Abstract
Blue-green algae is an environmental problem; they reproduce rapidly and threaten aquatic ecosystems. Therefore, research on their use as a new energy resource is being actively conducted in various fields. This study was carried out to apply the natural dye from blue-green algae to the textile dyeing industry. Two methods were used to dye cotton and silk fabrics using blue phycocyanin dye extracted from blue-green algae. Six mordants were also tested in the process. The dyed fabrics were evaluated through dyeing depth and color fastness (washing, rubbing, dry cleaning, and light) tests. Phycocyanin dyed both fabrics blue and the dyeing effect was better on silk than on cotton fabric. In addition, Al, Sn, and Fe mordants were most effective for dyeing fabrics and co-mordants were more effective than pre-mordants.
Introduction
A harmful algal bloom (HAB) is defined as the situation where the cell density in a water body is >1.0 × 106 cells/mL, 1 resulting in adverse impacts such as esthetic problems, terrible taste, bad odor, and the release of toxic matter. 2 HABs are a major threat to the world’s water supplies and adversely affect human health and ecosystems. HABs are mostly dominated by microalgae derived from blue-green algae, which are capable of releasing harmful toxins.
Various technologies have been developed,3–7 but they have not been able to eliminate HABs completely. Therefore, it is necessary to approach the challenge from a different point of view than the unconditional removal of HABs. Various studies8,9 have been reported on converting algal waste into biomass energy. Cyanobacterial biomass extracted from blue-green algae constitutes an important third and fourth generation biofuel and has great potential to synthesize a wide range of metabolites, including mainly carbohydrates. Recently, however, carbohydrate-based biofuels derived from cyanobacteria—such as bioethanol, biohydrogen, and biobutanol—are attracting substantial attention as sustainable alternatives.
Blue-green algae contain a wide range of photosynthetic pigments. Phycocyanin from Spirulina platensis, blue-green algae, is a protein composed of two kinds of subunits, α (20.5 kDa) and β (23.5 kDa), to form α3β3 and nine phycocyanobilin moieties as a chromophore 10 (Figure 1). These chromophores, also called phycobilins, are the open-chain tetrapyrroles that are attached to the protein through thioether bonds. As a result, rather than the protein backbone structure, these phycobilins contribute to the blue color of phycocyanin.11–13 This natural blue colorant is a natural blue color that has been tested in various ways as a food additive.14–16 Kratzer and Murkovic 17 tested the C-phycocyanin extract of Cyanobacterium Arthrospira platensis as a blue colorant in beverages, demonstrating that samples containing the protein extract had colors similar to those of commercial beverages containing synthetic dyes.
