Abstract
Sustainability has become a global requirement in all industries. In the textile sector sustainability can be achieved by developing innovative techniques, approaches, and machinery. The current study introduces a sustainable approach for linen dyeing with natural lac dye using microwave heating after the treatment with chitosan as a cationic bio-mordant. The chitosan treatment was carried out by a pad/dry-cure process with citric or acetic acid. Linen samples were characterized by scanning electron microscopy (SEM), and fourier transform infrared spectroscopy (FTIR). The results confirmed the deposition of chitosan on linen fabric with both citric and acetic acid. X-ray diffraction (XRD) was used to determine the change in the crystallinity of linen fabric after chitosan treatment and lac dyeing. The color strength results confirmed the successful application of chitosan as a bio-mordant and the efficiency of microwave heating in shortage the dyeing period in lac dyeing, where the K/S increased from 1.54 to reach 18.86 and 13 with acetic and citric acid, respectively in 7 min. Furthermore, the maximum color strength was achieved at pH 3 of lac dye and the presence of chitosan eliminated the usage of salt in the lac dyeing of linen fabrics. Fastness properties of the lac dyed linen fabrics to wash, light, and perspiration have been discussed. The functional properties (antibacterial activity and UV protection) of linen fabrics were assessed. The antibacterial activity was decreased after the chitosan-treated linen samples were dyed with lac dye. However, the UPF of linen samples was enhanced after lac dyeing.
Introduction
Nowadays, the environment issues and pollution control have a huge interest. The numerous loads of synthetic dyes waste in the aquatic system are considered one of the major environmental and human health problems. This serious problem led to growing interest in sustainable products. So, natural dyes are gaining more importance as they are eco-friendly, non-toxic, and obtained from a renewable resource. Furthermore, consumers are becoming more and more concerned about environmental issues and hence are demanding natural products incorporating natural ingredient.1,2
According to the origin of natural dyes, there are three major categories: vegetable, mineral, and animal origins. The anthraquinone structure lac, cochineal, and kermes dyes are from the animal source as they are obtained from insects. These insect red dyes can dye textiles in purple and red colors with good colorfastness to light in comparison with most vegetable dyes.3,4 Lac dyes (Laccaic acid) are obtained from the insect Coccus Lacca or Laccifer Lacca as a byproduct of the shellac industry. They are weakly acidic natural dyes and consist of polyphenolic anthraquinone compounds.3,5
Although the utilizing of metallic salts in natural dye has some benefits in producing different shades and hues of the same dye and improving the inferior properties of wash and lightfastness, it causes a lot of environmental problems.6,7 The replacement of metal salts with natural mordants becomes an essential matter in natural dyeing. Bio-mordants have been used for enhancing color strength and fastness properties of natural dyes in several studies. 8 Also, the replacement of metallic mordants with cationization can be considered a novel method for upgrading the properties of natural dyeing and minimizing the pollution.
Cationization is a novel technique for cellulosic fabrics which has been used to improve the color strength and fastness properties of dyed fabrics. Chitosan is considered one of the cationic agents.9 –12 Janhom et al. 13 utilized polyethyleneimine (PEI) as a cationic agent for cotton dyeing with lac dye. And found that PEI improved dye adsorption of cotton and the dye desorption from fibers was decreased. Chitosan is considered an inexpensive, non-toxic, and biodegradable compound; consequently, it can be used in numerous areas of application as wastewater treatment, food, textile industry, and recently in drug industry as a hydrating agent in cosmetics.14,15
The application of chitosan to textile materials can provide multifunctional properties. According to its chemical structure, chitosan not only affords antimicrobial properties but also enhances the color strength of the dyed materials. Also, chitosan is used as an auxiliary in the printing of textiles as they increase the color strength of fabrics with synthetic dyes.11,16
Chitosan is an amino polysaccharide [(1-4)-2-amino-2-deoxy-D-glucose] which is produced from chitin by the deacetylation process. Chitin is found in the shell of crabs and shrimps, is an insoluble copolymer of N-acetyl glucosamine and glucosamine units, and generally contains more than 90% glucosamine units. Chitosan has a chemical structure as cellulose with the same β-glucosidic linkages, but the main difference is the presence of primary amino groups at most of the C-2 position in chitosan. These amino groups are responsible for the antimicrobial properties of chitosan. 14 Several studies have been used chitosan for natural dyeing to obtain ecofriendly dyeing process as well as enhancing the color strength and functional properties. Saini et al. succussed in utilizing a layer-by-layer technique for chitosan and green tea extraction to dye and produce a multifunctional linen fabric. 17
In the textile industry, wet processes such as washing, dyeing, rinsing, desiccating, and bleaching expend considerable water and energy. The cost of energy is crucial in the development of market competition. Microwave (MW) and UV light radiation studies have attempted to reduce processing time and thus the energy consumed. Radiation could be employed in heating, drying, condensing, dyeing, pressing, finishing, changing surface textiles, and improving the product’s quality. Radiation methods are gaining massive interest because, they are naturally safe, clean, and eco-friendly. 18
MW affords fast, uniform, and effective heating by enabling the heating of all particles at the same time with its easy penetration property into the particles of the matter.19 –21 Microwave irradiation is a powerful technique of non-contact heating by causing vibration and rotation of permanent dipoles in the microwave field. In the previous studies, the microwave was used in desizing, scouring, or bleaching cotton fabrics to clean the cotton surface to prepare it for further dyeing processes. Compared to these interesting earlier studies, recently, the microwave is used directly during the application of the functional agents to the textile. 22 Furthermore, microwave heating has been introduced to obtain better results compared to conventional heating for the preparation of silver nano particles (AgNPs). 23
Microwave radiation was used also in various ways, as an alternative to traditional curing for improving the properties of crosslinked textile materials and for sustainable extraction of natural dyes. MW irradiation was used in recent work to separate natural color from tea leaves under alkali conditions. The ability of MW energy to separate colorant from tea leaves for dyeing cotton fabric at lower optimal dyeing conditions has been demonstrated. 2 In another recent study, Adeel et al. used MW heating as a sustainable heating tool for irradiating both cinnamon bark dye and silk fabric. It is found that this sustainable heating tool can successfully be employed for the exploration of therapeutic plants as a source of natural dye for natural fabric dyeing. 8 Adeel et al. used microwave in isolation of lac dye, the extract was prepared in acidic medium and stimulated through MW treatment up to 7 min and used to dye wool fabrics. 24 Another study used microwave energy to extract natural dyes from lac insects and apply them to nylon fabric. 25 Also, another study used microwave radiation as an extraction mode for Esfand (P. harmala) for polyamide (nylon) dyeing. 26
Natural dyes are not only eco-friendly but also more compatible with the environment, which is related to their excellent biological properties. So, the functionalities of natural dyes such as deodorant, cool finishes, anti-bacterial and anti-UV properties are highlighted as the value addition in recent days. 27
UV protection finishes, or UV shielding agents, are one of the most common chemical finishing processes used on textile materials to protect people and textile materials from the damaging effects of UV radiation. UV radiation, which has a much higher intensity than visible light, has the potential to cause a variety of chemical reactions that can be harmful to human health and damage textile fibers. Although moderate sun exposure offers health benefits, excessive UV exposure can cause serious harm because UVA (320–400 nm) and UVB (280–320 nm) radiation cause distinct cellular reactions that manifest as pigmentation, sunburn, skin aging, skin cancer, and DNA damage. 28
Many studies indicate the ultraviolet protective property of natural dyes toward textiles to the absorption characteristics of the functional groups such as tannin-containing polyphenols, flavonoids, anthraquinone, terpenoids, aromatic conjugated system, etc., present in the plants.24,27,29 Lac dye is one of the natural dyes that protects against UV rays. Mongkholrattanasit et al. dye silk fabrics by the post-mordanting process by the extracted natural dye from Laccifer lacca Kerr. And, it has been observed that very good to excellent UV protection is offered by metal mordanted silk fabrics dyed with lac dye but without mordant offering good UV protection with sufficient washing fastness.27,30
Although the advantages of M.W. rays of the extraction with high yield for natural dyes and time reduction, it has been observed that there is little information available on utilizing microwave in linen dyeing with lac dye. So, the novelty of this work focused on applying an eco-friendly and sustainable approach to linen dyeing and finishing with natural lac dye by microwave as a green heating tool and chitosan as a cationic bio-mordant. Moreover, the present work studied the effect of microwave irradiation and chitosan finishing on the dyeing properties and functional properties (anti-bacterial and anti-UV) of linen fabric. Another purpose is to assess the changes in the surface morphology in addition to the crystallinity of linen fabrics after chitosan bio-mordanting and microwave lac dyeing.
Experimental
Materials
Fabrics
100% linen fabric with plain weave (205 g/m2) was purchased from the local textile industry.
Chemicals
Chitosan powder (medium molecular weight-deacetylated chitin, poly(D-glucosamine) 75:85% deacetylated from Sigma-Aldrich Chemie GmbH), sodium sulfate (anhydrous powder), sodium hypophosphite (anhydrous powder), citric acid (monohydrate powder), and acetic acid (96% solution) were of laboratory grade chemicals from Sigma-Aldrich Chemie GmbH and non-ionic Detergent from dystar company.
Dye
The powder of Lac natural dye was purchased from ALPS Industries Ltd– Sahibabad– India to utilize in this study. The specification of Lac dye has been listed in Table 1.
Lac dye specification.

