is below 100°C for natural fibers, there are negligible changes if the dyeing temperature is controlled lower than the boiling point, which is suitable for the dyeing of both natural cellulosic and protein fibers.
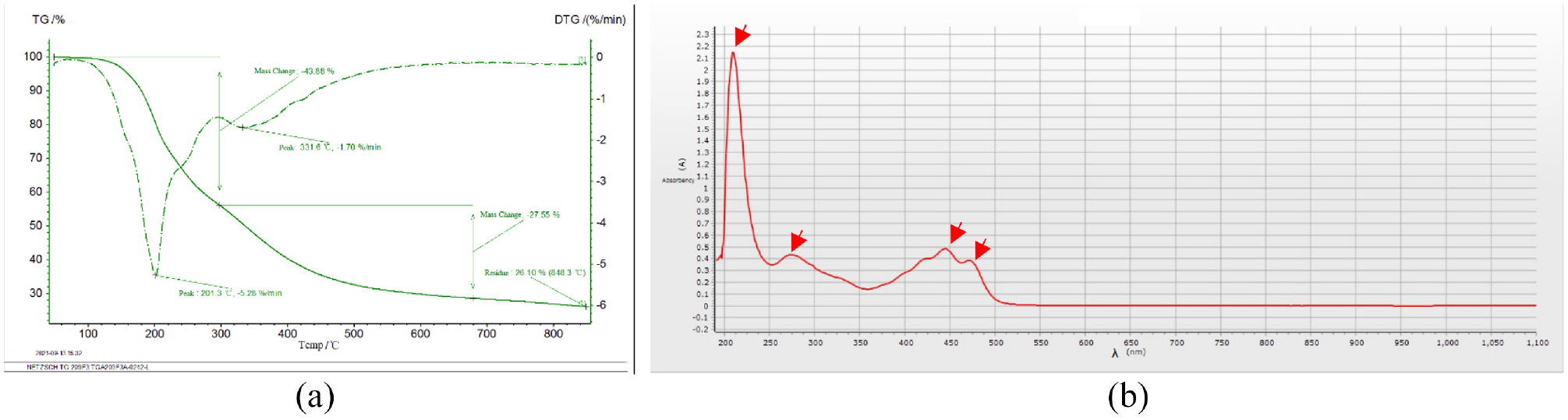
(a) Thermogravimetric analysis diagram of pumpkin extracts and (b) UV–vis spectrum of pumpkin extracts.
The general structure of the carotenoid and lutein is a polyene linear chain consisting of conjugated C=C bonds in trans-configuration and terminating in rings. This structure of conjugated double bonds leads to a high reducing potential, or the ability to transfer electrons throughout the molecule. The length of both structures plays a role in the pumpkin’s color, as the length of the polyene tail determines which wavelengths of light the plant will absorb. Specifically, in molecules with extended π systems, the highest occupied molecular orbital (HOMO)–lowest occupied molecular orbital (LUMO) energy gap becomes so small that absorption occurs in the visible rather than the UV region of the electromagnetic spectrum. Therefore, both carotenoid and lutein, with their system having extended conjugated double bonds, absorb light with wavelengths in the blue region of the visible spectrum while allowing other visible wavelengths—mainly in the red-yellow region—to be transmitted. 9 Wavelengths not absorbed are reflected and that portion is seen as the color of the pumpkin. 10
We could use the UV–vis region of the spectrum to identify typical groups in the chemical structure since the structure of carotenoid is not complex compared with most other dyes. From Figure 2(b), pumpkin extracts have one strong absorption peak at about 214 nm and one weak peak at around 270 nm, which indicates a slight degree of UV-protective function in that range. These two peaks provide evidence of a chromophore which consists of the π→π* transition of a long-conjugated system. In addition, two absorption peaks at approximately 445 and 470 nm could be found in the visible spectrum, which agrees with the previous publications, and these peaks mainly contribute to the plant color. 9 These two peaks at the higher wavelength could be explained by the existence of autochrome which has either an n-electron with a p-π conjugated system (e.g. −OH hydroxyl group in the case of lutein) or a ring structure (e.g. cyclopentane) or both. 18 A lengthened chromophore of 11 conjugated π-bonds leads to a much smaller energy difference between the π and π* molecular orbitals than from an isolated double bond. Both chromophore and autochrome sections could easily be identified from the chemical formulas of carotenoid and lutein in Figure 1.
