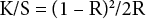Abstract
Natural dyes extracted from natural sources are likely to be less harmful and more eco-friendly than synthetic dyes. In this study, cotton fabric was dyed with natural dyes extracted from Java plum fruit (Syzygium cumini). Dyeing was accomplished with or without mordanting agents using simultaneous and post-mordanting methods. The color of the treated substrates was investigated in terms K/S and CIELAB values. In addition, the attenuation co-efficient and solubility in organic solvents of the extracted dyes were determined. The wash and rubbing fastness of the dyed fabrics were good to excellent. Deep shades were obtained by post mordanting method with ferrous sulfate, whereas alum treated fabrics produced brighter shades with excellent fastness. Java plum fruit is available in many countries and offers a possible substitute for synthetic dyes.
Introduction
Natural dyes extracted from natural sources are often found to be less harmful and more eco-friendly, as the synthetic dyes cause environmental pollution during their processing and application. 1 Interest in using natural dyes on textiles is increasing day by day. 2 The shades produced by natural dyes/colorants are exceptional and harmonious, and usually soft and gentle to the human eye. 3 The present work aims at studying the extraction and application of a new source of natural dye for cotton fabric.
Syzygium cumini, the scientific name of Malabar plum, Java plum, Black plum, Jambolan or Jamun, is a common fruit tree in India and other countries in the Asian subcontinent. The plant is also found in other tropical and subtropical regions of the world. 4
The S. cumini (Myrtaceae) tree is a traditional medicine plant used for the treatment of various diseases. 5 The tree has a great importance as most of the parts of the tree, like the bark, leaves, seed, and fruits are used as medi-cine. 6 Overall, the plant is rich in anthocyanins, glucoside, ellagic acid, isoquercetin, kaemferol, and myrecetin. 7 The java plum seeds contain the alkaloid, jambosine, and the glycoside jambolin or antimellin, which can lower blood pressure and can impeed the conversion of starch into sugar as it contains ellagic acid, 8 which is a well-known antioxidant. 9 The seeds are rich in flavonoids, contain small amounts of protein and calcium, and have a high total phenolics content with significant antioxidant activity. 10 So java plum seeds are used extensively in medicine, whereas the fruit pulps can be used as a natural colorant for textile dyeing.
Java plums are rich in sugar, mineral salts, vitamins C, anthocyanins, and flavonoids. 11 The fruit pulps are also rich in raffinose, glucose, fructose, citric acid, malic acid, and gallic acid. The sourness and color of the fruit might be due to the presence of gallic acid and anthocyanins respectively. 4 Fig. 1 shows the structure of anthocyanin. The peel powder of java plum can be used as a colorant for foods and pharmaceuticals due to the high antioxidant activity and relatively high stability of the pigments. 12
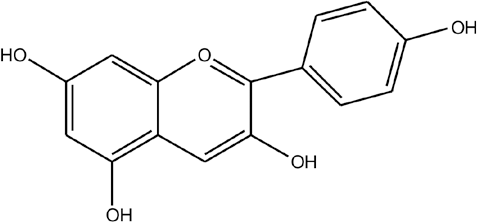
Structure of anthocyanin.
Dyeing behavior of natural dye extracted from the bark of S. cumini has been studied on silk fabric. A range of shade was obtained by using various mordants and mordanting techniques. 13 Extraction of natural dye from black plum fruit using various solvents and its staining property on red hepatic tissue were studied. 14
S. cumini seeds are used as bio-absorbents to remove Acid Red YG dye from textile industry wastewater; a 92.6% dye biosorption was achieved at pH 2.0. 15 The active functional group of jamun pulp dried at various temperatures was studied and found the presence of typical functional groups such as alkyl halides, aromatic rings, phosphine, carboxylic acids, alkenes, phenyl rings, thiocarbonyl, a bioactive fraction, nitro compounds, and amines. 16 Silk and cotton fabrics were dyed at room temperature (RT) using natural black currents and the properties of the dyed fabrics were investigated. 17
Most of natural dyes have less affinity for the fibers and require a mordanting agent, which has an affinity both for the dye and fiber. Some natural dyes are substantive and capable of being fixed directly, but the mordant helps to produce faster shades by forming an insoluble compound. 3 Common mordants used in natural dyeing are the metal salts of aluminum, chromium, iron, copper, and tin. 18 Alum and ferrous sulfate are the most widely-used mordants as they are relatively safe and have less or zero impact on the environment.19,20 Depending on the final color formed with the natural dyes, the alum and iron salts are known as brightening and dulling mordants respectively. 18 In the present study, alum (KAl2(SO4)2·12H2O) and iron (II) sulfate (FeSO4·7H2O) were used as mordanting agents as they are safer to the environment.
