Abstract
The escalating concerns over the environmental impacts of synthetic dyes have catalyzed a shift toward sustainable and eco-friendly alternatives in the textile industry. Within this framework, the purpose of this research article is to investigate the environmentally friendly application of brown pods generated from amaltas (Cassia fistula) as a natural pigment for the purpose of dyeing wool yarn. The purpose of this study is to investigate the possibility of obtaining the reddish-brown anthraquinone-based pigment that is found in Cassia fistula pods by the application of microwave-assisted extraction process. The efficacy of microwave radiation in enhancing the extraction process is evaluated, highlighting its potential for expediting and improving the yield of the natural colorant. In addition, the response surface methodology is utilized in order to optimize the dyeing conditions. Particular attention is paid to characteristics like as temperature, duration, pH, and salt content. In addition, the study investigates the function of mordants, notably salt of Fe, salt of Al, and tartaric acid, in the process of improving colorfastness and generating shades that are both brilliant and long-lasting. The scope of the work is expanded to include the impacts of plant-based extracts as a biomordants, such as pomegranate, red sumac, and hulela zard, for the purpose of producing a wide variety of colors. Additionally, the impact of microwave treatment on wool yarn dyeability is explored, emphasizing improvements in surface morphology and dye absorption. The yarn analysis encompasses color strength, shade variation, and surface morphology, employing techniques such as Fourier transform infrared spectroscopy (FTIR) to elucidate chemical interactions. The study’s findings contribute to the growing body of knowledge on sustainable dyeing practices, highlighting the viability of amaltas-based natural pigments and environmentally friendly mordants in textile applications. Ultimately, this research underscores the significance of adopting ecologically conscious dyeing methods and paves the way for a more sustainable approach to coloration in the textile industry.
Introduction
Dyes are naturally colored organic substances commonly utilized for coloring papers, fabrics, leather, and other materials. A significant amount of synthetic dyes are suspected of containing intermediates that are harmful and carcinogenic, which, upon their release, pose threats to both the environment and human health, consequently contributing to pollution. 1 Synthetic dyes have detrimental impacts on the environment, even at low concentrations. 2 Furthermore, the discharge of colored wastewater contains highly harmful substances that exacerbate environmental problems.3,4 The introduction of synthetic dyes into aquatic ecosystems is known to diminish sunlight penetration, elevate the biological (BOD) and chemical (COD) oxygen demand, inhibit photosynthesis, and impede plant growth. 5 Dye effluents contain substantial quantities of organic and inorganic pollutants, such as chlorinated compounds, heavy metals, dyes, and pigments, contributing to elevated BOD and COD. 6 The adverse impact of the industrial revolution has had a negative influence on ecosystems, leading to decreased biodiversity. 7 Anthropogenic activities are a significant factor in environmental contamination. The textile industry, for instance, generates hazardous waste streams comprising various chemicals, including dyes and heavy metals, resulting in increased environmental pollution. Synthetic dyes, due to their non-biodegradable nature, pollute the environment and have harmful consequences for the atmosphere. Transitioning to natural or bio-based dyes is a promising alternative. Plants, animals, and bacteria can produce cost-effective and environmentally friendly dyes suitable for application in textiles. 8
As global regulatory agencies have heightened their vigilance toward the detrimental aspects of synthetic dyes, the promotion of environmentally friendly alternatives like bio-colorants has gained significant momentum across various sectors. The ensuing enumeration highlights some benefits of using natural dyes. 9 Throughout history, natural dyes have been extensively utilized for imbuing color into a range of substrates, encompassing food, leather, and natural fibers like wool, silk, and cotton. 10 Natural dyes find applications in diverse fields such as flavors, cosmetics, foods, pharmaceuticals, and even solar cells. These dyes exhibit diminished toxicity and pollutants, rendering them non-carcinogenic, less harmful to health, and environmentally benign. 11 A notable feature of many natural dyes is their non-hazardous impact on human health, with certain variants even demonstrating anti-allergenic properties, thereby ensuring safety during skin contact. 12 While synthetic dyes tend to fade over time, natural dyes exhibit an enhancement in color quality as they mature. Furthermore, the majority of natural dyes are considered ecologically sound, as they are sourced from renewable resources. In stark contrast, synthetic dyes are derived from non-renewable petroleum resources and are synthesized through intermediate processes fraught with numerous chemical hazards. 13 The enduring color retention and vibrant hues of natural dyes have propelled their widespread use, firmly establishing them as indispensable tools in the realm of textile dyeing. Therefore, naturally occurring pigments have been collected by our research team from a wide variety of plant sources, including gardenia yellow, 14 Triadica sebifera, 15 black rice, 16 Rheum emodi plants, 17 Dillenia indica leaf, 18 tulsi leaves, 19 Butea monosperma plants, 20 Pterocarpus santalinus, 21 roots of Alkanna tinctoria, 22 flower of Coral Jasmine, 23 and extractions of neem leaves. 24