Furthermore, a WatersTM HPLC system was used for the purpose of identification. The volume injected was 20 µL. Quantitative determination was performed at 445 nm on a Fisher C18 5 µm, 150 mm × 4.6 mm column with an eluent of a 60:40 mixture of methanol and acetonitrile. The flow rate was 0.8 mL/min. The solvent was filtered and degassed before use. Peaks were identified by comparison of standards and sample peak retention times. The results of HPLC analysis for pumpkin extracts are shown in Figure 3. According to Figure 3(c) and (d), β-carotene and lutein are the colored compounds identified, which are shown as large peaks at about 3.5 and 35 min, respectively. These peaks match the standards of these two compounds, which are shown in Figure 3(a) and (b), respectively.
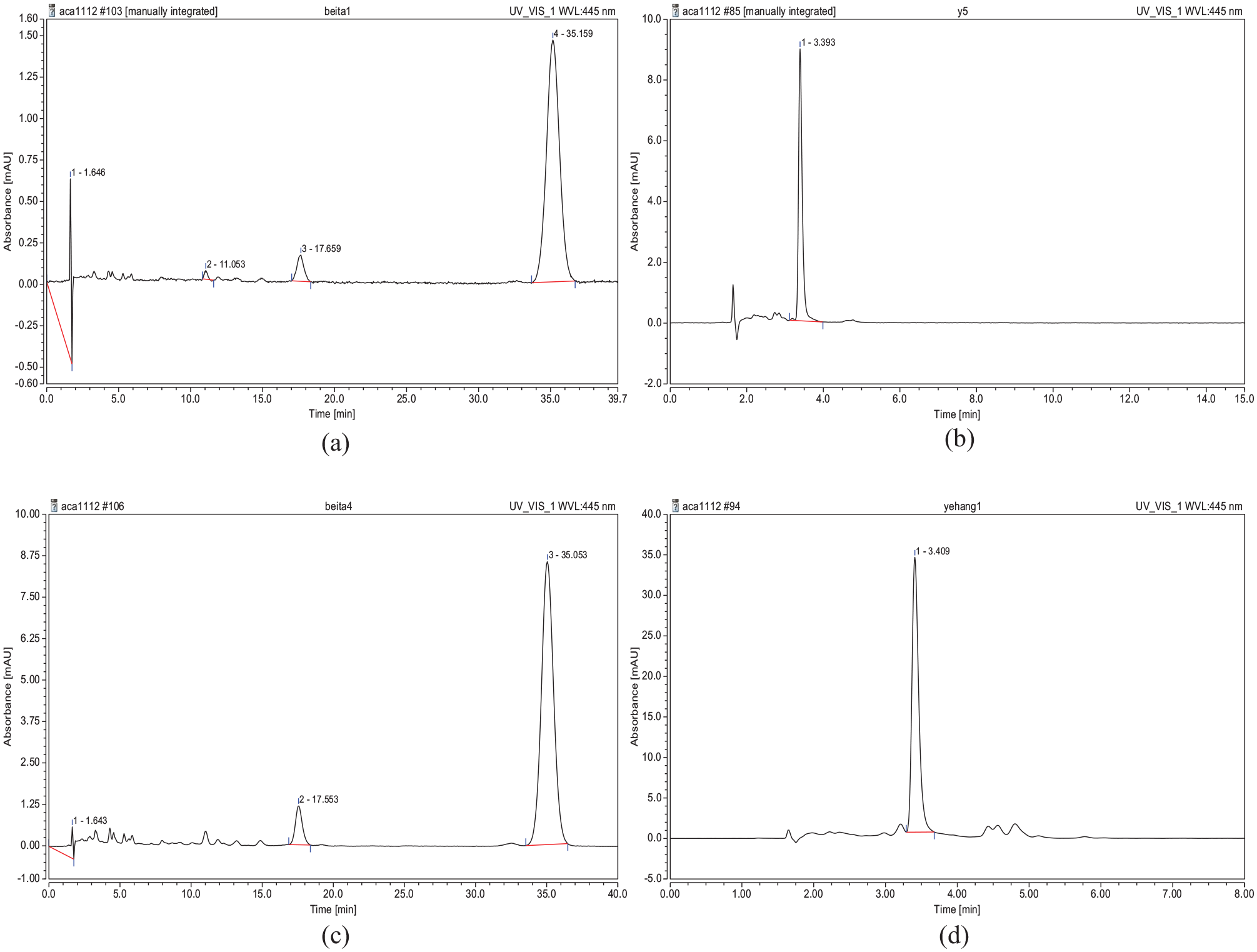
HPLC chromatogram of pumpkin extracts compared to β-carotene and lutein standard: (a) β-carotene standard, (b) lutein standard, (c) β-carotene from the pumpkin extracts, and (d) lutein from the pumpkin extracts.