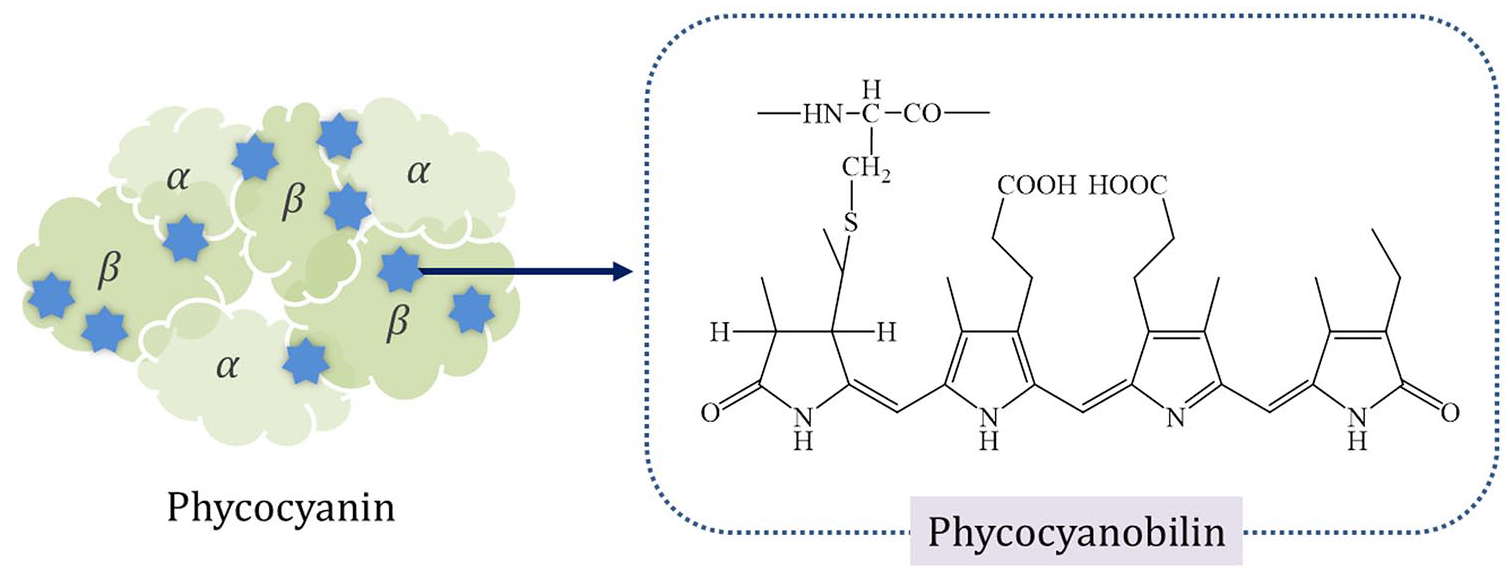
The structures of phycocyanin and phycocyanobilin.
Recently, algae have been tested in various ways to dye textiles, showing the possibility of its application in the textile industry. García et al. 18 dyed cotton fabrics with aqueous, alkaline, acidic, or organic extracts of four algal species: (1) the brown algae, Iyengaria stellata, (2) Sargassum muticum, (3) Colpomenia sinuosa, and (4) the red alga Laurencia obtusa at various temperatures, pH, and exhausting agent concentrations. They found that pre- and post-mordanting of algal powder dyed fabric using chemical mordants, particularly tannic acid, produced other colors, such as light and dark brown. El-Khatib et al. 19 dyed wool with dyes extracted from green algae and eco-friendly pre-treatment with chitosan and tannic acid to improve the dyeability of wool fibers. They reported that wool fibers pretreated with chitosan and tannic acid lead to high color strength compared to the untreated fibers.
Natural blue colorant is a rare natural dye for dyeing fabrics. Especially, blue-green algae are known as an environmental waste that reproduces rapidly and threatens the aquatic ecosystem. Applying the blue dye produced by recycling these blue-green algae to the dyeing of fabrics is an extraordinary challenge. Phycocyanin is a natural blue dye and has already been proven to be a food coloring agent. However, it is only stable at low temperatures and very sensitive to heat treatment, losing the intensity of its color when heated up to 60 °C for 30 min in a neutral solution.12,20 These issues create a particular challenge for the use of phycocyanin in dyeing fabric, where color stability is important for consumer preferences. In this study, we tested the dyeability of cotton and silk fabrics using phycocyanin blue dye extracted from blue-green algae. In order to improve the low color stability of natural pigments, six types of chemical mordant were tested by pre- or co-mordanting.
Materials and Methods
Materials
Cotton (97 g/m2) and silk fabric (68 g/m2) were supplied by Wujiang HMT Textile Co., Ltd (China). The blue-green algae (Microcystis aeruginosa) used in the experiment was obtained from Shanghai Guangyu Biotechnology Co., Ltd (China). The sample of blue-green algae was cultured in the BG11 medium at 25–30 °C. BG11 medium was chosen as the isolation medium due to its selectivity in promoting the growth of blue-green algae. 8 The preparation formula for BG11 medium is shown in Table 1. All the other chemicals used in this study were commercially available and of analytical grade.
Preparation formula of BG11 medium.