For the purpose of getting natural colors from plants, the microwave-assisted extraction approach has been shown to be significantly more successful than the conventional method. This is mostly due to the fact that it reduces the amount of solvent that is used and speeds up the processing time. 25 Microwave radiation can disintegrate larger particles into smaller ones, facilitating their deeper penetration into yarns. In contemporary practices, microwave radiation serves as an economical and efficient heating source, displaying commendable performance. By minimizing energy, solvent, and time requirements, microwave radiation contributes to heightened extraction yields. 26 Furthermore, microwave radiation plays a role in enhancing shade fastness by modifying the yarn’s surface and augmenting its absorption capabilities. While certain metals are utilized to achieve desired hues, their byproducts could have adverse effects on the environment. A mordant, a substance that enhances color fastness, interacts with dyes to improve their adherence to fibers. In order to increase the color intensity and fastness of the dye, natural dyeing mordants are utilized. This helps to improve the interaction between the dye and the fabric. 27 The trend toward mordant utilization has transitioned toward the adoption of bio-mordants, eschewing hazardous chemicals in favor of a more eco-friendly and pollution-free natural dyeing process, thereby enhancing safety and environmental friendliness. 28
The present study aims to investigate the dyeing behavior of golden shower pods (Cassia fistula) as a natural source of reddish-brown anthraquinone colorants for dyeing wool yarn, utilizing the advantages of microwave radiation. In addition to its biological and pharmacological characteristics, the Fabaceae family, which is also known as the golden shower or amaltas, is widely recognized for its golden shower. For centuries, it has been utilized as a laxative, anti-inflammatory, anti-fungal, antioxidant, antibacterial, antidiabetic, and even anti-cancer agent for the purpose of treating skin conditions. 29 The reddish-brown anthraquinone-based dye present in the pods of this plant is utilized for coloring both natural and synthetic fibers. 30 Anthraquinone, the primary colorant in Cassia fistula pods, features a planar three-ring aromatic structure with two carbonyl groups as integral components (Figure 1). 31 Anthraquinone is found in many plants as an active ingredient, often with laxative and estrogenic properties. Additionally, several of these plants exhibit anti-bacterial, anti-parasitic, antiviral, and even anticancer attributes. 32 In earlier research, Hosseinnezhad et al. 33 used ultrasound method for the dyeing of wool yarn using Rubia tinctorum natural dyes. Baseri 34 demonstrated the utilization of date seeds as an environmentally friendly mordant and Zenian as a dye for the purpose of coloring wool yarns. Ammayappan and Shakyawar 35 focused on the process of dyeing carpet woolen yarn using a natural dye derived from cochineal. Imani et al. 36 demonstrated the utilization of zinc sulfate and magnesium sulfate as additives for simultaneously mordanting and dyeing wool yarns using madder pigments extracted using ultrasound. However, till date, no research has been conducted for the dyeing of wool yarns using natural dyes extracted from golden shower pods (Cassia fistula).

Structure of anthraquinone.
Therefore, this research endeavors to explore the potential of utilizing golden shower pods (Cassia fistula) as a natural source of reddish-brown anthraquinone colorants for dyeing wool yarn. Leveraging the benefits of microwave radiation, our study delves into the dyeing behavior of these colorants and their interaction with wool yarn. Through a comprehensive investigation, we aim to uncover the efficacy of this eco-friendly and sustainable dyeing approach, offering insights into the utilization of natural resources for textile applications. Our research was conducted with the intention of making a contribution to the development of dyeing methods that are both ecologically sensitive and practical. This was accomplished by integrating the conventional wisdom of using botanical sources with contemporary technology.
Experimental
Materials
Plants such as amaltas (Cassia fistula), serving as a source of natural brown colorant, and for the bio-mordant, the plants of red sumac, pomegranate, and hulela, were procured from Faisalabad’s local herbal market, Pakistan. Each plant material underwent meticulous washing with distilled water, followed by drying in shaded conditions. The plant sources were subsequently finely ground and then passed through a 20-mesh sieve to ensure a consistent particle size in the resulting powder. The sheep wool was employed for the dyeing process. Notably, all chemicals engaged in the processes of extraction, dyeing, mordanting, and fastness assessment were sourced locally.
Extraction and irradiation process
The process of extracting the natural colorant that is reddish-brown in color entailed heating a specified quantity of powder, which was 4 g, along with 100 mL of aqueous solutions that had both acidic and basic compositions. The extraction procedure maintained a Material-to-Liquid (M:L) ratio of 1:25, with the leaf powder percentage being 4%. Following the boiling process, the mixture underwent filtration to separate the reddish-brown pigment, which naturally comprised anthraquinone-based constituents. The resulting filtrate was subsequently utilized in the dyeing procedure. Upon separation, the reddish-brown filtrate, along with pre-treated wool yarn, underwent a 6-min exposure to microwave radiation using a microwave oven. Here, high-power microwave rays were applied for durations of 2, 4, 6, 8, and 10 min to both the yarn and the extract before being utilized in the dyeing process. Both microwave-treated and untreated Cassia fistula extracts were employed in the subsequent dyeing of wool yarn. In order to ensure that the correct proportion of Cassia fistula extract to surface-modified wool yarn was maintained during the dyeing process, it was performed out at a temperature of 80°C for a period of 45 min.