Extraction Profile
To achieve the optimized dyes extraction conditions for the pumpkin colorants, we designed an orthogonal experiment and chose four main factors that affect extraction efficiency. These factors are solvent concentration, temperature, time, and liquor ratio. Preliminary experiments were performed to determine the range of these factors. Based on the results from the spectra analysis, the absorption rate was measured at a wavelength of 445 nm. The exact levels for each parameter are shown in Table S2 in the Supplemental Information file. The results of absorption for the orthogonal experiment are shown in Table S3 in the Supplemental Information file.
The SPSS software was used to compute the Sums of Squares Type III values (SSTYPE III) for the relative importance of each parameter. The SSTYPE III effect of each variable is evaluated again for all other factors in the design, which is shown in Table S4 in the Supplemental Information file. The higher the SSTYPE III value, the greater the relative importance of the factor to the performance. From Table S4, it could be found that the order of impact for these parameters is C > A > D > B, which means the liquor ratio and solvent concentration have the major influence while extraction temperature and time are less important compared with the other two factors. From Figure 4, the order of significance of extract parameters contributing to pumpkin colorant extraction was found to be A3B2C1D3. To confirm that the selected parameters have the maximum absorbance, we also conducted an additional experiment with the same optimized condition. The result is 0.621, which confirmed the selected parameters. In conclusion, the optimized extraction condition is 100% ethanal, 60 min, 70°C, and 1:10 material:liquor ratio.
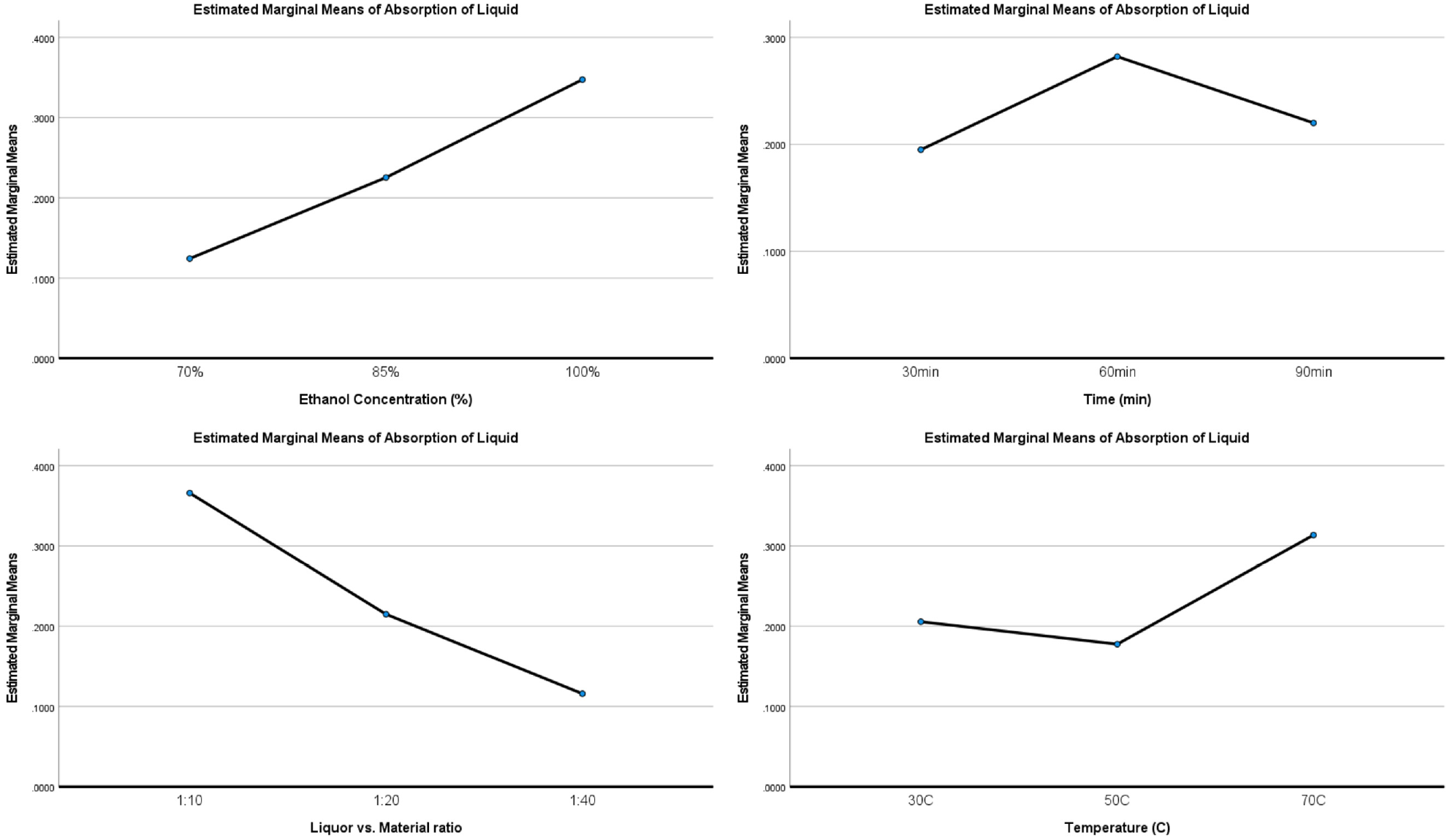
Optimized parameters of solvent concentration, temperature, time, and liquor ratio.
Dyeing Profile for Silk Fabrics