Preparation of Phycocyanin Dye Extracted from Blue-Green Algae
Blue-green algae (100 mL) were grown in 1 L of GB11 medium, a freshwater medium. Algae samples were incubated for 72 h at 25–30 °C under LED lighting (2000 lx). This study prepared blue-green algae collected from the culture aquarium as a dried powder. The algae samples were harvested by centrifugation at 15,000g at 20 °C for 15 min. The harvested algae samples are filtered using a filter membrane and washed with 50 mL of methanol and 25 mL of ethanol. Then, washed algae samples were prepared by grinding them into a powder using a freeze dryer. 21 Five volumes of distilled water were added to 50 g of the obtained algal powder, and then a pressure of 1300 bar was applied using a high-pressure cell disrupter (TS21401AA1AA, England) at 20 °C three times to obtain blue phycocyanin in blue-green algae. 22 After high-pressure treatment, the sample was centrifuged at 5000 rpm for 15 min to recover the blue supernatant and then left in a refrigerator at 4 °C for 12 h. Then, after vacuum filtration using filter paper, the blue phycocyanin dye powder was collected through freeze-drying at −20 °C (Figrue 2).

Preparation of phycocyanin dyes used to dye fabric.
Dyeing of Fabrics
In this study, a total of six mordants were used as follows: copper sulfate (CuSO4), calcium acetate (Ca(CH3COO)2), aluminum sulfate (Al2(SO4)3), aluminum potassium sulfate (KAl(SO4)2), iron sulfate (FeSO4), and tin chloride (SnCl2). A swatch of silk or cotton fabric sample (0.5 g) was immersed in acetate buffer 25 mL (0.1 M, pH 4) containing phycocyanin powder (0.5 mg/mL) and mordant (0.3 mg/mL) at a liquor-to-goods ratio of 50:1 and treated at 65 °C for 2 h. The treatment was performed under agitation at 100 rpm in a shaking water bath. After the dyeing process, the samples were thoroughly washed several times with deionized water to remove any unfixed dye. The fabric samples were dried in an oven at 55 °C prior to conducting color measurements. In the case of pre-mordanting, the mordant and fabric were treated at 95 °C for 20 min prior to the dyeing reaction. The detailed dyeing process is shown in Figure 3.
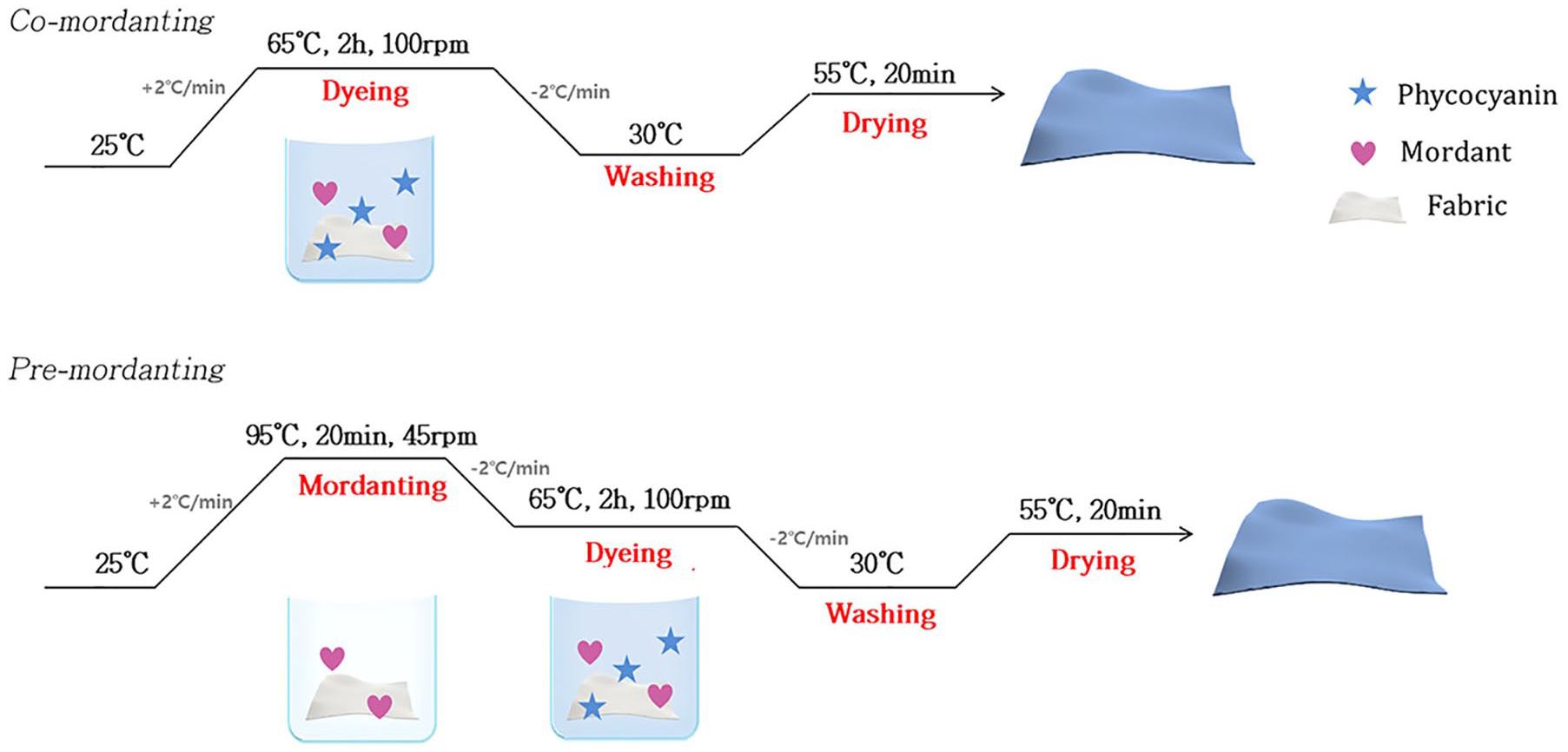