Dyeing condition optimization
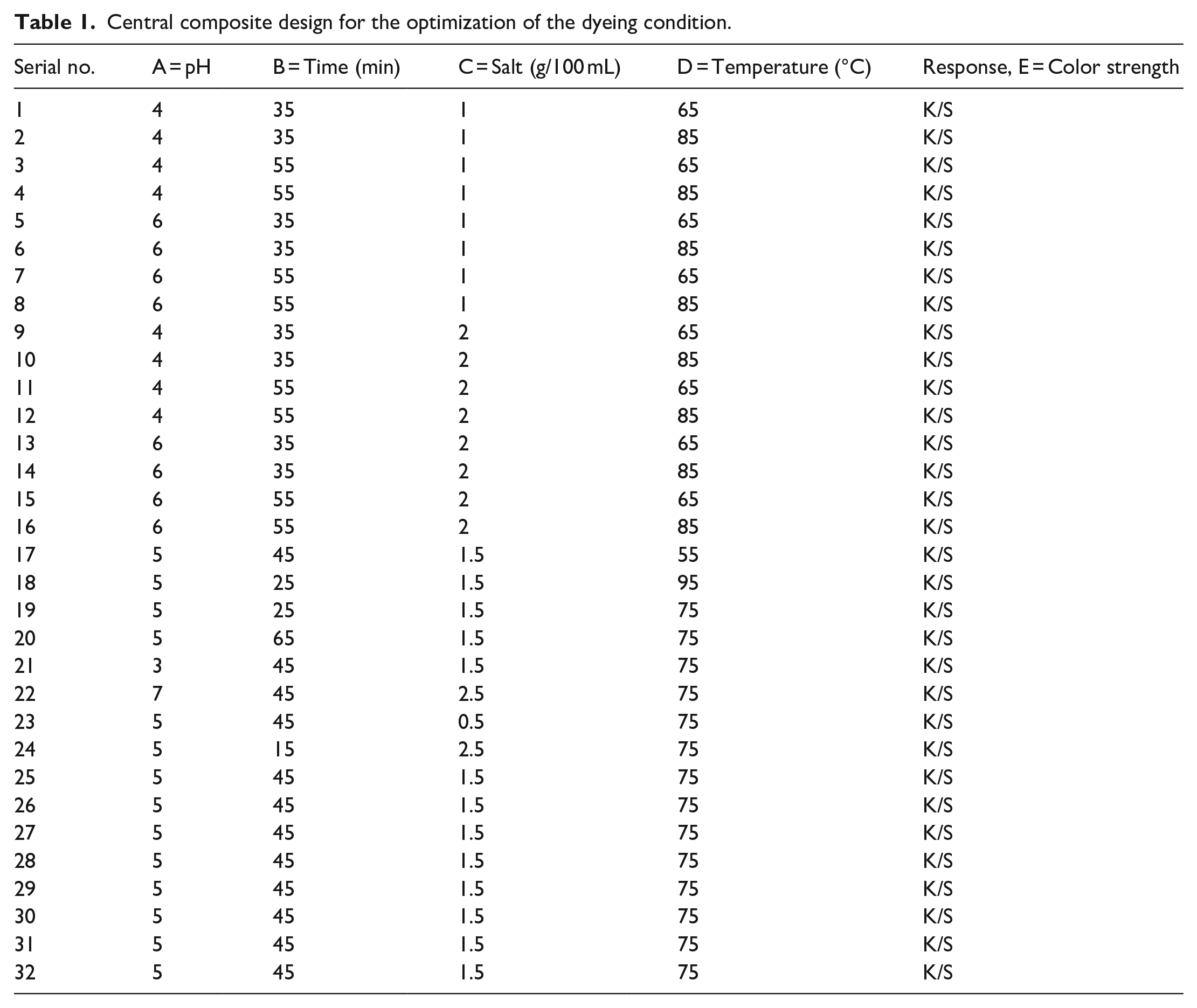
The optimization of dyeing conditions was conducted through the application of the Response Surface Methodology (RSM). Various dyeing parameters, namely temperature, duration, pH, and salt concentration, were subjected to optimization. In order to do this, a Central Composite Design (CCD) was developed, which included a total of 32 different tests. Within this design, different levels of temperature, time, salt concentration, and pH were systematically varied to establish optimal conditions for dyeing wool yarn effectively. Upon completing the experimental phase, each yarn sample was carefully evaluated, and the outcomes were subjected to thorough statistical analysis. The appropriateness of the applied models was assessed to attain desirable and meaningful results. The different parameters for the dyeing condition optimization are shown in Table 1.
Central composite design for the optimization of the dyeing condition.
Shade enhancement process
The study employed radiated Cassia fistula extract (RCFE) to generate novel colors on wool yarn in a controlled environment. The procedure entailed mordanting the yarn at a temperature of 80°C for a duration of 45 min, maintaining a mordant-to-yarn ratio of 25:1. In each instance, 25 mL of the mordant solution was employed to mordant both before and after the dyeing process, involving 1 g of yarns under selected conditions. This research focused on the augmentation of color fastness and shade quality through the utilization of plant extracts containing promising bio-active constituents, all within an ideal operational context. 37 To prepare the plant extract, a mixture of mordant powders (comprising Fe, Al, and Tartaric Acid) and water was combined and subjected to boiling. After the boiling process, the extract that was produced was filtered before it was applied, and the amount of the extract that was utilized was proportional to the weight of the wool yarn both before and after the dyeing process (Table 2).