Cotton fabrics have attractive wearing qualities, having relatively hydrophilic, water absorbent fibers. 21 In this study, steps were taken to find a replacement for synthetic dyes on cotton. The cotton knitted fabric was dyed with natural dyes extracted from Java Plum with or without mordanting agents. Simultaneous and post-mordanting methods were applied for the application of mordanting agents. The dyeing behavior and fastness properties of Java plum extract on cotton were investigated. The roles of alum and iron sulfate as mordanting agents on dyeing were also studied.
Experimental
Fabric
Factory bleached 100% cotton single jersey plain knit fabric (170 g/mβ, Mymun Textile Ltd.) was used in this study.
Extraction of Java Plum
Java plum pulp (300 g/L) separated from seeds was used to prepare extracts for dyeings. The Java plum pulp was boiled in 1 L of distilled water for 1 h and filtered. After extraction, the lost volume was brought up 1 L with distilled water and the extract solution was cooled down to RT. This solution served as the stock solution for dyeing.
Mordanting
The simultaneous and post-mordanting methods were used in the dyeing process. Alum (KAl2(SO4)2·12H2O) and iron (II) sulfate (FeSO4·7H2O) were used as mordanting agents at a concentration of 1.6 g/L. Post-mordanting was carried out for 30 min at 60 °C and a liquor ratio (LR) of 1:30. Simultaneous mordanting was carried out along with the dyeing process. Pre-mordanting was also attempted, but did not give favorable results compared to the other two mordanting methods.
Dyeing
Dyeing was accomplished with simultaneous and post-mordanting methods in an Ahiba IR laboratory sample dyeing machine at a LR of 1:50 for 60 min at 100 °C. For simultaneous mordanting, dyeing was carried out along with mordanting, and for post-mordanting, dyeing and mordanting were done separately. Salt (NaCl, 10 g/L) was used for better exhaustion. After dyeing, the dyed samples were washed properly and soaping was carried out with 2 g/L soap solution at 60 °C for 10 min and dried at RT. Fig. 2 presents the time-temperature profile of the simultaneous dyeing and mordanting processes.
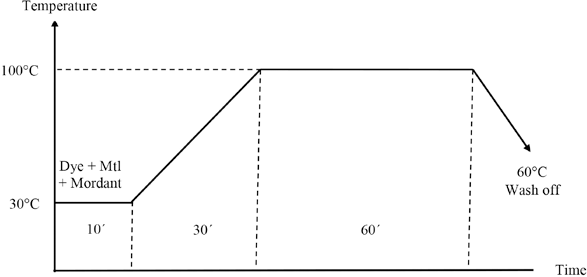
Time-temperature profile of simultaneous dyeing and mordanting process.
Testing
Dye Solubility
To test the solubility of aqueous extract of Java plum in organic solvents, 100 mL of the Java plum extracted liquor was placed in an evaporating dish and heated at 70 °C for 32 h. If heated over 70 °C, the extract tend to form char. The semi-solid thus obtained at 70 °C was used for the solubility test.
For this assessment, 10 mL of organic solvent and 0.5 g of Java plum (semi solid) were mixed in a test tube, shaken for 2 min, and allowed to stand for 2 min for observation. The solution was also heated to see any change in results. The solubility test was done using four different organic solvents: toluene, benzene, rectified spirit, and dimethyl formamide (DMF).