We have experimented with different dyeing methods using pumpkin peel extracts, and meta-mordanting (mordant is added in the dye bath itself) resulted in the highest rate of dye uptake rate and the highest K/S value. Additional single-factor dyeing experiments show that the four most important factors that affect color depth are concentration, time, pH, and temperature. Initial trials were taken to decide the range of each factor concerned. Based on pre-experiments, investigations of K/S value and dye uptake rate (%) were conducted in order to determine the optimum dyeing profile. The K/S value and the dye uptake rate were measured at the maximum absorption wavelength of 445 nm. Thereby, orthogonal design with three levels was used for this procedure and is shown in Table S5 in the Supplemental Information file, and results for K/S and dye uptake rate are presented in Table S6 in the Supplemental Information file.
Taking the analysis of variance (ANOVA) multivariate analysis, K/S and dye uptake data were evaluated in the SPSS software. The results, presented in Table S7 in the Supplemental Information file, revealed the fact that in order to achieve the greatest K/S value and highest dye uptake on silk fabrics, the influence orders of four factors are A > C > D > B for K/S value and A > D > C > B for dye uptake. The SPSS range analysis function also generated Figure 5 for K/S evaluation and Figure S1 for dye uptake evaluation. The trend in Figure S1 exhibits that there are slight variances of dye uptake rate for each parameter, which further means that changes in the dyeing parameters have very little impact on dye update rate. Considering this fact and the high dye update rate (>90%), as well as a clear residue liquid after dyeing, it is reasonable to use the K/S value to decide the best dyeing profile. Therefore, the optimum dyeing profile could be obtained with A3B3C3D2 referring to Figure 6, which is 95°C, 90 min, pH 5.5, and 1:60 material:liquor ratio.