Relation between sample formulation and obtained color strength and tonal values using Cassia fistula pods extracts.

NRAE: non-radiated amaltas extract; RAE: radiated amaltas extract; RWY: radiated wool yarn; NRWY: non-radiated wool yarn; MAD: microwave assisted dyeing. K/S: Color strength; L*: lighter/darker; a*: redder/greener; b*: yellower/bluer tone; c*: chroma; h: hue.
Yarn analysis
The analysis of the yarn encompassed several aspects. Color strength was assessed using the Kubelka-Munk equation, while shade variation was evaluated utilizing the CMC (L*, a*, b*, c*, h) equation within the Data color SF600 instrument. 38 In addition, the surface morphology of the yarn was investigated using scanning electron microscopy (SEM), with pictures being acquired at a resolution of 2000 times. In addition to this, Fourier Transformed Infrared Spectroscopy (FTIR) was utilized in order to determine the chemical makeup of the dye, fiber, and colored components. Furthermore, the evaluation of colorfastness properties for both dyed mordanted yarn and un-mordanted dyed yarn was conducted. The assessment involved adhering to ISO standard methodologies for light (ISO 105 B02), rubbing (ISO 105 X-12), and washing (ISO 105 CO3) fastness. 39 The Department of Applied Chemistry at Government College University Faisalabad, Pakistan, was the location where these assessments were carried out.
Results and discussion
Selection of radiation and extraction
As the utilization of natural dyes becomes increasingly prevalent across various aspects of life, with researchers continually exploring novel avenues for their extraction from newfound plants through economical and energy-efficient techniques. Microwave rays, being an affordable and readily available resource, have emerged as a promising approach. 40 These rays exhibit the capability to uniformly penetrate the plant structure during extraction, disrupting cell walls, and facilitating the amalgamation of solvent molecules with the inherent plant colorants through an efficient powdered solvent mechanism. 41 This dynamic process ensures a consistent supply of heat up to an optimal level, consequently enhancing mass transfer kinetics. The outcome is a higher yield at the chosen level, contingent on the employed solvent. 42 The alteration of the inherent scale structure of wool yarn after microwave treatment is due to the interaction between microwaves and the structural constituents of the wool fibers. Microwaves stimulate molecular motion and produce thermal energy within the yarn by reacting with water molecules contained in the fiber. 43 The localized heating might cause various alterations in the structure of the wool yarn. Specifically, it can impact the hydrogen bonds and disulfide connections present in the keratin protein structure of wool fibers. 44 The cuticle scales that are located on the surface of the fiber can be opened by the application of heat and subsequent cooling that occurs during the microwave process. This results in an increase in the amount of dyeing that occurs in wool yarn.
In this study on wool yarn, microwave rays were employed for durations of up to 10 min, resulting in the acquisition of aqueous extracts under neutral, acidic, and basic conditions. However, the basic medium was excluded due to the nature of wool keratin (amido), as it did not readily accept the basic nature of the colorant, resulting in a low yield. The effectiveness of the colorant was assessed both before and after microwave treatment, focusing on color strength. The investigation indicated that aqueous radiation up to 4 min yielded a favorable outcome (K/S = 4.5701), while acidic medium exhibited a productive result with radiation extended to 6 min (K/S = 9.8190) (Figure 2). Remarkably, the acidic extract yielded exceptional results when applied to woolen yarn. The analysis of color coordinates for shade variation revealed that employing the aqueous extract led to a vivid red shade (L* = 74.33, a* = 10.21) with a hue of yellow (b* = 13.64) (Table 2). With an extension of treatment time to 6 min, the shade deepened (L* = 46.25), adopting a more intense reddish-yellow hue (a* = 14.49, b* = 12.58, c* = 19.19). On the other hand, using an acidic medium, the initial shade appeared more luminous (L* = 67.80), with a reddish-yellow hue (a* = 8.850, b* = 36.24). However, with radiation treatment extended to 6 min, the shade intensified in strength (K/S = 9.8190), grew darker in appearance (L* = 47.32), and took on a more pronounced reddish tone (a* = 10.76), while retaining lesser yellowish tint (b* = 29.08).