Dyeing process of pre- or co-mordanting.
Evaluation of Color Depth
A Color-Eye 7000A Spectrophotometer (GretagMacbeth, USA) with an aperture diameter of 7.7 mm was used to determine the color values, color strength, and differences between dyed fabric samples, represented by the CIE L*a*b* color space system. Each sample was foldedinto three and measured four times. All values were measured and calculated under controlled conditions with the illuminant and observer conditions of D65 and 10°, respectively. The color depth (K/S value) was calculated based on the Kubelka–Munk equation, as shown in equation (1) as follows:
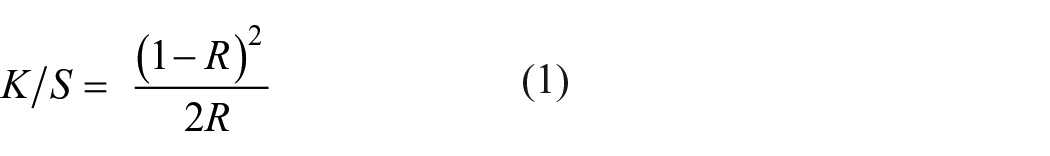
where K is the absorbance coefficient, S is the scattering coefficient, and R is the reflectance. 18
Color Fastness Tests
The washing colorfastness and sunlight fastness were tested based on ISO 105-C06:2010 and AATCC-16.3:2014, respectively. The dry cleaning fastness was measured according to AATCC-132:2013. The dry and wet rubbing fastnesses were tested based on AATCC-8:2016 by mounting the fabrics to a panel and applying 10 strokes on both the dry and wet fabrics, respectively.
Results
Dyeing Effect of Phycocyanin Dyes in the Presence of Chemical Mordants
The dyeing depth of cotton fabric is shown in Table 2. The overall K/S values were low, but the co-mordant showed a slightly higher K/S value than the pre-mordant. When dyeing with phycocyanin dye only, without the chemical mordant, the L value was 87.2, which was dyed light blue, and the K/S value was also very low at 0.5. However, in the case of the cotton samples with added mordant, they showed relatively high K/S values, proving that most chemical mordants have a good effect on the dyeing of phycocyanin dyes. When analyzing the effects of mordants, the Al mordants were the best, while the Sn and Fe mordants also showed relatively good effects. However, the K/S values (0.4) of the cotton fabrics pre-mordanted with Cu and Ca were lower than those of the no-mordant sample. When Cu mordant was used, the b value was 1 or more, and the color was green. However, considering the low K/S value of the Cu mordant, it was difficult to use as a mordant.
CIELab values obtained for the dyed cotton samples.