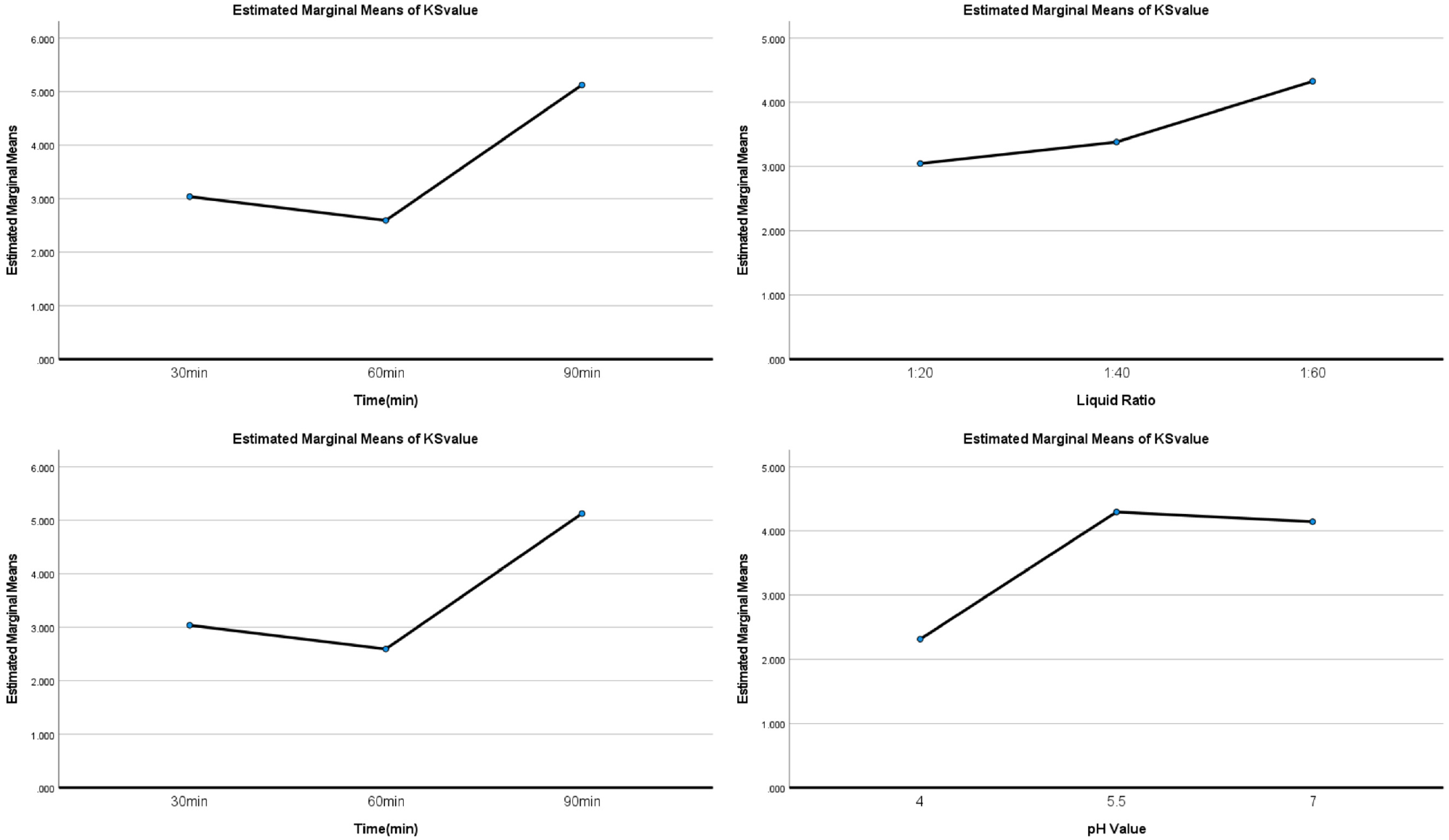
Optimized parameters of solvent concentration, temperature, time, and liquor ratio against K/S values of silk fabrics.