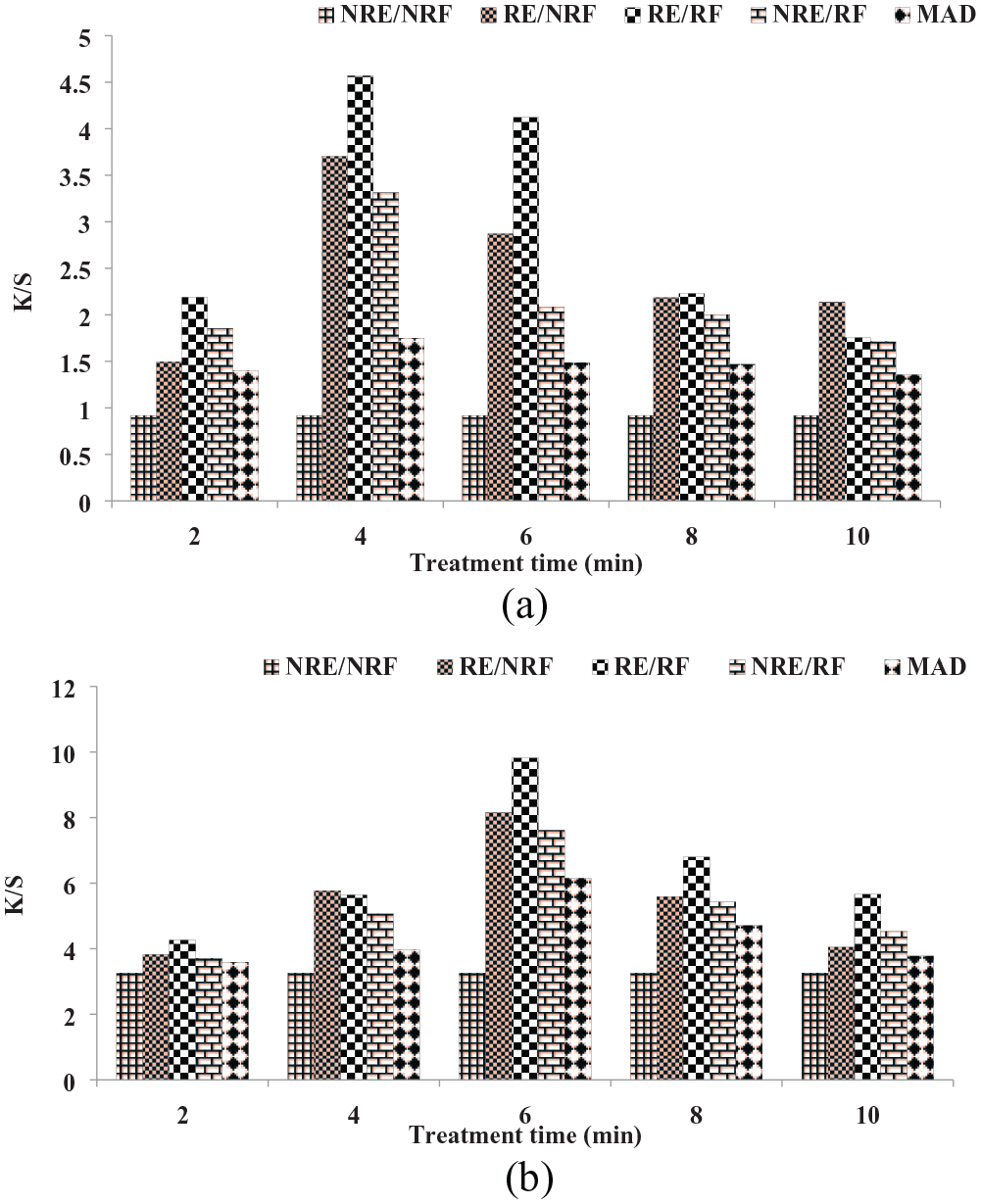
Radiation versus color strength comparison using aqueous (a) and acidic extract (b) of amaltas for wool yarn dyeing.
Physiochemical analysis of yarn
The FTIR analysis was harnessed to scrutinize the functional groups present on the surfaces of both wool yarn and Cassia fistula pod extract. Figure 3 exhibits the FTIR spectra of untreated wool yarn, microwave (MW)-treated wool yarn, untreated dyed wool yarn, MW-treated dyed wool yarn, untreated Cassia fistula pod extract, and MW-treated Cassia fistula pod extract. Prominent peaks emerged in the spectra of untreated wool yarn, untreated dyed wool yarn, MW-treated wool yarn, and MW-treated dyed wool yarn at 3273.79, 3280.06, 3279.66, and 3278.40 cm−1, respectively, corresponding to the stretching of -OH groups. Peaks at 1634.02, 1626.3, 1633.54, and 1625.72 cm−1 were attributed to the C=C bonds of the aromatic ring, while those at 1515.78, 1515.06, 1515.58, and 1514.15 cm−1 indicated C=O stretching. Notably, peaks at 3278.40, 1625.72, and 1514.15 cm−1 corresponded to the -OH group stretching, C=C in the aromatic ring, and C=O stretching, respectively. Distinctive peaks were also evident in the untreated and MW-treated Cassia fistula pod extracts at 3291.47 and 3334.46, 2924.34 and 2919.06, 1722.51 and 1728.86, and 1517.33 and 1515.5 cm−1, indicative of the -OH, benzene, and C=O functional groups. Previous research aligns with these findings, substantiating the presence of anthraquinone in Cassia fistula extract. 45 A planar aromatic structure is formed by the presence of two aromatic rings that are connected by carbonyl groups. Anthraquinone, which is the principal colorant found in Cassia fistula pods, is made of these two rings. A significant shift in the bands was seen in the FTIR analysis of the acidic extract obtained both before and after the application of radiation. The presence of a light band at 2924.13 cm−1, which was attributed to C=O ketones, was seen in addition to the appearance of a strong and broad band at 3273.79 cm−1. Post-radiation, the bands shifted to 3279.66 and 2924.7 cm−1. This suggests that for optimal colorant yield on wool yarn, both the extract and yarn should be irradiated for 6 min. The observed peaks in both the wool yarn and cassia pod extract spectra remained consistent with prior studies. 46

Spectral analysis of (a) un-treated wool yarn, (b) MW treated wool yarn, (c) un-treated dye wool yarn (d), MW treated dyed wool yarn (e), un-treated Cassia fistula pod extract (f), MW treated Cassia fistula pod extract.