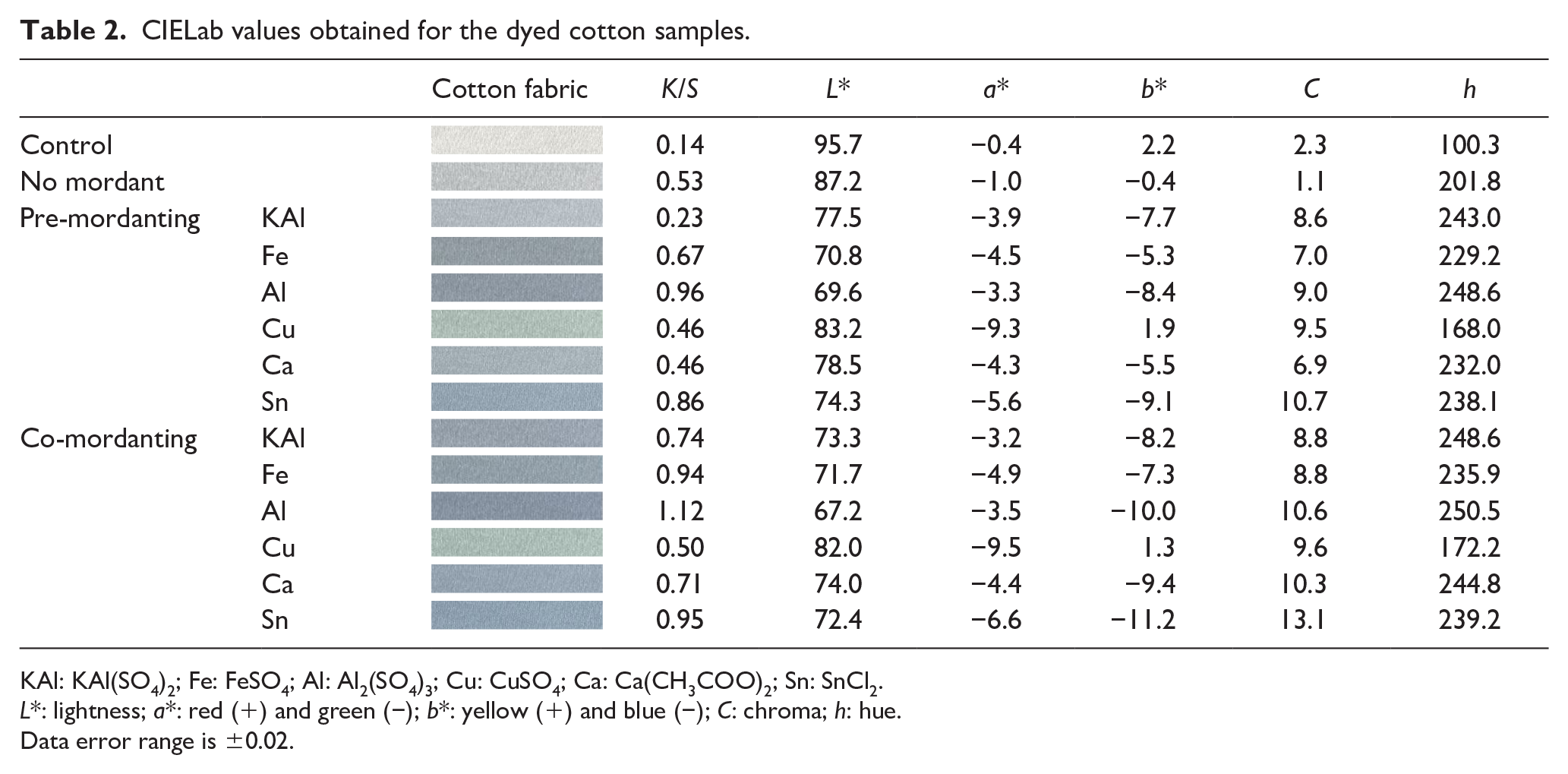
KAl: KAl(SO4)2; Fe: FeSO4; Al: Al2(SO4)3; Cu: CuSO4; Ca: Ca(CH3COO)2; Sn: SnCl2.
L*: lightness; a*: red (+) and green (−); b*: yellow (+) and blue (−); C: chroma; h: hue.
Data error range is ±0.02.