Methods
The chitosan treatment for linen fabric
Linen fabric was treated with chitosan by the pad-cure method on a laboratory padder (ROACHES, UK). The treatment bath was consisted of either [1% (o.w.b.) chitosan, 6% (o.w.b.) citric acid and 5% (o.w.b.) sodium hypophosphite] or [1% (o.w.b.) chitosan and 2% (o.w.b.) acetic acid]. Linen samples were padded through two dips and two nips to 100% wet pick up Then, the fabric was (dry-cure) one step in the oven (ROACHES, UK) at 120° C for 5 min. After that, the samples were washed with 2 g/L non-ionic detergent at 40°C for 8 min, rinsed, and air-dried.
Dyeing
Linen samples were immersed in a dye bath composed of 13% (o.w.f) Lac dye, sodium sulfate (0%, 2%, 4%, 6%, 8%, 10% (o.w.f.)), and the M: LR was (1:50). The dyeing process was carried out in a microwave oven (Samsung, model GE614ST May 2017, with 900 w out power operating at frequency of 2450 MHz) through different intensities (900, 600, 450, 300, 100 W) for different times (3, 5, 7, 9, 11 min) and the pH was adjusted to (2, 3, 4, 5, 6). After that, the dyed samples were washed using 2 g/l non-ionic detergent at 60°C for 10 min, rinsed, and air-dried.
Testing and analysis
Characterization of linen fabric
In this study various characterization techniques were used to evaluate linen fabric after chitosan finishing and lac dyeing. The surface morphology of linen fabric was estimated by using A JEOL-Model JSM T20 scanning electron microscopy (SEM) operating at 19 kV. 31 The functional groups present in linen fabric were studied by fourier-transform infrared spectroscopy (FTIR) using a VERTEX 70 v FT-IR spectrometer (BRUKER, Billerica, MA, USA) with a resolution of 4 cm−1 and a spectral range of 4000–400 cm−1. 32 X-ray diffractometer (Shimadzu XRD-6000) was used to determine the crystallinity of the linen samples. The XRD patterns are obtained in the range of 2θ from 4° to 90° at room temperature. Cu Ka is used as a radiation source of wavelength λ = 0.15408 nm, the scan rate 8°/min, and the operation voltage and current were 50 kV and 40 mA, respectively.
The resultant graphs were printed out by ORIGIN PRO 2018 graph plotting software and the degree of crystallinity was then calculated by the following equation (1). 33