Subsequent to the microwave treatment, the surface of the wool yarn exhibited discernible improvements, as depicted in the scanning electron microscope (SEM) analysis (Figure 4). Evidently, the microwave radiation triggered alterations in the fibers’ surface structure, potentially mitigating pre-existing surface imperfections or scratches. This phenomenon led to the smoothening of the wool yarns surface, rendering them more receptive to dye uptake (as observed in Figure 4(b)). After MW treatment, noticeable scratches have been observed, indicating that the scaling process has contributed to the physical modification of the fiber. 47 These scratches suggest an enhanced surface area and texture, which, in turn, facilitates a more secure adhesion of the dye to the fiber. The enhanced dye uptake capability of wool yarn following microwave treatment carries substantial implications for diverse applications, particularly within the textile industry. The improved dye penetration facilitates vibrant and enduring dyeing effects on the wool yarn (depicted in Figure 4(d)). This outcome underscores the significance of microwave treatment in augmenting the dyeing process, consequently enhancing the quality and appeal of wool yarn-based products.

Scanned images of un-treated wool yarn (a), MW treated wool yarn (b), un-treated dyed wool yarn (c), MW treated dyed wool yarn (d).
Selection of dyeing variables and statistical analysis
To optimize dyeing conditions, the irradiated extract selected earlier was utilized to dye the radiated yarn, employing variable conditions prescribed by the central composite design methodology. 48 By systematically varying each parameter within its designated range, it was observed that the fitted model followed a linear trend (Table 3). In terms of statistical significance, the pH (p = 0.017) and salt (p = 0.039) played pivotal roles. This can be attributed to the presence of amide units in the wool yarn, serving as binding sites that interact effectively under acidic conditions to establish bonds with the dye molecules. 49 Similarly, the addition of salt contributes to effective coloration by facilitating the exhaustion of colorant from the dye bath-treated yarn, functioning within a narrow range of attractive forces that aid in the development of color shades. 50 This approach aimed to enhance sorption, resulting in the maximum color strength of the dyeing process. Thus, the individual contributions of pH and salt prove to be crucial for achieving desired outcomes. In terms of two-way interactions, the interplay between pH and time (p = 0.185), salt (p = 0.039), and temperature (p = 0.604) also demonstrated significant impacts. This suggests that time and temperature synergistically contribute to the dyeing process when combined with other parameters. Consequently, the application of 25 mL of irradiated extract with a pH of 4, supplemented with 1 g/100 mL of salt, at a temperature of 65°C for a duration of 35 min, yielded optimal and effective results. This comprehensive approach highlights the intricate interdependence of various factors in achieving the desired dyeing outcome.
Statistical analysis for optimization of dyeing variables.

Mordanting for shade development
Mordanting is a crucial process for securing the steadfast fixation of natural colors onto yarn, as most natural dyes exhibit poor fastness characteristics. In our study, environmentally friendly chemical anchors were employed both before and after the dyeing process, specifically aluminum and iron salts, as well as tartaric acid (TAA). Prior to dyeing, proportions of 2% iron salt solution, 1% aluminum salt solution, and 1.5% TAA solution, relative to the yarn weight, produced deeper shades (Figure 5(a)). Following dyeing, the application of 2% iron salt, 1% aluminum salt, and 1% TAA, again based on yarn weight, yielded excellent color strength on the yarn (Figure 5(b)). Consequently, for Cassia fistula dyeing, iron mordanting is recommended, as it is challenging for this dye to absorb into yarn voids and remain unfixed. 51 After washing, a majority of the complex is stripped, leading to a diminished shade. Comparatively, both before and after dyeing, the use of 1% iron salt solution is advisable. Iron salt serves as a safe and environmentally friendly mordant, producing dark and colorfast shades. This is attributed to the formation of a metal complex between iron and the dye on the yarn through a coordinate covalent bond. 11 The uniform formation of this complex on the yarn requires an optimal mordant quantity. Below the selected amount (<1%), a less complex is formed, while above (>1%) leads to an accumulation of complex clusters.

(a) Pre-chemical mordant, and (b) post-chemical mordanting of wool yarn with Cassia fistula pod at optimized conditions.
Within our study, red sumac, pomegranate, and hulela zard were employed to attain darker shades both before and after wool yarn dyeing (Figure 6). Pre-dyeing proportions of 0.5% red sumac, 1% pomegranate, and 1.5% hulela zard, and post-dyeing proportions of 2% red sumac, 0.5% pomegranate, and 1.5% hulela zard, yielded outstanding outcomes. Comparatively, for both pre-dyeing and post-dyeing phases, pomegranate proved to be the optimal choice for bio-mordanting. This bio-mordant exhibited promising results for achieving deep, robust shades when dyeing wool yarn with cassia pods. From our comprehensive study, both environmentally friendly chemical and bio-mordants were identified and employed. Iron salt, as a chemical mordant, and pomegranate, a bio-mordant rich in tannins, were found to be particularly promising for achieving the desired high-strength shades.