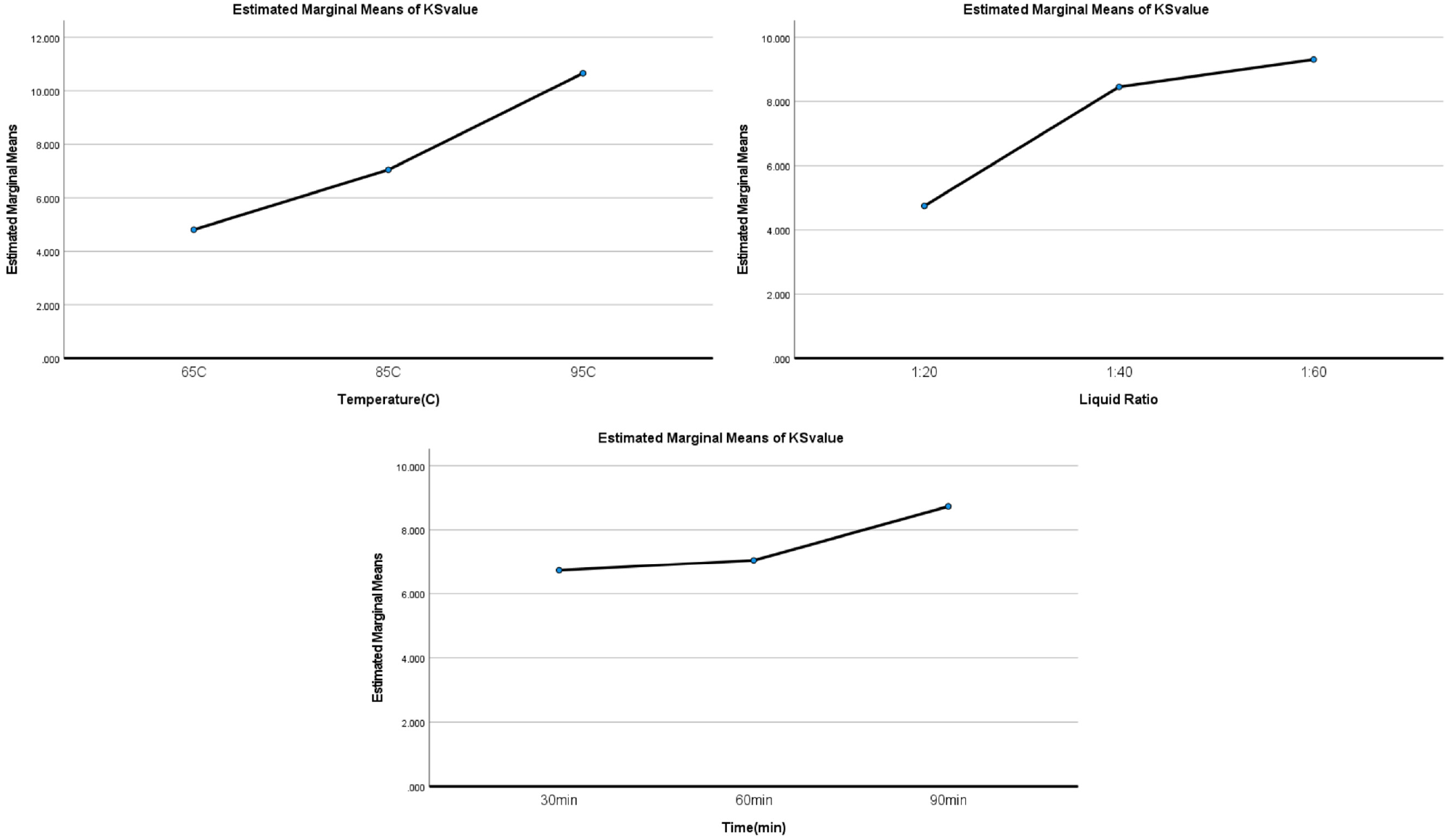
Optimized parameters of solvent concentration, temperature, time, and liquor ratio against K/S values of cotton fabrics.
Dyeing Profile for Cotton Fabrics
Since meta-mordanting resulted in the best color performance, we utilized the same dyeing method as the silk dyeing. The initial trial for different pH values resulted in a greenish liquid with sediment under the alkaline conditions. Therefore, cotton fabrics were dyed in a neutral condition. Based on this fact, we only chose three factors (temperature, liquid ratio, and dyeing time) to study the parameters that influence the cotton dyeing process using an orthogonal design of the experiment, as shown in Table S8 in the Supplemental Information file. The range of each factor was evaluated with a pre-experiment and three levels were assigned to each factor. The results of fabric K/S value and dye update rate are recorded in Table S9 in the Supplemental Information file.
The K/S and dye uptake data were processed using SPSS software. The results are presented in Table S10 in the Supplemental Information file, and optimum parameters are identified in Figure 6 and Figure S2. From Table S10, the influence order of the three factors was A > C > B for dye uptake rate and A > B > C for K/S value. As far as optimum dyeing conditions are concerned, since the dye uptake rates were high (90%) for most of the residue dye bathes, which were clear liquid, we choose to use the K/S factor to decide the optimum conditions. When referring to Figure 6, we conclude the optimum dyeing conditions are A3B3C3: 95°C, 90 min, and 1:60 material:liquor ratio.