as the ratio of the area of the crystalline peaks to the whole area,
The crystallite size was obtained from XRD peaks according to Scherrer’s equation (2), as shown below:

Where: λ is the wavelength of the radiation (0.1548), θ is the Bragg angle of the diffraction peak, β (FWHM) is half the width of the peak (002) (in radians), and K is a constant that is usually considered to be 0.89. 34
Tensile strength
The tensile strength of linen fabrics was evaluated before and after treatment with chitosan according to ASTM: Breaking Strength/Elongation Strip Method 5035.2016 Standard test method.
UV/Vis absorption spectra of lac dye
The Uv/Vis absorption spectra of lac dye were recorded on a shimadzu UV2401 spectrophotometer at the wavelength of maximum absorption (λmax). 35
Color strength (K/S)
Color strength (K/S) of the dyed samples was measured on Mini Scan XE spectrophotometer using Hunter lab universal software. The color strength is calculated based on Kubelka – Munk equation (3), which stated below.

Where: K, S, and R are the absorption coefficient, scattering coefficient, and reflectance, respectively.
Dye fixation
The fixation of the lac dye in percentage was calculated by the following equation (4). 36

Color fastness
The light, wash, and perspiration fastness properties of the dyed linen samples were evaluated according to AATCC: Colorfastness to Light ‒ 16-2004.2005 test method, AATCC: Colorfastness to Laundering, Home and Commercial Accelerated 61-2003.2005, and AATCC: Colorfastness to Perspiration 15-2002.2005 test method, respectively.
Antimicrobial activity (turbidity method)
The antimicrobial activity of linen samples was determined by turbidity method according to Bhat et al. 37 and Balouiri et al. 38 against two Gram-positive bacteria (Staphylococcus aureus NRRL B-767 and Salmonella typhimurium ATCC 25566), and a Gram-negative bacterium (Escherichia coli ATCC 25955).37,38 Briefly, 30 µl of the tested microorganism (106 colony-forming units (CFU)/ml) were inoculated into 3 ml of nutrient broth in standard test tubes containing linen samples (2 ×2 cm2). Test tubes were incubated at 35 ± 2°C for 24 h under shaking conditions. Antimicrobial activity was evaluated by measuring the optical density at 620 nm and comparing it to the blank sample.
The bacterial reduction percentage was calculated according to equation (5):
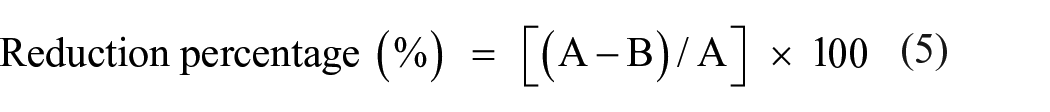
Where A and B are the optical density of the bacterial culture at 620 nm in the presence of blank and treated linen samples, respectively. The data was the mean of three replica.
Ultraviolet protection factor (UPF) evaluation
The ultraviolet protection factor (UPF) of linen fabric was calculated according to the Australian/ New Zealand Standard test method (AS/NZS-4399-1996) using UV-Shimadzu 3101-PC-Spectrophotometer. The UPF was calculated according to the following equation (6) which based on the percent ultraviolet radiation transmittance (UVR) through the specimen. Depending on the UPF value, the protection categories are excellent with the UPF values are 40–50, <50 and very good with the UPF values in the range of 25–39, and good with the UPF values in the range of 15–24. 15