The dyeing depth of silk fabric is shown in Table 3. The dyeing effect of silk fabric was better than that of cotton when observed with the naked eye. In addition, the dyeing depth of all mordants showed higher K/S values than those with no mordants. When dyeing silk with phycocyanin, the Sn mordant was more effective than the Al mordants, and co-mordanting was more effective than pre-mordanting. The KAl and Cu mordants showed relatively low K/S values when compared to other mordants, and the Cu mordant was dyed light green, as observed in the dyeing of cotton fabric.
CIELab values obtained for the dyed silk samples.
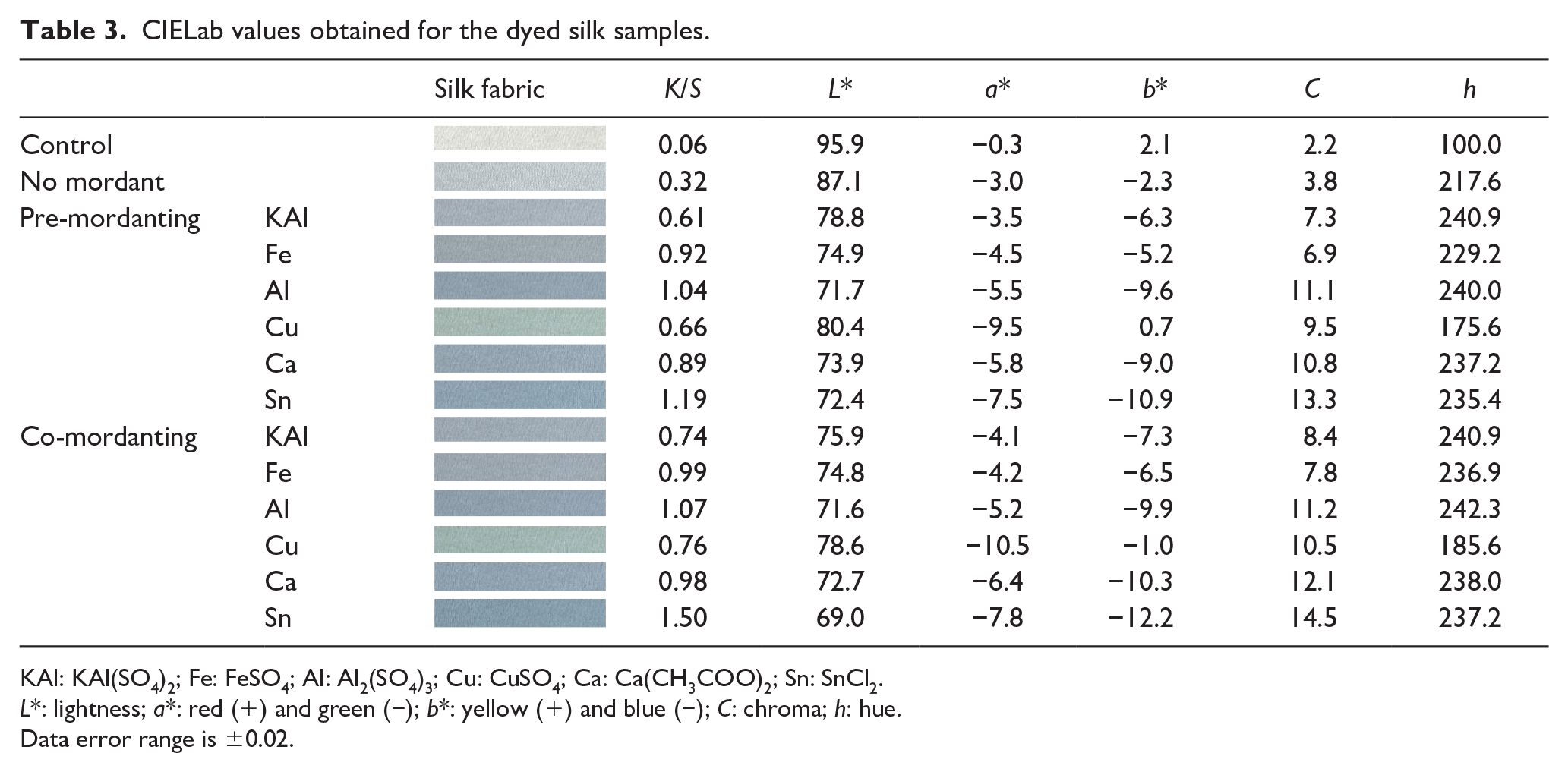
KAl: KAl(SO4)2; Fe: FeSO4; Al: Al2(SO4)3; Cu: CuSO4; Ca: Ca(CH3COO)2; Sn: SnCl2.