Colorfastness and Dyeing Principles
The color fastness results for optimally dyed silk and cotton fabrics are shown in Table 1. Silk fabrics dyed with pumpkin peel extracts exhibited excellent colorfastness to sunlight, as well as staining properties of laundry. However, both wet crocking and washing colorfastness were graded as “3,” which indicates a “noticeable color staining” and “noticeable color change,” respectively. However, all colorfastness results of silk fabrics meet or exceed the Chinese National Standard for “acceptable” colorfastness ratings of 3 or higher.
Colorfastness of silk fabrics and cotton fabrics dyed with pumpkin extracts.
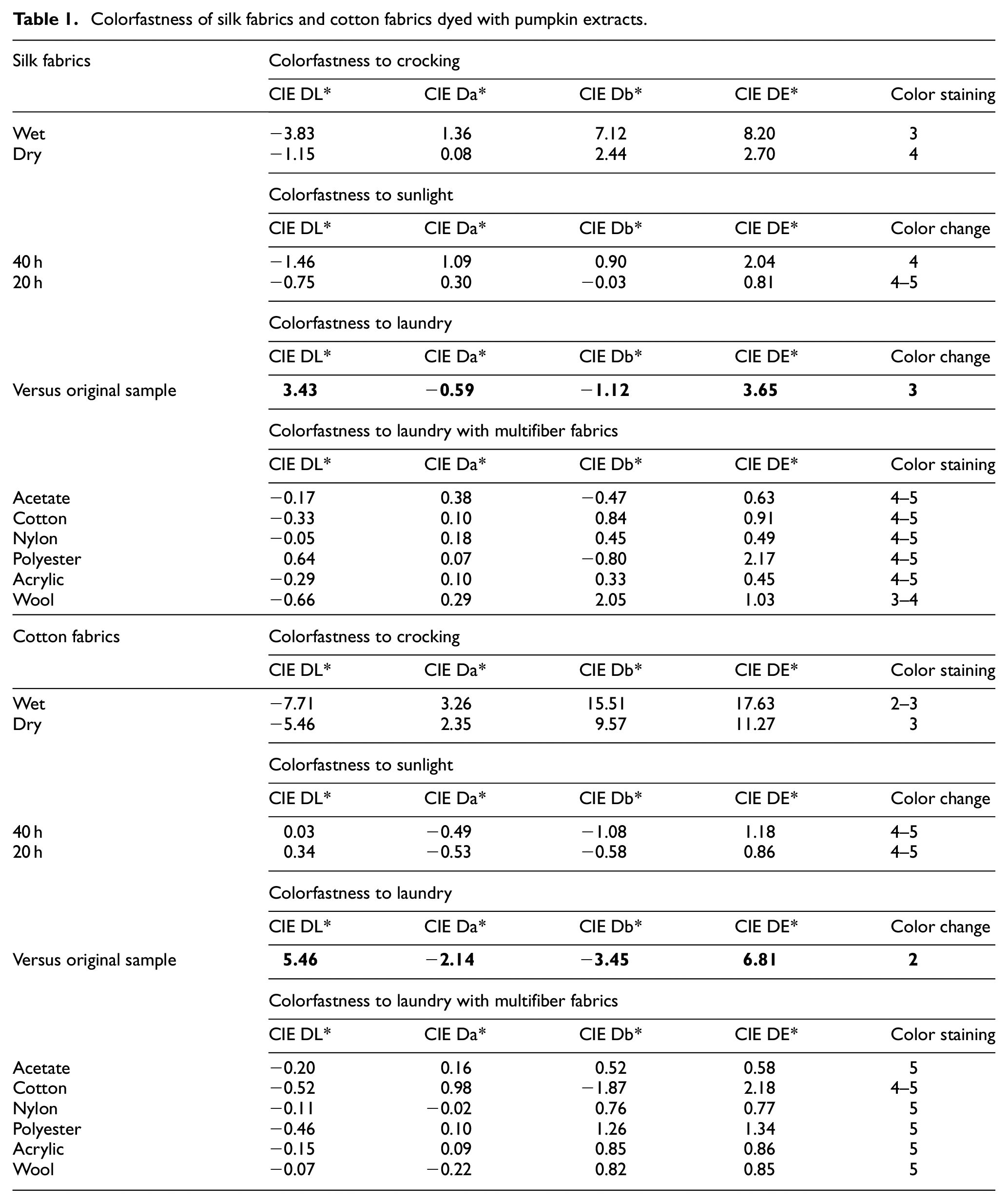
For dyed cotton fabrics, colorfastness to sunlight and colorfastness to laundry (multifiber fabrics staining) were rated as “very good” and “excellent,” respectively. Outstanding colorfastness issues from dyed cotton fabrics are wet crocking and color change to laundry with grade of 3 or 2, which indicate “noticeably” or “considerably” color change respectively. Grades of these two properties are found lower than the acceptable tolerance of the Chinese National Standard. Considering the sustainable aspects of natural dyeing, the acceptable tolerances for natural dyeing could have a bit room of leniency for natural dyes, and some specific textile end uses might be proper where a specific colorfastness would not be so critical.