Where: Sλ is the solar spectral irradiance (W/cm2/nm), that is, a function of the amount of solar energy that reaches the surface of the earth of each wavelength; Eλ is the relative erythemal spectral effectiveness, that is, a weighting spectrum of the action of UVR on the skin for each wavelength; Τλ is the spectral transmittance of the specimen (measured); and Δλ is the measured wavelength interval or bandwidth (nm).
Results and discussion
Chitosan treatment and microwave lac dyeing
Linen samples were treated with 1% chitosan by pad /dry-cure method to afford cationic charge and allow the fabric to dye with lac dye without metallic mordants. The choice of chitosan concentration for linen treatment in this work is related to the effect of chitosan on the hand properties as it makes the fabric stiffer. 39
Table 2 shows the effect of chitosan treatment with both acids (citric and acetic) on the color strength (K/S) of linen fabrics dyed with lac dye in a microwave oven for 5 min at 450 W. From the results, it was observed that the treatment of linen samples with chitosan in the case of both acids affected the color strength of lac dye. The color strength of the blank sample was (1.54) and increased to (2.6, 3.64) after treatment via chitosan with citric and acetic acids, respectively. The role of chitosan as a bio-mordant may be related to the presence of amino groups which get protonated and offer cationic sites for attachment with the anionic lac dye.1,39
The effect of chitosan on the color strength of lac dye.

Moreover, the color strength of chitosan/acetic treated linen samples was higher than that of chitosan/citric treated. This result may be interpreted as, there is not any chemical bond between chitosan and linen fabric in the case of acetic acid, so the most of amino groups in chitosan are free and can be attached with lac dye. But in the case of citric acid which acts as a crosslinking agent for linen and chitosan, the free carboxylate group of citric acid will react with amino groups of chitosan to form a salt linkage between citric acid and chitosan. Therefore, a fewer number of free amino groups exist. 40
Characterization of the finished and dyed linen fabric
Surface morphology of linen fabric
Figure 1 shows the SEM photographs of linen fabrics before and after treatment with 1% chitosan and citric/acetic acids (a: blank linen, b: the chitosan/citric treated linen, and c: the chitosan/acetic treaded linen). From these photos, it was observed the deposition of chitosan on the treated fabrics with both citric and acetic acids (b and c) in comparison to blank fabric (a). These photos also affirm the results in Table 2 of increasing the K/S of lac dye after chitosan treatment. Chitosan makes a surface film on linen fabrics that has a cationic character subsequently, attracts the anionic lac dye and increases the color strength of linen fabrics.