L*: lightness; a*: red (+) and green (−); b*: yellow (+) and blue (−); C: chroma; h: hue.
Data error range is ±0.02.
Color Fastness
Dyed fabrics are required to have sufficient resistance and durability against the conditions of use. Natural dyes have the disadvantage of low fastness when compared to chemical dyes. Therefore, the fastness properties—including washing, rubbing, dry cleaning, and light—were studied for the dyed cotton and silk fabric (Table 4). A fastness test was conducted on fabrics co-mordanted with the Al, Sn, and Fe mordants, which had the highest K/S values among the fabric samples.
The color fastness of the dyed fabrics.
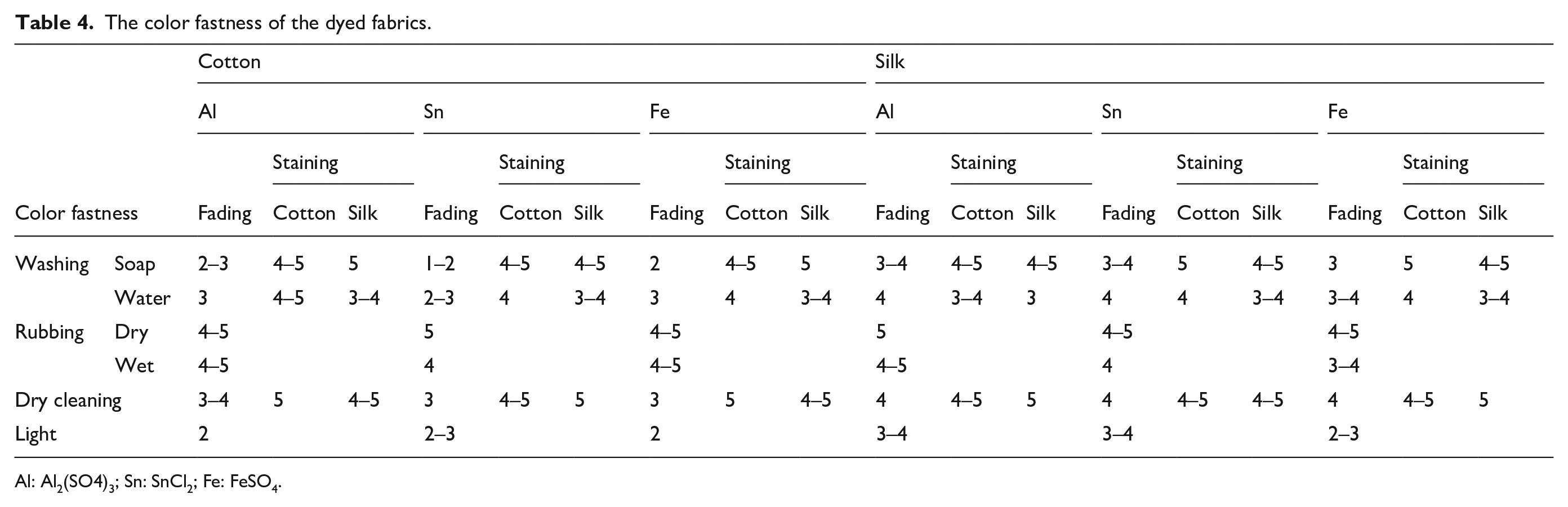
Al: Al2(SO4)3; Sn: SnCl2; Fe: FeSO4.
The washing fastness test was used to determine the degree of discoloration and degree of transfer to other fabrics when washed. For both cotton and silk fabric, it was found that most of the dye was removed even after one wash with soap, so that the color became lighter with the naked eye. Cotton fabrics showed more severe dye loss than silk fabrics, and in particular, cotton fabrics dyed with the Sn and Fe mordants showed a clear difference after washing. In addition, when washed with water, the cotton fabric showed a lower staining fastness than silk fabric. In particular, severe transfer of dyes to silk fabrics has been observed. It was possible that the phycocyanin dye removed by washing with water was transferred to another fabric next to it. In addition, the fading fastness of the silk fabrics was higher than that of cotton fabrics, which indicated that phycocyanin, a protein dye, has a higher dyeability with silk fabric than cotton fabrics.