Absorbance of Dyed Solution
The semisolid extract of Java plum was dissolved in water at ten different concentrations to measure the λmax absorbance, which was 540 nm as recorded from the scan of a dye solution across a 400-700 nm wavelength range. A DataColor 650 dual-beam spectrophotometer was used.
K/S Values of Dyed Cotton Fabric
Color strength values (K/S) of the dyed samples were measured using the DataColor 650 dual-beam spectro-photometer pulsed xenon filtered to approximate D65 illumination and 8° viewing measurement geometry at the individual wavelengths of maximum absorption. The Kubelka-Munk equation (Eq. 1) was used to measure K/S values with the help of relevant software.
K is the coefficient of absorption, S is the coefficient of scattering, and R is the reflectance of the dyed fabric.
Determination of Color Fastness
Wash fastness was determined according to ISO 105 C06 (C2S): Color fastness to domestic and commercial laundering, 22 and ISO 105 C03: Color fastness to washing. 23
Color fastness to rubbing of dyed cotton fabrics was determined according to ISO 105 X12:1993. 24
Results and Discussion
The Java plum extract was found to be insoluble in toluene and benzene, but slightly and fairly soluble in rectified spirit and DMF, respectively. This clearly indicated the polar nature of the extract as it was only soluble in polar solvents.
The UV-Vis absorbance/concentration curve is shown in Fig. 3 and Table I. The straight line of best ft was drawn by use of the least square method and the attenuation coefficient of the dye at λ = 540 nm was 722 × 10−4 L/g·cm. From the data, the amount of dye present in the extract was very low.