(a) Pre-bio mordant, and (b) Post bio-mordant of wool yarn with Cassia fistula pods powder at optimized conditions.
The pursuit of standardizing natural dye methods continues, with an emphasis on enhancing their fastness properties. To make the process environmentally conscious, plant-based extracts are being advocated. This approach boasts dual advantages: the creation of new shades and the infusion of their biological activities when combined with the dye on the yarn. Phenolic (-OH) groups are primarily utilized for binding with the functional sites of the amide in wool yarn, while anthraquinone components (-C=O, -OH) participate in forming hydrogen bonds with the -OH or -C=O & -OH groups of natural dyes (Figure 7). This newly introduced hydrogen bonding, coupled with the addition of conjugation systems, aids in producing high-strength, colorfast shades.

Mode of reaction between colorant, yarn stuff and eco-safe chemical (a) and bio (b) mordants.
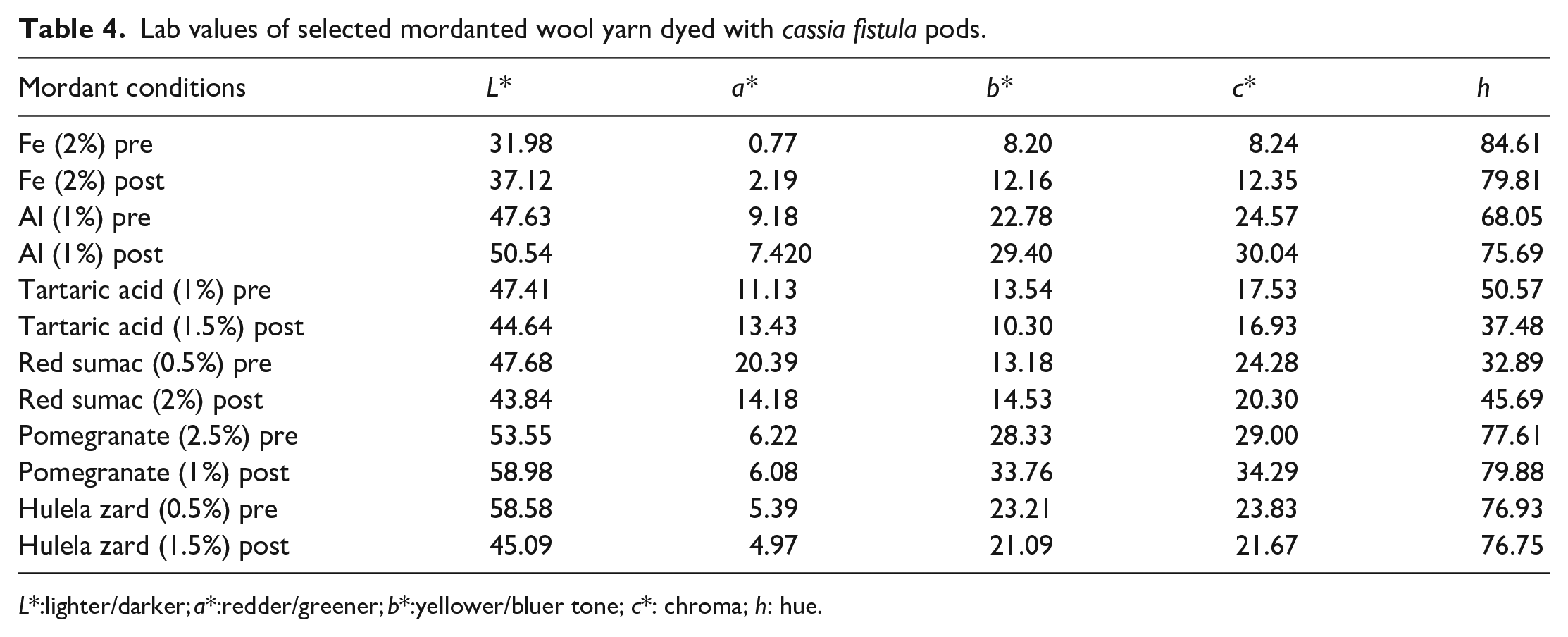
Shade appearance, revealing tonal variations, demonstrates that pre-dyeing mordanting with iron salt (2% relative to yarn weight) resulted in deeper shades (L* = 31.98) with a reddish-yellow hue (a* = 0.77, b* = 8.20, c* = 8.24) (Table 4). Similarly, post-dyeing with iron salt (2% relative to yarn weight) yielded a darker shade (L* = 37.12) with a more pronounced reddish-yellow hue (a* = 9.18, b* = 22.78). In contrast, aluminum (Al) mordanting produced darker shades (L* = 47.63) with less reddishness (a* = 9.18) and diminished yellowness (b* = 22.78, c* = 24.57). After dyeing with aluminum salt (1% relative to yarn weight), the resultant shade appeared darker (L* = 50.54) yet exhibited reduced reddishness (a* = 7.420) and heightened yellowness (b* = 29.40, c* = 30.04). The utilization of tartaric acid (TAA) as a mordant led to darker shades (L* = 47.41) with diminished reddishness (a*= 11.3, b* = 13.54) and yellowness (c* = 17.53). Following dyeing with TAA, the yarn exhibited a darker shade (L* = 44.46) with less reddishness (a* = 13.43) and yellowness (b* = 10.30, c* = 16.93).