SEM photographs of linen fabrics: (a) blank linen, (b) chitosan/citric acid treated linen, and (c) chitosan/acetic acid treated linen.
FTIR of linen fabric
FTIR analysis showed the structural bond changes in the linen fiber after the treatments with chitosan in case of citric and acetic acids.
Figure 2 shows the FTIR of raw, treated, and lac dyed linen fabrics. As shown in figure a broad peak for raw linen fabric was observed between 3000–3200 cm−1 which related to O-H stretching vibration of linen fabrics. The asymmetric C-H stretching is observed in range of 2900 cm−1 and the peaks at 1433 and 1340 cm−1 are related to C-H bending vibration. A strong peak is appeared at range 1000–1100 cm−1 is concerned to C-O-H bending of alcohol and a peak at 914 is related to C-O-C stretching of cellulose ring.
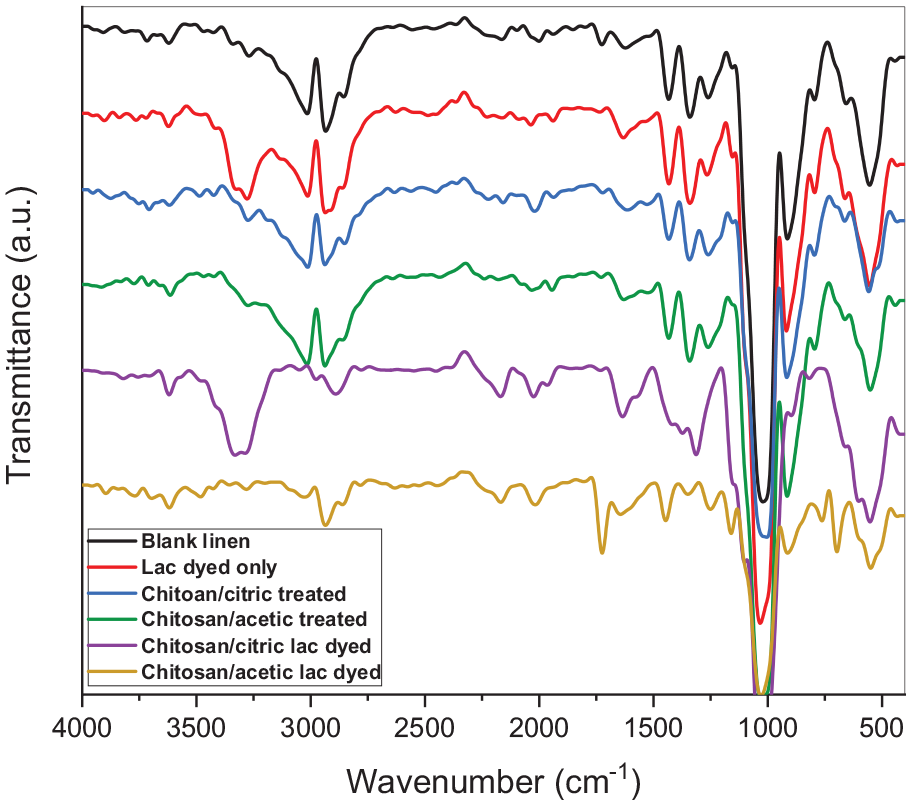
FTIR spectra of linen fabrics.
After chitosan treatment the appearance of peaks at 3600–3700 cm−1 is attributed to the stretching vibration of -OH overlapped with NH stretching due to the hydrogen bond for chitosan. 41 Another peak was observed at 2020 cm−1 due to stretching within the C-H group. 42 But after lac dyeing the band of N-H appeared at 1600–1700 cm−1 range which is attributed to the bonds between chitosan and linen fabrics.43 –45
These IR spectra confirm the presence of chitosan and the characteristic change in linen fabric after chitosan treatment and lac dye.
XRD and crystallinity behavior of linen fabric
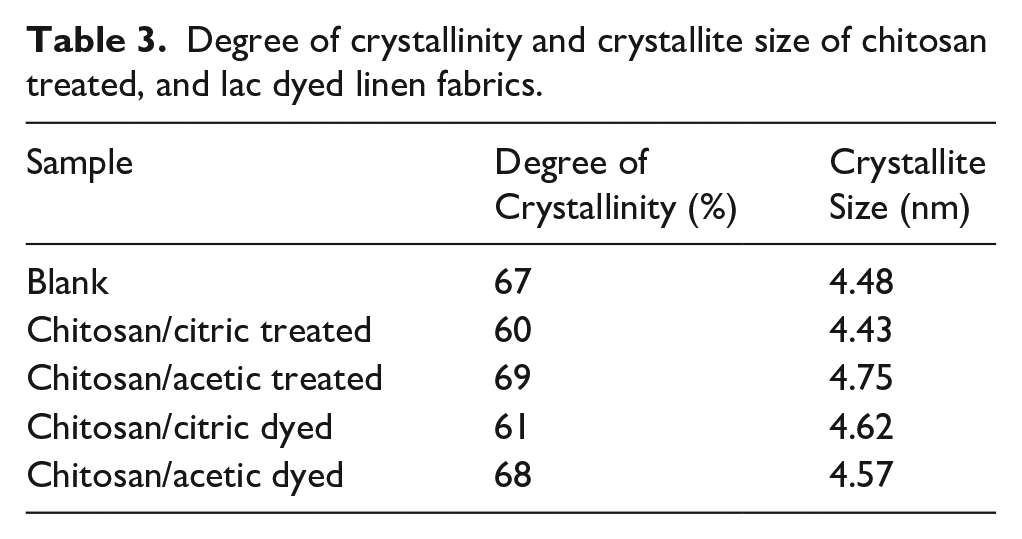
The XRD was utilized to measure the crystallinity of linen samples, Figure 3 shows the spectra of raw, treated, and dyed linen samples. The strongest peaks at 2Θ = 14.1°, 15.56°, and 21.9° are related to the crystallinity of cellulose. The crystallinity degree and crystallite size of blank linen fabric is 67% and 4.48 nm in Table 3. The results show a slightly increase in the peak intensity and in the crystallinity degree of the chitosan/acetic treated linen samples which is related to the occupancy of pores available in the amorphous region and hydrogen bonding between linen and chitosan. 17 But in case of citric acid both the peak intensity and the crystallinity degree are reduced due to the crosslinking reaction of chitosan with linen under acidic conditions. 46 This result is parallel with the tensile strength results in Table 5. Also, it was observed from the results that there is a little change in the crystallinity of linen fabrics after lac dyeing. In addition to that, the crystallite size has not remarkable change with the different treatments.