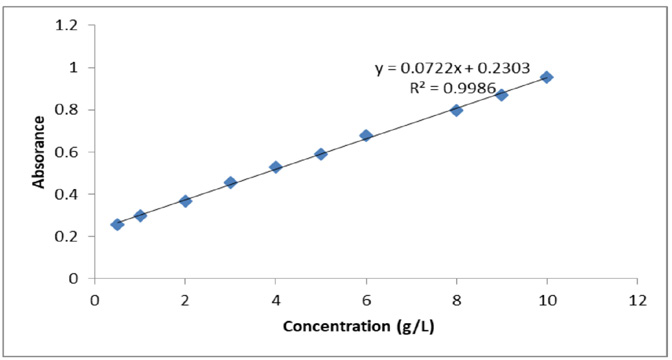
Absorbance vs concentration curve.
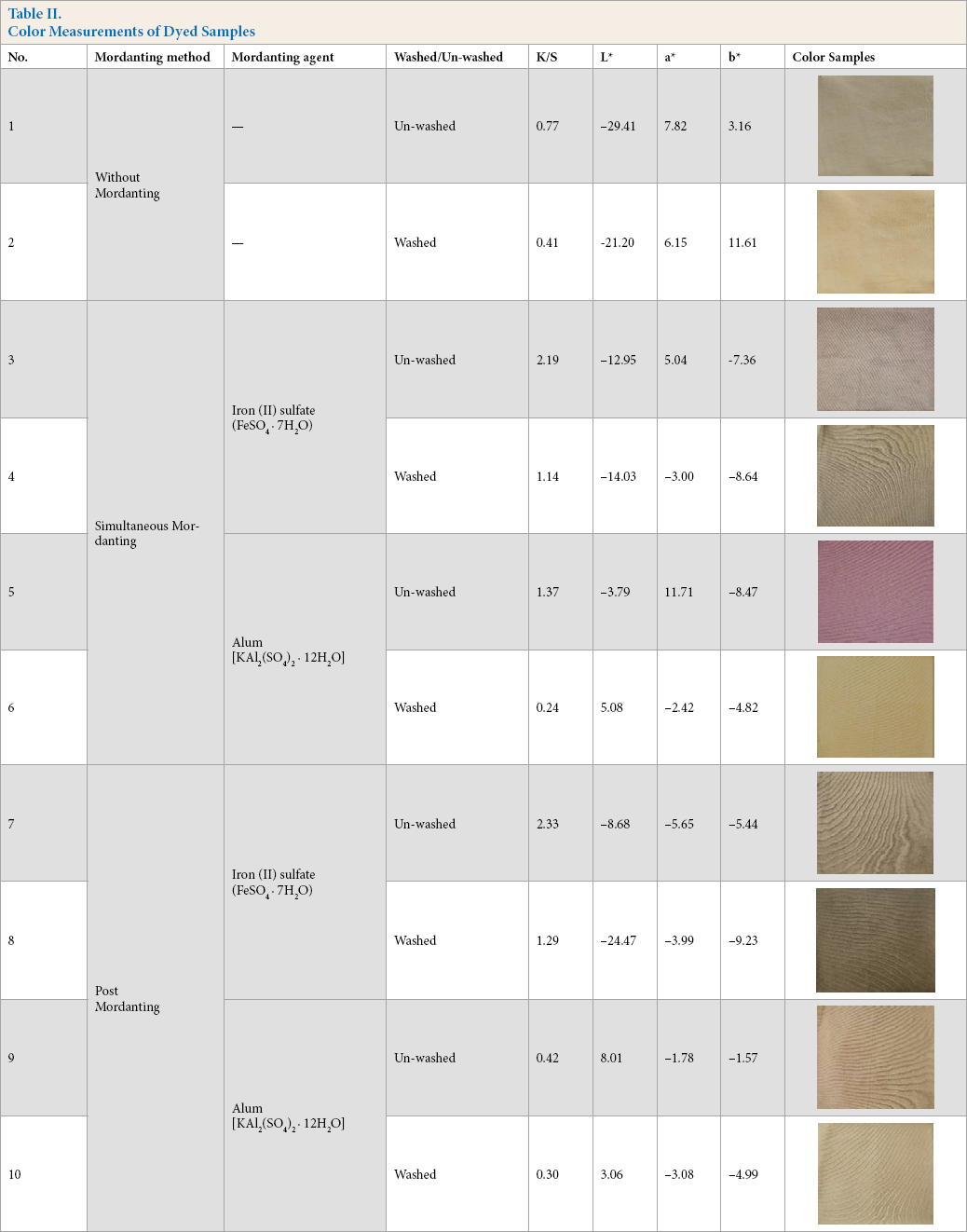
Table II and Fig. 4 present the color strength and shades of the dyed samples. After dyeing with Java plum extract, the produced shades were mainly olive and greenish with alum and iron mordanting agent respectively. There were significant shade changes found among the dyed samples after washing. Simultaneous mordanting with alum only generated the magenta-pink color, which was very similar to the Java plum color, but did not remain after washing.
Color Measurements of Dyed Samples
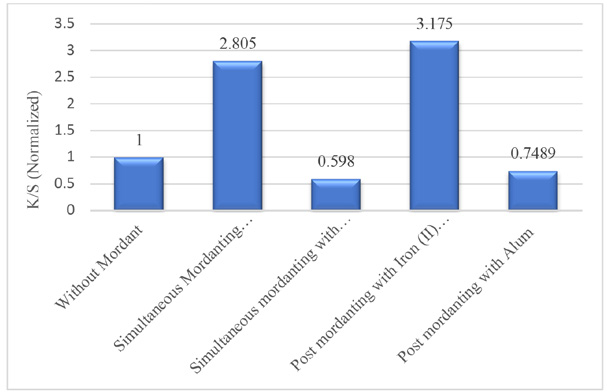
Normalized K/S value of dyed samples (washed).
The samples treated without mordanting agent showed typical shades with more reddish and yellowish tints. Samples treated with mordanting agents produced different types of hues, perhaps due to the formation of metallic salts. Simultaneous and post-mordanting samples followed similar hue trends. Iron-treated samples produced deep shades. Heating the ferrous form may have oxidized it to the ferric form, which is brown to black, whereas the alum treated samples showed brighter shades.
For color strength (K/S values), iron-treated samples provided the highest color yield in both simultaneous and post-mordanting compared to the other samples. Non-mordanted samples produced distinctive shades with low color strength compared to the iron-treated samples, but gave better results than the alum-treated samples.