Lab values of selected mordanted wool yarn dyed with cassia fistula pods.
L*:lighter/darker; a*:redder/greener; b*:yellower/bluer tone; c*: chroma; h: hue.
The plant extract also yielded a range of shades, with a tendency toward increased darkness and yellowness. For instance, using red sumac extract prior to dyeing (0.5% relative to yarn weight) generated a darker shade (L* = 47.68) with a more reddish-yellow tone (a* = 20.39, b* = 13.18, c* = 24.28). Similarly, post-dyeing application of red sumac (2% relative to yarn weight) resulted in a deeper shade (L* = 43.84) with reduced redness (a* = 14.18) and heightened yellowness (b* = 14.53, c* = 20.30). In the case of pomegranate, the yarn exhibited a darker shade (L* = 53.55) with less pronounced reddish yellow (a* = 6.22, b* = 28.33, c* = 29.00). After dyeing with pomegranate (1% relative to yarn weight), the shade became even darker (L* = 58.98) with decreased redness (a* = 6.08) and increased yellowness (b* = 33.76, c* = 34.29). Using Hulela zard extract before dyeing (0.5% relative to yarn weight) resulted in a darker shade (L* = 58.58) with diminished redness (a* = 5.39) and heightened yellowness (b* = 23.21). Following dyeing, the shade remained darker (L* = 45.09) with reduced redness (a* = 4.97) and increased yellowness (c* = 21.67). In essence, the shades achieved through mordanting, and plant extract application vary in darkness and tonal properties, providing a broad spectrum of color possibilities for different applications.
Colorfastness grading of selected shades
Colorfastness is the primary need for any colored textile, especially those colored with natural dyes. Wool yarn that was dyed prior to being mordanted, whether with chemical or biological mordants, produced results that were satisfactory in terms of lightfastness, soaping, and rubbing, as shown by the ratings that are presented in Table 5. After mordanting, the materials, on the other hand, demonstrated ratings that ranged from high to beyond extraordinary. This improvement can be attributed to the formation of stable complexes on the yarn as a consequence of chemical mordanting, as well as the additional hydrogen bonding that is made possible by the OH groups that are present in bio mordants. 52 When it comes to the formation of colorfast colors, the properties of the yarn, the atmosphere in which the dyeing takes place, and the mordant that is used are all important elements. The conjugation system that is supplied by bio mordants and the coordinate bonding that is made possible by metals both contribute to the formation of deeper colors that are resistant to fading, as stated by the standards of the ISO standards. Because of this, the creation of colorfast colors and the improvement of the natural dyeing process in terms of its environmental friendliness and sustainability are dependent on the utilization of microwave treatment under certain conditions and the utilization of the appropriate quantity of mordants.
Colorfastness grading of wool yarn dyed with amaltas pod extract according to ISO standards.

Conclusion
This research paper delved into the utilization of natural dyes derived from amaltas (Cassia fistula) pods as a sustainable and eco-friendly alternative for dyeing wool yarn. The study explored the application of microwave-assisted extraction for obtaining the natural colorant from the plant material. The investigation demonstrated that microwave radiation significantly enhances the extraction process, facilitating the release of the reddish-brown anthraquinone-based pigment present in Cassia fistula pods. Optimization of dyeing conditions was conducted using response surface methodology, taking into account variables such as temperature, duration, pH, and salt concentration. The findings highlighted the significance of pH and salt in influencing the dyeing process. Additionally, mordanting with iron salt, aluminum salt, and tartaric acid were examined to improve colorfastness. Iron salt mordanting emerged as a promising choice, offering robust color strength and improved fastness properties. The study also delved into the effects of various plant extracts, including red sumac, pomegranate, and hulela zard, as sources of bio-mordants. The utilization of these extracts exhibited varying shades, showcasing the potential for creating diverse color palettes through natural means. Furthermore, the role of microwave treatment in enhancing the dyeability of wool yarns was explored, unveiling improvements in surface morphology and dye uptake. Ultimately, this study lays the path for the adoption of ecologically friendly processes in the textile sector, which ultimately fosters a greener and more sustainable approach to dyeing.
Footnotes
Author contribution
Dr. Shahid Adeel is the supervisor of the work in whose lab practical work has been done, whereas Mariam Younis conducted the experiments. Dr. Meral Özomay and Somayeh Mirnezhad have helped in characterization of the materials, whereas Rony Mia and Dr. Muhammad Imran have technically guided in running experiments and writing the manuscript.
Data availability
The work is from M.Phil studies.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors express their appreciation to the Deanship of Scientific Research at King Khalid University, Saudi Arabia, for this work through research group program under grant number RGP-2/523/44.
Ethical approval
N/A
Consent to participate and publish
We give consent to publish the work a part of M.Phil studies and is jointly contributed by all authors.