In general, dry cleaning is used to wash silk, wool, fur, and so on, which are not suitable for washing with water. The dry cleaning fastness was tested considering the low washing fastness of 3–4 grade. Both fabrics exhibited higher grades than washing fastness in dry cleaning. Therefore, dry cleaning was suitable for washing phycocyanin-dyed fabrics.
As can be seen from the table, the light fastness of the silk fabric was slightly higher than that of the cotton. However, both fabrics dyed with phycocyanin dye showed a low grade of 2–3. Therefore, follow-up studies are needed to increase the light fastness of phycocyanin-dyed fabrics. In the rubbing fastness tests, both fabrics were similar, or the cotton fabric showed higher fastness than the silk fabric.
Discussion
It is common knowledge that algal blooms in freshwater systems can harm people and livestock due to the production of toxic compounds. Although various studies have tried to address this problem, algal blooms still damage ecosystems. Therefore, we must make efforts to turn blue-green algae into a new resource. Among the natural dyes that are used to dye textiles, there are relatively few blue dyes. Thus, blue phycocyanin was tested for textile dyeing, to demonstrate how environmentally problematic blue-green algae can be utilized in the textile industry.
In this experiment, the dyeing performance of cotton and silk fabrics was tested using phycocyanin dye extracted from blue-green algae. The dyeing effect of phycocyanin was better with the co-mordant than with the pre-mordant. As a result of testing six chemical mordants, three mordants (Al: aluminum sulfate, Fe: iron sulfate, and Sn: tin chloride) were the most effective. In addition, the cotton fabric showed high color depth (K/S value) on the order of Al > Sn > Fe, and silk fabric showed high K/S value in the order of Sn > Al > Fe. Silk fabric exhibited a better dyeing effect than cotton fabric, but neither fabric showed a K/S value greater than 2.0. As a result of testing the color fastness, the washing and light fastnesses were not good, but the rubbing fastness was good. It is presumed that the main component of phycocyanin dye is composed of protein, and thus, it is not properly combined with the fabric fibers. Therefore, a follow-up study is required to increase the textile dyeing effect of phycocyanin, a protein dye.
Most natural dyes lack dyeability with fibers and have low color fastness. Therefore, it is common to use inorganic or organic mordants to increase the stability of dyed fabrics.23–25 In this experiment, the metal mordants were used to dye fabrics using phycocyanin dye, which has poor color stability. However, several studies have reported that these metal mordants affect the metal content of wastewater.26–28 The use of metal mordants such as Cu and Sn seriously counteracts the potential sustainability of the natural colorant phycocyanin. Various studies have reported that the effect of bio-mordants is comparable to that of metal mordants.29–31 Therefore, if a bio-mordant is used, it is thought that it can replace metal mordants such as Cu and Sn. As a follow-up study, we are studying various substances, including bio-mordants, to improve the stability of phycocyanin dye extracted from blue-green algae.
Conclusion
This study was carried out to apply the natural dye from blue-green algae to the textile dyeing industry. Two methods were used to dye cotton and silk fabrics using blue phycocyanin dye extracted from blue-green algae. Six mordants were also tested in the process. The dyed fabrics were evaluated through dyeing depth and color fastness (washing, rubbing, dry cleaning, and light) tests. Phycocyanin dyed both fabrics blue and the dyeing effect was better on silk than on cotton fabric. In addition, Al, Sn, and Fe mordants were most effective for dyeing fabrics and co-mordants were more effective than pre-mordants. Cotton fabric showed high color depth in the order of Al > Sn > Fe, and silk fabric showed high K/S value in the order of Sn > Al > Fe. However, neither fabric showed a K/S value greater than 2.0. The fastness of the fabrics dyed with phycocyanin was not ideal, and thus, further studies are needed to understand how to dye them effectively.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (21674043).