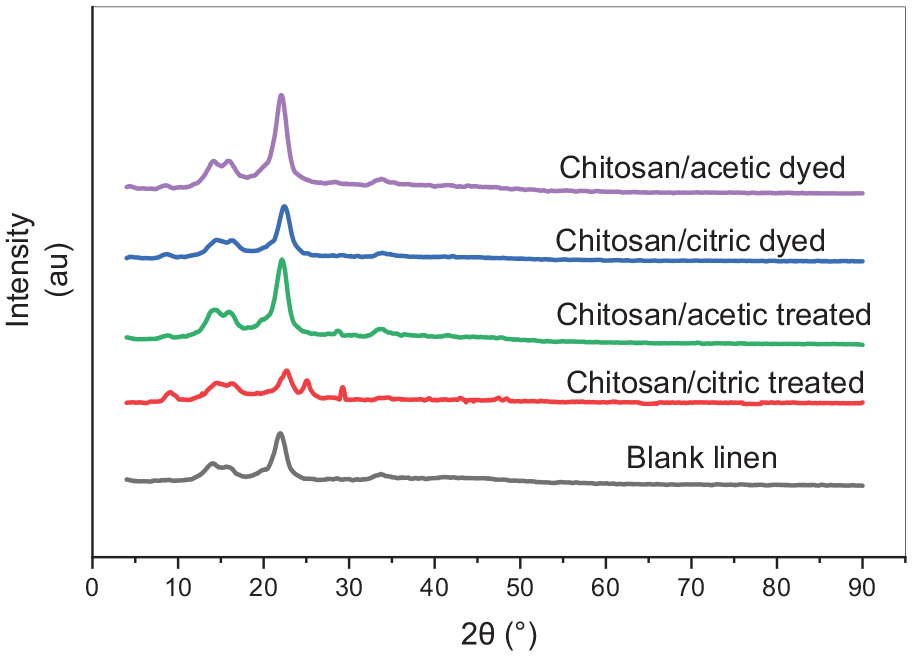
X-ray diffraction pattern of linen fabrics.
Degree of crystallinity and crystallite size of chitosan treated, and lac dyed linen fabrics.
The optimization of microwave lac dyeing
Microwave power and dyeing time
Microwave (MW) radiation is a volume heating compared to conventional heating, which heats the material surface only. When MW fields of electromagnetic radiation are varied, the polarized molecule (such as water) will be affected and reoriented according to the field. Their orientation leads to interfacial friction with other molecules and produces enormous amounts of heat. 47 This heat penetrates the dye material and enables the dye components to transfer from the dye bath and bond with the fabric. Heating levels at a particular point always accelerate the dye bath to an excessive extent consequently, the rate of the radiation & the rate of dyeing reach an equilibrium where the maximum fixation is observed, and excellent color strength is obtained. 8 Microwave gives better color depth in a short period as compared to the conventional.
The chitosan-treated linen fabrics (with citric and acetic acids) were dyed with lac dye in a microwave oven at different power intensities (100–900 W) over different periods (3–11 min). The results in Figure 4 show the K/S of lac-dyed linen fabrics (chitosan/citric acid treatment) at 100 and 300 W increased by extending the dyeing time. But the color strength of linen samples at 450 and 600 W reached the maximum value (6.2 and 3.85) respectively through 7 min and decreased with prolonged periods. While The color strength of linen samples decreased by growing the dyeing duration at 900 W. But, in the case of acetic acid, the K/S of linen fabrics at 100, 300, and 450 W increased by the dyeing time, and reached the maximum value at 600 W (7.4) through 7 min as presented in Figure 5. While the high level of microwave power (900 W) decreases the color strength as in the case of citric acid. This result may be attributed to the inability of microwave irradiation to stimulate the colorant toward the fabric surface in a minimum time while the long heating periods degrade the dye. 48 The low heating level cannot accelerate dye molecules to rush toward the surface-modified fabric, whereas high heating either cause desorption of dye molecule or cause colorant degradation due to which low tint strength is observed. 8
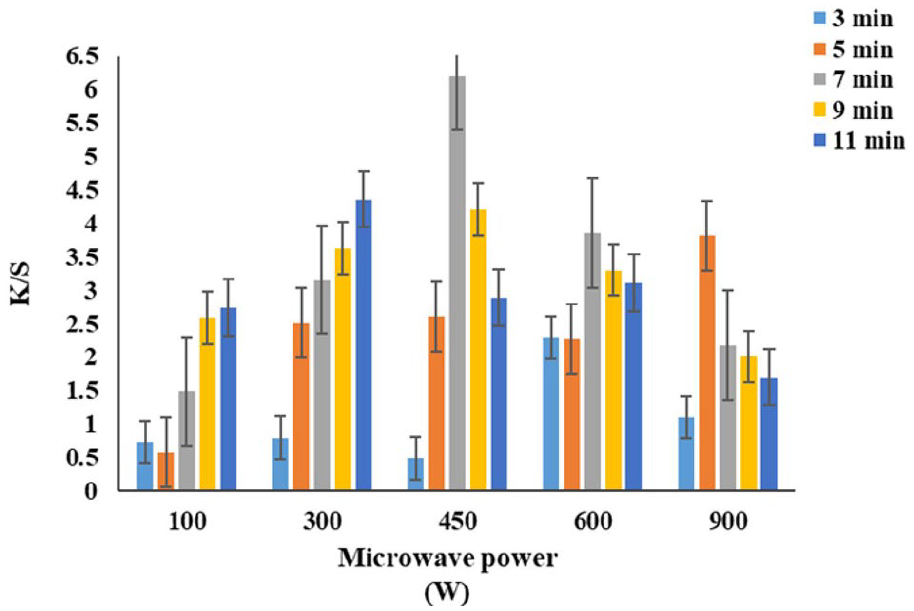
The effect of microwave power and dyeing time on the color strength of lac dyed linen fabric in case of chitosan/citric acid treatment.