Overall, the colorfastness performance of natural dyeing using plant dyes is comparatively lower than with their synthetic dye counterparts. Instead of ionic and covalent bonding of the primary forces, the affinity of natural pumpkin dyes to fibers would largely depend on the secondary forces of attraction, namely van der Waals forces, such as dipolar forces, hydrogen bonding, π-H bonding, or dispersion forces. Among these bonds, hydrogen bonding between the dyestuffs and fibers molecules is expected based on the structures of these two components, even though lutein may have a little from the hydroxyl but should be minimal. Probably, the simplest of the dyeing processes is diffuse adsorption in which the dye is not adsorbed on specific sites and may form multilayers within the polymer matrix of silk and cotton fibers. The surface morphology of the fabric, undyed and dyed, was evaluated by scanning electron microscopy (SEM) and is shown in Figure 7. The morphology of silk and cotton fiber has a few slight changes without any apparent rupture due to the coloration process. It is estimated that the majority of pumpkin dyes penetrate the fiber and stay among polymers. The surface is a bit rough but overall uniform and coherent under the SEM, which indicates still a small portion of dyes located on the surface of the fiber, resulting in the inferior crocking colorfastness for both types of fabrics. In addition, the morphological structure images exhibit an intact physical property of fiber and hence indicate that the physical or mechanical properties of fibers remain unaffected.

SEM microscope images of undyed and dyed silk fabric (top) and dyed cotton fabric (bottom), and a color picture of each dyed fabric.
Conclusion
It is concluded that the colorants extracted from pumpkin have excellent thermal stability under normal dyeing conditions, as well as good stability in acid and neutral environments. HPLC and UV–vis spectra verified several outstanding structural features of the extracts. To obtain the maximum rate of extraction, the optimum extraction conditions for the pumpkin colorants involved a temperature of 70°C for 60 min with a material:liquor ratio of 1:10. Based on an overall consideration of dye uptake and K/S value performance, we have identified the optimum dyeing conditions for silk fabric to involve the meta-mordanting dyeing, a pH value of 5.5, and dyeing at 95°C for 90 min with a 1:60 liquor ratio. Under these conditions, dyed silk fabrics showed the highest dye uptake rate with K/S values, and the colorfastness of the fabric to washing, crocking, and light meeting acceptable standards. Similarly, the optimum dyeing conditions of cotton fabrics show 95°C, 90 min, and a 1:60 liquor ratio. Most of the colorfastness grades tested are acceptable for dyed cotton fabrics, but wet crocking and laundry (color change) are two under-performing properties. A combined secondary force between dye molecules and fiber polymers with some surface coating results in the color and colorfastness of pumpkin dyeing. Affinity and fastness properties of natural pumpkin dyes on textiles might be improved by structural modification through introducing new functional groups or the use of mordants during the dyeing process.
Supplemental Material
sj-docx-1-aat-10.1177_24723444221147980 – Supplemental material for Extraction and Application of Pumpkin Peel Colorants for Natural Textile Dyeing
Supplemental material, sj-docx-1-aat-10.1177_24723444221147980 for Extraction and Application of Pumpkin Peel Colorants for Natural Textile Dyeing by Jiangning Che, Xinghua Teng and Junling Ji in AATCC Journal of Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Partial funding for this project has been provided by the “Science and Technology Innovation Grants of Changzhou (2021)” (CZ20210018).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.