It was observed that the depth of shades and K/S values of the dyed samples were higher for the post-mordanting samples using either mordanting agent. This may be due to the better complex forming ability of the metals with the dyes in this method.
Wash fastness of all the dyed samples were tested by two methods (i.e., color fastness to washing and color fastness to domestic and commercial laundering). The non-mordanted samples showed good wash fastness (3) ratings, which were slightly improved with alum mordanting. The simultaneous and post-mordanted samples with alum showed improved fastness (4 and 4-5 respectively). No improvement was observed for the ferrous sulfate treated samples.
Figs. 5 and 6 present the grey scale ratings for color staining on multi-fiber fabric. In all cases, not much staining was observed, but mainly color stained on bleached cotton, polyamide, and wool. The staining behavior indicated the ionic characteristics of the dyes extracted from Java plum as it was stained on ionic fibers only.
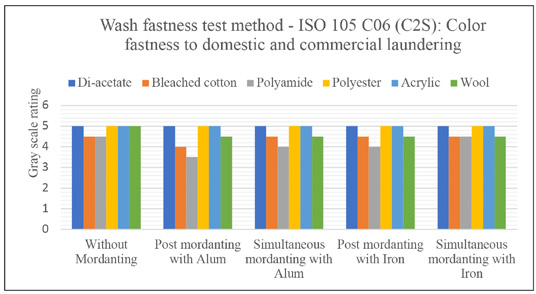
Color staining rating of dyed samples to domestic and commercial laundering.
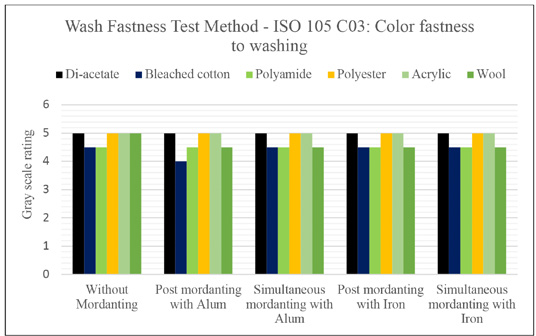
Color staining rating of dyed samples to washing.
Fig. 7 shows the results obtained from the rubbing fastness test. All samples, with or without mordanting agents, showed good rubbing fastness properties. Mordants affect the wet rubbing fastness slightly, but not significantly.
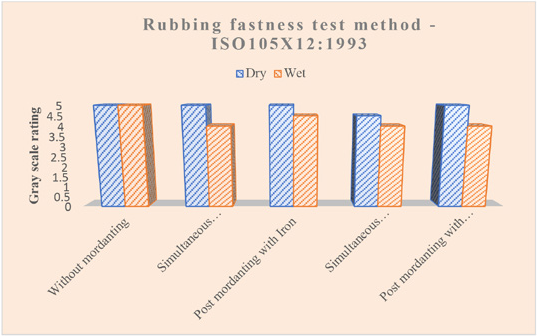
Rubbing fastness ratings of dyed samples.
The solubility in polar solvents and the staining characteristics of dyes extracted from Java plum indicated its polar nature. As it was attracted to bleached cotton, polyamide, and wool to some extent indicated its anionic nature.
Conclusion
In this study, dyeing of cotton with S. cumini pulp extracted from Java plum was found to be effectively accomplished at neutral and slightly acidic pH. Dyeing without or with different mordants resulted in differences in color hues that clearly indicate the influences of mordanting agents. Samples without mordanting agents produced typical shades with low K/S values. The alum-treated samples showed better fastness properties with brighter shades. Post-mordanted samples with ferrous sulfate showed the highest color yield with good fastness. The solubility of dyes in ionic solvents and color staining on ionic fibers indicated the ionic characteristics of the dyes. Since S. cumini fruit is available in many other countries, the colorant may be considered as a possible replacement for synthetic dyes. This dye can be used on commercial basis, focusing on environmental issues and user friendliness. Java plum is a niche substance that has unique uses. The useful properties of the natural dyes reported in this study will encourage others to find the novel advantages of many other plants.