The effect of microwave power and dyeing time on the color strength of lac dyed linen fabric in case of chitosan/acetic acid treatment.
Hence, the dyeing for 7 min at 450 and 600 W with citric and acetic acid respectively had chosen for the next experiment.
Salt concentration
The results in Figure 6 demonstrated the increase of sodium sulfate concentration accompanied by reducing the color strength of lac dyed linen samples, and the maximum color strength was obtained with no- salt dyeing (12.4, 18.86) for both (chitosan/citric) and (chitosan/acetic) respectively. This result is related to the presence of chitosan biopolymer, which acts as a polycationic agent on linen samples and improves the affinity of anionic lac dye to cellulosic materials. Chitosan modification affords linen fabric cationic amino groups that reduce the repulsion between linen fabrics and lac dye moreover facilitate binding between them. Consequently, chitosan biopolymer can offer the complete elimination of electrolytes (salts) in lac dyeing. 49

The effect of salt conc. on the color strength of lac dyed linen fabrics.
The effect of pH
The pH of the dye bath is an important influencing factor for dyeing linen fabrics with lac dye. In this study, the effect of pH on the color strength of lac-dyed linen fabrics was studied with the range of (2–6 pH). The results in Figure 7 shows that raising dye bath pH from 3 to 6 is accompanied by decreasing the color strength of the lac dyed linen samples with both cases (chitosan/citric acid and chitosan/acetic acid). The maximum color strength of linen samples was obtained at pH 3, where it reached 18.86 with (chitosan/acetic acid) and 13 with (chitosan/ citric acid). This result can be interpreted in the view of two facts; the first one is that the raising of lac dye adsorption on cellulosic fabrics is accompanied with lower pH of dye bath. 39 The second one is that chitosan behaves, in acid solutions, as a cationic polyelectrolyte due to the protonation of the amino groups. Furthermore, most of the amino groups, (−NH2) of chitosan will be in the protonated cationic form (−NHC3+) in an acidic solution which enhances the ionic attraction between the anionic dyes and cationic groups of the chitosan and fibers. Hence, increasing the protonated amino groups of chitosan reflects on the amount of dye exhaustion on the chitosan treated fabrics.50 –52 A similar conclusion was reached by Ibrahim et al. who also found that the appropriate pH for the chitosan treatment of linen fabric is pH 3. 11 Figure 8 shows the linen dyed samples with lac dye after the optimization of the dyeing conditions.

The effect of pH on the color strength of lac dyed linen fabrics.

Lac dyed linen samples.
The dye fixation and the fastness properties of lac dyed linen fabric
The fastness properties of lac dyed linen samples to wash, light, and perspiration were estimated for both (chitosan/citric) and (chitosan/acetic) treated samples. From the results in a Table 4, it is clear that the substantial role of chitosan modification on the wet fastness properties of lac dyed linen fabric where the wash fastness rating of lac dyed samples is very good (4–5) and those of color change for (chitosan + citric) are good (4). 50 The results of dye fixation may interpret these findings where the dye fixation of chitosan-treated linen fabrics reached 97% and 90% for chitosan/citric and chitosan/ acetic, respectively. Also, these results are matched with the findings of Rastogi et al. who used (Discofix DBA) as a cationic agent for cotton fabric to dye with lac dye. And it was found that the cationized cotton which was dyed with lac dye exhibited a good color yield and wet fastness properties even without mordanting. 53 But the lightfastness rate of lac dyed linen is good (4) and moderate to good (3–4) for chitosan/citric and chitosan/acetic treated samples, respectively. Furthermore, the rating of perspiration fastness is acceptable where it ranged from good to very good (3–4) and very good to excellent (4–5). The lower rating (3) and (3–4) may be due to the sensitivity of the chitosan dyed samples to pH degree. 16
The dye fixation and fastness properties of linen fabric dyed with lac dye.

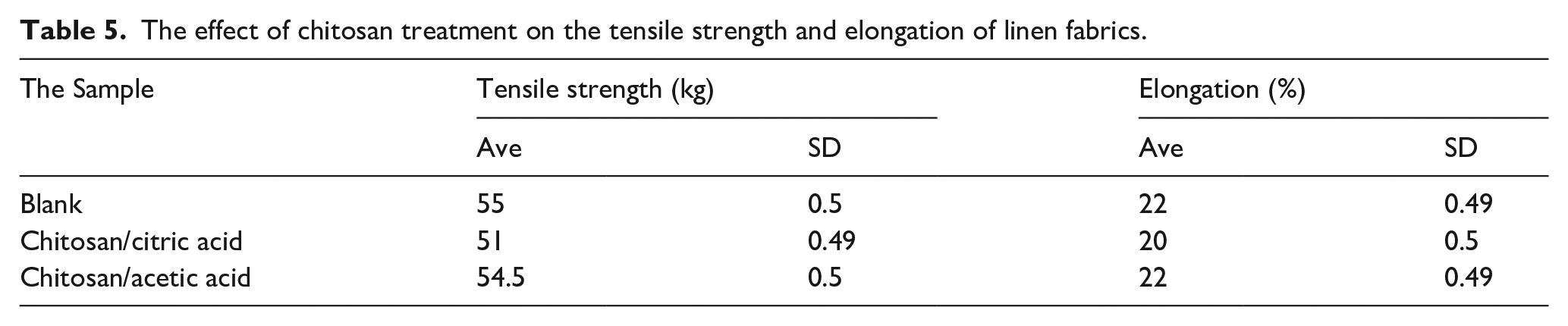
The effect of chitosan treatment on the tensile strength of linen fabrics
The tensile strength and elongation (%) of linen fabric were evaluated before and after the treatment with chitosan. It was observed from the results in a Table 5 a little decrease in tensile strength of linen samples after the treatment with chitosan. As well as the decrease in the tensile strength was more with chitosan/citric treated samples because of the chitosan/citric crosslinking mechanism, which decreases the tensile strength of linen fabrics.40,54 This result is confirmed by the findings of Sunder et al. who used different polycarboxylic acids to crosslink chitosan with cotton fabric and reported that there was a significant loss in the cotton strength after the treatment with chitosan and polycarboxylic acids. And observed that chitosan and citric acid combination show a minimum strength loss in both tensile and tearing strength. 54
The effect of chitosan treatment on the tensile strength and elongation of linen fabrics.
Evaluation of functionalization potential of lac dyed linen fabric
The antibacterial properties
The antibacterial activity of linen fabrics before and after dyeing with lac dye was evaluated against two strains of Gram-positive bacteria (Staphylococcus aureus and Salmonella typhimurium) and one Gram-negative bacteria (Escherichia coli). The chitosan-treated samples have the highest antibacterial properties along with the three tested strains for both chitosan/citric and chitosan/acetic treated linen samples as can be seen from the results in Table 6. This result is related to the polycationic structure of chitosan which causes death to the cell by interacting with anionic proteins of the microorganisms. It causes extensive alterations to the cell surface and alters the permeability of the cell. This will lead to the leakage of intracellular materials such as electrolytes, UV-absorbing material, proteins, amino acids, glucose, and lactate dehydrogenase. Consequently, chitosan inhibits the normal metabolism of microorganisms and finally causes the death of these cells.16,54,55 Moreover, the linen samples with lac dye only have antibacterial activity in the order of (S. aureus> S. typhimurium > E. coli). Although both chitosan and lac dye have antibacterial properties, the antibacterial properties decreased after the chitosan-treated linen samples were dyed with lac dye. This result may be explained by the occupation of most amino groups in chitosan polymer (which is responsible for the antibacterial properties) with lac dye.
The antibacterial properties of chitosan treated and the dyed lac dye linen fabrics.

UV protection
Linen fabric has been afforded ultraviolet protection properties after the dyeing with lac dye as presented in Table 7. The UPF of linen samples was enhanced after chitosan treatment followed by lac dyeing and the protection grade reached very good and excellent for (chitosan/citric/lac dye) and (chitosan/acetic lac dye) respectively.
The ultraviolet protection properties of lac dyed linen fabrics.

The UV protection of natural dyes depends on the absorption characteristics of the functional groups such as tannin-containing polyphenols, flavonoids, anthraquinone, terpenoids, and aromatic conjugated systems. 27 The ability of lac dye to the UV protection is related to its anthraquinone structure. The high UV absorption of lac dye can be seen in the UV visible spectrum Figure 9, which identifying the cause of the increased UPF of colored fabrics.35,56

The UV-Vis spectra of lac dye.
Also, Mongkholrattanasit et al. found the ultraviolet protection factor (UPF) values of lac dyed silk fabric with and without metal mordants ranged between very good and excellent. 30 In addition to that, chitosan has a considerable role in these results, where it improved the affinity of linen to lac dye and enhance the uptake of the dye so, increasing the UV protection property. These results are supported by findings of Kim SH, who assumed that chitosan increased the uptake of active moiety in green tea which would be responsible for the UV protection of cotton fabric. 52 Also, the results of current study are found in line with Verma et al. who studied the dyeing characteristics and UV protection property of onion skin cotton dyed after biopolymer chitosan treatment and reported that the UPF value was found higher for the chitosan treated onion skin dyed fabric than alum treated onion skin dyed fabric. 10
Conclusion
The current study introduces an eco-friendly approach for linen dyeing and finishing with natural lac dye through utilizing a microwave oven as a green heating tool and chitosan bio-moderating. The present findings confirm the validity of using a microwave for linen dyeing with natural lac dye and saving energy by cutting down the dyeing time to only 7 min. The K/S of lac-dyed linen fabric reached from 1.54 to 18.86 and 13 for acetic and citric acids, respectively, along with good fastness properties and no-salt dye technique. Also, these findings provide the optimum pH of lac dyeing for linen fabric is pH 3, which is considered the ideal pH for both the protonation of chitosan to allow the attachment of linen fabric with anionic lac dye and for the higher adsorption of lac dye. The tensile strength of linen fabric decreased after the modification with chitosan/citric acid, but there was no change in the case of acetic acid. This result concerns the crosslinking mechanism of citric acid with chitosan. The antibacterial properties of chitosan-treated and the lac dyed linen fabric decreased after integration of both. That may be related to the linkage between the functional groups of chitosan and lac dye. However, lac dye improved the UPF of linen fabrics and shifted their protection from no category to very good and excellent. Future research should consider the potential effects of other bio-mordants for lac dyeing in a microwave and compare it with chitosan and the validity of using a microwave in both mordanting and dyeing processes.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
